Abstract
Despite the particular susceptibility of the rabbit to experimental infection with Human herpesvirus 1 (HHV-1) and the high seroprevalence of HHV-1 in human beings, reports of natural infection in pet rabbits are rare. The current report describes 2 cases of HHV encephalitis in pet rabbits in North America. Antemortem clinical signs included seizures, ptyalism, and muscle tremors. Results of complete blood cell count and plasma biochemistry panel were unremarkable except for a mild leukocytosis in both cases. Both rabbits died after a short period of hospitalization. Rabbit 1 presented mild optic chiasm hemorrhage on gross examination, while rabbit 2 had no gross lesions. Histologic findings for both cases included lymphocytic and/or lymphoplasmacytic encephalitis with necrosis and the presence of intranuclear inclusion bodies in neurons and glial cells. Polymerase chain reaction (PCR) analysis of affected brain tissue using primers specific for Human herpesvirus 1 and 2 confirmed diagnosis of HHV encephalitis for rabbit 1. Immunohistochemical staining (poly- and monoclonal) and PCR analysis using primers specific to HHV-1 confirmed the diagnosis of HHV-1 encephalitis for rabbit 2. The owner of rabbit 2 was suspected to be the source of infection due to close contact during an episode of herpes labialis. Given the high susceptibility of rabbits to experimental HHV-1, high seroprevalence of HHV-1 in human beings, and severity of clinical disease in this species, clinician awareness and client education is important for disease prevention. Human herpesvirus 1 encephalitis should be considered as a differential diagnosis for rabbits with neurologic disease.
Keywords
Herpesvirus disease in rabbits includes both naturally occurring and experimental infections. The most common naturally occurring herpesvirus infections in rabbits are caused by Leporid herpesvirus 2 (syn. Herpesvirus cuniculi) and Leporid herpesvirus 3 (syn. Herpesvirus sylvilagus), both gammaherpesviruses.6,7,14,19,24,28,33 Leporid herpesvirus 4, an alphaherpesvirus, was identified in published accounts from 2008 as pathogenic to domestic rabbits.9,10
Rabbits are susceptible to Human herpesvirus 1 (HHV-1; order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Simplexvirus) and are used as experimental models for HHV encephalitis in human beings.13,17,18,21,23 Human herpesvirus 1 in rabbits is exclusively neurotropic. The different proposed mechanisms of infection and virus spread into the central nervous system (CNS) in people with HHV encephalitis have been investigated in rabbits. Intranasal or olfactory bulb virus inoculation is considered the most important route of infection for human beings.12,29 With experimental inoculation in rabbits, the virus migrates through the olfactory nerves into the frontal and temporal lobes.22,23 The trigeminal nerve pathway has been proposed as an important route of infection in cases of human HHV encephalitis and can be experimentally induced in rabbits with cornea scratching and inoculation. The virus migrates through the trigeminal system to the brain stem and cerebral hemispheres, especially the ventral parts of the limbic system. 21 Cell to cell spread of HHV has also been investigated in rabbits by intraocular inoculation of the virus. Following inoculation, the virus migrates through the optic nerve and chiasm to the corpus geniculatum.17,18 Rabbits can present neurologic signs as early as 2 days postinoculation. Such signs include changes in activity (decreased or increased), ataxia, head jerking movements, and/or circling.13,17,18,21,30 Ptyalism has been described in rabbits following intranasal virus inoculation. 30 Neurologic disease can progress to seizures, moribund state, or death with intranasal or intraocular virus inoculation.17,18,30 Mortality rates vary with inoculation route being the highest for intranasal inoculation (up to 95%) 30 and the lowest following intraocular inoculation (10%).17,18 Development of latent state of infection has been described for experimental infection in rabbits following corneal and intranasal inoculation.13,21,30 Characteristic histologic lesions include mononuclear cell inflammation, perivascular inflammation and necrosis, and intranuclear inclusion bodies. Lesions are routinely first noted along the virus track and entry site into the CNS.13,17,18,21–23
Reports of spontaneous HHV encephalitis in rabbits are relatively rare, with current reports describing disease in pet rabbits in Europe.4,5,16,25,32 Human beings are considered the source of infection for pet rabbits, as close contact with an owner with active facial and labial lesions before the onset of clinical signs has been reported.4,16,32 Although horizontal transmission of HHV from rabbit to rabbit is possible, 11 spontaneous HHV encephalitis has been described in single rabbit households.4,16 Clinical and histologic features of spontaneous HHV encephalitis in rabbits are similar to what has been described for experimental infection, with the exception of 100% mortality reported for cases of natural infection.4,16,32 Considering rabbits’ susceptibility to experimental HHV encephalitis, high prevalence of disease in people, and the popularity of domestic rabbits as pets and laboratory animals, a higher incidence of spontaneous disease would be expected. The relative low incidence of clinical disease could be due to several factors, including rare occurrence of transmission from human beings to rabbits, infection followed by complete recovery or development of latent infection, and/or low virulence of the human virus.4,16 The rare occurrence of natural cross-infection between human beings and other species may be due to a non-immunologic protective mechanism against HHV infection in animals. 27 The current report describes fatal HHV encephalitis in 2 pet rabbits in North America.
