Abstract
Systemic necrotizing polyarteritis was diagnosed in three 7–11-mo-old lambs from the same flock. Aneurysmal dilation and rupture of the gastroduodenal artery in 1 lamb resulted in fatal hemorrhage. All lambs had severe necrotizing vasculitis involving the small intestine, abomasum, mesentery, kidney, and heart with concurrent lymphocytic enteritis. Immunohistochemistry for T- and B-lymphocytes and macrophages found a T-cell– and macrophage-dominant transmural vascular infiltrate and T-cell–associated enteritis. PCR analysis for pestivirus failed to identify infection in 1 lamb, and more extensive viral microarray techniques applied to the second and third lamb failed to detect viral nucleic acid. The identification of 3 cases within 1 flock raises the possibility of a common etiology; however, no cause was established. A genetic etiology was not considered likely as not all of the lambs were related. The presence of concurrent T-lymphocyte–associated enteritis raises the possibility of an immune-mediated disease process linking the vasculitis and enteric lesions.
Idiopathic systemic necrotizing vasculitis is reported in a number of different species including pigs, 10 cats, 1 dogs, 2 rats, 9 cattle, 5 sheep,4,6 and humans. 7 In humans, the term polyarteritis nodosa (PAN) is used to describe a necrotizing arteritis of medium or small arteries without glomerulonephritis or vasculitis in arterioles, capillaries, or venules. 8 This term has also been applied to reported cases in the veterinary literature showing a similar pattern of vasculitis. Acute lesions are often typical of immune-complex–induced arteritis; however, as lesions progress, a mononuclear cell–dominated infiltrate is seen. 17 Vascular occlusion and rupture (secondary to micro-aneurysm formation) lead to a wide range of nonspecific clinical signs in humans. The etiology of the condition is unknown, but, in humans, PAN-like lesions have been associated with chronic viral infections such as hepatitis B virus, human immunodeficiency virus, and cytomegalovirus. 7 In rats, it is seen most commonly in naturally occurring or drug-induced hypertension, and is used as a model for investigating the human disease. 9 In sheep, it is a rare and usually sporadic condition. We describe necrotizing polyarteritis in 3 lambs from the same flock, 1 of which died as a result of spontaneous aneurysmal rupture of the gastroduodenal artery.
Three lambs, arbitrarily identified as A, B, and C, from the same flock in northern England were submitted for postmortem examination. The flock comprised 20 breeding Beltex and 12 breeding Texel ewes. All ewes lamb indoors in February and are bred using a mixture of artificial insemination/embryo transfer and natural service. Lamb A was a 10-mo-old Beltex ewe lamb (submitted in January 2015), lamb B was a 7-mo-old Texel ram lamb (submitted in September 2015), and lamb C was an 11-mo-old Beltex ram (submitted January 2016) of the same parentage as lamb A. Lamb A clinically had a “hunched” appearance. Immediately prior to death, the animal had been examined for lameness with physical handling for foot trimming. The history provided for lamb B was of chronic ill thrift and diarrhea. Lamb C also showed ill thrift and diarrhea prior to death. An EDTA blood sample was collected from lamb C during the clinical phase, and a complete blood count was undertaken. Significant findings included thrombocytosis (platelet count 1,028 × 109/L, reference interval [RI]: 250–750 × 109/L), and leukocytosis (white blood cell count 16.3 × 109/L, RI: 5.0–11.0 × 109/L) with absolute neutrophilia (13.0 × 109/L, RI: 0.4–5.0 × 109/L) and left shift (band neutrophils 0.2 × 109/L, RI: 0.0–0.0 × 109/L), and mild monocytosis 0.7 × 109/L (RI: 0.0–0.6 × 109/L). These findings are consistent with the presence of suppurative bronchopneumonia, which was diagnosed following postmortem of lamb C.
On gross postmortem examination, notable features in lamb A included poor body condition, hemoabdomen, and marked periduodenal hemorrhage dissecting the adjacent mesentery and extending from the pyloric region of the abomasum distally for ~20 cm. Serial sectioning of this area identified a 5-cm fusiform aneurysmal dilation of up to 1 cm diameter of the gastroduodenal artery along the mesenteric border (Fig. 1). In addition, the myocardium was mottled tan-white, most apparent in the right ventricular free wall. Multifocal 1 mm diameter pale renal cortical foci were present. No other gross lesions were apparent. Examination of lamb B found poor body condition, moderate subcutaneous edema, moderate generalized lymphadenopathy (the mesenteric lymph nodes being most notable), and diffuse thickening of the small intestinal wall. Lamb C was also in poor body condition, had consolidation of the right middle lobe of the lung, and marked mesenteric lymphadenopathy.
