Abstract
Porcine circovirus-2 (PCV-2) serology is frequently used to determine PCV-2 status and optimal timing of PCV-2 vaccination in the field. The objectives of the current study are to determine the diagnostic accuracy of 3 currently available commercial anti-immunoglobulin G (IgG) PCV-2 enzyme-linked immunosorbent assays (ELISAs) and to compare the ability of the 3 assays to detect and differentiate between anti—PCV-2a and anti—PCV-2b antibodies, as well as anti—PCV-2 and anti—PCV-1 antibodies. Fifty-five 3-week-old, conventional pigs were randomly allocated to 7 groups: 1) negative controls (n = 7), 2) PCV-2a (n = 8; inoculated with PCV-2 ISU-40895), 3) PCV-2b (n = 8; inoculated with PCV-2 NC-16845), 4) PCV-1 (n = 8), 5) vaccine A (n = 8; Ingelvac® CircoFLEX™), 6) vaccine B (n = 8; Circumvent® PCV2), and 7) vaccine C (n = 8; Suvaxyn® PCV2 One Dose). Blood samples were collected weekly, and all sera were tested by 3 different anti-PCV-2 IgG ELISAs. The results indicated that all ELISAs had area under the receiver operating curve values greater than 0.94, detected both anti-PCV-2a and −2b antibodies with no differentiation, and did not detect anti-PCV-1 antibodies in infected animals. One of the ELISAs was able to distinguish pigs vaccinated with vaccine B from pigs inoculated with either PCV-2a or PCV-2b.
Introduction
Porcine circovirus (PCV; family Circoviridae; genus Circovirus) was first identified as a contaminant of the permanent porcine kidney cell line PK-15 in 1974. 26 The virus is nonenveloped and composed of a single-stranded, circular DNA genome. Two major types of PCV have been identified, PCV type 1 (PCV-1) and type 2 (PCV-2). 19 Porcine circovirus-1 has been shown to be nonpathogenic in pigs under experimental conditions. 1 , 27 In contrast, infection with PCV-2 has been associated with respiratory disease, enteritis, reproductive failure, porcine dermatitis and nephropathy syndrome, and systemic infections, which are known as Porcine circovirus—associated disease or PCVAD. 22 While the pathogenesis of PCVAD remains unclear, it can result in high levels of PCV-2 viremia in 5% to 30% of a swine population. In affected animals, mortality can reach 70% to 80%. 22
Based on sequence analyses of PCV-2, type 2 can be further divided into 2 main phylogenetic clusters, PCV-2a and PCV-2b. 5 The PCV-2 genome is composed of 2 known open reading frames (ORFs) that code for functional proteins. The ORF1 gene encodes for a replication-associated protein (Rep), which is essential for virus replication. 4 The ORF2 gene encodes for the capsid protein, which has been shown to be immunogenic 20 , 21 and which has a greater nucleotide variation than ORF1. 15
Several commercial PCV-2 vaccines became available in North America in 2006. As of January 2008, 3 products are fully licensed and available in the United States for use in healthy pigs. Two of the vaccines contain the PCV-2 capsid protein expressed in a baculovirus (BA) system as an antigen. 6 , 8 The third vaccine is composed of an inactivated PCV-1–2 chimera virus. 7 Other notable differences between the 3 vaccines include the dose and timing of administration: 2-ml versus 1-ml injections and 1-time administration versus booster vaccination. 22
The release of PCV-2 vaccines provides an impetus for assessing the PCV-2 status of herds to determine the best time for vaccination. One method of assessing when and if PCV-2 is circulating in a herd is to conduct a cross-sectional serological survey to determine the prevalence and levels of anti—PCV-2 antibodies over time using 1 or more of the serological tests for PCV-2 including various enzyme-linked immunosorbent assays (ELISAs). 3,16,20,25,29,30
Most ELISA tests use the ORF2 region as antigen since it is specific and immunogenic for PCV-2. 28 Although most assays are based on PCV-2 ORF2, the methods of antigen production differ among assays. Specifically, antigen has been produced by expression of the capsid protein in a BA vector system, 3 , 16 , 20 in live virus preparations in PK-15 cells, 29 or through expression in Escherichia coli. 25 , 30 While reports indicate that ELISAs based on the ORF2 region of PCV-2 are useful, a direct comparison of the diagnostic accuracy of currently available ELISAs has not yet been performed to the authors' knowledge. In addition, reports on the ability of the assays to detect and differentiate between anti—PCV-2a, anti—PCV-2b, or vaccine-induced antibodies are lacking.
To address the gaps in knowledge on current PCV-2 ELISAs, the current study had the following objectives: 1) to determine the diagnostic accuracy of 3 commonly used ELISAs, 2) to determine whether each assay can detect and/or differentiate between anti—PCV-2a and anti—PCV-2b antibodies, and 3) to determine whether PCV-2—based ELISAs can detect anti—PCV-1 antibodies.
Materials and methods
Animals and housing
Animals were purchased from a PCV-2—negative herd, weaned at 2 weeks of age, and transported to the Iowa State University Livestock Infectious Disease Isolation Facility (Ames, IA). On the day of delivery, the pigs were randomly assigned to 1 of 7 rooms, each containing a 1.2 × 2.4 m raised wire deck pen equipped with 1 nipple drinker and 1 self-feeder.