Case 1 was a 2.5-year-old intact male rabbit presented for evaluation of seizures and ptyalism following a minor head trauma accident at home. The rabbit had no past medical history and was housed indoors only, with 3 other rabbits in the household. The rabbit appeared blind. Ophthalmologic examination findings included bilateral incomplete pupillary light reflex, optic nerve hemorrhage and edema in the right eye, mild optic nerve edema in the left eye, and bilateral horizontal nystagmus with fast phase to the right. Blood was collected for a complete blood cell count and biochemistry panel. The results revealed leukocytosis (50 × 103/µl; reference [ref.] interval: 5.2–12.5 × 103/µl) 3 with heterophilia (93%; ref. interval: 20–75%) 3 and lymphopenia (5%; ref. interval: 30–80%) 3 ; the presence of large reactive and occasional atypical lymphocytes was also reported. Significant biochemistry panel abnormalities included hyperglycemia (331 mg/dl; ref. interval: 75–155 mg/dl) 3 and elevated blood urea nitrogen (60 mg/dl; ref. interval: 13–29 mg/dl), 3 creatinine (2.9 mg/dl; ref. interval: 0.5–2.5 mg/dl), 3 and phosphorus (13.4 mg/dl; ref. interval: 4–6.9 mg/dl). 3 Calcium levels were within normal limits (9.7 mg/dl). The patient was treated with dexamethasone (0.5 mg/kg, intramuscularly, once), phenobarbital (2 mg/kg, orally, twice a day), and diazepam (1 mg/kg per rectum when seizure activity was noted). The seizures worsened, and rabbit died after 48 hr of treatment and monitoring.
Necropsy revealed a small amount of hemorrhage in the meninges around the optic chiasm. The left kidney was approximately one-half the size of the right kidney and was pale and firm with a pitted cortex. Histologic examination of the brain revealed laminar cortical necrosis and neuron loss in addition to neuronal necrosis in the basal nuclei and thalamus throughout the cerebrum. Many of the remaining neurons contained intranuclear viral inclusion bodies. Heterophils were present within and around hypertrophic parenchymal blood vessels and areas of necrosis. Inflammatory cells within the meninges were predominantly lymphocytes. Within the brainstem, fibrin and leukocytes were present in the fourth ventricle. Other significant histological findings included acute necrotizing interstitial pneumonia with congestion and edema and interstitial fibrosis and atrophy of the left kidney. DNA extracted from paraffin-embedded sections of brain from the rabbit was subjected to polymerase chain reaction (PCR) using HHV-1 and 2 specific primers, along with a thermostable DNA polymerase and deoxyribonucleotide triphosphates. Formation of a PCR product was detected by polyacrylamide gel electrophoresis (PAGE) and ethidium bromide staining of the gel. Human herpesvirus types 1 and 2 viral DNA were detected in the sample due to the presence of PCR products of the expected size as visualized on the PAGE gel after staining, strongly supporting the diagnosis of HHV encephalitis.
