Abstract
Leptospirosis is a zoonotic disease of global importance. Very little information is available on Leptospira infection in nonhuman primates. We report herein a high seroprevalence (49.4%; 95% confidence interval: 41.6–57.2%) to Leptospira serovars in vervet monkeys (Chlorocebus sabeus) on the Caribbean island of Saint Kitts. Monkeys bred in captivity (n = 81) had a significantly higher seroprevalence compared to wild-caught monkeys (n = 81; p < 0.05). Seroprevalence to serovar Bataviae was significantly higher in monkeys bred in captivity and was higher to serovar Bratislava in wild-caught monkeys (p < 0.05). Our data confirm that exposure to various Leptospira serovars and seroconversion occurs in wild and captive vervet monkeys on the Caribbean island of Saint Kitts. Further studies are warranted to better understand epidemiology, transmission, pathology, and possible reservoir status in this species.
Leptospirosis is a widespread zoonotic disease that may result in fatal illness as the result of renal, hepatic, and pulmonary diseases in both humans and animals. 8 Leptospira spp. inhabit the renal tubules of a wide variety of mammals, and urinary shedding contributes to widespread contamination of the environment, mainly water. 8 Leptospirosis is endemic in the Caribbean region. 15 The estimated annual data on human morbidity and mortality caused by leptospirosis places the Caribbean near the top, based on World Health Organization Global Burden of Disease regions (annual number cases: 22,300; morbidity: 50.7%; deaths: 1,300; mortality: 2.9%). 5
The federation of Saint Kitts and Nevis is a small dual-island Caribbean country located in the Leeward Islands in the Lesser Antilles. A large number of feral vervet monkeys (syn. African green monkeys; Chlorocebus sabeus) inhabit and share the landscape with humans and other wild and domestic animals. These animals are descendants of a small number of monkeys brought as pets from Africa to the Caribbean islands of Saint Kitts, Nevis, and Barbados in the 17th and 18th centuries and, in the absence of predators, established feral populations at levels >50,000.9,13 The free-roaming population is considered to be an agriculture pest by local people given their destructive habits. They also are widely used as pets for entertaining tourists.
Limited published data are available on Leptospira infection in nonhuman primates. Experimental infection in a variety of monkeys has resulted in various outcomes, such as severe lymphocytic meningoencephalitis and nephritis in grivet monkeys; colonization in various tissues with no signs of severe clinical disease or macroscopic lesions in grivet monkeys; and patterns comparable to those seen in the severe forms of human leptospirosis, including pulmonary hemorrhage and severe tubulointerstitial nephritis in marmoset monkeys.11,14,16 Fatal cases of leptospirosis have been reported in the squirrel monkeys infected with serovar Copenhageni. 17 An outbreak of severe leptospirosis manifesting as jaundice and pulmonary hemorrhage was reported in capuchin monkeys housed in a Colombia wildlife rehabilitation center. 19 The status of Leptospira infection in vervet monkeys inhabiting Saint Kitts is currently unknown. Because clinical signs of leptospirosis similar to those seen in humans have been reported in nonhuman primates, it is critical to understand whether natural infections leading to clinical disease or reservoir status occur in this abundant monkey species roaming the island. We assessed the Leptospira seroprevalence in island vervets as a first step towards understanding the occurrence of Leptospira infection in this population.
Monkey serum samples were collected in 2013 and 2014 using protocols approved by the Animal Care Committee of the Behavioral Science Foundation (collaborating Institution), acting under the auspices of the Canadian Council on Animal Care, and the Ross University School of Veterinary Medicine Institutional Animal Care and Use Committee. Sera (n = 162) from feral monkeys in quarantine at the Behavioural Science Foundation facility (n = 81) and from a captive breeding colony (n = 81) were screened against 20 Leptospira serovars (kindly provided by the Center for Disease Control, Atlanta, GA; Table 1) belonging to 17 serogroups using the microscopic agglutination test (MAT) using protocols described previously. 7 Briefly, 4–7-d-old cultures in Ellinghausen–McCullough–Johnson–Harris medium (BD Diagnostic Systems, Sparks, MD) standardized at a transmittance rate of 60–70% at 425 nm was used for MAT. Fifty microliters of each serum sample at 1:50 dilution in phosphate-buffered saline were added to the wells of a 96-well flat-bottom microtiter plate. Standardized Leptospira cultures (50 µL) were added to the wells. The plates were incubated for ~2 h at 29°C; plates were read using a light microscope equipped with a dry dark-field condenser and a long 5× objective. Initially, all of the samples at a final dilution of 1:100 were screened for the agglutination reaction to each of the serovars. The agglutination reaction was graded at 1+ (1–25% agglutination), 2+ (25–50% agglutination), 3+ (50–75% agglutination), or 4+ (75–100% agglutination). Samples showing ≥50% agglutination were recorded as positive, and endpoint titers were determined for positive serovars. Data were analyzed using R software (http://www.R-project.org/). Seroprevalences were computed with 95% confidence intervals (CIs). Prevalences between groups were compared using a Fisher exact test. Age distribution between positive and negative animals was compared using a Student t-test. The threshold of significance was determined at a p value of 0.05.
