Abstract
Neospora caninum is a parasite responsible for abortion in cows and neuromuscular disease in dogs. Serology is the most widely used technique to evaluate the prevalenceof N. caninum in different host populations. A sandwich enzyme-linked immunosorbent assay (ELISA) based on the use of an anti-SRS2 monoclonal antibody was evaluated against the indirect fluorescent antibody test for 100 canine sera and against a well-characterized ELISA for 102 bovine sera. In cattle sera, the relative sensitivity and relative specificity were 100%. In dog sera, the relative specificity and relative sensitivity were 94% and 86%, respectively. The kappa value was 1 for bovine sera and 0.77 for canine sera. The seroprevalence was 3.9% in bovine sera and 21–23% in canine sera. The SRS2 sandwich ELISA was considered a valuable tool in both species.
Neospora caninum (order Eucoccidiorida, suborder Eimeriorina, family Sarcocystidae) is an apicomplexan parasite responsible for abortion in cows and neuromuscular disease in dogs. This heteroxenous parasite uses the dog and the American coyote (Canis latrans) as definitive hosts. Cattle are considered as the major intermediate host. 9 Serology is commonly used to study the prevalence of N. caninum in animals. Different methods were developed, including indirect fluorescent antibody test (IFAT), 7 direct agglutination, Western blot, or enzyme-linked immunosorbent assays (ELISAs). 10 Whole tachyzoites 18 or purified antigens 15 also can be used for the detection of specific antibodies. Because intact tachyzoites are used as antigen in the IFAT, this test mainly detects antibodies directed to antigens present on the cell surface of the parasite. 2 Additionally, in different experimentally infected animal species, the N. caninum IFAT shows little cross reactivity with other coccidian parasites, including Toxoplasma gondii. 6 Consequently, IFAT is considered as a reference technique in N. caninum serology. 10 The aim of the present study was to compare the performance of a new commercially available SRS2 (also referred to as p38) 19 sandwich ELISA kit to the standard IFAT for canine sera (n = 100) and to a previously validated and commercially available ELISA for bovine sera (n = 102).
Canine sera (n = 100) were obtained from an animal control facility in Algiers, Algeria. Blood samples (5 ml) were collected from the cephalic vein into a dry glass tube. a After clotting, the tubes were centrifuged at 3,000 × g for 5 min, and the sera were separated and kept at −20°C until further use.
The IFAT was performed as previously described. 7,12 Each slide b comprised a positive and a negative control. For the canine sera, the positive control was the serum of a dog experimentally infected with N. caninum, whereas the negative control was obtained from a noninfected specific pathogen–free (SPF) dog. 14 Slides were observed under epifluorescence at a magnification of 400×. A sample was considered as positive when the majority (>50%) of the tachyzoites fluoresced uniformly. The cut-off used for canine sera was 1/100 as recommended 8 for an optimal specificity to avoid cross reactions with closely related T. gondii without affecting the sensitivity. The sensitivity and specificity for this cut-off were 82% and 100%, respectively. 7 The specificity of the method is difficult to address because neosporosis in dog is mostly asymptomatic; thus, sera from SPF dogs are necessary to guarantee their seronegativity.
The SRS2 sandwich ELISA kit c was used following the manufacturer's recommendations. The odd columns were coated with a monoclonal antibody specific to SRS2 (p38), 17 and a crude extract of tachyzoites was then deposited to specifically capture the SRS2 antigen. The even columns were left uncoated (negative controls). One hundred microliters of each diluted serum sample was added to twin wells (a coated and an uncoated well for each sample). The plates were incubated for 1 hr at room temperature and then washed 3 times with the washing solution provided with the kit. For the canine sera, horseradish peroxidase conjugate (anti–dog immunoglobulin G) d was diluted 1:10,000 in the appropriate dilution solution, whereas for the bovine sera, the kit conjugate was diluted according to the manufacturer's recommendations. One hundred microliters of the conjugate was added to each well and was incubated for 1 hr at room temperature. The plates were then washed 3 times, and 100 μl of the substrate solution (3,3′,5,5′-tetramethylbenzidine, H2O2) was added to each well and incubated for 15 min in the dark at room temperature. The reaction was stopped by the addition of 50 μl of stop solution (H3PO4, 1 mol). The optical density at 450 nm (OD450) was read using a plate reader. e For each serum sample, the difference between the ODs recorded on the coated and uncoated wells was calculated (ΔOD450). The ratio between each ΔOD450, and the ΔOD450 of the positive control, was calculated and multiplied by 100 (S/P). A ratio value >15 or <10 was considered as positive or negative, respectively, whereas a ratio value between 10 and 15 was considered as borderline.