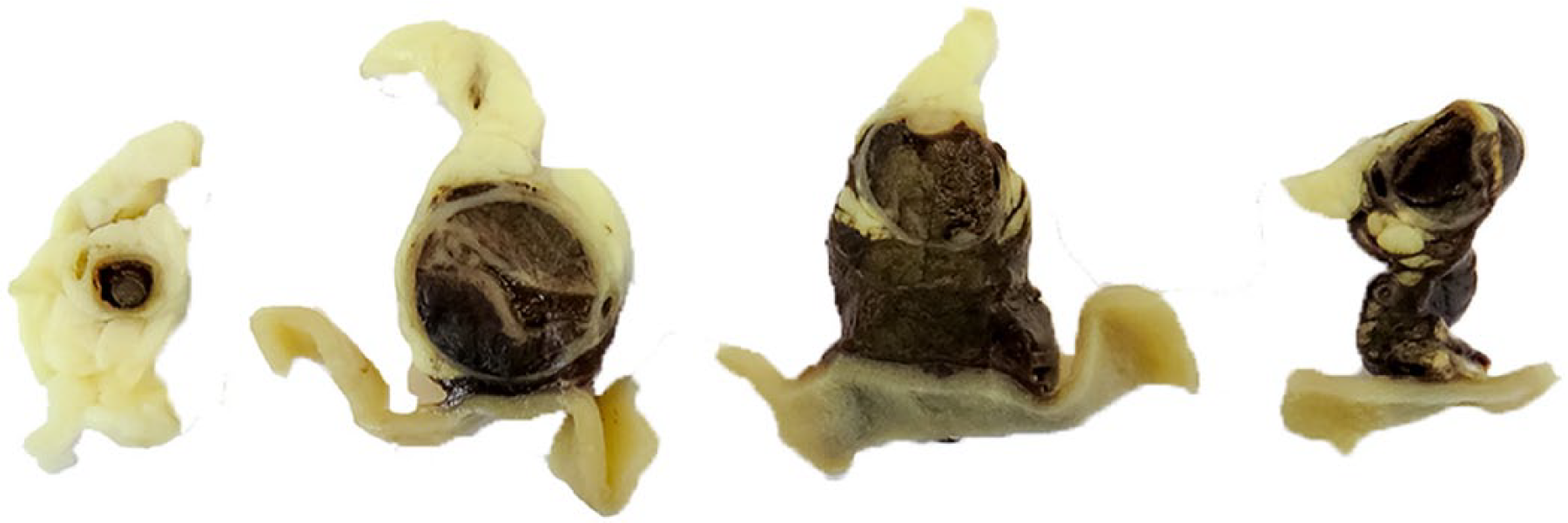
Serial sections (cranial to caudal – left to right) of the gastroduodenal artery of lamb A demonstrating fusiform aneurysm formation and adventitial hemorrhage. Opened duodenum is ventral to the artery.
The following tissues were collected and fixed in 10% neutral-buffered formal saline for histologic examination: the pyloric region of the abomasum and proximal duodenum (lamb A); esophagus (B and C); rumen (C); abomasum (B and C); representative portions of the jejunum, ileum, and large intestine (A–C); mesenteric lymph node (B and C); and liver, kidney, heart (A–C); plus lung, skin, skeletal muscle, brain, and eye (C). Tissues were routinely processed, embedded in paraffin, and 4-µm sections were stained with hematoxylin and eosin. Additional periodic acid-Schiff– and Masson trichrome–stained sections of the mesoduodenal aneurysm in lamb A were also produced. Sections of mesentery including arteries and small intestine of all lambs were immunohistochemically stained with cluster of differentiation (CD)3 (Dako UK, Ely, Cambridgeshire, UK), CD79a (AbD Serotec, Kidlington, Oxfordshire, UK), and lysozyme (Dako; Table 1).
Antibodies and immunohistochemical staining procedures.*
Sources: CD3, lysozyme, REAL EnVision = Dako UK; CD79a = AbD Serotec.
