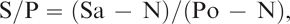Abstract
Fourteen first-calf heifers were tested free of antibodies against Bovine viral diarrhea viruses (BVDV) by serum neutralization and free of BVDV by polymerase chain reaction. Twelve were exposed to BVDV-1b strain CA0401186a at 84–86 days of gestation, and 2 were exposed to mock inoculum and served as negative controls. Fetuses were harvested by cesarean section at 115–117 days of gestation. The 12 fetuses removed from the BVDV-exposed heifers were BVDV positive based on virus isolation from kidney, thymus, cerebellum, and spleen. It can be assumed that these fetuses would have developed into persistently infected calves had they been allowed to go to term. Virus was not isolated from the fetuses of control animals. Ear punch samples were collected from all fetuses at time of harvest. Antigen capture enzyme-linked immunosorbent assay (ACE), using a commercial kit, was performed on ear punch samples that were frozen within 5 hr of collection and stored at −20°C until tested, tested after storage for 7 days at room temperature (18–25°C), or tested after storage for 7 days at 37°C. Samples stored for 7 days at room temperature or 37°C lost an average of 34% of their starting weight. All samples from BVDV isolation–positive fetuses tested positive by ACE, whereas samples from nonexposed fetuses tested negative, regardless of storage conditions. These results suggest that ACE testing of skin samples collected from aborted fetuses and stillborn calves found in the field may represent a practical surveillance method for BVDV-induced reproductive disease.
Bovine viral diarrhea virus 1 and 2 (BVDV-1 and -2) are recognized as economically important pathogens of cattle worldwide. 2,10,12,18 Although infection with BVDV may be associated with enteric and respiratory disease, it is generally recognized that the greatest economic impact of BVDV infections is related to reproductive disease. 1,4,10 Reproductive losses are the result of infection of the fetus, which may result in fetal reabsorption, mummification, abortion, congenital defects, and the birth of calves persistently infected (PI) with BVDV. Persistently infected animals are a major vector in the introduction, maintenance, and circulation of BVDV in cattle populations. 11 Accurate diagnosis of BVDV-induced reproductive disease is important to herd health management at the individual producer level and to the development of BVDV control programs at the national level.
Detecting BVDV in field submissions can be problematic for the diagnostic laboratory. Virus isolation (VI) is considered the gold standard for diagnosis but relies upon the existence of viable virus in tissues. Autolysis, which is common in aborted fetuses or stillborn calves found in pens or pastures, reduces the recovery of live virus, leading to false negatives. 5 Such tissues as thymus, spleen, and brain, which give reliable results for BVDV diagnosis when collected soon after death, may be poorly preserved in aborted or stillborn fetuses due to exposure to heat, light, and the autolytic microenvironment in the time between fetal death and expulsion and the time between expulsion and discovery in pens and pastures. Tests based on detection of BVDV antigen in sections of haired skin of cattle have become popular due to the convenience of sample collection (ear notching) and the relative stability of the sample. 6,8,11,14 Skin samples, particularly biopsies derived from the ear, are easier to collect from aborted and stillborn fetuses than organ samples and are less likely to be affected by autolysis. Because aborted and stillborn fetuses found in pastures and pens are seldom collected immediately after expulsion and frequently may be exposed to the elements for 1 or more days, the question of stability of the antigen in these samples has been raised. The purpose of the current study was to compare the detection of BVDV in fetal ear punch samples by antigen capture enzyme-linked immunosorbent assay (ACE) between samples frozen at −80°C after collection. After 2 days, the samples tested were processed and held for 7 days at −20°C, room temperature (18–25°C), or 37°C. The range of temperatures (−20°C to 37°C) reflects the range of temperatures that would be observed in a Midwest feedlot over the course of a year.
Before the study, mixed-breed first-calf heifers were prescreened for virus neutralizing titers in serum in a microtiter plate assay, using the cytopathic strains BVDV2-296c and BVDV1-Singer as reference strains. Serum samples were also collected for polymerase chain reaction (PCR) testing as a means to confirm freedom from persistent infection with BVDV before the start of the study. The BVDV PCR testing was conducted at the Veterinary Diagnostic Laboratory of Iowa State University in Ames, Iowa, using a method described previously. 15 At the laboratory, the individual serum samples were pooled into groups of up to 5 for testing. All pools tested negative.
Prior to purchase, the dams were housed under commercial-type cattle feeding conditions. After purchase, the dams were held in controlled outdoor facilities until inoculation with BVDV. Melengestrol acetate, a synthetic progesterone, was added as a top dressing to the dams' feed 34 days before artificial insemination (AI). The AI process was performed over 3 days to ensure the dams were cycling and maximum conception was obtained. Dams were examined for pregnancy 30 days after AI by ultrasound and 62 days after AI by rectal palpation. Twelve of the dams were exposed to 105.4 tissue culture infective dose (TCID) of BVDV-1 strain CA0401186a by the nasal route. 16 The inoculum virus was tested for extraneous agents and was confirmed as BVDV-1 by indirect fluorescent antibody test using a BVDV-specific monoclonal antibody. a The volume of the inoculation dose was 4 ml total, divided into 2-ml aliquots per nostril, using a glass atomizer. b A plastic bag was placed over the nose of the dam for approximately 1 min prior to administration and for 1 min after administration of the inoculum to induce hyperventilation. The 2 control animals were inoculated with the same volume of mock inoculum. After inoculation, the pregnant dams were individually penned in an indoor facility.