Experimental design
Three-week-old, conventional pigs were randomly allocated to 7 groups and inoculated with either PCV-1, PCV-2a, or PCV-2b; vaccinated with commercially available PCV-2 vaccines; or left as negative controls as follows: 1) negative controls (n = 7), 2) PCV-2a (n = 8), 3) PCV-2b (n = 8), 4) PCV-1 (n = 8), 5) vaccine A (n = 8), 6) vaccine B (n = 8), and 7) vaccine C (n = 8). Upon arrival, pigs were bled, and serum was tested by quantitative real-time PCV-2 polymerase chain reaction (PCR) 23 to detect PCV-2 DNA and by a PCV-2 fluorescent antibody test (FAT) 24 to detect anti—PCV-2 antibody. The experimental inoculations and vaccinations were performed on trial day 0, at which time the pigs were 3 weeks of age. All pigs were bled at weekly intervals until trial day 49. All serum samples were tested by 3 different ELISAs, and results were compared. The experimental protocol was approved by the Iowa State University Institutional Animal Care and Use Committee.
Inoculation
On trial day 0, pigs in the PCV-2a, PCV-2b, and PCV-1 groups were inoculated with 2 ml intramuscularly (IM) and 3 ml intranasally of 104.5 50% tissue culture infective dose (TCID50)/ml of the appropriate virus inoculum. Animals were inoculated with the following vaccines: PCV-2a group (ISU-40895; GenBank accession number AF264042), PCV-2b group (NC-16845; GenBank accession number EU340258), and PCV-1 group (well-characterized PCV-1 clone). 7 To confirm that the pigs in the PCV-1 group were infected with PCV-1, serum samples were analyzed by PCR for PCV-1 DNA. 13 To confirm that PCV-2—inoculated pigs were infected, serum samples from all pigs were tested by PCR for PCV-2 DNA. 23 In addition, a PCR product recovered on trial day 49 from a PCV-2a— and a PCV-2b—inoculated pig was sequenced and compared with the respective inoculum.
Vaccination
Pigs in vaccine groups A, B, and C were vaccinated with the respective vaccines according to the recommendations of the manufacturers. Briefly, animals in the vaccine A group a received a 1-ml IM injection on trial day 0. Pigs in the vaccine B group b received a 2-ml IM injection on day 0 and a second 2-ml IM injection on day 14. Pigs in the vaccine C group c received a 2-ml IM injection on trial day 0.
Serology
All serum samples collected from the pigs over time were tested by a previously described indirect PCV-2 FAT 24 and by each of the following 3 ELISAs.
ELISA 1. This in-house, modified, indirect PCV-2 ELISA was performed as previously described. 20 For this assay, alternating rows on the ELISA plate were coated with an ORF2 antigen (expressed in a BA expression vector) or a BA antigen (BA expression vector without the PCV-2a ORF2 insert). The PCV-2 isolate used for antigen production, ISU-31 (GenBank accession number AJ223185) had 99% similarity to the PCV-2a isolate used for inoculation. The following modifications were made to the previously described protocol: following incubation, the plates were washed 5 times with 0.1 mol of phosphate buffered saline d (pH 7.2) and 0.1% Tween 20 e using a microplate washer. For visualization of results, 100 μl of 3,3′,5,5′-tetramethylbenzidine f was added to the wells. Following a 15-min incubation at 25°C, the reaction was stopped by adding 50 μl of 1 mol H2SO4. Results were reported as sample-to-positive (S/P) ratios. All samples were run in duplicate.
Initially, a corrected optical density (OD) value was calculated for the 2 replicates of each sample and the positive control: [(ORF2 OD + ORF2 OD)/2] — [(BA OD + BA OD)/2], where ORF2 is ORF2 antigen and OD is the optical density value at 450 nm. To generate the S/P ratio, the sample-corrected OD was divided by the positive control—corrected OD 20 ; S/P ratios < 0.2 were considered negative, and S/P ratios ≥0.2 were considered positive. The previously determined diagnostic sensitivity and specificity of this method using a 0.2 cutoff were 79.9% and 99.6%, respectively. 20
Area under the receiver operating curve (AUC) values (±standard error) for each of the 3 enzyme-linked immunosorbent assays ELISAs) on different trial days.*
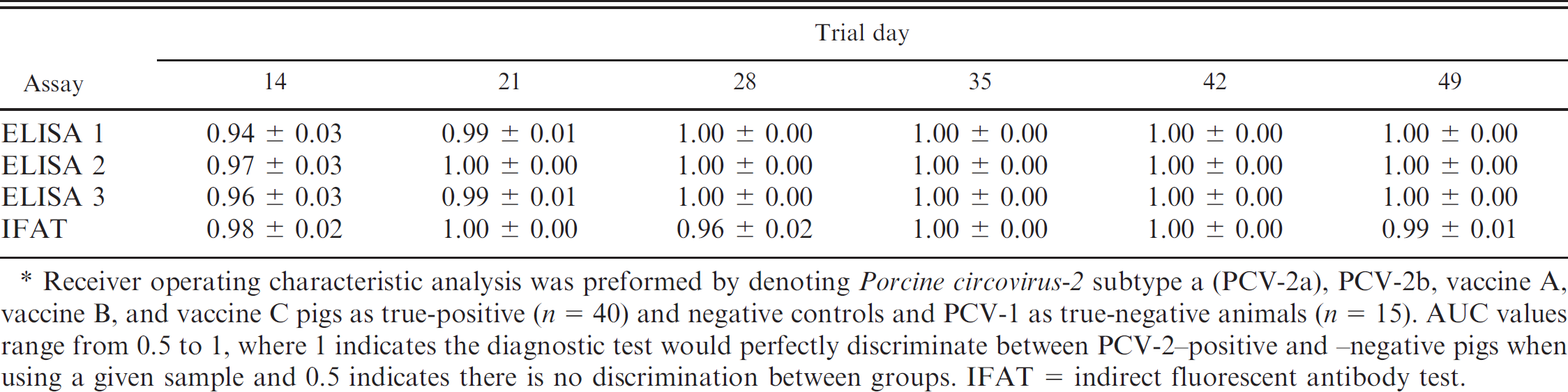
Receiver operating characteristic analysis was preformed by denoting Porcine circovirus-2 subtype a (PCV-2a), PCV-2b, vaccine A, vaccine B, and vaccine C pigs as true-positive (n = 40) and negative controls and PCV-1 as true-negative animals (n = 15). AUC values range from 0.5 to 1, where 1 indicates the diagnostic test would perfectly discriminate between PCV-2-positive and -negative pigs when using a given sample and 0.5 indicates there is no discrimination between groups. IFAT = indirect fluorescent antibody test.
