Abstract
Bluetongue virus (BTV) and epizootic hemorrhagic disease virus (EHDV) have both been reported in mainland Ecuador, but their occurrence was unknown in the Galapagos Islands, an Ecuadorian province. We aimed to detect BTV or EHDV in cattle from the 3 main cattle-producing Galapagos Islands at a between-herd design prevalence of 20% and a within-herd design prevalence of 15%. Blood samples were collected from 410 cattle in 33 farms and tested for antibodies against BTV and EHDV by competitive ELISAs. All results were negative, suggesting that BTV and EHDV are not present in the Galapagos Islands.
Bluetongue virus (BTV) and epizootic hemorrhagic disease virus (EHDV) are orbiviruses of the Reoviridae family. The viruses are transmitted by biting midges of the genus Culicoides (order Diptera, family Ceratopogonidae).18,24 Infection by these viruses occurs in livestock in tropical and temperate regions of the world. Several serotypes of BTV have been isolated in the United States and Canada (serotypes 2, 10, 11, 13, 17). 24 Although BTV infection in the United States causes severe and frequently fatal disease in sheep and white-tailed deer, clinical disease in cattle is mild and observed infrequently. 24 In Central America and in the Caribbean islands, different BTV serotypes have been reported (serotypes 1, 3, 4, 6, 8, 12, 14, 17), although no clinical signs have been described in ruminants in these tropical and subtropical zones.9,24 In South America, BTV has been reported in Argentina (serotype 4), Brazil (serotypes 4, 6, 14, 17, 19, 20), Colombia (serotypes 12, 14, 17), Chile, French Guiana (serotypes 1, 2, 6, 10, 12, 13, 17, 24), Guyana (serotypes 14, 17), Peru, Suriname (serotypes 6, 14, 17), and Venezuela.8,9,23 BTV presence in Ecuador has been described for 30 y (serotypes 9, 13, 18).11,22
EHDV has been reported mainly as infecting wild ungulates in North America, causing a hemorrhagic disease (serotypes 1, 2, 6),12,15,16 and has also been identified in the Caribbean Islands and in Central and South America (EHDV-1, EHDV-2).8,12,23 There appears to be no correlation between virulence and EHDV serotype. For example, Ibaraki virus, long known as a strain of EHDV-2, has been reported to be pathogenic for cattle in Japan, although the homologous North American strains are not.12,18 EHDV-1 has been identified in cattle in mainland Ecuador. 22 The average homology of the Ecuadorian EHDV-1 strain with the USA EHDV-1 segment sequences (available in GenBank) was <95%, suggesting that the EHDV-1 isolated in Ecuador was not a result of a North American EHDV-1 transfer (French Reference Laboratory for bluetongue and epizootic hemorrhagic disease, pers. comm., 2018).
The Galapagos archipelago belongs to the insular territory of Ecuador and is made up of 13 islands comprising >13 km², and more than 100 islets. The islands are located ~960 km off the coast of Ecuador. 21 Although 97.7% of the islands’ territory has been allocated to a national park, agriculture has also been developed on ~26,356 ha of the 4 colonized islands: San Cristóbal, Santa Cruz, Isabela, and Floreana. In 1832, there was massive introduction of productive domestic species (cattle, goats, pigs, among others) that could have carried parasites or pathogens and led to the degradation of ecosystems. 19 In 1997, a first serologic survey of bovine diseases was carried out. 13 BTV was monitored using the agar gel immunodiffusion (AGID) test in 407 cattle sampled in the 4 islands; results were negative. 13 A ban on the entry of animals was only implemented in 2003. 5 Therefore, the introduction of orbiviruses through the importation of susceptible animals between 1997 and 2003 cannot be excluded. The objective of our cross-sectional study was to detect antibodies against the viral protein 7 (VP7) of both orbiviruses BTV and EHDV in cattle, the main ruminant species raised on these islands. Indeed, it has been reported that antibodies against BTV remain detectable several years after natural infection in cattle (up to 6 y in a study conducted in Germany after the introduction of BTV-8). 7 Detection in goats in our study was not attempted because cattle are much more attractive to Culicoides than other domestic species 1 and thus more frequently infected by these viruses.
