Abstract
Four species of ruminant pestivirus are currently circulating in the United States: Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2; predominant host: cattle), Border disease virus (BDV; predominant host: sheep), and pronghorn virus (sporadically detected in wild ruminants). A third bovine pestivirus called HoBi-like virus has been detected in cattle and water buffalo in South America, Asia, and Europe. To date, no isolations of HoBi-like viruses from U.S. cattle have been reported. To assess exposure, 2,000 cattle sera, collected between 2014 and 2015 as part of the U.S. brucellosis surveillance program, were tested for antibodies against BVDV-1, BVDV-2, and HoBi-like viruses. In addition, RNA was extracted and tested by reverse transcription–polymerase chain reaction for the presence of pestiviruses; all samples tested negative. The percent of VN-positive samples was 91.3% for BVDV-1, 89.3% for BVDV-2, and 84.9% for HoBi-like viruses. Because the 3 bovine pestiviruses are antigenically cross-reactive, the comparative level of antibody against each pestivirus species was determined. Based on comparative titers, samples were segregated into 6 categories: no titers (7.6%), titers clearly higher against BVDV-1 (22.2%), titers substantially higher against BVDV-2 (9.1%), BVDV-1 and BVDV-2 titers equivalent but substantially higher than HoBi titers (25.7%), titers substantially higher against HoBi-like viruses (0%), and equivocal (35.4%). Titers tended to be higher against BVDV-1 than BVDV-2. However, the overall percentage of animals with titers below levels considered protective against acute bovine pestivirus infection were ~11% for BVDV-1, 12% BVDV-2, and 18% for HoBi-like virus.
Introduction
The Pestivirus genus (family Flaviviridae) includes 4 recognized or “classic” species: Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2), Border disease virus (BDV), and Classical swine fever virus (CSFV), as well as 5 putative species.10,22 As their names suggest, BVDV-1 and BVDV-2 were first isolated from cattle and are recognized as economically significant pathogens in both the dairy and beef industries.1,13 A third classic pestivirus species, BDV, has been described in the United States, 17 although information regarding prevalence is scarce. Although sheep are the primary host of BDV infection, cattle infection has been described.5,7 Pronghorn virus (PV) is the only putative ruminant pestivirus that has been reported in the United States to date. Although PV is apparently circulating in free-ranging ruminants, the potential for it to infect and cause disease in cattle is unclear. 29 Another putative pestivirus species, provisionally named HoBi-like virus, has been detected in cattle and/or water buffalo in South America, parts of Asia, and Italy. 4 HoBi-like viruses share genetic and antigenic characteristics with the BVDV species3,21 and, on infection, these viruses lead to clinically indistinguishable syndromes in cattle.14,19
The circulation of BVDV-1 and BVDV-2 in the United States has led to the development of vaccines containing strains of BVDV-1 and/or BVDV-2 available either as killed or modified-live virus (MLV) formulations. Studies have shown that >87% of cattle received in feedlots in the United States are vaccinated for BVDV, and 68% of dairy operations vaccinated cows against BVD in 2013.25,27 In contrast, HoBi-like virus has not been reported in U.S. cattle herds, and currently no commercial vaccine is available.
The objective of our study was to test bovine sera for neutralizing antibodies against BVDV-1, BVDV-2, and HoBi-like viruses. In addition to pestivirus antibody level, RNA was extracted from the serum samples and assayed using reverse transcription polymerase chain reaction (RT-PCR) assays: 2 that detect a wide range of pestiviruses and another specific for HoBi-like viruses.