ELISA 2. This commercially available competitive ELISA g was preformed according to the manufacturer's recommendations. The plates provided by the manufacturer were coated with purified PCV-2 antigen. Results were reported as an OD ratio of the sample OD value to the negative control OD value. A plate-specific cutoff was used such that samples with OD ratios ≤0.15 were considered positive, samples with an OD ratio of 0.15 to 0.20 were considered suspect, and samples with an OD ratio ≥0.20 were considered negative as recommended by the manufacturer.
ELISA 3. This commercially available capture PCV-2 ELISA h was also preformed according to the manufacturer's recommendations. The plates provided in the assay came coated with monoclonal antibodies specific for swine IgG. The antigen provided in the assay was a recombinant PCV-2b protein produced through expression in a BA vector (J. Delbecque, personal communication, March 5, 2008). Results were reported as an OD ratio of the sample OD value to the positive-control OD value. The plate-specific cutoff was based on the OD 450 nm value of the positive control × 0.3 as recommended by the manufacturer.
Statistics
Diagnostic accuracy of each assay was evaluated using receiver operating characteristic (ROC) analysis. This method results in area under the ROC curve (AUC) values ranging from 0.5 to 1, where 0.5 indicates the test is not discriminating between positive and negative animals and 1 indicates perfect discrimination. 31 Area under the ROC values were compared pairwise between tests using a previously described method. 11 All ROC calculations were preformed on continuous data with infection status as the indicator of true infection status using MedCalc. i Summary statistics were calculated for all groups to assess the overall quality of the data, including normality. Continuous data were analyzed using multivariate analysis of variance. If a significant (P < 0.05) change over time was noted for the outcome variable, a nonparametric Kruskal-Wallis 1-way analysis of variance (ANOVA) was performed at each time point. If this nonparametric ANOVA test was significant (P < 0.05), then pairwise Wilcoxon tests were used to assess differences between groups. Statistical analysis was performed using JMP software. j
Results
Diagnostic accuracy of assays
All assays had AUC values greater than or equal to 0.94 for all trial days using ROC-generated optimal cutoffs; no statistical differences were noted among the assays (Table 1). When data were dichotomized using the manufacturer's recommended cutoff values (suspect samples were considered negative), the sensitivity of all 3 assays was substantially reduced at trial day 14 (Table 2). In contrast, the specificity of ELISAs 2 and 3 was improved. When the manufacturer's recommended cutoff was used at trial day 49, the sensitivity of ELISAs 2 and 3 was substantially reduced (Table 2). The number of pigs classified as positive or negative based on S/P ratios of 0.2 (ELISA 1) or the manufacturer's recommendations (ELISA 2 and 3) by the 3 ELISAs at trial days 14 and 49 is presented in Table 3.
Detection of PCV-2a and PCV-2b
Infection status for PCV-2a and PCV-2b groups was confirmed by PCR and sequence analysis (Opriessnig T, Ramamoorthy S, Madson DM, et al.: 2007, Experimental comparison of the virulence of PCV2a and PCV2b isolates in a conventional pig model. Proc Conf Res Workers Anim Dis 88:145). To assess whether PCV-2a could be differentiated from PCV-2b on any of the 3 ELISAs, the following null hypothesis was tested: there is no difference between PCV-2a and PCV-2b ELISA S/P or OD values on ELISAs 1, 2, or 3. Statistical analysis using a Wilks' lambda multivariate test indicated a significant (P < 0.0001) difference over time between control and PCV-2a and PCV-2b groups. A nonparametric ANOVA at each time point revealed a significant difference between negative controls and PCV-2a and PCV-2b groups (P < 0.05) for all tests. Further analysis using pairwise Wilcoxon tests indicated that no significant differences were noted between PCV-2a and PCV-2b groups on any of the assays (Fig. 1).
