Abstract
Malignant catarrhal fever (MCF) is a severe, systemic, lymphoproliferative disease affecting domestic ruminants, caused by a group of MCF viruses in the genus Macavirus. Infection of cattle and bison with ovine herpesvirus 2 (OvHV2) is economically significant in North America. Sheep are the reservoir host of the virus, and only rarely manifest disease. Cattle and bison, however, frequently have lymphoproliferation, mucosal ulceration, and systemic vasculitis. OvHV2-induced MCF in cattle and bison is often fatal, with clinical recovery reported only rarely. Chronic cases are uncommon, but vascular changes of variable severity and ocular lesions have been described. Here we present a case of chronic MCF in a cow with proliferative arteriopathy, systemic vasculitis, and OvHV2-associated hypophysitis. We demonstrated OvHV2 nucleic acid in affected tissues with in situ hybridization.
Keywords
Six gammaherpesviruses belonging to the malignant catarrhal fever virus group (Herpesviridae, Gammaherpesvirinae, Macavirus) are known to cause malignant catarrhal fever (MCF) in several species of ungulates. Of these, alcelaphine herpesvirus 1 (AlHV1; Alcelaphine gammaherpesvirus 1) and ovine herpesvirus 2 (OvHV2; Ovine gammaherpesvirus 2) are associated most commonly with natural outbreaks. 12 Each of the MCF-inducing gammaherpesviruses is well-adapted to a natural reservoir host, including wildebeest (Connochaetes sp.) for AlHV1 and domestic sheep (Ovis aries) for OvHV2, which are generally affected subclinically and shed the virus.12,19 In domestic sheep, OvHV2 is shed in nasal secretions and transmitted to susceptible hosts via aerosolization.11,17 Susceptible hosts infected with OvHV2, including domestic cattle (Bos taurus, B. indicus) and American bison (Bison bison), develop severe, systemic disease that is often fatal.12,19 Systemic necrotizing vasculitis in sheep, formerly diagnosed as polyarteritis nodosa, has been linked to OvHV2 infection. 14
Infection with OvHV2 in cattle and bison often leads to systemic disease characterized by a triad of lesions including lymphoid proliferation, vasculitis, and erosions and ulcerations in the skin and mucosal surfaces, resulting in spontaneous death or euthanasia 1–18 d after the onset of clinical signs.9,12,19 Other reported clinical presentations include persistent mild ocular disease of several months duration with progression to terminal disease, and partial-to-complete clinical recovery from systemic disease, with vascular changes of variable severity and ocular lesions persisting.9,10,12,16,19
A 3-y-old Brangus cow was presented for evaluation of a 4-mo history of bilateral uveitis and severe keratitis that failed to respond to treatment with topical antibiotics and systemic NSAIDs. PCR on whole blood was negative for bovine alphaherpesvirus 1, bovine leukemia virus, bovine viral diarrhea virus (BVDV; Pestivirus), bluetongue virus, OvHV2, and epizootic hemorrhagic disease virus. A chemistry panel and urinalysis revealed mild renal azotemia and pyuria, respectively. Despite supportive care, the cow developed hyporexia, hematuria, stranguria, and dark, foul-smelling feces. Given the lack of response to treatment and continued decline, euthanasia was elected.
At autopsy, mucopurulent exudate coated the conjunctivae bilaterally; in the cornea of the right eye was a focal area of edema. In the interdigital space of the left forelimb, right hindlimb, and on the udder were 1-mm to 3-cm diameter ulcers. In the mucosa of the oral cavity, including tongue, hard palate, and gingiva, as well as esophagus, were multifocal-to-coalescing ulcers of 0.5–2-cm diameter (Fig. 1). In the mucosa of the abomasum, omasum, and cecum were multifocal erosions. On the capsular surface of both kidneys were pale-tan, pinpoint to 2-mm, slightly raised, soft nodules; on cut section, areas of necrosis and inflammation extended from the medulla into the cortex, consistent with suppurative pyelonephritis. The left renal pelvis and proximal ureter contained thick, tan-yellow, malodorous, opaque exudate. BVDV antigen testing in a sample from the right external pinna was negative. Aerobic culture of the left renal pelvis yielded 4+ growth of Proteus mirabilis, 2+ growth of Escherichia coli, and 1+ growth of Enterococcus faecalis, consistent with an ascending pyelonephritis.