In 2014, the Galapagos Islands had ~10,000 cattle distributed among the islands of Santa Cruz (~6,500 cattle), Isabela (~1,500 cattle), San Cristóbal (~1,500 cattle), and Floreana (~100 cattle). Most herds are formed by mixed cattle breeds (Bos taurus × Bos indicus). Santa Cruz holds the majority of cattle farms and is the main milk producer of the region. Floreana has only a small number of cattle farms, which are mostly located within the Galapagos National Park area. The number of farms for the 3 islands with the largest cattle population was estimated at 111 for Santa Cruz, 65 for San Cristóbal, and 38 for Isabela, with an average number of cattle per herd of 62, 23, and 40, respectively. Cattle on farms are raised in an extensive system of free grazing on open grasslands.
Because a limited number of animals could be sampled considering costs and practical implementation of the survey, we sampled only in the islands with the largest cattle population (i.e., Santa Cruz, Isabela, and San Cristóbal). In South American countries, where BTV and EHDV have been reported, seroprevalence was high in cattle.10,22,23 In France, when BTV-8 was introduced in 2007, between-herd seroprevalence was 60–100% and within-herd seroprevalence was 10–100% in cattle in regions where the virus circulated before winter. 6 Therefore, to detect BTV or EHDV antibodies in the Galapagos Islands, a between-herd seroprevalence of 20% and a within-herd seroprevalence of 15% were considered as reasonable values for vector-borne diseases in a South American tropical area. For each island, a sample size for freedom-from-disease was calculated to detect those seroprevalence values with imperfect tests and with 95% level of confidence using EpiTools (http://epitools.ausvet.com.au/). This yielded herd sample sizes of 11 in San Cristóbal, 11 in Isabela, and 17 in Santa Cruz, with 28 animals per herd in the largest herds and 14 in the smallest herds. For practical reasons, it was decided to randomly select 17 herds in Santa Cruz, 8 herds in Isabela, and 8 herds in San Cristóbal, and to test 15 animals >6-mo-old in each herd. In herds with <15 animals, all animals were tested.
In August 2014, blood was collected from cattle by coccygeal vein venipuncture. Sera were tested for antibodies against BTV by a competitive ELISA (ID Screen bluetongue competition kit, IDVet, Grabels, France), specific to anti-VP7 antibody detection with a sensitivity of 100% (95% confidence interval [CI]: 99.5–100%) and specificity of 99.9% (95% CI: 99.8–100%; values estimated from the validation file supplied by the manufacturer and from results available at the French Reference Laboratory for this serologic test). The detection of EHDV VP7 antibodies was performed using a competitive ELISA (PrioCHECK EHDV Ab serum kit, Applied Biosystems, Lissieu, France) with a specificity of 99.4% (95% CI: 98.0–100%) and sensitivity of 100% (95% CI: 96.7–100%; values estimated from the validation file supplied by the manufacturer). Both tests were performed according to the manufacturer’s instructions for these orbiviruses at the French Reference Laboratory for bluetongue and epizootic hemorrhagic disease (Maisons-Alfort, France).
Survey sensitivity and probability of freedom were estimated. 14 The prior probability of freedom was set to 0.5 for EHDV as recommended when there is no prior information about disease status. For BTV, because the survey conducted in 1997 in the Galapagos Islands yielded negative results, we chose a most likely value of 0.7, a minimum value of 0.5, and a maximum value of 0.8 for the Pert distribution assigned to this variable. A stochastic process was generated using a Monte-Carlo approach with 1,000 iterations in PopTools (http://www.poptools.org/) to take into account the variability of test performance and uncertainty of the prior probability of freedom.