Comparison of the sensitivity and specificity of each of the 3 enzyme-linked immunosorbent assays (ELISAs) at trial days 14 and 49 using various cutoffs for data dichotomization.
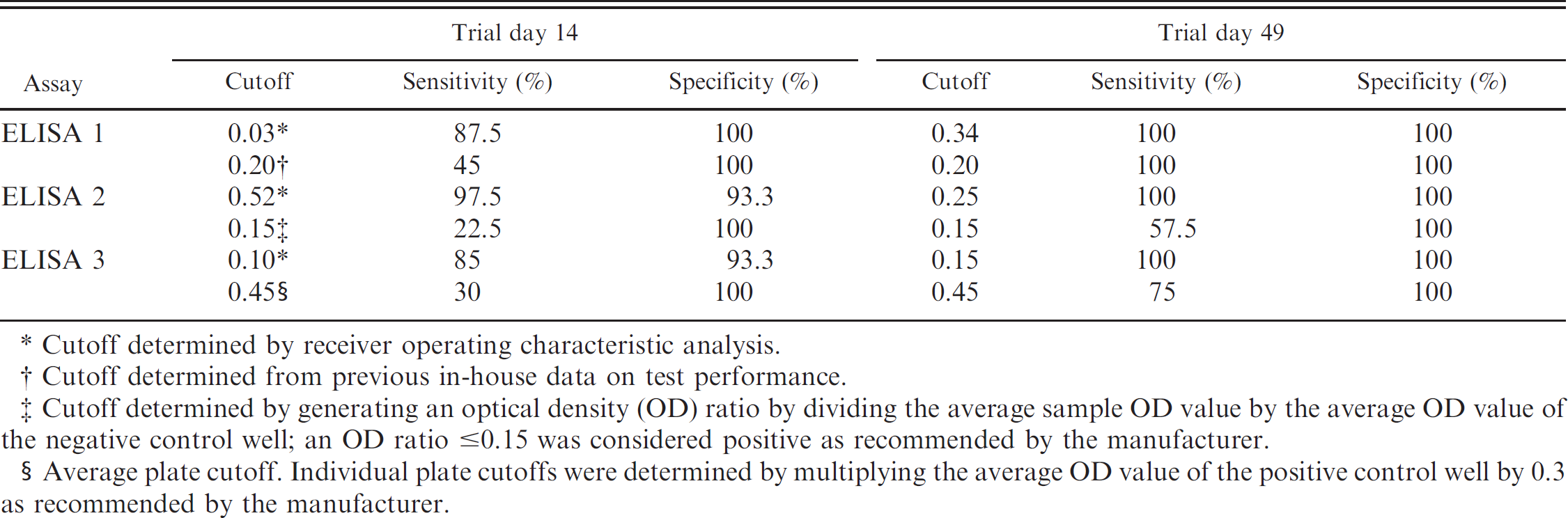
Cutoff determined by receiver operating characteristic analysis.
Cutoff determined from previous in-house data on test performance.
Cutoff determined by generating an optical density (OD) ratio by dividing the average sample OD value by the average OD value of the negative control well; an OD ratio ≤0.15 was considered positive as recommended by the manufacturer.
Average plate cutoff. Individual plate cutoffs were determined by multiplying the average OD value of the positive control well by 0.3 as recommended by the manufacturer.
Detection of PCV-1
Serum samples from pigs in the PCV-1 group were positive for PCV-1 DNA by PCR starting on trial day 7 (data not shown). No positive reactions were detected in serum samples from PCV-1-inoculated animals on any of the 3 ELISAs at any time during the trial (data not shown).
Detection of antibodies in vaccinated animals
Mean and standard error S/P and OD ratios for each group are provided in Figure 2. Results from ELISA 1 indicated that animals in the vaccine A group had significantly lower mean S/P ratios in comparison with PCV-2a- and PCV-2b—inoculated animals from trial days 14 to 49 (P < 0.0005 for all days; Fig. 2a). Animals in the vaccine B group also had significantly lower mean S/P ratios from trial days 28 to 49 (P < 0.03 for all days) in comparison to PCV-2—inoculated animals. In contrast, animals in the vaccine C group had a significantly (P = 0.01) higher mean S/P ratio at trial day 28 in comparison to PCV-2-inoculated animals (Fig. 2a). In contrast to ELISA 1, no significant difference was noted between animals in either the vaccine A or B group and the PCV-2—inoculated animals based on ELISA 2 (P > 0.05 for all days; Fig. 2b). Animals in the vaccine C group had a significantly lower (more positive) OD ratio than that in challenged animals at trial day 28 and 35 (P < 0.008 for both days; Fig. 2b). Results from ELISA 3 were similar to ELISA 1 (Fig. 2c); animals in the vaccine A group had significantly lower mean OD ratios in comparison to PCV-2—inoculated animals at trial days 14 to 49 (P < 0.0006 for all days).