Materials and methods
Source of samples
The 2,000 cattle sera, collected between August 2014 and September 2015 as part of the U.S. brucellosis surveillance at time of slaughter, were used for this seroprevalence study. The details of this surveillance system have been described previously. 26 Samples from 7 slaughter plants were collected from cattle raised in the contiguous 48 states. Regions were defined based on the state of origin (based on backtag number) of the animal (Pacific West: California, Oregon, and Washington; Mountain West: Montana, Idaho, Wyoming, Nevada, Utah, Colorado, Arizona, and New Mexico; Upper Midwest: Nebraska, North Dakota, Minnesota, Wisconsin, South Dakota, Iowa, Kansas, Missouri, Illinois, Indiana, Michigan, and Ohio; South Central: Oklahoma and Texas; Southeast: Arkansas, Louisiana, Mississippi, Alabama, Georgia, Florida, South Carolina, North Carolina, Tennessee, Kentucky, West Virginia, Virginia, Maryland, and Delaware; Northeast: Pennsylvania, New York, Vermont, Connecticut, New Jersey, Rhode Island, Massachusetts, New Hampshire, and Maine; Fig. 1). The USDA Brucellosis Surveillance Program collects blood at slaughter from cattle (both male and female) at least 2 y of age at slaughter plants (Fig. 1). Serum is then submitted to 2 central laboratories for testing (Kentucky Regional Laboratory, Frankfort, Kentucky, and Florida Department of Agriculture, Live Oak Laboratory, Live Oak, Florida).
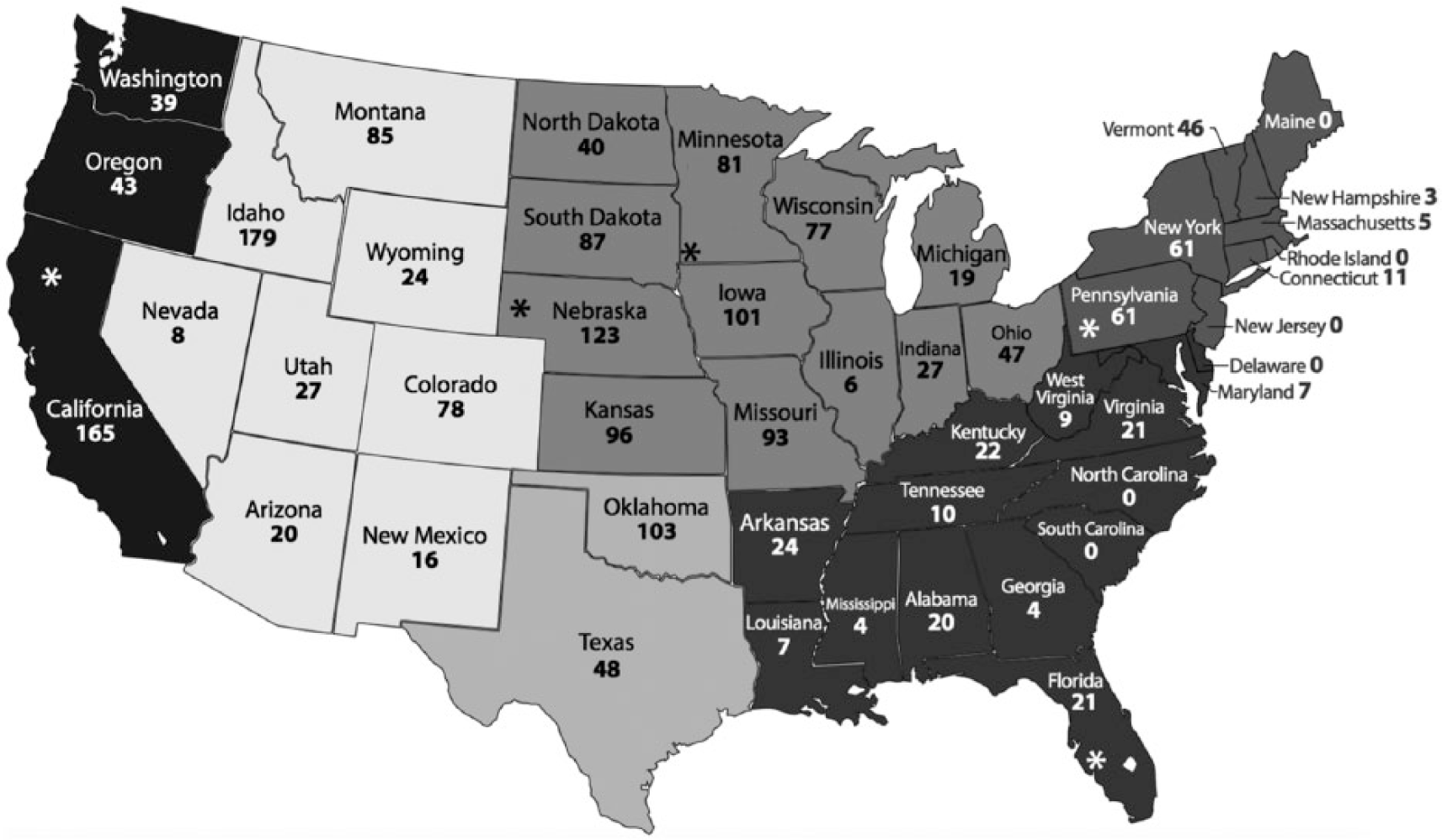
Number of samples for each state in the contiguous United States. Gray scale determines the following 6 U.S. regions—Pacific West: California, Oregon and Washington; Mountain West: Montana, Idaho, Wyoming, Nevada, Utah, Colorado, Arizona, and New Mexico; Upper Midwest: Nebraska, North Dakota, Minnesota, Wisconsin, South Dakota, Iowa, Kansas, Missouri, Illinois, Indiana, Michigan, and Ohio; South Central: Oklahoma and Texas; Southeast: Arkansas, Louisiana, Mississippi, Alabama, Georgia, Florida, South Carolina, North Carolina, Tennessee, Kentucky, West Virginia, Virginia, Maryland, and Delaware; Northeast: Pennsylvania, New York, Vermont, Connecticut, New Jersey, Rhode Island, Massachusetts, New Hampshire, and Maine. States with processing plants contributing samples to our study are marked with asterisks and include California, Florida, Minnesota, Nebraska, and Pennsylvania.
For the purpose of this study, the Kentucky Regional Laboratory sent a total of 2,000 samples, received in 10 lots of 200 samples every 30–45 d. Samples were randomly collected from 5 slaughter plants listed by state of location (California, Florida, Nebraska, Pennsylvania, and Minnesota). The number of sera selected for each slaughter plant was proportional to the total annual number of cattle 2 y and older slaughtered in that plant. The serum samples selected for this study tested negative for brucellosis. After testing, sera were shipped on ice to the USDA:ARS National Animal Disease Center (Ames, Iowa) for subsequent testing as described below.
Viruses and cells