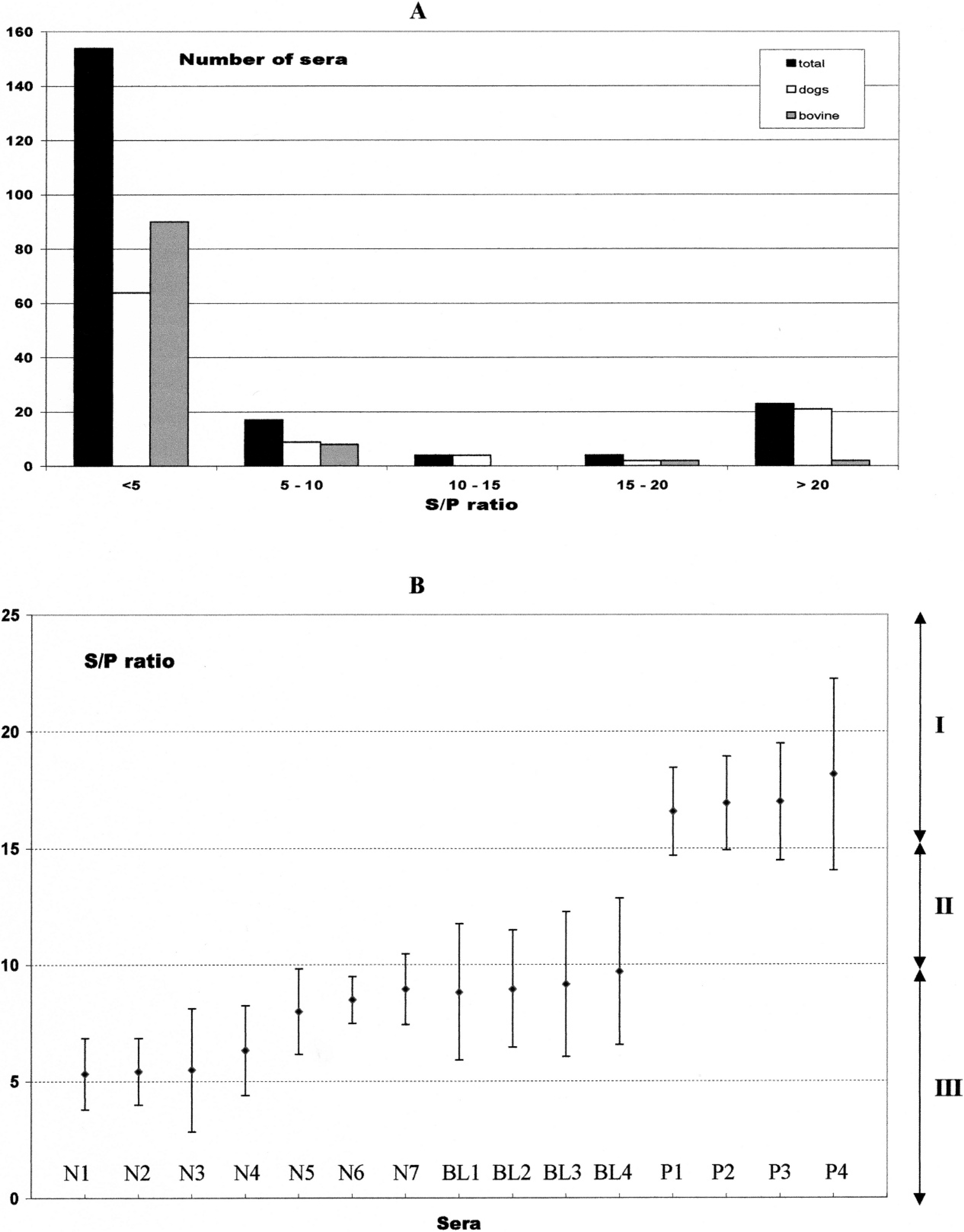
Serological status of the sera tested using the SRS2 sandwich enzyme-linked immunosorbent assay.
Among the 100 canine sera, using the SRS2 sandwich ELISA, 23 were positive (S/P >15), 73 were negative (S/P <10), and 4 sera were classified as doubtful (S/P = 10.2, 10.5, 10.9, and 11.5, respectively; Fig. 1A). The borderline sera were repeated twice, resulting in negative results (serum BL1 average S/P = 8.83, coefficient of variation (CV): 16.5%; serum BL2 average S/P = 8.97, CV:13.9%; serum BL3 average S/P = 9.17, CV: 16.7%; serum BL4 average S/P = 9.7, CV: 16.2%; Fig. 1B).
The seroprevalence was 21% (95% confidence interval [CI]: 13–29%) using IFAT and 23% (95% CI: 14.7–37.7%)using SRS2 sandwich ELISA. The relative specificity and sensitivity were 94% and 86%, respectively. The relative accuracy was 97%, indicating a good general agreement between the 2 methods, confirmed by a kappa value of 0.77 (calculated with the Win Episcope 2.0 software f ). McNemar's test was nonsignificant (P = 0.72). Therefore, the results of canine sera were similar for IFAT and SRS2 sandwich ELISA.