Number of pigs classified as positive (+) or negative (−) by the 3 enzyme-linked immunosorbent assays (ELISAs) and a Porcine circovirus-2 fluorescent antibody test (FAT) at trial days 14 and 49.*
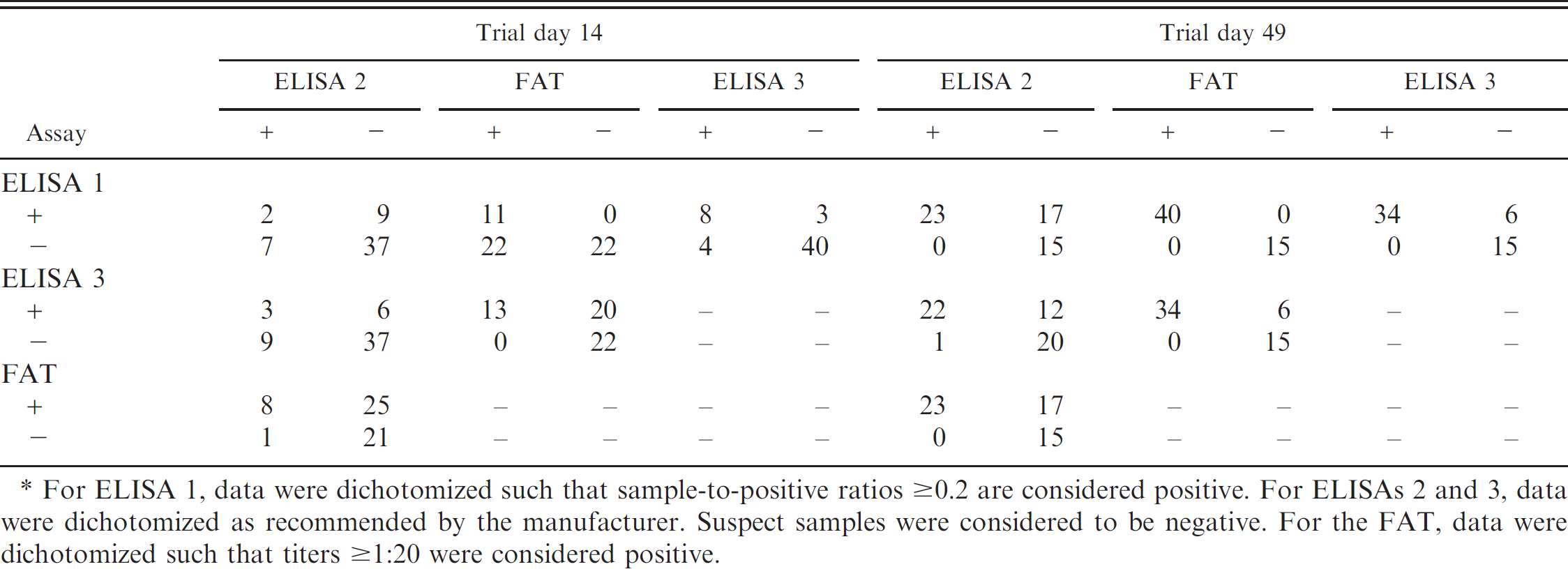
For ELISA 1, data were dichotomized such that sample-to-positive ratios ≥0.2 are considered positive. For ELISAs 2 and 3, data were dichotomized as recommended by the manufacturer. Suspect samples were considered to be negative. For the FAT, data were dichotomized such that titers ≥1:20 were considered positive.
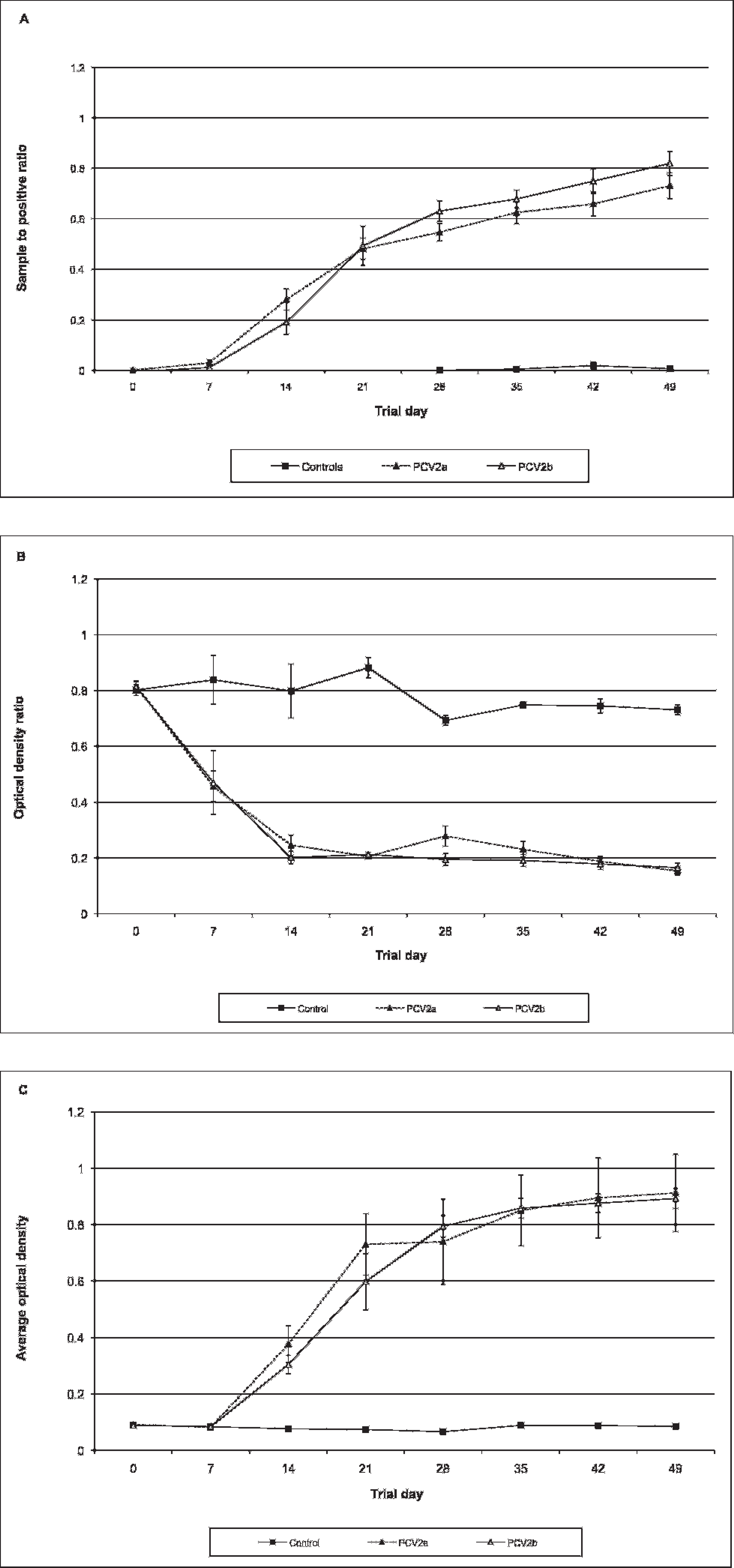
Mean sample-to-positive ratio and standard error for enzyme-linked immunosorbent assay (ELISA) 1 (
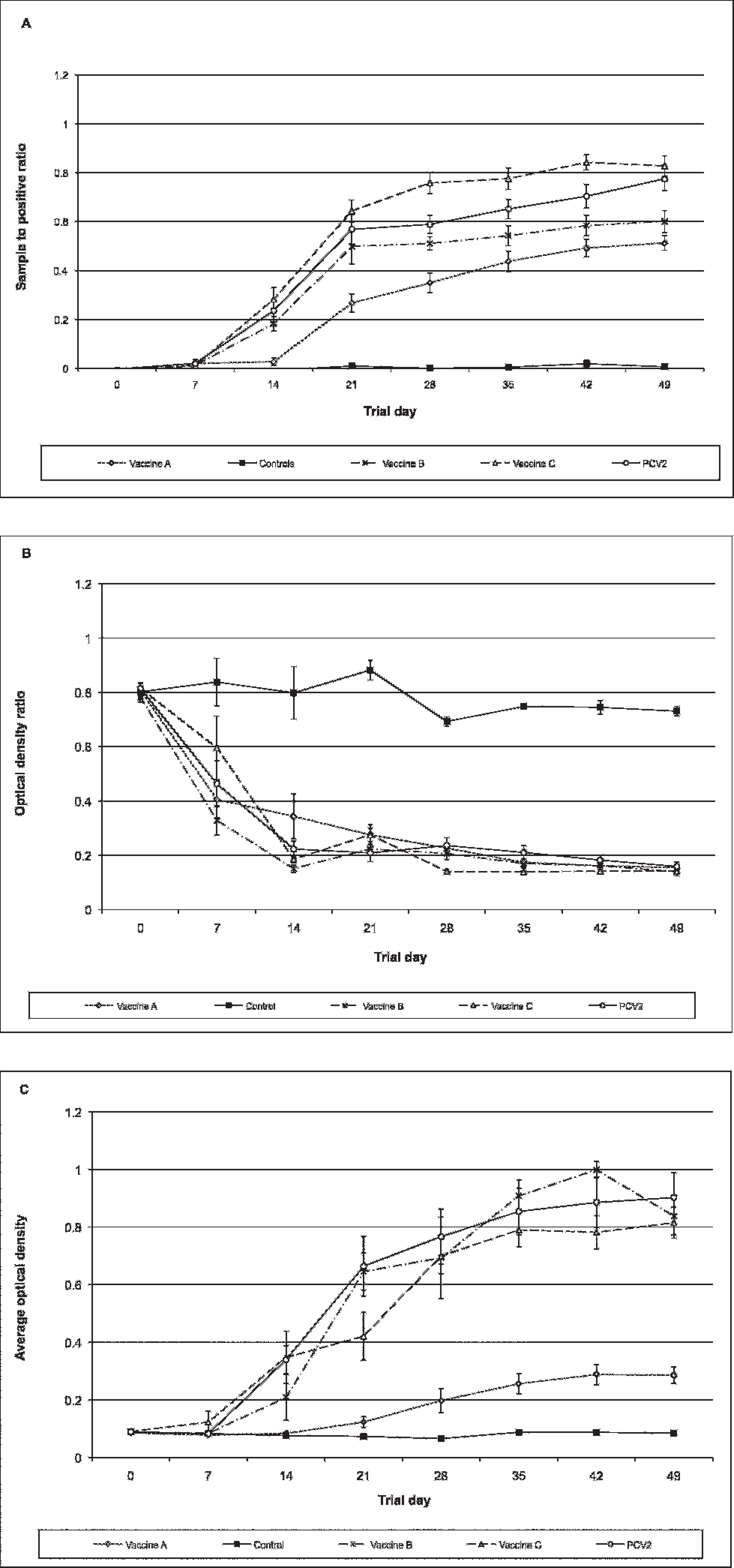
Mean sample-to-positive ratio for enzyme-linked immunosorbent assay (ELISA) 1 (