Pestivirus isolates used included the cytopathic (cp) strains BVDV-1a Singer, BVDV-2a 296c, HoBi-like virus Italy-1/10-1, and BDV-CB5, and the noncytopathic (ncp) PV isolate. The BVDV isolates and HoBi-like virus were propagated and titered in Madin–Darby bovine kidney (MDBK) cells; the PV and BDV-CB5 isolates were amplified and titered in ovine turbinate (OFTu) cells. Cell lines tested free for pestivirus antigen by RT-PCR. 2 Cell lines used during the study were grown in minimal essential medium (MEM), a supplemented with L-glutamine b (1.4 mM), 1% of antibiotic-antimycotic 100×, c and 10% fetal bovine serum (FBS). FBS was tested free for pestivirus RNA and antibodies by RT-PCR and virus neutralization (VN) assay, respectively.2,3
Antibody detection assays
On arrival at the USDA:ARS, serum samples were filtered through a 0.2-μm membrane d and divided into 2 aliquots. One set of aliquots was used for RNA extractions as described below. The other set was placed in a water bath at 60°C for 60 min for inactivation of the complement system. Inactivated samples were then stored at 4°C until tested. Sera were initially tested in triplicate against the species of pestiviruses (BVDV-1, BVDV-2, and HoBi-like viruses) as described. 3 Sera were tested by VN performing 2-fold serial dilutions in MEM from 1:32 to 1:4,096. A total of 100 tissue culture infectious dose of virus was added to each well. Plates containing serum dilutions and virus were incubated for 100–120 min at 37°C in 5% CO2. Following incubation, 50 µL/well of cell suspension containing 105 cells/mL was added to all wells. Plates were incubated for another 96 h at 37°C in 5% CO2. Serums of convalescent animals with known titers for BVDV-1, BVDV-2, and HoBi-like virus were used as controls. Control sera were diluted and added to plates and then diluted as described for the samples. Plates were read using an optical microscope for cytopathic isolates. Titer was determined by the reciprocal of endpoint serum dilution that prevented viral replication at a particular dilution. Samples that neutralized virus at a dilution of 1:4,096 or that showed no neutralization at a dilution of 1:32 were then retested in dilutions from 1:4 to 1:32,768. Following the test for BVDV-1, BVDV-2, and HoBi-like virus, samples with unclear titer differentiation among the 3 viruses were also tested by VN against PV and BDV-CB5. VN procedures were the same as described for the bovine pestiviruses with the exception that immunoperoxidase was used to determine endpoint rather than observation of cytopathic effect for PV as follows. VN plates were fixed after 96 h of incubation and stained. Plates were blocked for 15 min at room temperature with 100 µL/well blocking solution (0.01% polysorbate 20 e and 1% bovine serum albumin f in phosphate-buffered saline [PBS]) before adding 50 µL/well of the primary antibody (hyperimmune serum against PV produced in goat) diluted 1:5 in blocking solution and incubated for 1 h at room temperature. Following 3 washes with PBS–Tween-20 (PBST) polysorbate 20 in PBS, 50 µL/well of the secondary antibody anti-goat g (2.5 μL/mL) diluted in PBST was added and incubated for 60 min at room temperature. Following 3 washes with PBST, 50 µL/well of the protein G h (1.4 µL/mL) was added, and plates were incubated for 60 min at room temperature. Three final washes were performed, and plates were stained using 3,3′-diaminobenzidine. i
RNA extraction and RT-PCR