Small- to medium-sized arteries commonly showed 1 or more of the following changes: endothelial activation, segmental-to-circumferential medial fibrinoid change and necrosis, and a moderate-to-marked lymphocyte and macrophage transmural infiltrate (Fig. 2). Organs affected included the small intestine (lambs A–C), abomasum (B), mesentery (A–C), kidney (A and B), and heart (A and C). Within the mesoduodenum of lamb A, the gastroduodenal artery had marked ectasia with multifocal mural and occlusive thrombosis and vasculitis as described previously. Additional features included disruption of the internal elastic lamina and marked expansion of the surrounding mesentery by dissecting hemorrhage. The pale foci in the heart and kidney corresponded to microscopic vascular changes in these organs.
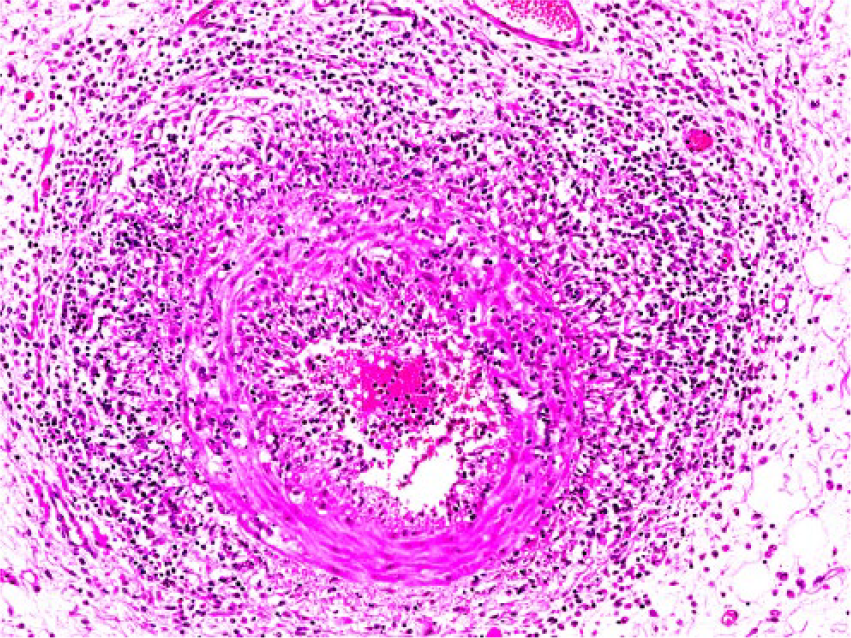
Circumferential vasculitis with marked lymphohis-tiocytic transmural infiltrate in a mesenteric artery in lamb B. H&E. 100×.
Small intestinal sections from all lambs had segmental (lambs A and C) and diffuse (B) moderate-to-marked, lymphocytic lamina proprial infiltrate leading to crypt separation, and villus widening with atrophy. Small numbers of crypt abscesses were present. Small numbers of submucosal and serosal blood vessels had segmental-to-circumferential vasculitis as described previously (Fig. 3). Moderate suppurative bronchopneumonia was identified in lamb C.
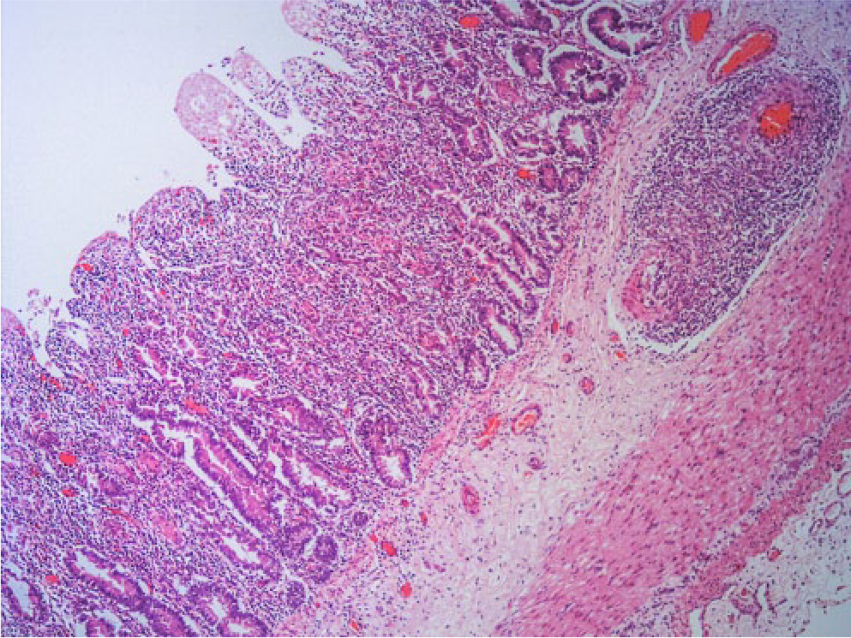
Lymphocytic enteritis and submucosal vasculitis (loss of villus enterocytes was the result of autolysis) in the small intestine of lamb A. H&E. 40×.