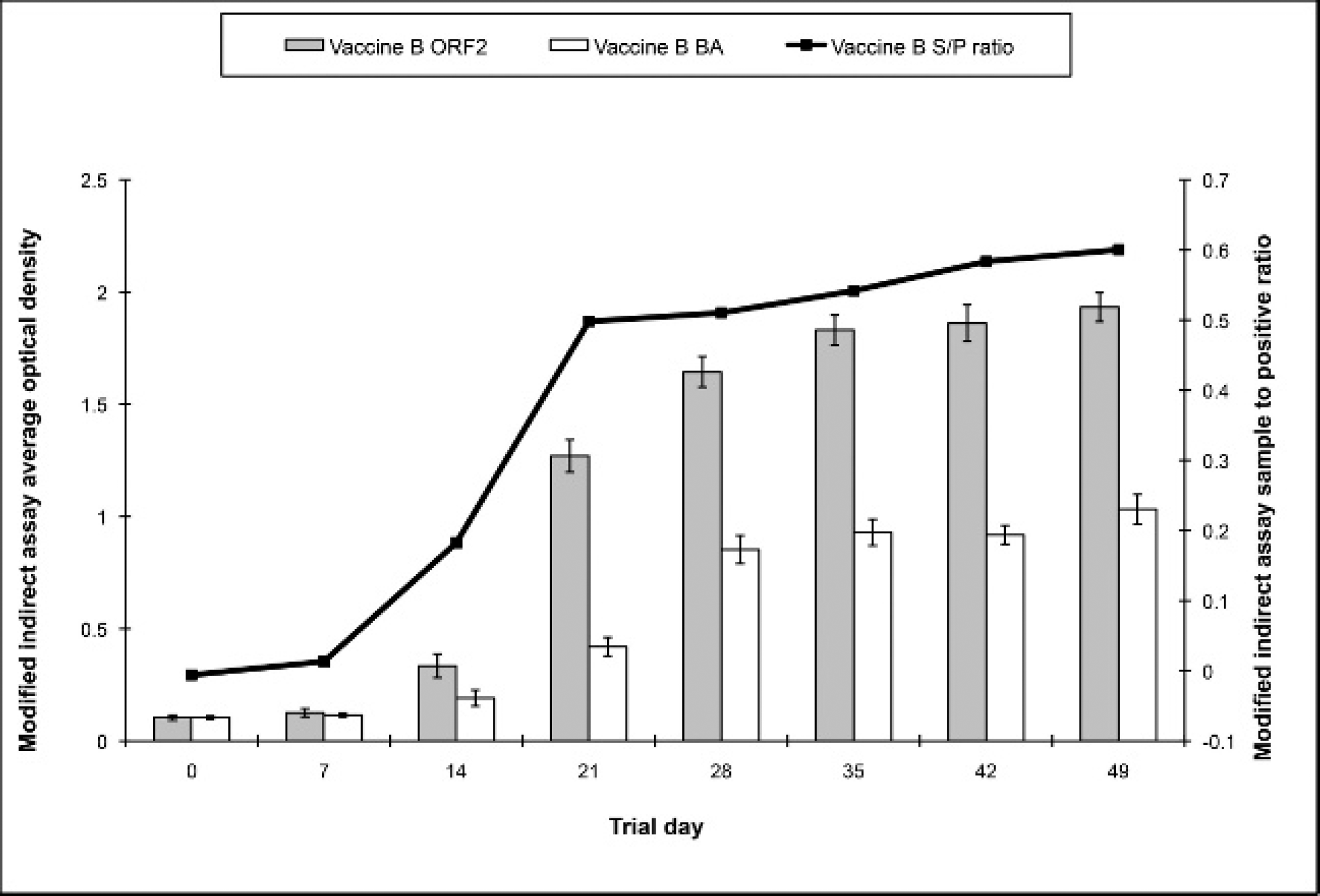
Mean and standard error sample-to-positive (S/P) ratio and average optical density for the baculovirus (BA) antigen and open reading frame 2 (ORF2) wells on enzyme-linked immunosorbent assay (ELISA) 1. All values are for animals in the vaccine B (n = 8) group.
Discussion
Receiver operating characteristic analysis has become an increasingly valuable tool for comparison of diagnostic tests. 9 , 31 Unlike the kappa statistic, ROC analysis determines the sensitivity and specificity of the test based on the classification of true status (infected vs. negative controls in the current study) at multiple cutoffs by using the complete set of continuous data. 9 Interestingly, when the 3 ELISAs were compared by ROC analysis, AUC values were very high (≥ 0.9). This indicated that all tests were able to distinguish between negative and positive animals using ROC analysis—generated optimal cutoffs (cutoffs that provide the highest sensitivity and specificity) based on continuous data from the experiment. In contrast, when the manufacturer's cutoff values were used, sensitivity was substantially reduced. While the current study was limited by a small sample size, ROC analysis data suggest that altering the cutoff value will increase the sensitivity of ELISAs 2 and 3. Further studies with larger sample sizes are necessary to determine an appropriate cutoff. The manufacturers' sensitivity and specificity for ELISAs 2 and 3 at the recommended cutoffs were not indicated on the package insert and could not be compared with the results in the present study.