Using 50 µL of serum from each sample, pools of 10 samples each were prepared making a total of 200 pools. An aliquot of 140 µL of the pooled serum was used for RNA extraction using a robotic workstation j for automated RNA purification by a spin-column system. k RNA extracted from supernatant of cells infected with BVDV-1 Singer or Italy-1/10-1 was used as positive control; RNA from FBS and MEM were used as negative controls. The 2 primer pairs HCV90-368 18 and 324-326 28 with ability to detect a wide range of pestiviruses were used. HoBi-like virus–specific RT-PCR was performed as described previously. 2 The PCR products were detected by electrophoresis in 1.0% agarose gel, stained, l and visualized under ultraviolet light.
Data analysis
Data on the collection site and animal source information were entered into an electronic spreadsheet m along with the test results. For the purpose of this analysis, the backtag on the animal was used to assign the animals to the state in which the backtag was applied prior to the animal arriving at the slaughter plant. The results of the 3 wells for each of the 3 pestiviruses were converted to a log2 value according to the methods of Spearman–Kärber. 16 The log2 value for each of the pestiviruses is hereafter referred to as the summary titer for each sample. A summary titer of more than 2.83 was considered positive for each of the pestiviruses.
Although sera frequently had titers against all 3 pestivirus species, it was uncommon that the levels of antibodies were equal. In order to compare the predominance of titers against the different species, a formula for determining the comparative ratio (R) for each serum sample was generated as follows; RspeciesX = (titer against species X + titer against species Y + titer against species Z)/(3 × titer against species X). Based on the calculated ratios, antibody-positive sera were segregated into 5 cross-reactive antibody categories. If the ratio value for 1 pestivirus was >0.2 above the values of the ratios for the other 2 pestiviruses, the sample was considered to be substantially higher against that pestivirus. If the ratio for the 2 BVDV viruses was similar to each other but both were at least 0.2 greater than the ratio for the HoBi-like virus, they were considered to be substantially higher against BVDV than HoBi-like virus. If the HoBi-like virus ratio was at least 0.2 greater than both the BVDV ratios, the sample was considered to be substantially higher against HoBi-like virus. If none of the previous conditions was observed, the sample was considered equivocal.
Background information collected at sampling did not include information on whether animals originated from dairy or beef production units. However, general information was available on the predominant production type making up the cattle processed in each slaughter plant contributing samples. Based on the annual profile of animal types processed, the source slaughter plants were classified as dairy (≥70% of animals processed were dairy), beef (≥70% of animal processed were beef), or mixed. Of the 5 slaughter plants contributing samples, 2 were classified as dairy (n = 728 total samples), 2 were classified as beef (n = 920), and 1 was classified as mixed (n = 352). Contingency tables for qualitative data were compared using software. n A chi-square p value <0.05 was considered significant.