Case 2 was a 4-year-old male neutered rabbit presented for evaluation after a 3-day history of anorexia and gastrointestinal stasis and 1-day history of periocular swelling and ptyalism. The rabbit was housed indoors only and had contact with several other rabbits. Main physical examination findings on presentation included bilateral blepharitis with purulent ocular discharge, and severe ptyalism with saliva noted on the chin, neck, and forelimbs. Tachypnea with dyspnea and harsh lung sounds and a cardiac arrhythmia characterized by occasional dropped heartbeats were also noted. Blood was collected for a complete blood cell count and biochemistry panel. The results revealed mild leukocytosis (15.1 × 103/µl) with high normal heterophil count (75%), lymphopenia (9%), and monocytosis (13%; ref. interval: 1–4%). 3 Biochemistry panel abnormalities were limited to hyperglycemia (220 mg/dl; ref: interval: 75–155 mg/dl) 3 and elevated blood urea nitrogen (43 mg/dl; ref. interval: 13–29 mg/dl). 3 Radiographic images of the thorax demonstrated a severe increased soft tissue opacity in the cranioventral thorax, suggestive of pneumonia. An electrocardiogram was performed, and the rabbit was diagnosed with sinus bradycardia with premature ventricular contractions (VPCs). Echocardiogram findings included mitral regurgitation and mild thickening of the mitral valve leaflets. The cause of the described bradycardia and VPCs could not be identified. The rabbit was treated with supplemental oxygen, intravenous crystalloid fluids, a enrofloxacin b (15 mg/kg subcutaneously every 24 hr), famotidine (0.5 mg/kg subcutaneously every 24 hr), albendazole c (10 mg/kg orally every 24 hr), and sucralfate d (25 mg/kg orally every 8 hr). Ptyalism and dyspnea worsened, and the rabbit was found dead 12 hr after being admitted for evaluation.
Significant gross necropsy findings included conjunctivitis and blepharitis of the left eye and locally extensive bronchopneumonia and rhinitis. Histologically, the brain had severe, multifocal, lymphocytic, meningopolioencephalitis with gliosis and neuronal satellitosis (Fig. 1A). Mild lymphocytic vasculitis was also noted (Fig. 1B). Many neurons contained intranuclear inclusion bodies (Fig. 1C). The lesions were more severe in the temporal and frontal cortex regions of the brain. The epineurium and perineurium of multiple nerves within the nasal cavity and olfactory bulbs contained dense lymphocyte infiltrates. Similar infiltrates were present within the bulbar conjunctiva and adjacent cornea, the epineurium of the optic nerve, and adjacent sclera and skeletal muscle. Additional relevant histologic findings included severe suppurative bronchopneumonia and mild myocardial degeneration and fibrosis.
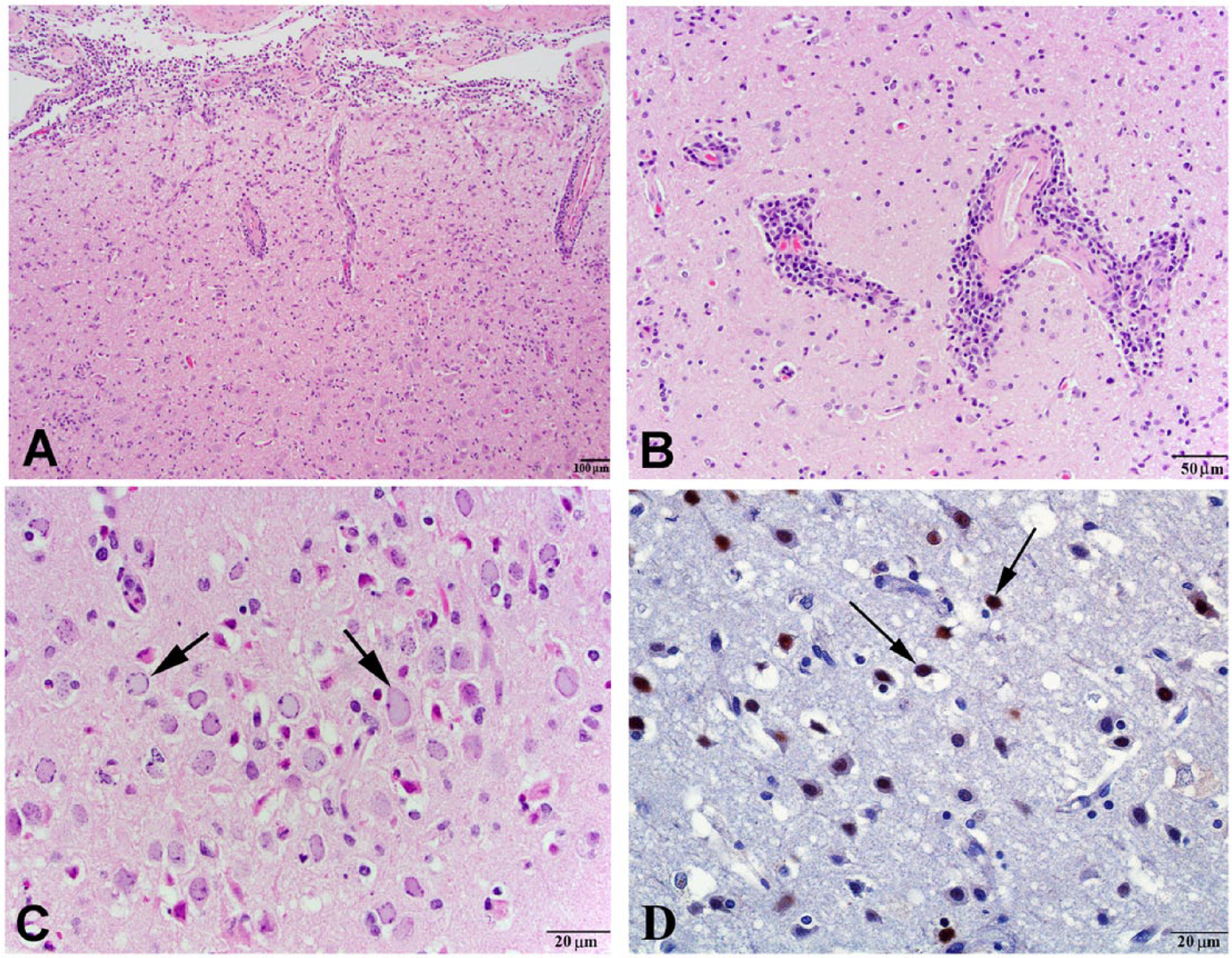
Human herpesvirus encephalitis in a rabbit.