Samples were collected from 410 cattle in 33 farms (Table 1) in the 3 sampled Galapagos Islands (Fig. 1). Most farms had more adult animals (with a median of 56–60%) than calves. All animals had negative results in the BTV ELISA and EHDV ELISAs. We obtained optimal survey sensitivity and probability-of-freedom values for Santa Cruz Island; the values were lower for Isabela and San Cristóbal (Table 1). This difference may be explained by the sampling of fewer herds and hence fewer animals in Isabela and San Cristóbal.
Number of farms and cattle sampled, survey sensitivity, and probability of freedom from bluetongue virus (BTV) and epizootic hemorrhagic disease virus (EHDV) in the 3 main cattle-producing Galapagos Islands.
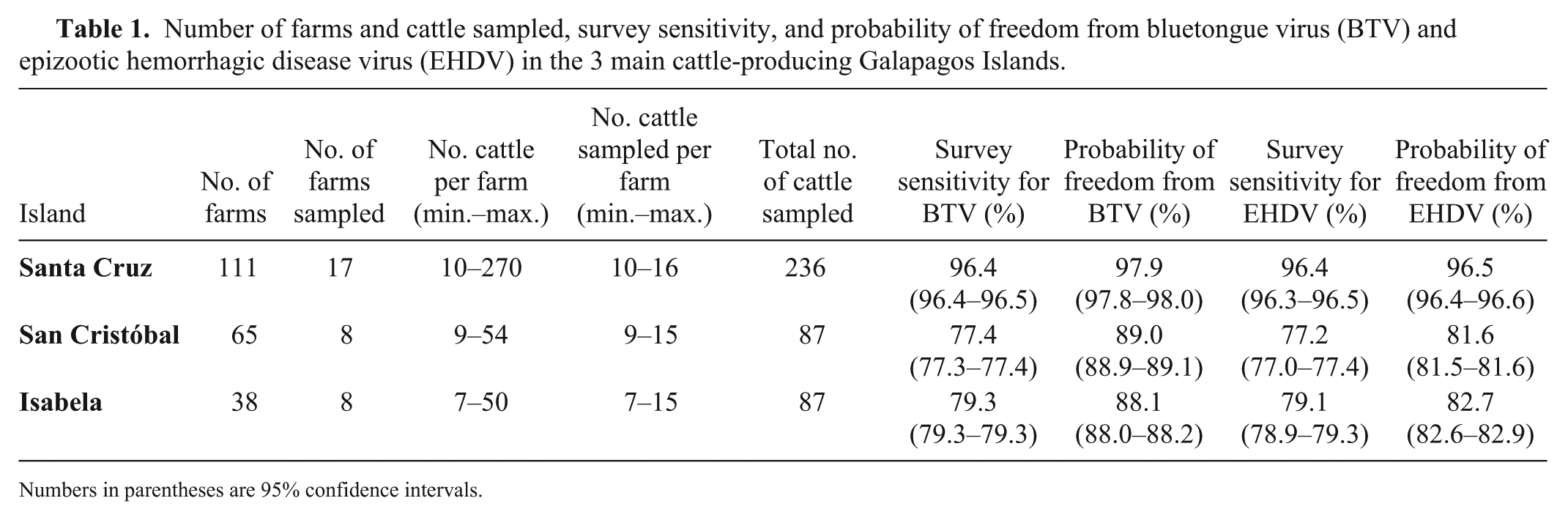
Numbers in parentheses are 95% confidence intervals.