Results
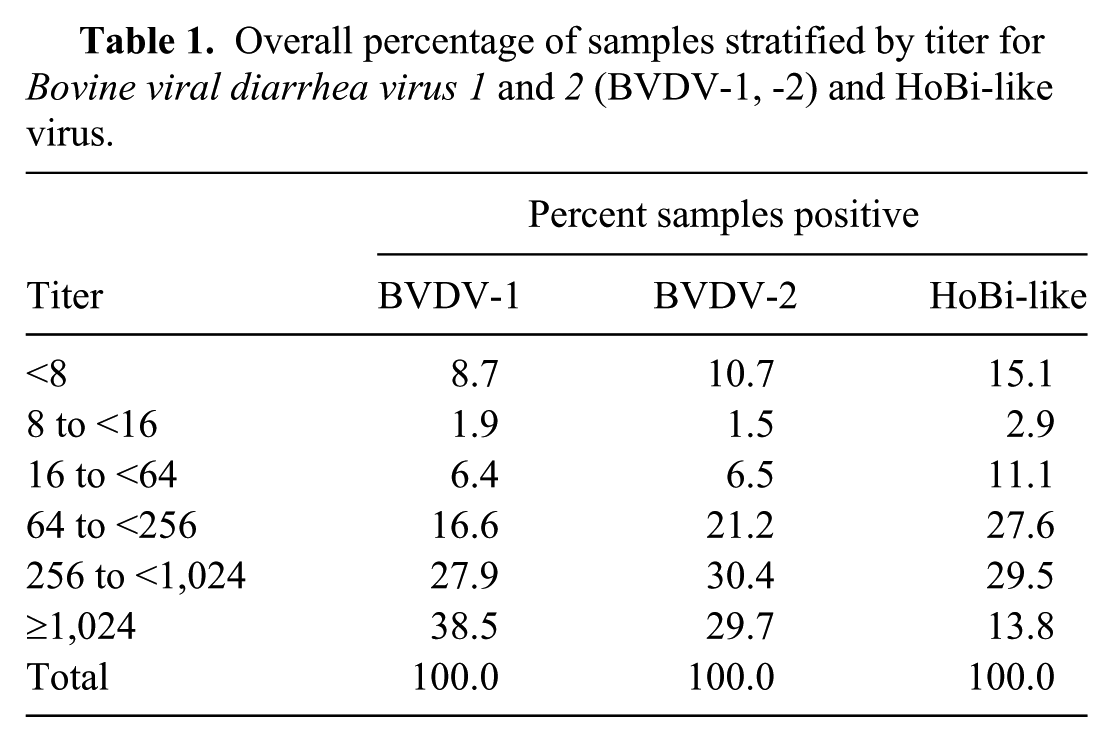
None of the 200 serum pools were positive using either of the RT-PCR tests. Of the 2,000 samples, 1,968 samples were acceptable for VN testing (Table 1). Samples with summary titers <8 were considered to be negative. Overall, a higher percentage of samples were negative (<8) or had low levels of antibodies (<16) against HoBi-like viruses (18.0%) compared to BVDV-1 (10.6%) or BVDV-2 (12.2%). Conversely, the percentage of samples that had high titers (>1,024) against HoBi-like viruses (13.8%) was lower than the percentage that had high titers against BVDV-1 (38.5%) or BVDV-2 (29.7%). Separating the sera by predominant titer, 7.6% had no titer for any pestivirus, whereas BVDV-1 titer predominated in 22.2% of the samples and BVDV-2 in 9.1%. Samples with titers similar for BVDV-1 and BVDV-2 but higher than to HoBi-like virus were 25.7%. Equivocal samples, with no predominant titer, were 35.4%, and no sample had a titer predominant for HoBi-like viruses. Sera with results falling into the equivocal category were further tested by VN against BDV and PV; titers were lower than or similar to the titers against BVDV-1 and/or BVDV-2 and/or HoBi-like viruses as determined in the previous VN testing (data not shown).
Overall percentage of samples stratified by titer for Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2) and HoBi-like virus.