Fetuses were 84–86 days of gestation at the time of the dam's inoculation and 115–117 days of gestation at the time cesarean sections were performed. After cesarean section, fetal tissue samples were collected from the thymus, kidney, spleen, and cerebellum. Samples were transferred to a collection bag containing 10 ml of isolation media (minimal essential media c supplemented with 3% adult bovine serum, d 1.05 g sodium bicarbonate per liter, and 0.25% amphotericin B) and frozen at −80°C until processing 2 days later.
Samples were processed by thawing in a 37°C water bath for 10 min and transferred to a disposable tissue grinder e and homogenized per the manufacturer's instructions. Samples were centrifuged at 1,500 × g for 20 min at 4°C, and 1 ml of supernatant from each sample was added to each of 4 wells of a 24-well plate f that had been seeded with MDBK (Madin-Darby bovine kidney) cells per well. One milliliter of isolation media was added to an additional preseeded well as a negative control, and 1 ml of 104.5 TCID/ml BVDV1-Singer was added to a second well as a positive control. The plates were incubated at 37°C in a humidified incubator under 5% CO2. After 7 days, 100 μl of cell culture fluid was removed from each well and transferred to a well of a 96-well plate preseeded with MDBK cells and incubated as described above. After 5 days, plates were fixed with 80% acetone in H2O for 20 min at room temperature. An aliquot of a directly conjugated commercial polyclonal anti-BVDV antibody f was added to each well, and the plate was incubated for 1 hr at 37°C. Samples were classified as positive or negative for BVDV based on fluorescent staining as described above.
Three circles, 0.64 cm in diameter, were punched from the ears of each fetus after delivery by cesarean section. One sample from each fetus was frozen in individual snap-cap vials at −20°C for 7 days. A second sample from each fetus was placed in a well of a 24-well tissue culture plate, which was covered with an air-permeable clear membrane g and kept at room temperature under normal ambient laboratory lighting (combination of fluorescent, incandescent, and sunlight) for 7 days. A third sample from each fetus was placed in a well of a 24-well tissue culture plate, which was covered with an air-permeable clear membrane g and placed in a 37°C walk-in, nonhumidified incubator. These samples were exposed to fluorescent lights 10 hr/day for 7 days. Samples were weighed before and after storage. After 7 days, 2 ml of 0.1 M phosphate buffered saline (pH 7.4) containing 0.2% biocide preservative h was added to each sample. Samples were then incubated for 2 hr at room temperature. Detection of BVDV antigen in samples was performed using a commercially available kit, i according to the manufacturer's instructions. Positive and negative controls supplied with the kit were run in duplicate and analyzed at the same time as the fetal ear notch samples. The presence or absence of BVDV antigen in each sample was determined by calculating the sample-to-positive (S/P) ratio for each sample. The calculation of the S/P ratio was determined using the following formula:
where Sa is the sample optical density (OD), N is the mean OD of negative control, and Po is the mean OD of positive control. Guidelines provided with the kit state that S/P ratios <0.20 indicate a negative BVDV antigen status, and S/P ratios>0.39 indicate a positive BVDV antigen status. Samples with S/P ratios between 0.20 and 0.39 are graded as “suspect” for BVDV antigen.
The mixed models analysis of variance procedure was used to compare group means. Statistical analysis was performed using a commercial software package. j Statistical significant difference was set at P<0.01.
Bovine viral diarrhea virus was isolated from thymus, kidney, spleen, and cerebellum collected for VI from the 12 fetuses harvested from dams exposed to BVDV. No viruses were isolated from any of the tissues collected from the 2 fetuses harvested from dams that were not exposed to BVDV. Virus isolation from the phosphate buffered saline extraction of ear notches used for ACE was unsuccessful regardless of storage condition. Ear punch samples held at room temperature and 37°C appeared desiccated at the end of the 7-day storage period and lost an average of 34% of their starting weight. There was no statistically significant difference between the weight lost by samples held at room temperature and samples held at 37°C. All ear punch samples harvested from fetuses carried by dams exposed to BVDV tested positive by ACE regardless of storage conditions (Table 1). Ear punch samples from fetuses carried by dams not exposed to BVDV tested negative regardless of storage conditions. However, there were significant differences (P <0.01) between the average S/P values observed for samples stored under different conditions. The lowest S/P values were observed in samples stored at −20°C, and the highest S/P values were observed in samples stored at 37°C (Table 1). The average S/P ratios of samples held at room temperature and 37°C for 7 days were 11.4% and 22.2%, respectively, higher than samples stored at −20°C. No experiments were done to determine the origin of the increased signal seen with samples stored at room temperature and 37°C compared with frozen samples. One possible explanation is that desiccation observed with tissues stored at room temperature and 37°C may have contributed to release of Erns viral protein from cell membranes.