Leptospira serovars tested against vervet monkey (Chlorocebus sabeus) sera and the detection of a positive titer.
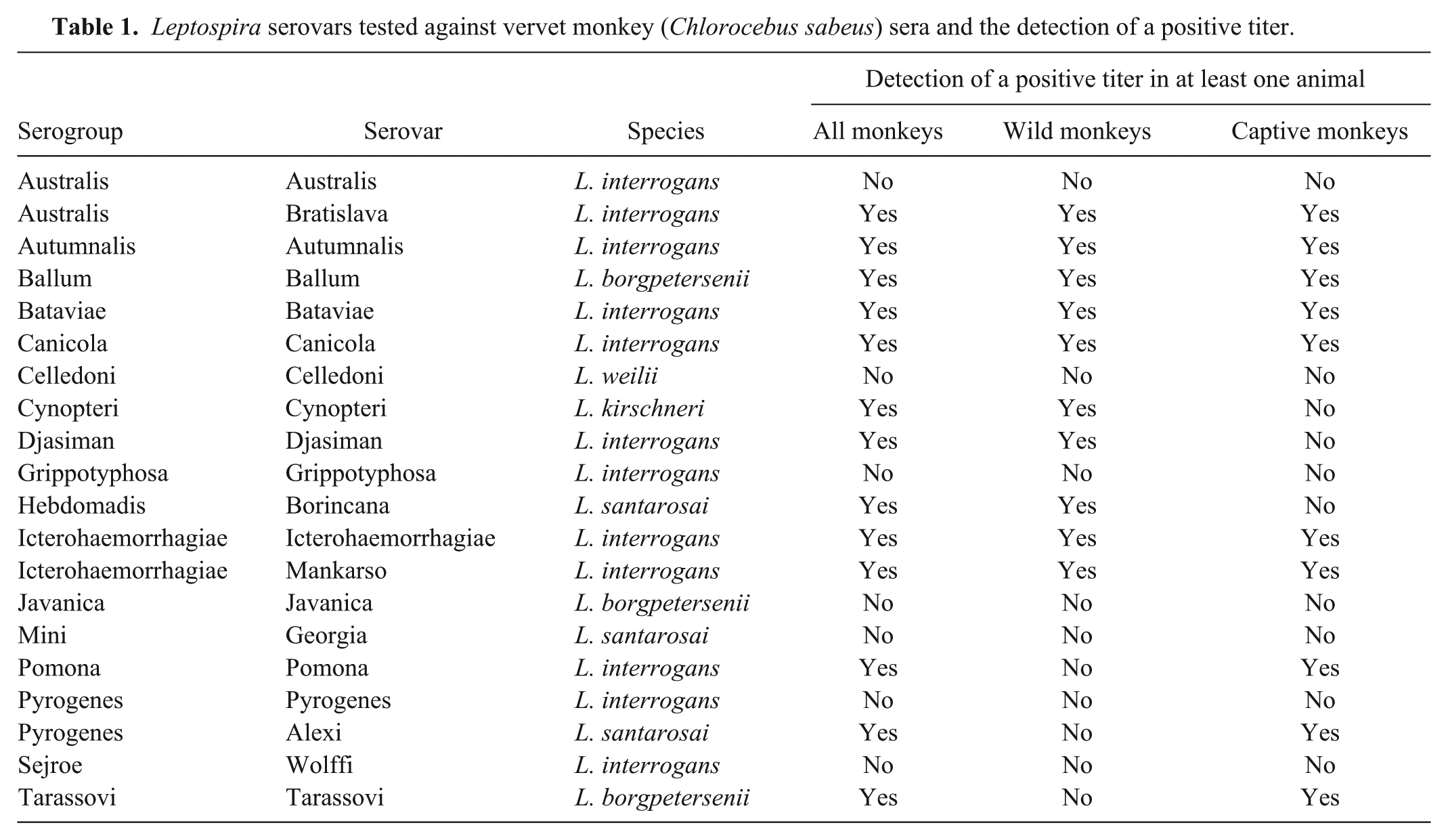
Various locations of capture and seroprevalence corresponding to the location are shown in Figure 1. An overall Leptospira seroprevalence of 49.4% (95% CI: 41.6–57.2%) distributed among 13 serovars was observed. Captive monkeys had a significantly higher seroprevalence (60.5%; 95% CI: 49.6–71.4%) compared to wild monkeys (38.3%; 95% CI: 27.5–49.1%; p < 0.05).