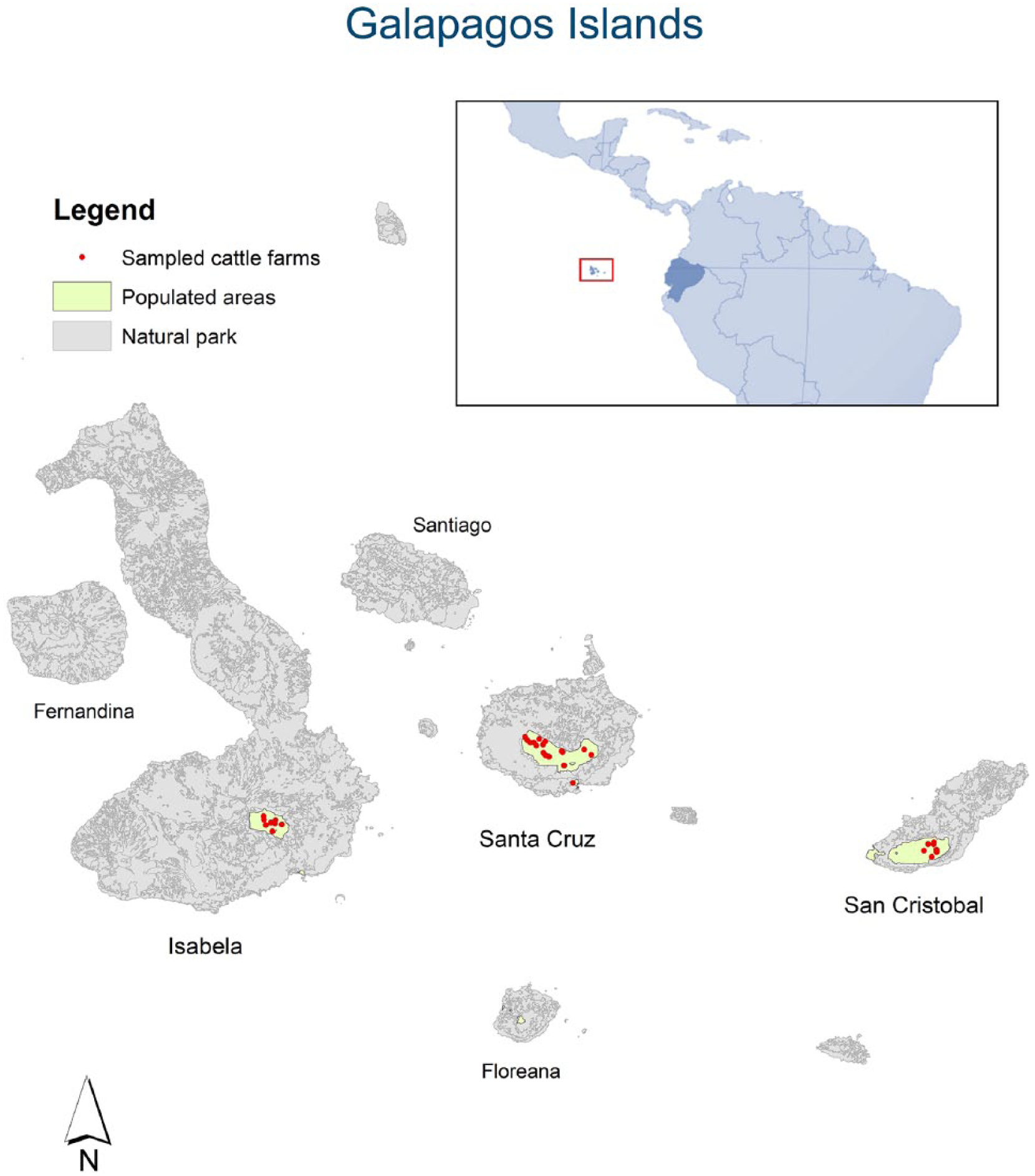
Map of study area and sampling location in the Galapagos Islands.
Our BTV and EHDV negative results suggest that these orbiviruses are not circulating among cattle of the 3 Galapagos Islands sampled at the freedom-from-disease sample size calculation used, and that these viruses could be absent in these islands for at least 5 y (average age of adult cattle). For BTV, this absence of circulation had been reported in 1997, 13 but the AGID test used at that time is considered less accurate than the ELISA used in our study. 25 The high sensitivity of ELISAs used in our survey makes the misclassification of negative animals almost impossible.