Large numbers of T-lymphocytes (CD3+) and macrophages (lysozyme+) were present within the tunica media and adventitia of affected arteries. Only rare B-lymphocytes (CD79a+) were seen in the tunica adventitia. Within the small intestine, there were large numbers of T-lymphocytes expanding the lamina propria and small-to-moderate numbers present within crypt epithelium. Small numbers of B-lymphocytes were admixed with the dominant T-lymphocyte lamina propria infiltrate.
EDTA blood, feces, spleen, and mesenteric lymph node tissues from lamb B were analyzed using an in-house–developed, pan-viral microarray, 3 which contains ~47,000 probes to ~2,500 virus species (Agilent Technologies, Stockport, UK). The array was 10–100 times less sensitive than corresponding virus-specific PCRs when tested using samples positive for influenza viruses and parvoviruses (unpublished data). In brief, total nucleic acid from the samples was extracted and amplified in a random PCR. The amplified target was fluorescently labeled and hybridized to probes on the pan-viral microarray chip before being statistically analyzed. To verify the microarray results, the potentially positive signals were tested with virus-specific PCR and, in this case, with the pestivirus-specific, short-target reverse transcription (RT)-PCR. 10 Subsequently, fresh kidney tissue from lamb A was also tested for the presence of pestivirus nucleic acid using a nested RT-PCR. This multiplex assay uses 3 probes, a bovine viral diarrhea virus 1 and 2 (BVDV-1, -2) probe, and 2 other probes specific for BVDV-2 and border disease virus (BDV), respectively. 13 Whole genome sequencing was undertaken on the total nucleic acid extracted from splenic tissue of lamb C using the Nextera XT extraction kit (Illumina, San Diego, CA) to fragment and subsequently amplify the nucleic acid primers targeted to adaptor sequences linked to the nucleic acid. Paired end sequencing of this tagged and amplified nucleic acid was performed (MiSeq, Illumina, San Diego, CA). Host genome sequence reads were removed through mapping to the ovine genome, and the consensus contigs were obtained by de novo assembly of the non-host sequence reads using Velvet 1.2.10 software. 19
No BVDV or BDV viral nucleic acid was detected using PCR on kidney tissue from lamb A. No specific viral nucleic acid was detected using microarray on EDTA blood, feces, spleen, and mesenteric lymph node tissue from lamb B. However, spleen and mesenteric lymph node produced very low averages of signal intensities for a pestivirus, directing towards potential presence of a virus related to ruminant pestiviruses. This microarray finding could not be confirmed by the pestivirus-specific, short-target RT-PCR. No parasitic or virus-like sequences were detected following whole genome sequence analysis of the splenic sample from lamb C; however, bacterial sequence reads from Psychrobacter spp. were present.
The clinical and pathologic features of these cases are similar to those reported previously, 4 except for the fatal hemorrhage associated with polyarteritis. Clinically, this includes ill thrift and weight loss, and lameness (in lamb A only). The vasculitis was almost identical to previous reports, and aneurysm formation was also a feature of those reported cases. 4 However, the authors did not report evidence of arterial rupture and hemorrhage in any of the affected blood vessels and organs. Hemorrhage has been recorded in other species involving the mesenteric arteries in the rat, 9 and the leptomeninges of pigs 11 and dogs, the latter associated with juvenile polyarteritis syndrome leading to progressive neurologic signs. 2 In humans, non-fatal retroperitoneal aneurysm formation and rupture involving the gastroduodenal artery has been reported. 15 The weakened arterial wall and aneurysm in lamb A may have ruptured following manual handling for foot trimming and led to fatal hemorrhage.