Enzyme-linked immunosorbent assay results for ear punches stored at −20°C, room temperature, and 37°C.*
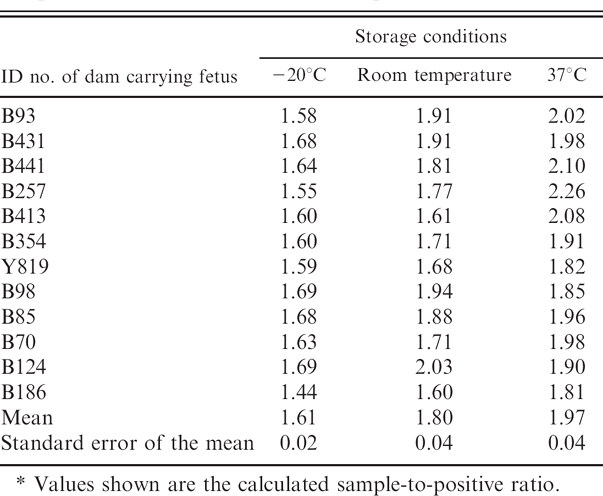
Values shown are the calculated sample-to-positive ratio.
Screening for BVDV in cases of reproductive disease is important to health management of individual herds and to the development of BVDV control programs at the regional and national level. Most of the effort in BVDV diagnosis has focused on the detection of neonatal or adult PI animals. 3,6,8,9 However, in addition to the birth of live PI animals, fetal exposure to BVDV may also result in mummification, abortion, or stillbirth. Validation of methods for detection of BVDV in fetal tissue is limited by the expense, availability of naïve dams, and biologic security required to generate infected fetuses of known gestation and infection history. The current study is unique in that the materials tested (the 12 infected fetuses) were generated under controlled experimental conditions.
Virus isolation is commonly considered to be the gold standard for diagnosis of BVDV. 7 When tissues are collected immediately after death, VI is a highly specific and reliable method. However, VI may give rise to false-negative results when used with tissues that have been exposed to such conditions as heat and light that do not support the recovery of live virus. 5,17 Immunohistochemistry (IHC) using ear notch samples has been shown to be a reliable method for screening herds for PI animals, 3,6 and ear notches have been shown to be stable samples for IHC analysis. Studies have shown that ear notch results are not affected by delayed fixation (refrigeration up to 10 days) or long-term fixation (up to 30 days). 13 However, IHC is dependent on the preservation of recognizable cell structures. Poorly preserved tissues or those damaged by autolysis or drying hamper diagnosis by IHC. In contrast, ACE does not rely upon the preservation of recognizable cellular architecture.
The detection of BVDV infection in tissues that are not in optimal condition is particularly important in the diagnosis of BVDV-related fetal death. Tissue damage resulting from exposure to heat, light, and tissue breakdown in the time between expulsion and discovery in pens and pastures can lead to false negatives when using VI or IHC. The present study demonstrates that holding ear punch samples at room temperature or 37°C for 7 days did not adversely affect the detection of BVDV in fetuses. The tissues lost an average of 34% of their starting weight, which indicates that the tissues had experienced substantial drying. These tissues were also exposed to light. Both drying and light exposure will occur when abortions or stillborn calves are expelled and not immediately harvested for diagnostic samples. It should be noted that these fetuses probably represented PI animals. Persistently infected animals are detected more reliably by ACE using ear notch samples than acutely infected animals. Thus, this method may not work on fetuses infected after 125 days of gestation. Further, this method is dependent on viral protein's still being present. Abortion associated with BVDV, in which the virus was cleared prior to the death of the fetus, will not be detected by this method. However, in such cases, other means of detecting BVDV, such as VI and PCR, will also fail. With those caveats, these results suggest that ACE testing of skin samples collected from aborted fetuses and stillborn calves found in the field may represent a practical method of screening for BVDV infections.
Acknowledgements. The authors thank Patricia Federico and Kathy Fulk for technical assistance, Harold Ridpath for statistical analysis, and Sue Ohlendorf for manuscript preparation. Disclaimer: Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Footnotes
a.
National Veterinary Service Laboratory/APHIS/USDA, Ames, IA.
b.
Model 151, DeVilbiss Healthcare, Somerset, PA.
c.
Minimum essential medium with Earle's balanced salts, with 2.0 mmol L-glutamine, with nonessential amino acids, without sodium bicarbonate, SAFC Biosciences, Lenexa, KS.
d.
Adult bovine serum (sourced in USA, gamma irradiated, and screened for bovine viral diarrhea viruses contamination per 9CFR), SAFC Biosciences, Lenexa, KS.
e.
Kendell Precision disposable tissue grinder system (15 ml), LifeLine Medical Inc., Danburg, CT.
f.
VMRD Inc., Pullman, WA.
g.
AirPore tape sheets, Qiagen Inc., Valencia, CA.
h.
ProClin® 300, Supelco, Bellefonte, PA.
i.
IDEXX HerdChek BVDV antigen test kit, IDEXX Laboratories, Westbrook, ME.
j.
SAS proprietary software release 9.1, SAS Institute Inc., Cary, NC.