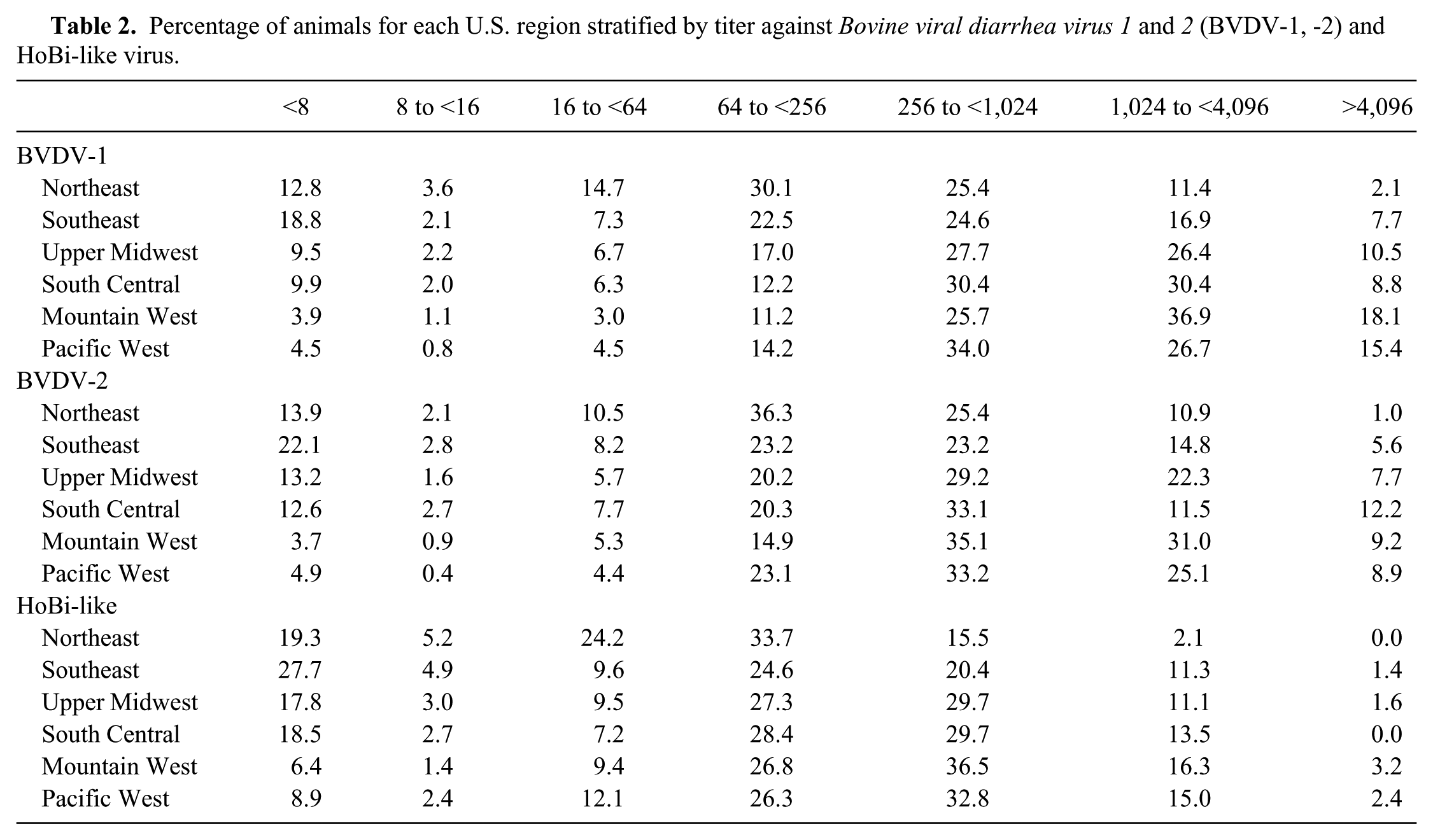
The titers detected against each of the 3 bovine pestivirus species varied significantly by region (Table 2). Sera from the Southeast region had the highest proportion of samples negative for all 3 bovine pestiviruses tested. Further, sera from the Northeast and Southeast regions tended to have lower titers compared to all other regions. In contrast, samples from the Pacific West and Mountain West had fewer negative results and tended to have higher titers.