Due to a strong suspicion of HHV encephalitis (owner had an episode of cold sores before the rabbit became sick), immunohistochemical staining (IHC) was performed to detect the presence of HHV in the brain. In the initial protocol, the primary antibody used was a polyclonal rabbit anti–HHV-1 antibody e and the secondary antibody was a goat anti-rabbit antibody. f A commercial staining kit f acted as the tertiary antibody, and the slides were developed with diaminobenzidine f as a chromogen. Positive (brain tissue from a common marmoset with HHV) and negative (brain tissue of a rabbit without neurologic signs or lesions) controls were used. To improve the specificity of the test, the IHC was repeated using a noncommercial monoclonal anti–HHV-1 antibody targeting the UL42 epitope of the polymerase processivity factor. g The secondary antibody was a commercial anti-mouse antibody, e with diaminobenzidine f also used as chromogen. The nucleus and cytoplasm of neurons and glial cells within the affected areas of the brain presented strong positive staining for HHV-1 with both IHC methods (Fig. 1D). The positive staining of the endothelial cells with the polyclonal antibody protocol suggested viral replication within these cells. The polyclonal IHC protocol was also performed on lung tissue, and no immunostaining was detected.
To confirm the presence of HHV-1 in the brain via PCR, DNA was extracted from paraffin-embedded brain tissue using a commercial kit. h The PCR reaction was performed using primers specific for the UL11 gene, which encodes a protein required for assembly and budding of HHV to the extracellular space. 2 The UL11 primers used were 5′-CTCCGGGGGTCCGCCGGT-3′ (forward) and 5′-TATGGGCCTCTCGTTCTC-3′ (reverse) based on the published DNA sequence for this gene. 15 For negative control, DNA was extracted from paraffin-embedded brain tissue from a rabbit without neurologic signs or lesions. Degenerate PCR primers targeting a highly conserved region within the DNA polymerase gene were used in order to detect a broader range of HHV strains and types. 31 The PCR products were visualized after ethidium bromide nucleic acid staining of the agarose gel electrophoresis product. The PCR amplification product, only detected in the specimen originating from the brain of the affected rabbit (Fig. 2), was submitted for sequencing. i The DNA sequence of the 258–base pair amplified product was identical to the UL11 gene of HHV-1 and homologous but not identical to UL11 gene of HHV-2. j
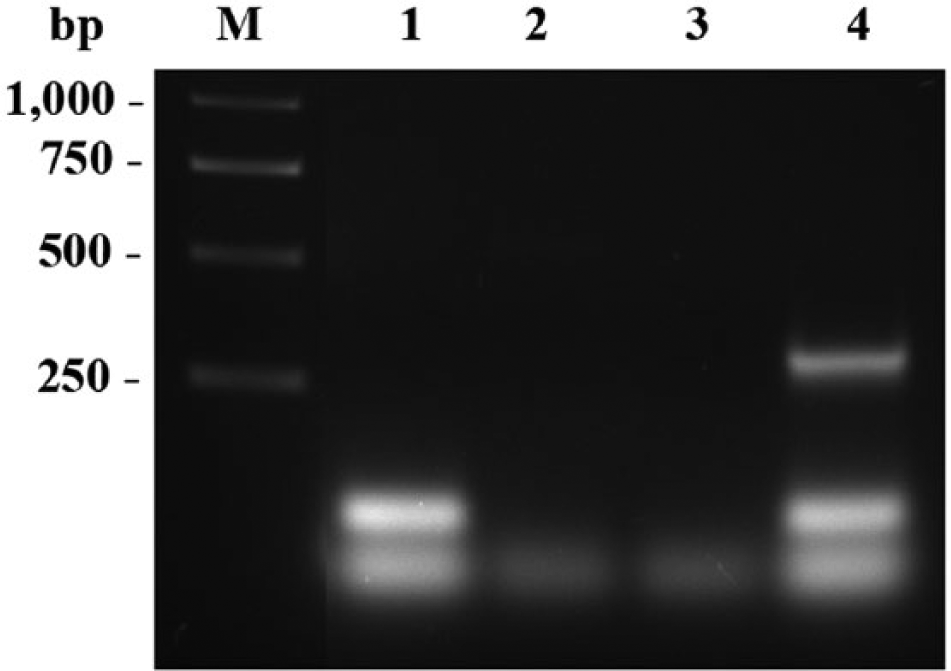
Results of polymerase chain reaction (PCR) assay used to confirm the presence of Human herpesvirus 1 (HHV-1) in the brain tissue of a rabbit with suspected HHV-1 encephalitis. DNA was extracted from paraffin-embedded brain tissue of the affected rabbit (lanes 2 and 4) and paraffin-embedded brain tissue from a rabbit without neurologic signs or lesions (control; lanes 1 and 3). The PCR reaction was performed using primers specific for the UL11 gene (lanes 3 and 4) and degenerate PCR primers targeting a highly conserved region within the DNA polymerase gene, used to detect a broader range of HHV strains and types (lanes 1 and 2). The PCR 258–base pair amplification product, only detected in the specimen originating from the brain of the affected rabbit, indicates HHV-1 infection. M = molecular weight marker.