Aneurysm formation is secondary to a combination of the acquired weakness of the arterial wall through medial necrosis and loss of integrity of the internal elastic lamina, and the hemodynamic effects of blood flow. The combination of vasculitis and turbulent blood flow associated with aneurysm formation predisposes to thrombosis, and it is these changes that are thought to account for a wide range of nonspecific clinical signs in humans. 7 The clinical appearance of lamb A suggested pain of nonspecific origin that may have been associated with aneurysm formation and thrombosis in the gastroduodenal artery or possibly as a result of vascular compromise leading to myopathy as has been described previously in humans 12 and sheep 4 but could not be confirmed in this case.
All other reports have failed to determine a cause for systemic necrotizing polyarteritis in sheep. In light of the viral-associated causes seen in humans, testing for BDV was undertaken in lamb A and more extensive microarray testing for viral nucleic acid (including whole genomic sequence analysis in one case) was performed on samples from lambs B and C. Results from these tests proved negative; however, the lack of detection of viral agents could be due to virus elimination as part of an immune-mediated response. It may, however, also reflect a non-viral cause for the lesions. Sequences compatible with Psychrobacter spp. were identified in the spleen of lamb C. This genus is found in a wide range of environments (often moist, saline, and cold) and rarely causes disease in humans and animals, with only a single report describing clinical disease in sheep. 18 It is unlikely this organism is implicated in the pathogenesis of the immune-mediated vascular lesions and may represent a contaminant.
A series of systemic vasculitis cases involving 6 of 7 sheep in a previous study has been reported. 16 All of these had ill thrift, mesenteric lymphadenopathy, and corrugated mucosal thickening of the small intestine with ulceration and erosion characterized by lymphocytic enteritis with vasculitis. This clinical and pathologic presentation compares favorably to that of all the lambs described in our report, although the enteritis may represent nonspecific change associated with an undefined cause. The pattern of change does raise the possibility of a common etiology and/or pathogenesis. The possibility of an immune-mediated disease process linking lymphocytic enteritis and polyarteritis should be considered, particularly because a significant T-lymphocyte enteritis was identified in all of the lambs reported herein. Rare examples of intestinal material from sheep with a similar lymphocytic enteritis and enteric vasculitis presenting with ill thrift and diarrhea, for which no cause was established, have been examined by one of the current authors (M Wessels).
Our report, and the report mentioned previously, 16 describe intestinal lesions; however, this likely reflects the limited number of tissues available to pathologists, as the nonspecific clinical presentation in sheep is a handicap for full investigation of the disease. Diagnosis is achieved via histopathology following postmortem examination, and the description in many reports is based upon a limited number of organs harvested at the time of autopsy and that were available for examination. The standard collection of a wide range of tissues at autopsy would partly overcome this problem. Suitable material for ancillary testing such as virology should also be collected, as it would also assist in further characterization of the disease and in establishing a cause. We were fortunate in obtaining blood for hematology from lamb C prior to death. Leukocytosis with absolute neutrophilia and left shift and monocytosis are seen in various conditions in other domestic species; however, thrombocytopenia, as a result of thrombotic consumption, is normally a feature that contrasts with the thrombocytosis in this lamb.1,2
In humans, recessive loss-of-function mutation of adenosine deaminase 2 has been established as a cause of PAN. 14 However, a genetic cause in our cases is unlikely as although 2 of the lambs were siblings, albeit born in different years, the remaining lamb was of a different breed. The finding of 3 cases within 1 flock does raise the possibility of a common etiology, which could not be established in our cases. In-depth multidisciplinary investigation of future cases is advised to help identify causes for this condition.
Footnotes
Acknowledgements
We thank the staff at Finn Pathologists, APHA Penrith, Lasswade, and Weybridge. We also thank Amanda Carson, APHA, for her input to the disease investigation. Richard Ellis provided assistance with the genomic analysis for lamb C. Photographic assistance by Richard Fox is acknowledged. 2017 Crown Copyright.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding from AHDB Beef and Lamb (previously EBLEX) for the fallen stock necropsy project, under which these carcasses were identified, is acknowledged. Assistance with funding for micro-array testing from the Defra scanning surveillance budget is acknowledged.