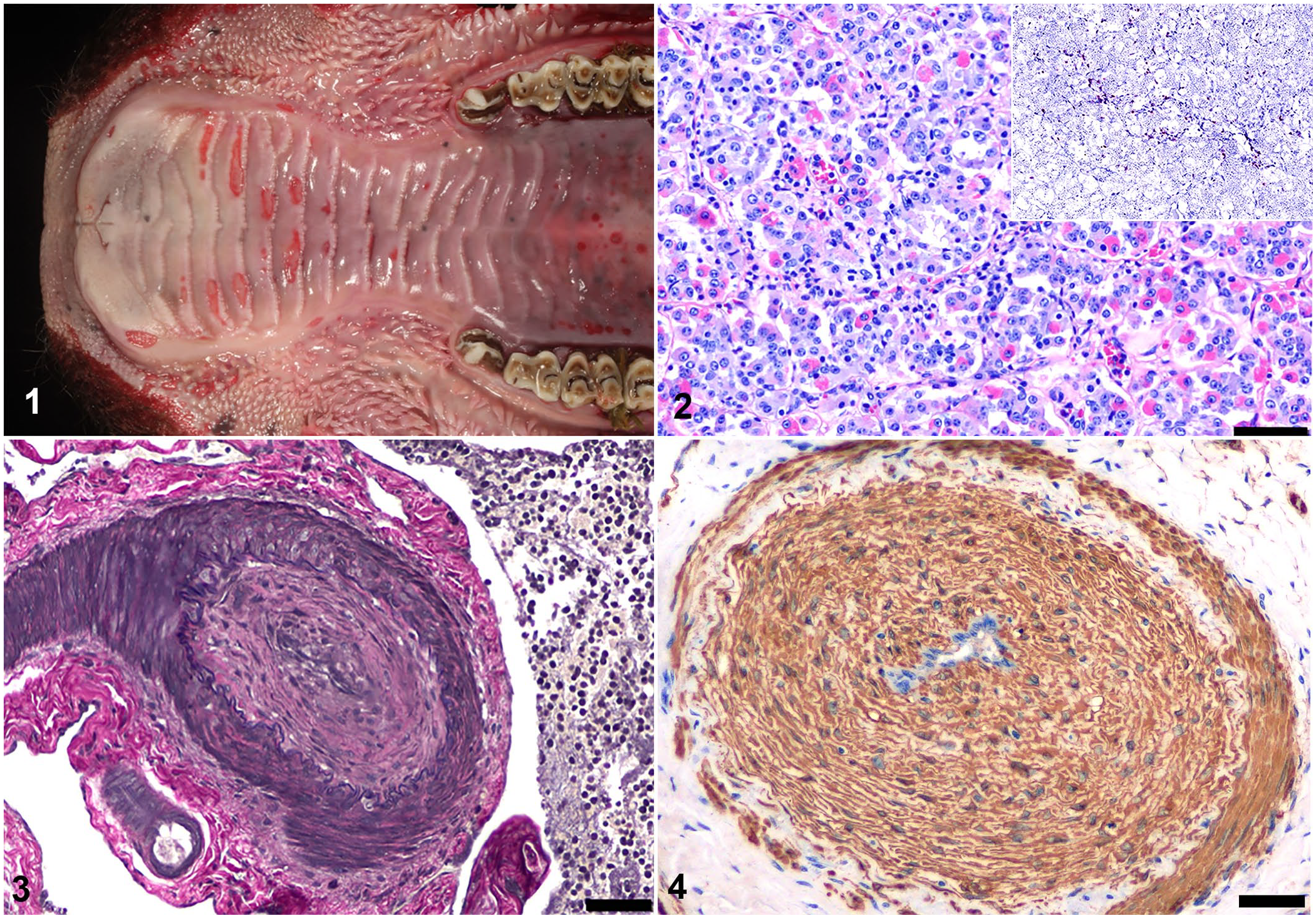
Chronic ovine herpesvirus 2 (OvHV2)-induced malignant catarrhal fever in a cow.
Histologically, small numbers of lymphocytes and rare plasma cells infiltrated the tunica muscularis of arteries of the rostral epidural rete mirabile (RERM, also known as the carotid rete), pituitary gland (Fig. 2), and small arterioles of the cerebrum and kidney. Between the endothelial cell layer and internal elastic lamina of arteries in the RERM and leptomeninges of the brain were moderate numbers of spindle-shaped cells (neointimal proliferation) with frequent narrowing of the lumen (obliterative arteriopathy; Figs. 3, 4). The cytoplasm of the spindle-shaped cells was strongly immunopositive for smooth muscle actin (SMA; Fig. 4). Examination of the kidneys revealed severe necrosuppurative pyelonephritis with gram-positive cocci, tubular necrosis, and mineralization, as well as lymphoplasmacytic vasculitis. Necrohemorrhagic cystitis with fibrin thrombi was observed in the urinary bladder.
OvHV2 was detected by real-time PCR in a retropharyngeal lymph node. Colorimetric (Red assay) in situ hybridization (ISH) to detect OvHV2 viral nucleic acids was performed on formalin-fixed, paraffin-embedded sections of spleen, liver, lymph node, lung, cerebrum, pituitary gland, and RERM, as described previously. 13 Probe hybridization was detected in lymphocytes in the pituitary gland (Fig. 2, inset), lumen of alveolar capillaries, and tunica muscularis of arteries of the RERM, spleen, lymph node, and liver.