General seroprevalence and seroprevalence to specific leptospiral serovars in vervet monkeys by the location of sampling on Saint Kitts.
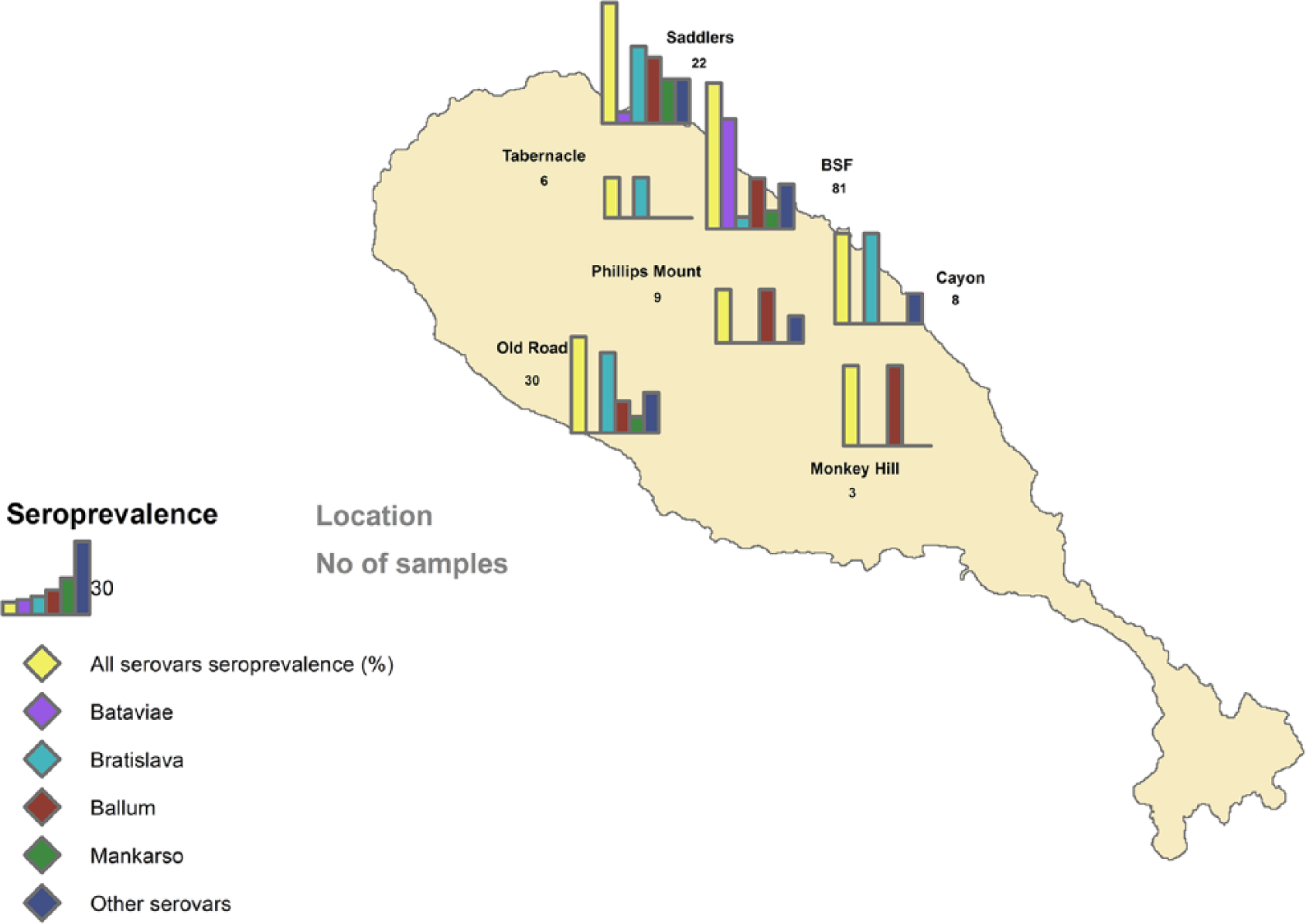
Predominant positivity was observed for serovars Bratislava (27.2%) and Ballum (16.0%) in wild monkeys, and Bataviae (45.7%) and Ballum (21.0%) in captive monkeys (Table 2). The majority of monkeys were females (97 females, 61%; 61 males; 39%). No significant difference of sex distribution was detected (p > 0.05 for all serovars). Although the ages of the captive monkeys were known (mean: 10 y, interquartile range [IQR]: 6–14 y), accurate age was