As most herds worldwide are seropositive for PCV-2, ELISAs are most commonly used in cross-sectional analyses to target optimal timing of vaccination. Recent trends indicate an increased incidence of detection of PCV-2b isolates and the association of these strains with high mortality in herds with increased incidence of severe mortality in swine populations. 5 , 12 This has stimulated interest in the use of serological tests to monitor PCV-2b. As such, there is a need to know whether commonly used ELISAs are detecting both anti-PCV-2a and anti-PCV-2b antibodies. Results of the current study indicate that all 3 ELISAs can detect both antibodies but cannot differentiate between anti—PCV-2a and anti—PCV-2b antibodies. This was independent of the origin of the coating antigen used since at least 1 assay used PCV-2a (ELISA 1) and 1 assay used PCV-2b (ELISA 3) for antigen production.
Nucleotide sequence homology between PCV-1 and PCV-2 ranges from 69% to 79.5%. 10,18,19 A previous report indicated that there was no serological cross-reactivity between the ORF2 region of PCV-2 and PCV-1 17 or between PCV-1 and PCV-2 polyclonal antisera and monoclonal antibodies using an indirect FAT. 12 Conversely, ORF1 regions of PCV-2 do exhibit serological cross-reactivity with PCV-1, 17 Results of the current study confirm previous reports and indicate that PCV-1-inoculated animals were not detected with any of the ELISAs evaluated. Although the PCV-1-inoculated animals were found to be PCV-1 positive by PCR, an anti—PCV-1 ELISA assay was not available to confirm the presence of anti—PCV-1 antibodies in these animals.
Following the introduction of vaccines into the U.S. market in 2006, the prevalence of seropositive animals has grown steadily. Therefore, an assay able to differentiate vaccinated and naturally infected animals would be a useful tool for veterinary practitioners in situations in which they are assessing vaccination protocol compliance issues. Although distinguishing vaccinated animals based on either an S/P or OD ratio would not be possible based on the results of the current study, using the OD value of the BA well on ELISA 1 may provide a means of differentiation for vaccine B. In contrast to the other 2 ELISAs, ELISA 1 uses BA derived from a baculovirus expression vector that does not contain the PCV-2 ORF2 insert to control for nonspecific background binding. 20 Serum samples from both vaccine A and B group pigs were expected to have increased OD values for the BA wells as both products use the baculovirus expression vector for antigen production. Interestingly, only the serum samples from the vaccine B group showed a significant increase in OD value in the BA well. Because the increase in the OD value of the BA well occurred following the administration of the second dose of vaccine B, it can be speculated that 2 doses of vaccine A may lead to the same response as seen with vaccine B. Alternatively, differences in vaccine preparation or type of adjuvant between the 2 products may account for the lack of cross-reactivity in BA well OD values of animals in the vaccine A group.
While providing a means of differentiating vaccinated and naturally infected animals, significant increases in OD values alone should be interpreted carefully as results could be confounded by vaccination with other products in which the BA vector is used, serum factors other than antibodies against BA, or other unknown nonspecific reactions. These nonspecific reactions could falsely elevate the OD value. As an S/P ratio is not generated when looking at the individual OD value from the BA well, no control for this is in place with the current assay.
The lack of increase of the S/P ratio in ELISA 1 for the vaccine B group cannot be interpreted as a lack of seroconversion because a high OD value for the BA could be lowering the S/P ratio. Therefore, the OD value for the BA well should be observed in addition to the S/P ratio, or 1 of the other 2 assays should be used to assess seroconversion with this product. Reasons for the decreased average OD on ELISA 3 for the vaccine A group may include vaccine preparation, type of adjuvant, or dose of administration. It should be noted that while antibody levels following vaccination were determined in the current study, there was no challenge of animals. Therefore, the ability of vaccination to protect against disease was not evaluated in this trial.
Acknowledgements
The authors wish to thank Paul Thomas for assistance with animal work and the staff at Iowa State University Veterinary Diagnostic Laboratory for assistance with serology work. This study was funded by the PCV-2 research award, Boehringer Ingelheim Vetmedica Inc., St. Joseph, Missouri.
Footnotes
a.
Ingelvac® CircoFLEX™, Boehringer Ingelheim Vetmedica Inc., St. Joseph, MO.
b.
Circumvent® PCV, Intervet Inc., Millsboro, DE.
c.
Suvaxyn® PCV2 One Dose, Fort Dodge Animal Health, Fort Dodge, IA.
d.
Fisher Scientific Co., Pittsburgh, PA.
e.
Sigma-Aldrich, St. Louis, MO.
f.
Kirkegaard & Perry Laboratories, Gaithersburg, MD.
g.
Serelisa® PCV2 Ab Mono Blocking, Synbiotics Europe SAS, Lyon, France.
h.
Ingezim Circovirus IgG, Ingenasa, Madrid, Spain.
i.
MedCalc® Software version 9.1.0.1, Mariakerke, Belgium.
j.
JMP® version 6.0.0, SAS Institute Inc., Cary, NC.