Percentage of animals for each U.S. region stratified by titer against Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2) and HoBi-like virus.
Discussion
The serologic results suggest that a large portion of U.S. cattle have been exposed to bovine pestiviruses. The source of this exposure, whether vaccination, natural exposure, or a combination of both, was not the purpose of our study. Although no sample had an antibody titer profile that was substantially higher for HoBi-like virus, this does not prove the absence of the virus in the United States. It does indicate that HoBi-like viruses, if present, are much less predominant than BVDV-1 or BVDV-2. Although the source of antibodies cannot be determined and the presence of HoBi-like viruses cannot be absolutely ruled out, the data yield important information on the susceptibility level of the national herd. The overall percentage of animals with titers below levels considered protective against acute bovine pestivirus infection (titer ≤16) 6 were ~11% for BVDV-1, 12% for BVDV-2, and 18% for HoBi-like virus. Control measures against BVDV in the United States have depended primarily on vaccination and removal of persistently infected (PI) cattle. Our results indicate that, despite the availability of BVDV vaccines since the 1960s and their widespread use, 1 in 10 animals reach breeding age with no protection against BVDV-1 or -2 infections. That number rises to nearly 1 in 5 animals that have no protection against HoBi-like viruses. When considering titers to protect against fetal infection (titer ≥256), 6 the percentage of animals not protected are 34% against BVDV-1, 40% against BVDV-2, and 57% against HoBi-like viruses. The numbers suggest that current vaccination efforts are not adequate to eliminate BVDV-associated losses.
It should be noted that our survey only takes into consideration B-cell responses, as expressed by neutralizing titers. T-cell response also plays an important role during animal infection with pestiviruses. 8 However, comparing titers against BVDV-1 and -2 to HoBi-like viruses yields important information regarding the predictive impact of the introduction of HoBi-like viruses into the United States. Various studies have estimated the economic impact of BVDV in the beef industry. The economic impact of BVDV infection in a cow–calf herd was estimated to be $14.85 to $24.84 per cow per year. 15 In feedlots, the presence of PI animals resulted in losses of $41.84 to $93.52 per exposed animal. 11 Our study indicates that there are ~50% more cattle naive to HoBi-like virus than to BVDV-1 or BVDV-2, and 25% more animals do not have titer levels sufficient to prevent fetal infection. This suggests that the introduction of HoBi-like viruses into the United States could result in a 25–50% increase in costs associated with pestivirus.
Although in ~60% of the samples the predominant titer was for BVDV (inclusive of BVDV-1 predominant, BVDV-2 predominant, and BVDV nondifferentiated), for almost half of those it was not possible to differentiate between BVDV-1 and BVDV-2. For those samples in which titers could be differentiated, ~23% were substantially higher for BVDV-1 and ~9% were substantially higher for BVDV-2. This is consistent with a higher prevalence of BVDV-1 strains in the field compared to BVDV-2 strains.9,20
Sera categorized as equivocal were evaluated by VN to BDV and PV to determine if the predominant response was to either of these 2 pestiviruses. No sample had substantially higher titers for either of these viruses, suggesting that although these viruses are present in the United States, their roles as bovine pathogens are limited.
Differences observed among regions in the percentage of samples with the highest titers (>4,096) for BVDV-1 and BVDV-2 could result from differences in vaccine use, differences in natural exposure, differences in the occurrence of PI animals, or variations in the BVDV species prevalence in the regions.9,20 Animals with high titers against BVDV-1 and BVDV-2 also had high titers against HoBi-like viruses. There were 6 animals that had slightly higher titers against HoBi-like viruses (average 9.6 log2) than BVDV-1 and BVDV-2 (average 8.2 and 8.3 log2, respectively). All 6 animals had titers >256 against all 3 bovine pestiviruses. The strength of these titers against these 3 antigenically divergent species suggest multiple and/or prolonged exposure to pestiviruses (natural or induced).