Cattle (n = 102) blood samples (10 ml) were collected from the tail vein of adult cows belonging to 7 different herds located around Algiers and processed as described above. A previously validated ELISA g , 23 was used to evaluate the performance of the SRS2 sandwich ELISA in bovine sera. The ELISA kit was based on whole antigens (WA ELISA) and was used following the manufacturer's recommendations, including the dilution of bovine sera (1/100). Using reference sera, the sensitivity and specificity of the WA ELISA were determined as 100% and 93.3%, respectively, in one study 23 and 100% and 99.7% in another study. 20 When compared to IFAT, the sensitivity and specificity were 93% and 96%, respectively. 21 Using sera from aborting and nonaborting cows, the sensitivity and specificity were 98% and 92%, respectively. 22 For bovine sera, the SRS2 sandwich ELISA resulted in 98 negative results (S/P < 10), 4 positive sera (S/P > 15), and no borderline serum. The results were identical for both ELISA kits with specificity, sensitivity, and agreement of 100%, respectively. The seroprevalence for WA ELISA and SR2 sandwich ELISA was 3.9% (95% CI: 0.1–7.7%).
Finally, the repeatability of the SRS2 sandwich ELISA kit was addressed as 4 borderline sera were retested and considered as negative. Therefore, 7 randomly selected (among the 17) negative sera with an S/P ratio ranging from 5 to 10 and the 4 positive sera with an S/P ratio ranging from 15 to 20 were repeated twice. The average S/P, CV, and CI at 95% were calculated and plotted (Fig. 1). Coefficient of variation values ranged from 6% to 24%. The negative samples stayed negative, and the positive samples stayed positive. However, for negative serum N7, the upper border of the CI was located in the borderline region (zone II on Fig. 1B). For positive sera P7, the lower border of the CI was located in the borderline zone. The presence of the borderline zone avoids the transformation of a positive into a negative and vice versa. In neosporosis, only high serological titers are of clinical relevance for borderline sera; thus, it is better to consider these borderline sera as negative, but a followup of the suspect animal is recommended. In a screening procedure, the borderline sera can be considered as positive since all positive samples will be confirmed by a confirmation test such as Western blot.
The present study was conducted to evaluate a new commercial sandwich ELISA for the detection of antibodies against N. caninum in both the dog and cattle. This kit uses a monoclonal antibody to capture SRS2 antigen from a crude lysate of tachyzoites. The performance of this kit was evaluated in comparison with the IFAT as the standard method for canine sera and in comparison with WA ELISA for bovine sera. Others studies have compared different ELISAs, based on the use of recombinant N. caninum–purified antigens, with the IFAT. 10 In cattle, previous studies 4,13 showed perfect agreement between ELISA and IFAT when the recombinant Ncp29 (NcsSRS1) antigen was used. ELISAs based on the use of rNcGra6 and NcGra7 also showed good correlation with the IFAT. 15 The RNcSRS2 antigen also gave similar results when compared to the IFAT. 11,17,19 In dogs, a competitive ELISA was validated against the IFAT for N. caninum–circulating antibodies with a specificity and sensitivity of 89.3% and 72%, respectively. 3 ELISAs were also validated against previously validated ELISA or Western blot. 23 ELISA validation can also be performed without reference method by using true positive and true negative sera previously confirmed by several methods. 1
Although it was not the main purpose of the current study, preliminary information about the potential impact of N. caninum in Algeria was collected for the first time. The seroprevalence in cattle (3.9%) was low compared to the authors' previous study conducted in Belgium (29%). 5 Interestingly, the 4 positive bovine sera originated from 3 herds with abortion problems. Nevertheless, a larger epidemiological control study should be performed to evaluate the seroprevalence in this region. The seroprevalence in Algerian dogs (21–23%) was similar to the prevalence obtained in other studies in rural or farm dogs. 16 This could be related to the fact that the dogs were stray dogs coming mainly from peri-urban areas. Once again, a larger epidemiological study must be performed in different canine populations (rural, stray, and urban dogs).
Acknowledgements. F. Ghalmi was financially aided by both the Belgian Technical Cooperation (Brussels, Belgium) and the Algerian Ministry of Higher Teaching via the National Veterinary School of Algiers.
Footnotes
a.
BD Vacutainer® Dry Tubes (368434), BD Diagnostics, Plymouth, Devon, UK.
b.
Black Teflon Slide (W0149D), Fisher Scientific Bioblock, Tournai, Belgium.
c.
Neospora caninum Antibody Detection Kit (BIO K 192), Bio-X Diagnostics SPRL, Jemelle, Belgium.
d.
Anti-Dog IgG (whole molecule)-Peroxidase Antibody (A6792), Sigma-Aldrich NV/SA, Bornem, Belgium.
e.
Multiskan Ascent®, Thermo Fisher Scientific BV, Breda, The Netherlands.
g.
HerdChek, anti-Neospora caninum, IDEXX Laboratories Inc., Westbrook, ME.