The pronounced neointimal hyperplasia in arteries of the RERM and leptomeninges of the brain, the mild nature of the vasculitis, and lack of lymphoproliferation are supportive of chronic MCF-induced vasculitis.9,10,12,15,16 Although the pathogenesis of MCF-induced lesions is incompletely understood, it is presumed to involve infection and transformation of CD4+ and CD8+ T lymphocytes resulting in T-cell dysfunction, abnormal lymphoid proliferation, and targeting of host tissues including arteries and mucosa of the gastrointestinal, respiratory, and urinary tracts.5,12,19 With prolonged MCF-induced clinical disease, and in cases of clinical recovery, variable vascular changes have been reported, including neointimal hyperplasia, neovascularization of the intima and media, and histiocytic and lymphocytic infiltration of the adventitia and intima.9,10,15,16
Neointimal hyperplasia, also referred to as proliferative arteriopathy, has been reported in cattle and bison with prolonged clinical MCF-induced disease and in recovered cases.9,10,12,15,16 Neointimal hyperplasia is thought to occur by either endothelial-to-mesenchymal transition induced by upregulation of transforming growth factor (TGF)-β signaling 7 or differentiation of resident fibroblasts in the adventitia into myofibroblasts in response to TGF-β.4,15 Both mechanisms involve the release of inflammatory cytokines and chemokines from recruited inflammatory cells. The question of what initiates recruitment of these cells remains unclear, although involvement of activated endothelial cells latently infected with OvHV210,15 and perivascular fibroblasts expressing open reading frame 25 major capsid protein of OvHV2 have been considered. 8 However, neointimal hyperplasia should be considered a hallmark lesion for identifying cattle, bison, and buffalo that have recovered from or experienced prolonged OvHV2-induced MCF.
Other vascular lesions identified in cattle and bison chronically infected with OvHV2 include disruption of the internal elastic lamina and segmental attenuation to complete loss of the tunica media. These lesions, in addition to neointimal hyperplasia and obliterative arteriopathy, have been reported in the carotid rete, leptomeninges, arcuate arteries of the kidneys, uvea, uterine broad ligament, and wall of the urinary bladder.9,10,15,16 The hypophysis, a highly vascularized organ, has surprisingly not been evaluated extensively in cows with OvHV2-induced MCF. Hypophysitis in domestic ruminants most commonly develops secondary to systemic or regional diseases, including but not limited to, extension of post-dehorning sinusitis and osteomyelitis, 6 extension of controlled suckling device–induced rhinitis, 2 and bacterial septicemia. 18 The susceptibility of the hypophysis to inflammation and infection in domestic ruminants is attributable to its dense fenestrated capillary network lacking a blood-brain barrier, integration with the extensive portal blood system within the median eminence, and proximity to the RERM. The pars distalis is the most vascularized region of the hypophysis, composed of an interconnected capillary portal system required for circulation of releasing hormones from the hypothalamus that stimulates release of trophic hormones produced by cells of the adenohypophysis. 1 In our case, OvHV2 probe hybridization was most consistent and remarkable in the pars distalis. OvHV2-positive, lymphocytic hypophysitis associated with OvHV2-induced MCF has not been reported previously in a cow, to our knowledge, nor has hypophysitis been evaluated with this ISH technique.
Other histologic findings in cattle and bison chronically infected with OvHV2 include lymphocytic interstitial nephritis, nonsuppurative meningoencephalitis, and lymphocytic panophthalmitis with corneal edema.3,9,10,16,19 In our case, the role of MCF in the progression of the ascending pyelonephritis cannot be confirmed because of the severity of lesions associated with bacterial pyelonephritis.
In our case, whole blood PCR testing for OvHV2 was negative; ISH for OvHV2 on formalin-fixed tissue was positive. Compared to tissues from 4 dairy cows naturally infected with OvHV2 with acute MCF examined with the same ISH technique for OvHV2, 14 our case had mid-to-low levels of hybridization. In our case, clinical signs persisted for ~120 d, likely resulting in a lower viral load and thus lower levels of hybridization, as well as negative OvHV2 PCR.
Given the severe and fatal nature of MCF, chronic OvHV2-induced MCF is rarely encountered by clinicians and pathologists, and neointimal hyperplasia and obliterative arteriopathy are thus observed and recognized infrequently. However, these arteriopathies are considered useful histologic hallmark lesions to identify chronic or recovered cases of MCF. Hypophysitis is not commonly described in cases of MCF yet was a prominent feature in our case; inflammation in tissues such as skin, alimentary tract, nervous system, and lymphoid tissues was underwhelming. Therefore, in suspected cases of MCF in cattle, the area containing the carotid rete and hypophysis should be collected during autopsy and examined carefully for lymphocytic arteritis-periarteritis with necrosis of tunica media or obliterative arteriopathy, and lymphocytic infiltration, respectively, given that both are useful for the diagnosis of acute and chronic MCF. 12
Footnotes
Acknowledgements
We thank the personnel of the diagnostic histopathology laboratory at the Texas A&M University Department of Veterinary Pathobiology for their excellent technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