Our study has 2 main limitations. First, sampling design did not allow detection of between-herd seroprevalence <20% or within-herd seroprevalence <15%. Therefore, prevalence under these values, as could be the case if the viruses had been recently introduced in the cattle population, could not be detected. Moreover, in San Cristóbal and Isabela, the confidence of freedom values indicate that the sample size was not optimal (<95%) to detect this seroprevalence. However, supposing that BTV or EHDV had been circulating for some time, and taking into account the tropical climate of the Galapagos Islands, it could be expected that, if present, seroprevalence would be well above those values as has been reported in cattle of tropical regions in South America: 85% for BTV and 60% for EHDV in French Guiana, 23 94–100% for BTV in Venezuela, 10 54% for BTV in Brazil, 10 98.9% for BTV and 81.3% for EHDV in Ecuador. 22 Second, we did not perform a survey on midges in the sampled farms to confirm their presence or absence, to determine the species, and to estimate their abundance.
Different hypotheses could be proposed to explain the apparent absence of these orbiviruses in the Galapagos Islands: first, they had not been introduced through cattle movement; second, there are no suitable vectors (midges) in the islands; third, even if midges are present they may not be competent for the transmission of these orbiviruses to cattle. Interestingly, in 1983, serologic evidence of BTV infection was found in one of the Ecuadorian provinces from which cattle were being exported to the Galapagos Islands (El Oro) before the ban on animal importation. 11 Regarding potential vectors, Culicoides pusillus is the only Culicoides species that has been identified in the Galapagos Islands, the last report dating from 2005. 20 BTV has been isolated from C. pusillus both in Central America and the Caribbean, but not in South America. 10 However, C. pusillus is considered one of the main representatives of Culicoides species in South America, 10 and its role in BTV transmission should not be ignored. Therefore, if either of these orbiviruses were to be introduced, their propagation could potentially be allowed by this Culicoides species.
One could argue that the semi-isolated condition of the Galapagos Islands and the ban on animal importation provide sufficient guarantees to prevent introduction of these orbiviruses. However, those conditions cannot prevent the introduction of infected midges in the future from South America that could arise in 2 ways: either by windborne transportation, as the midge’s small body size (1–3 mm body length) enables largely passive flights over water bodies for hundreds of kilometers, 3 or by infected vectors carried by airplanes or ships. 17
Considering that the weather conditions in the Galapagos Islands are suitable for the development of vector species, the spread of BTV or EHDV among cattle further to the introduction of infected midges could be likely. It has been estimated that 463 alien insect species have been introduced unintentionally into the Galapagos Islands. 4 Among the alien mosquito species is Culex quinquefasciatus, a vector for agents of diseases such as avian malaria and West Nile fever, which is now considered a serious risk factor for the archipelago’s endemic fauna. 2 Once infection is established in the animals, the Culicoides species present in the island could also act as vectors, provided the Culicoides are competent for its transmission. A survey could be implemented in cattle farms with the help of entomologists from the Galapagos National Park to determine Culicoides species that could be potential orbivirus vectors for cattle.
Despite the animal importation ban enforced in the Galapagos Islands, the introduction of BTV- or EHDV-infected midges cannot be excluded. The detection of cattle positive to one or both orbiviruses in future surveys could be the signature of such introduction. In this respect, cattle of the Galapagos Islands could act as sentinels for these vector-borne pathogens.
Footnotes
Acknowledgements
We thank the farmers who participated in the study. We also thank technicians from Agencia de Regulación y Control de la Bioseguridad y Cuarentena para Galápagos (ABG), Laboratorio de Entomología Médica y Medicina Tropical (LEMMT) and Centro de Transferencia y Desarrollo de Tecnologías (CTT) from the Universidad San Francisco de Quito (USFQ), and undergraduate students from USFQ for the collection and processing of bovine blood samples.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
