Abstract
A 5-y-old Holstein dairy cow had surgery for a suspected displaced abomasum 10 d postpartum, developed acute neurologic signs at day 19, and was found dead 21 d postpartum. At autopsy, there was a peri-incisional intramuscular abscess that communicated with the peritoneal cavity, as well as hemorrhage and malacia involving the brain, and multiple nodules in the liver, kidneys, and lungs. Fungal hyphae were seen histologically at the surgery site, on the surface of the liver, and in lesions of severe necrotizing vasculitis in the lungs, kidneys, brain, and liver. The uterus was free of fungal organisms. Pan-fungal PCR and DNA sequencing identified the fungus as Mortierella wolfii. Previously reported deaths from M. wolfii have been related to abortion, but in this case, there was no histologic evidence of fungal organisms in the uterus, calving was routine, and there was a several week delay between calving and development of neurologic signs. The findings suggested a unique case of surgical site infection with subsequent embolic mycosis.
Fungal infections resulting in encephalitis in cattle are rare, with most cases reported in aborted or neonatal calves rather than in adult cows.8,17,19 In the reported cases of mycotic encephalitis in adult cattle, all have had necrotizing vasculitis with intralesional zygomycete fungi.9,16,26 Encephalitis was associated with recent abortion or calving, and the reports in which definitive etiologic identification had been pursued established Mortierella wolfii as a consistent causative agent.9,16
M. wolfii is a saprophytic fungus in the class Zygomycota, which includes Mucor and Rhizopus spp. 20,21 Fungi in orders Mucorales and Mortierellales have a propensity for blood vessel invasion and hematogenous spread. 21 Lesions develop commonly in the lungs, 21 gastrointestinal tract,2,4,21 and reproductive system of cattle. 25 M. wolfii is a common environmental fungus, especially in silage,13,21 and soil and other contaminants pose a risk to surgical wounds. The morphology of colonies grown on Sabouraud agar can help differentiate M. wolfii from other zygomycetes. 21 The characteristic appearance of M. wolfii is white velvety colonies with distinct lobulated outlines. 21 Additionally, as in our case, molecular techniques can aid in etiologic diagnosis. 16
Bovine mycotic abortions occur worldwide and are generally sporadic with a propensity for the third trimester.1,11,25 Aspergillus spp. are the most common cause of mycotic abortions in cattle in North America, 12 whereas M. wolfii is the most common cause in areas of New Zealand. 3 Up to 20% of cows with abortion caused by M. wolfii may develop fatal mycotic pneumonia, which is considered a distinctive mycotic abortion-pneumonia syndrome. 7 The proposed pathogenesis involves initial inhalation followed by hematogenous translocation to the uterus, fungal abortion, and subsequent recirculation to cause embolic pneumonia.6,7 M. wolfii is widespread in the environment, and environmental factors, such as climate, as well as management practices, such as feeding moldy hay and silage, have been associated with infection.3,8,19,21
Disseminated mycosis in adult cattle may develop secondary to gastrointestinal or respiratory infection, 4 with possible predisposing factors including ruminal acidosis4,27 and antimicrobial therapy. 10 Previous cases of bovine zygomycotic encephalitis have hypothesized systemic debilitation and/or immunosuppression related to ketoacidosis, 16 traumatic reticulopercarditis, 5 and parasitic anemia 18 as potential contributing factors. We describe a unique case of disseminated Mortierella spp. mycosis associated with surgical site infection, in which previous pregnancy, suspected metritis, ketosis, and surgery may have contributed to a propensity for infection.
On the day after death, a 5-y-old Holstein dairy cow was presented to the Diagnostic Center for Population and Animal Health (Michigan State University, Lansing, MI) for autopsy. The cow was 3 wk postpartum and had a history of therapy for presumed metritis in the week following parturition. Surgery was performed 10 d postpartum for a suspected displaced abomasum, but none was found. The cow developed ketosis with continued clinical decline, and despite medical therapy, developed neurologic signs, including circling, 19 d postpartum. Treatments in the postpartum period included antimicrobials (ceftiofur, penicillin, and oxytetracycline), flunixin meglumine, dexamethasone, propylene glycol, thiamine, and probiotics; however, the cow was found dead 21 d after parturition. Her calf had no clinical evidence of disease.