Although the overall percentage of positive samples differs among the tested regions, interesting differences were also evident in the analyzed population when categorized by titers. In general, lower titers and a greater number of negative animals were found in the Northeast and Southeast regions. Although it is not possible to establish the cause of this phenomenon, lower use of vaccine (priming or boostering) and/or decreased viral circulation among the herds could also explain the lower number of animals in the high titer category (>1,024) compared with animals in the central (South Central and Upper Midwest) and western parts of the country. A previous study also indicated regional differences in the percentage of animals vaccinated in dairy herds in the country. 25 Although 85.6% of heifers and 82.2% of cows were vaccinated in the west, the percentage in the east was 72.8% and 74.4% for cows and heifers. 24 Another study of cow–calf operations showed that the Southeast region had the lowest percentage (59.8%) of operations using any kind of vaccine, compared with 76.3% and 90.7%, respectively, for the western and central parts of the country. 23
Variation was observed among packing plants that were considered primarily dairy, beef, or mixed dairy and beef cattle; however, there was also large variation between plants processing the same kind of cattle. Part of this variation may have been the result of regional differences. There appeared to be a trend for fewer negative sera among samples collected at plants that primarily processed dairy cattle.
Although no sample was positive for virus by RT-PCR, it was not a completely unexpected finding. The percentage of PI animals detected in beef calves <4 m of age was 0.3% and 0.2%,12,30 which suggests that 4–6 PI animals might exist in a sample of 2,000 cattle. However, the prevalence of PI animals tends to decrease with age because of attrition. Although some PI animals survive to adulthood and beyond, PIs have a higher mortality rate as neonates and tend to succumb at significantly higher rates to disease than non-PIs. PI animals are also more likely to be culled earlier because of decreased production values. 1 Samples used in our study are from apparently healthy animals >2 y of age, thus the prevalence of PI animals in the sampled population may have been relatively small.
Our nationwide serologic study included testing of samples against HoBi-like virus strains. Although these results show no evidence for the circulation of HoBi-like viruses in the United States, the absence of the virus cannot be confirmed. Our results do indicate that, if HoBi-like viruses are present, they are less prevalent than BVDV-1 or BVDV-2 strains. Regular monitoring of exposure rates is necessary to track the introduction and impact of nonendemic bovine pestiviruses and to assess the effectiveness of control programs.
Footnotes
Acknowledgements
We thank the staff at the Kentucky Federal Brucellosis Laboratory, including Christopher Cracraft, Shanna Wilburn, April Shannon, Lisa Burke, Megan Zinner, Phyllis Bays, and Jeffery Gifford for logistic support in identifying and shipping sera to the laboratory for testing. We also thank Dr. Mark Camacho for his technical assistance in setting up the collaboration between USDA:ARS and the laboratory. We thank the staff at the National Animal Disease Center, including Kathryn McMullen, Patricia Federico, and Renae Lesan. Thanks also to Dr. Kathy Simmons and the National Cattlemen’s Beef Association for their interest in this project and their work to promote cattle health research projects at USDA:ARS.
Authors’ note
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Authors’ contributions
All authors contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted and critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Minimum essential media, Sigma-Aldrich, St. Louis, MO.
b.
Gibco L-glutamine, Thermo Fisher Scientific, Rockford, IL.
c.
Gibco Antibiotic-Antimycotic (100X), Thermo Fisher Scientific, Rockford, IL.
d.
Millex–GV filter unit, Millipore Ireland, County Cork, Ireland.
e.
Tween20, Sigma-Aldrich, St. Louis, MO.
f.
Albumin from bovine serum, Sigma-Aldrich, St. Louis, MO.
g.
Pierce Goat anti-mouse IgG (H+L), peroxidase conjugate, Thermo Fisher Scientific, Rockford, IL.
h.
Rec-protein G-peroxidase conjugate, Invitrogen, Camarillo, CA.
i.
3,3′-diaminobenzidine, Sigma-Aldrich, St. Louis, MO.
j.
QIAcube, Qiagen, Hilden, Germany.
k.
QIAamp viral RNA mini kit, Qiagen, Hilden, Germany.
l.
GelRed, Biotium, Hayward, CA.
m.
Excel 2010, Microsoft, Redmond, WA.
n.
SAS Institute, Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by cooperative research and development agreements with Novartis Animal Health and Zoetis Animal Health.