The current report describes 2 cases of spontaneous HHV encephalitis in domestic rabbits in North America. Viral encephalitis was suspected based on histologic lesions, with confirmation of the presence of HHV antigen in the affected tissues with IHC techniques and/or PCR.
Natural HHV infections in pet rabbits are suspected to arise from close contact with human beings with active herpes skin lesions, and this was proposed for case 2 in this report. Isolation of virus from the owner and comparison with virus isolated from the rabbit would be required to confirm the source of virus. Horizontal transmission was not considered for the 2 cases in this report, as other rabbits in the same household were not affected. As for human beings, it is unknown if spontaneous HHV encephalitis in rabbits results from infection immediately following exposure or reactivation of latent disease.
Antemortem clinical signs for the 2 rabbits in the present report were similar to what has been described for experimentally induced and spontaneous HHV encephalitis in rabbits. Ptyalism was noted in both of the cases described in this report, and it has been reported in both natural4,16,32 and experimental 22 infections with HHV. Ocular signs were seen in both cases and in a previous report. 16 Ocular signs and significant ptyalism are usually not reported in rabbits with neurologic disease caused by more common conditions in rabbits (Encephalitozoon cuniculi, Pasteurella multocida). 5 No signs specific to the CNS were noted in case 2, which is different from what has been reported in previous cases of rabbits with natural HHV encephalitis, and not expected considering the lesions in the CNS for this case were similar to what has been described in previous cases. It is possible that the respiratory disease, later confirmed as bronchopneumonia, accelerated the course of disease and caused death before neurologic signs developed. Hemogram abnormalities for both cases in the current report included leukocytosis with heterophilia and lymphopenia. Monocytosis was reported for case 2. These findings are similar to what was described in a case of natural disease 16 and, although nonspecific, can occur with viral infection in rabbits. 8 The severe suppurative bronchopneumonia reported for case 2 can also support the hemogram abnormalities. Clinical disease progressed fast for both cases in the present report despite aggressive supportive care and symptomatic treatment. Fast progression of the disease appears to be a feature of spontaneous HHV encephalitis in rabbits, with rabbits dying or being euthanized soon after clinical signs develop.