available only for 37 wild monkeys (47%; mean: 6 y, IQR: 5–7 y). Age of positive captive monkeys was significantly higher than negative captive monkeys (p < 0.05). The mean number of serovars detected per positive monkey was 1.7 (IQR = 1–2). No difference in the number of positive serovars was observed between wild and captive animals (p > 0.05). Serum MAT titers were 100–3,200 (Fig. 2). Twenty-four (20.5%) of the sera had titers ≥400. The titers to serovars Ballum and Bataviae were associated with each other 9 times. Other combinations of serovars were Ballum–Mankarso (6 times); Mankarso–Bataviae, Mankarso–Bratislava, Ballum–Bratislava, Autumnalis–Bratislava, and Bataviae–Icterohaemorrhagiae (5 times); none of the combinations belong to the same serogroup, indicating possible exposure to each of the serovars.
Seroprevalence per serovar and comparison between wild and captive monkeys.
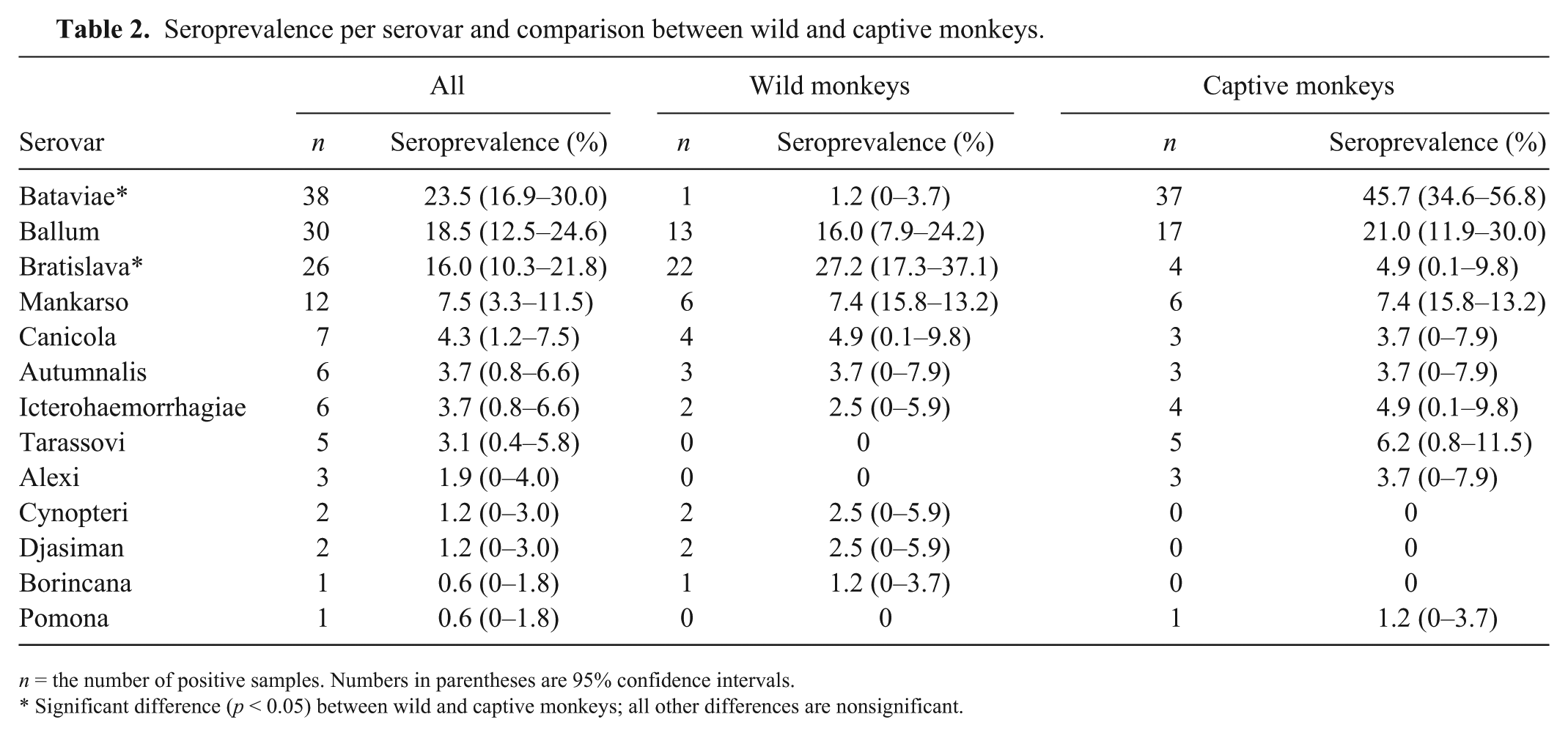
n = the number of positive samples. Numbers in parentheses are 95% confidence intervals.
Significant difference (p < 0.05) between wild and captive monkeys; all other differences are nonsignificant.
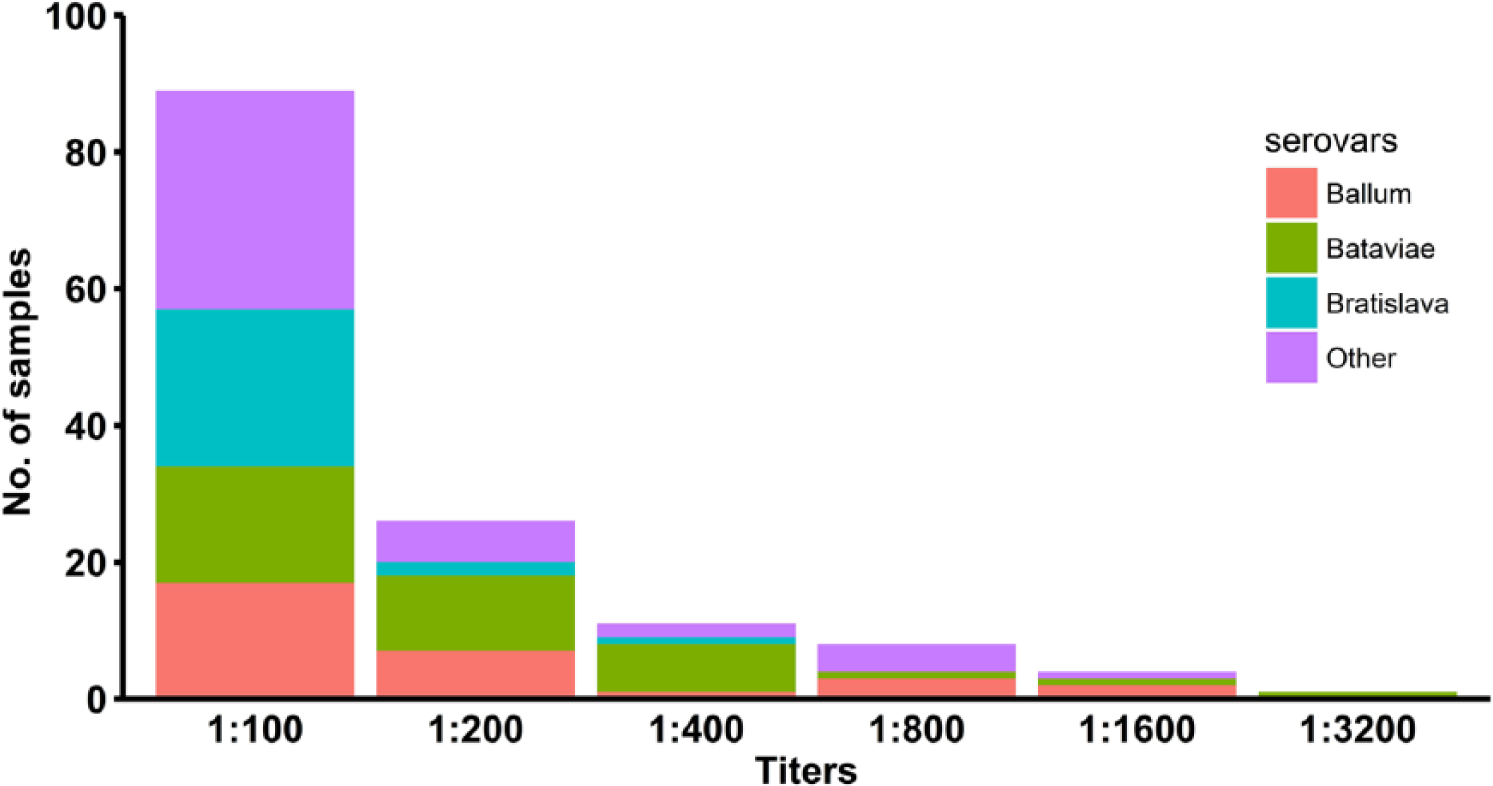
Distribution of titers in vervet monkeys to various leptospiral serovars.