At autopsy, a 10-cm closed surgical incision was present in the right paralumbar fossa and associated with a subjacent 8 × 10 × 10 cm intramuscular abscess. The abscess was filled with fetid, opaque, pale-brown, pink-tinged pus and lined by a friable wall that communicated with the peritoneal cavity. The liver was diffusely pale, tan, and friable with rounded margins; ~15 firm, tan, flat, 6–12 mm nodules extended into the parenchyma. The kidneys had six 3–5 mm, raised, tan cortical nodules. The lungs had scattered, firm, tan-brown, slightly raised, 3–12 mm nodules, and tan fibrinous adhesions between lung lobes. The uterus contained opaque fluid. A blood clot was present in the right lateral ventricle of the brain, and there was malacia with multifocal-to-coalescing hemorrhage affecting the walls of the lateral and third ventricles, the midbrain, and rostral brainstem (Fig. 1). All other organs were grossly unremarkable.
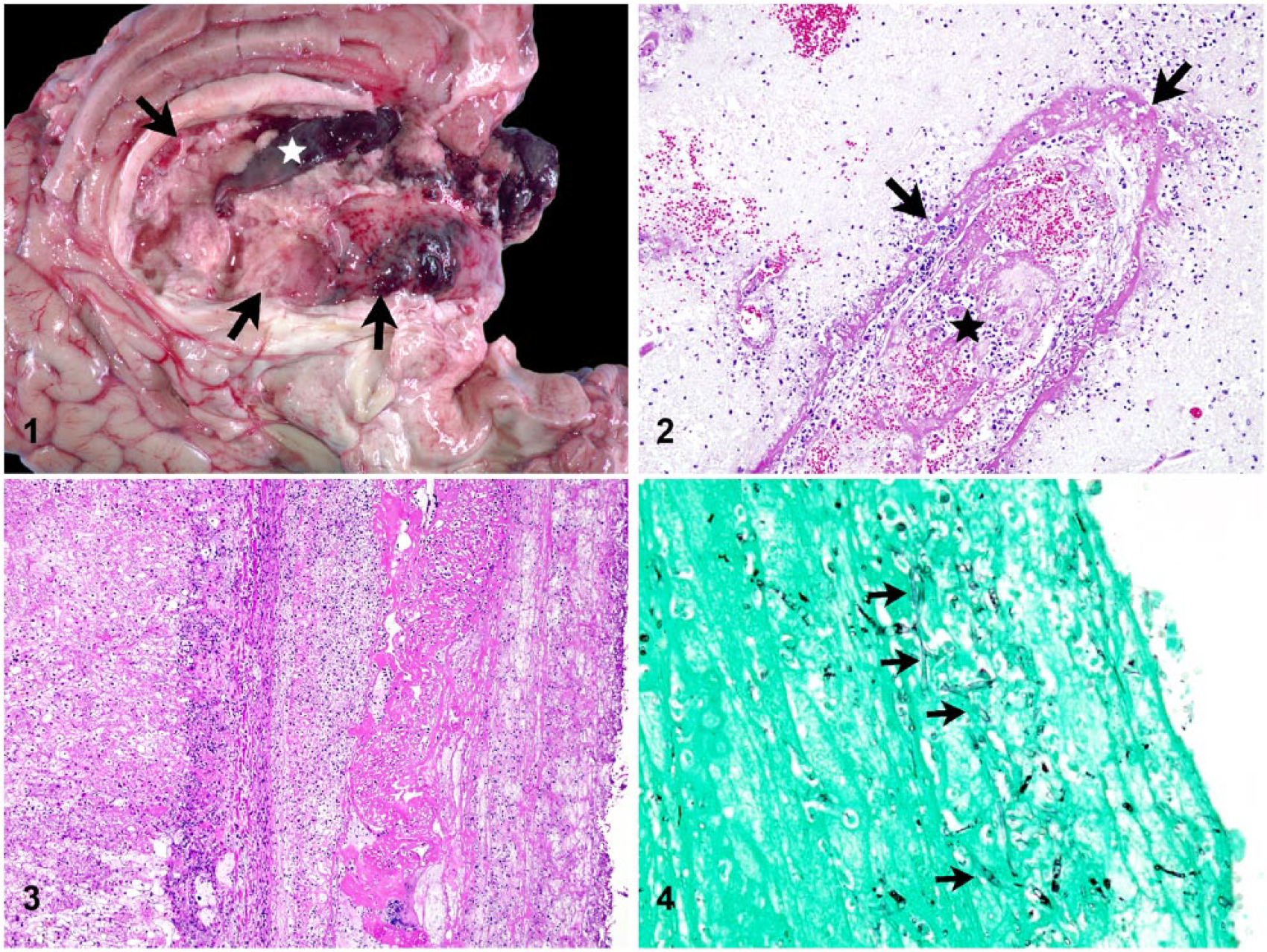
Five-year-old Holstein cow with embolic mycotic encephalitis and hepatitis.