The histologic lesions for both cases are similar to cases of natural and experimental infection with HHV. As in previous reports of natural infection with HHV, the distribution of the lesions within the CNS for both cases is different than what is reported with experimental HHV infection, making identification of route of infection difficult. This could be due to 1) alternative or combined route of infection; 2) unknown period between exposure to the virus, development of clinical signs, and death or euthanasia for cases of spontaneous disease (well characterized for experimental infection); and/or 3) different methodologies for postmortem sectioning of the brain and histologic evaluation of the CNS between previous reports and even between the 2 cases in the current report.
Pneumonia was reported for both cases in the present report, with each case presenting different histologic lesions (acute necrotizing interstitial pneumonia with congestion and edema for case 1 and severe suppurative bronchopneumonia for case 2). The histologic description associated with HHV encephalitis rabbit model studies is limited to the CNS, and previous reports of natural HHV disease in rabbits have no reference of gross or histologic lung lesions. Human herpesvirus 1 has been implicated in pulmonary disease in human beings. Immunosuppression and/or concurrent diseases may cause reactivation of latent infection and shedding of the virus into the oropharynx, with the virus reaching the lung by aspiration or continuous spread. 26 However, HHV-1 does not appear to have a primary role in pulmonary disease in affected individuals. 1 In addition, no immunostaining was detected in the lung with IHC using polyclonal antibody against HHV-1 IHC. A primary upper respiratory–derived bacterial infection was suspected, as histologic changes were consistent with suppurative inflammation. Activation of P. multocida infection has been described in rabbits experimentally exposed to a novel herpesvirus. 20
In conclusion, rabbits are susceptible to natural infection with HHV, and the organisms should be considered as a differential diagnosis for rabbits presenting with signs of CNS disease, especially indoor rabbits in close contact with persons with active herpes lesions. Clinical signs of HHV encephalitis are nonspecific and can be seen with more common neurologic diseases of rabbit including E. cuniculi, P. multocida, neoplasia, and trauma. 5 Spontaneous HHV encephalitis in rabbits progresses fast, with no response to treatment, resulting in death or euthanasia. In most cases of spontaneous disease, no gross lesions are present in the CNS. Histologic changes are characterized by nonsuppurative encephalitis, necrosis, and presence of intranuclear inclusion bodies. The pattern of distribution of the lesions with natural infection does not appear to be as precise as with experimental infection but may suggest route of infection in some cases. Given the high susceptibility of rabbits to experimental HHV-1, high seroprevalence of HHV-1 in human beings, and severity of clinical disease in this species, clinician awareness and client education is important for disease prevention.
Footnotes
Acknowledgements
The authors would like to thank Drs. Cindy Fishman and Evan Janovitz for postmortem and histologic evaluation of case 1; Drs. Jim Casey and Joel Baines, and Jakob Trimbert and Elizabeth Wills from the Department of Microbiology and Immunology, Cornell University College of Veterinary Medicine for performing the PCR for case 2; Dr. Deborah Parris from the Department of Molecular Virology, Immunology, and Medical Genetics, The Ohio State University College of Medicine for providing the monoclonal HHV-1 antibody; and Florinda Jaynes from the Department of Veterinary Biosciences, The Ohio State University College of Veterinary Medicine for performing the HHV-1 monoclonal antibody IHC protocol.
a.
PlasmaLyte, Baxter Healthcare Corp., Deerfield, IL.
b.
Baytril (22.5 mg/ml injectable solution), Bayer Healthcare Corp., Whippany, NJ.
c.
Valbazen (113.6 mg/ml oral suspension), Pfizer Animal Health, New York, NY.
d.
Sucralfate (suspension 1 g/10 ml), Sanofi-Aventis, Kansas City, MO.
e.
Dako North America Inc., Carpinteria, CA.
f.
ABC Elite, Vector Laboratories Inc., Burlingame, CA.
g.
Courtesy of Dr. Deborah Parris, The Ohio State University, Columbus, OH.
h.
QIAamp DNA blood mini kit, Qiagen Inc., Germantown, MD.
i.
Biotechnology Resource Center, Cornell University, Institute of Biotechnology, Ithaca, NY.
j.
BLAST, National Center for Biotechnology Information, National Institutes of Health, Bethesda, MD. Available at: blast.ncbi.nlm.nih.gov/
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