It was surprising to see the difference in seroprevalence to Leptospira serovars between captive and wild-caught monkeys living on this small island. A high percentage of exposure to specific serovars in monkeys may be as a result of a one-time exposure occurred at a specific time in a group of monkeys or a multipoint exposure consequent to contact with a specific reservoir or contaminated environment harboring this serovar in the vicinity. Any illness as a result of Leptospira infection is unknown given the retrospective nature of our study, but no obvious clinical signs were seen in captive animals. Previous exposure resulting in retention of antibody titers may have occurred, given that naturally acquired antibodies to Leptospira have been shown to persist for at least 2.5 y in vervets in Barbados. 3 Housing conditions in captivity might increase the access to reservoirs or the contaminated environment as in the case of urban human leptospirosis. A previous study suggested that vervets transmitted Leptospira infection among themselves independently of other groups of animals, and were not a major source of infection for humans. 3 The source infection on Saint Kitts is unknown for the predominant serovars Bataviae, Ballum, and Bratislava. In Peninsular Malaysia, serovar Bataviae persisted in urban rats, and serovar Ballum in common house mice; both rats and mice are abundant on Saint Kitts.4,12 In Barbados, the only other Caribbean island inhabited by vervets, seroprevalence to serovar Ballum (61%) was predominant in wild-caught monkeys. 3
Human leukocyte antigen (HLA) DQ6 has been identified as an independent risk factor for the development of leptospirosis. 10 The captive monkeys kept at the Behavioural Science Foundation are pedigree stocks, and a possible association of a similar genetic determinant cannot be excluded. In any case, prospective studies to identify clinical consequences of infection are certainly in order.
The vervet monkey is a valuable and widely used nonhuman primate model, and the wild Caribbean populations of this species are the largest global source for biomedical research. 9 For that reason, factors related to Leptospira infections in this species such as the source of infection, mode of transmission, disease spectrum, and the nature of immune response deserve further investigation. As deeper understanding of vervet system biology is near completion, exploring this species as an experimental animal model for studying leptospirosis may be rewarding. 9
Notification practices are highly limited for diseases such as leptospirosis in the Caribbean region. 6 Laboratory-confirmed cases of human leptospirosis have been reported with a frequency of seropositivity of 11.2% in Saint Kitts and Nevis. 1 A high estimated human morbidity and mortality of 133 and 7 per 100,000 population, respectively, is reported for Saint Kitts and Nevis. 5 A high Leptospira seroprevalence in a nonhuman primate species that is numerous on the island warrants improved human surveillance plans, which are currently nonexistent in many of the Caribbean islands. Valuable clues can be obtained from comparative studies on these closely related species that share the same landscape and environmental conditions and may potentially have similar transmission dynamics. Leptospira DNA has been detected in various environmental water sources in Saint Kitts, which could be a possible source of infection for both species. 18 In addition, the potential role of vervets as asymptomatic reservoirs and source of infection to humans cannot be underestimated. This is especially significant as travel-associated leptospirosis is on the increase, and Saint Kitts is one of the top tourist destinations in the Caribbean. 2
Footnotes
Acknowledgements
We thank Shamara Welcome for technical assistance, and CDC Atlanta for kindly providing the Leptospira panel for MAT testing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The project was funded by the One Health Center for Zoonosis and Tropical Medicine at the Ross University School of Veterinary Medicine.