Microscopic examination of the brain revealed marked, multifocal, fibrinosuppurative and necrotizing vasculitis, and associated encephalitis with thrombosis and abundant hemorrhage (Fig. 2). Throughout the brain and meninges, affected blood vessels had fibrinoid mural necrosis with large numbers of viable and degenerate, intramural and perivascular neutrophils, and perivascular hemorrhage and edema. The adjacent neuropil was rarefied and infiltrated by neutrophils extending from inflamed vessels. Small numbers of fungal organisms were found within inflamed vessel walls and free in the surrounding neuropil. Skeletal muscle and adipose tissue bordering the abscess at the incision site had multifocally extensive severe myodegeneration and regeneration with vascular thrombosis, and regional dissecting myositis and steatitis with intralesional fungal hyphae. The grossly observed pale liver nodules consisted of regionally extensive necrotizing hepatitis with vasculitis, thrombosis, and mats of fungal organisms and fibrin on the hepatic surface (Fig. 3). Fewer fungal organisms were scattered amid the devitalized hepatic parenchyma. Sections of lung and kidney had similar histologic changes with vasculitis, thrombosis, and small numbers of fungal organisms centered on blood vessels. The fungal organisms in the brain, abscess, liver, lung, and kidney were comparable in morphology; hyphal forms were poorly staining, non-septate, and ~4–8 µm wide with irregularly parallel walls, and occasional variable branching. The fungal organisms stained variably positive for periodic acid–Schiff and Grocott methenamine silver (Fig. 4). The uterine mucosa was autolytic with aggregates of mixed bacteria along the devitalized endometrial surface; vasculitis, thrombosis, and fungal organisms were not identified in the uterus.
Moderate numbers of Escherichia coli and Enterococcus faecalis were isolated on bacterial culture of the abscess. Small numbers of coliforms, Lactobacillus, and Trueperella spp. were identified from the liver and brain, consistent with contaminants and/or perimortem-to-postmortem bacterial growth. There was no growth on fungal culture of any of the submitted tissues. Growth may have been inhibited by moderate postmortem autolysis, and the presence of cycloheximide, an inhibitor of zygomycete fungal growth, 21 in one of the culture media. Fungal DNA was detected in grossly abnormal liver tissue by a pan-fungal polymerase chain reaction (PCR) assay using a previously published primer 24 targeting the 28S large subunit ribosomal RNA. Nucleic acid sequencing of the PCR amplicon was performed at the Research Technology Support Facility at Michigan State University (Lansing, MI) for confirmation. The nucleic acid sequence was 100% identical to 2 published M. wolfii isolates (GenBank accessions HQ667382, AB154774). Although the fungus seen microscopically in our case was not isolated in culture, it was confirmed as M. wolfii by molecular techniques.
To our knowledge, our case represents only the third report of systemic and encephalitic mycosis attributable to M. wolfii in an adult cow, and the second case in North America. Both of the previous reports were related to recent abortion,9,16 whereas in our case there was no evidence of predisposing mycotic endometritis or placentitis. Calving was routine, the calf was healthy, there was a several week delay postcalving prior to the onset of neurologic signs, and there were no fungal-associated histologic lesions in the uterus. Moreover, the distribution of fungal organisms within the incision site abscess and heavily along the surface of the liver supported wound-associated infection with peritoneal translocation and subsequent embolic spread. The pattern of lesions in the lungs supported hematogenous spread rather than an inhaled infection, and no gastrointestinal source was identified.
Other differentials for necrotizing and hemorrhagic lesions affecting the bovine central nervous system, as seen in our case, include a bacterial septicemia, such as Histophilus somni, and a neurotropic virus, such as bovine alphaherpesvirus 5 (BoHV-5, bovine encephalitis virus).14,15,22,23 Infectious thrombotic meningoencephalitis (ITME) caused by H. somni is similarly characterized by random foci of hemorrhage and necrosis with distinctive vasculitis and thrombosis.14,15 However, ITME is most prevalent in feedlot cattle in North America, 14 and H. somni was not identified by bacterial culture in our case. Bovine necrotizing meningoencephalitis caused by BoHV-5 is also a sporadic disease in adult cattle. However, gross lesions are centered on the gray matter, and characteristic histologic findings, including necrotic neurons, perivascular cuffs, and intranuclear viral inclusion bodies, were not identified in our case.22,23
Mycotic encephalitis should be included in differential considerations for neurologic signs in cattle, and may have gross and histologic lesions resembling other bovine encephalitides. Furthermore, although M. wolfii is a rare cause of systemic mycosis, it should be especially considered in mature cows with systemic disease following abortion or, possibly, wound infection.
Footnotes
Acknowledgements
We thank the DCPAH histology laboratory for their help with sample processing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This report was supported by the Diagnostic Center for Population and Animal Health, Michigan State University.
