Abstract
Rabbit hemorrhagic disease virus 2/genotype GI.2 (RHDV2/GI.2;
Keywords
The order Lagomorpha
The order
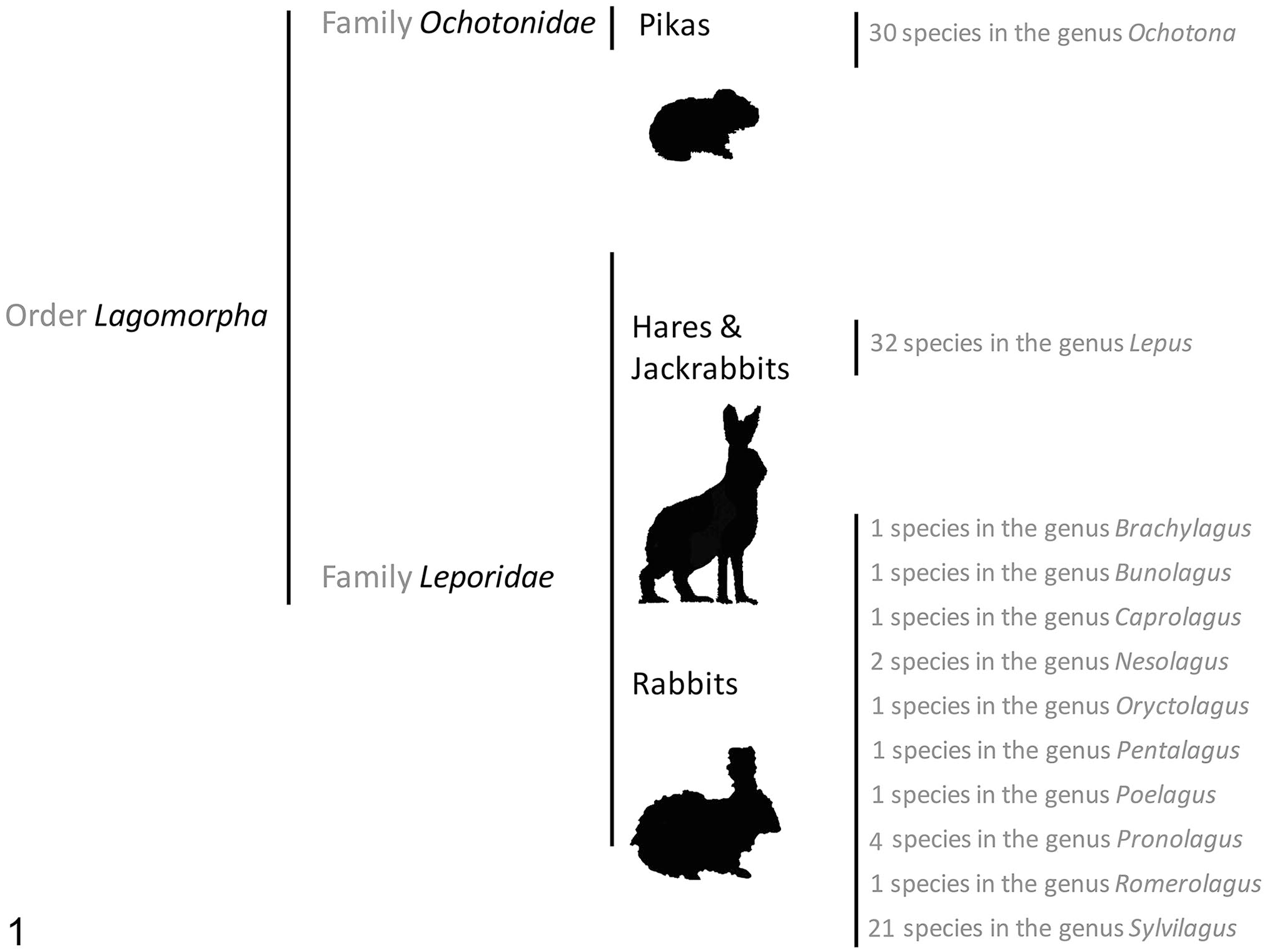
The order
Leporids play a crucial role in many ecosystems. They are primary consumers that convert plant protein into animal protein of different weight ranges depending on the species, providing an ideal biomass intake for predators of various sizes, including genets, wild felids and canids, and eagles.56,161,167,173 The decline of native
Overview of lagoviruses
The genus
In 2017, several authors proposed a new classification based on phylogeny (Fig. 2), specifically by aligning
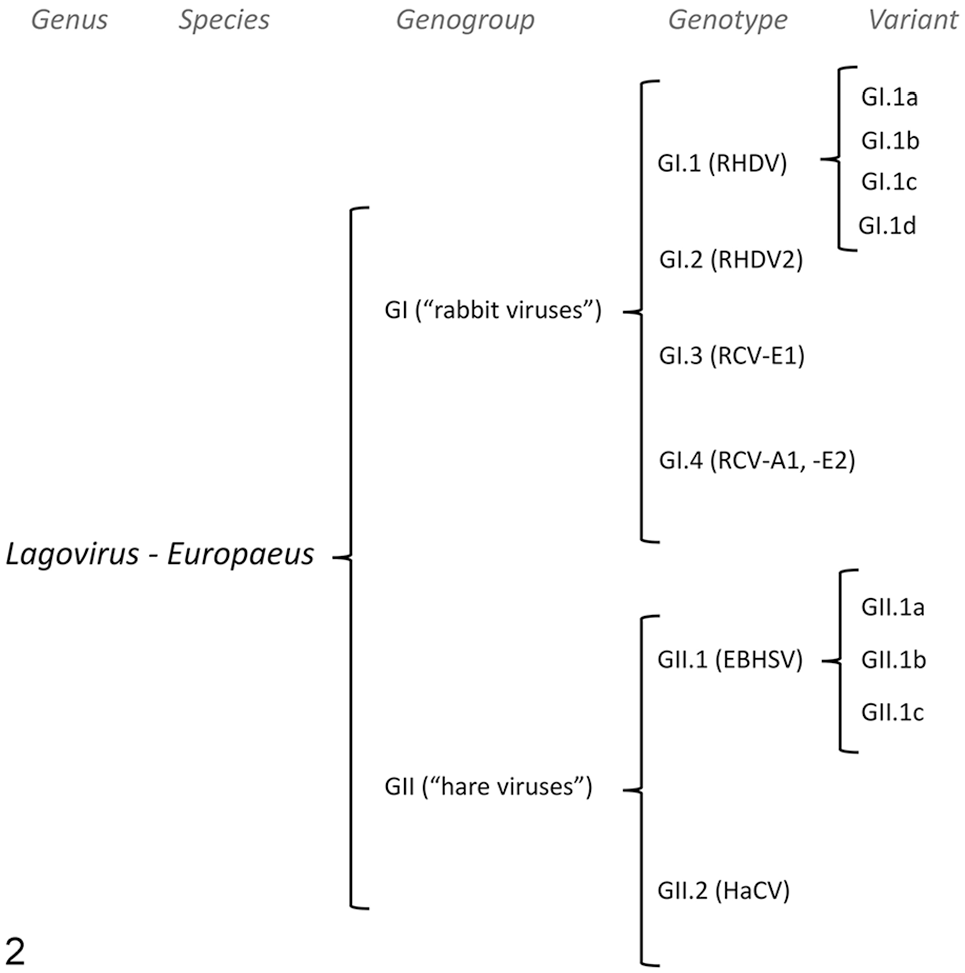
Organization of the
Lagoviruses are single-stranded, positive-sense RNA viruses with an icosahedral capsid, a spherical shape, and no envelope; the viral capsid is composed of 90 arch-like dimers of the capsid protein VP60 and has 32 cup-shaped depressions reminiscent of a calyx, which gives the viral family the name
As with other members of the
Typical clinical signs of RHD are the result of severe hepatic failure and dysregulation of the coagulation system. The main differences between RHDV and RHDV2 are that the latter has a much wider host range within the
Objectives and methodology
In this literature review, we gathered data on RHDV2 detections reported worldwide, focusing on reports in different lagomorph species, pathology and genetic differences, and diagnostic advances made in the context of the global spread of this virus in each geographic location. We searched the scientific literature (PubMed, Google Scholar, Web of Science, and Scopus) and the websites of official international, national, and regional animal health regulatory agencies and authorities (e.g., WOAH, U.S. Department of Agriculture–Animal and Plant Health Inspection Service [USDA-APHIS], or as indicated below).
Global detections
By October 2023, RHDV2 had been detected almost globally (Fig. 3). It is actively spreading to new territories and readers are encouraged to check the official reporting agencies’ websites for the latest information on reports of detection of this virus (e.g., https://wahis.woah.org/#/event-management).
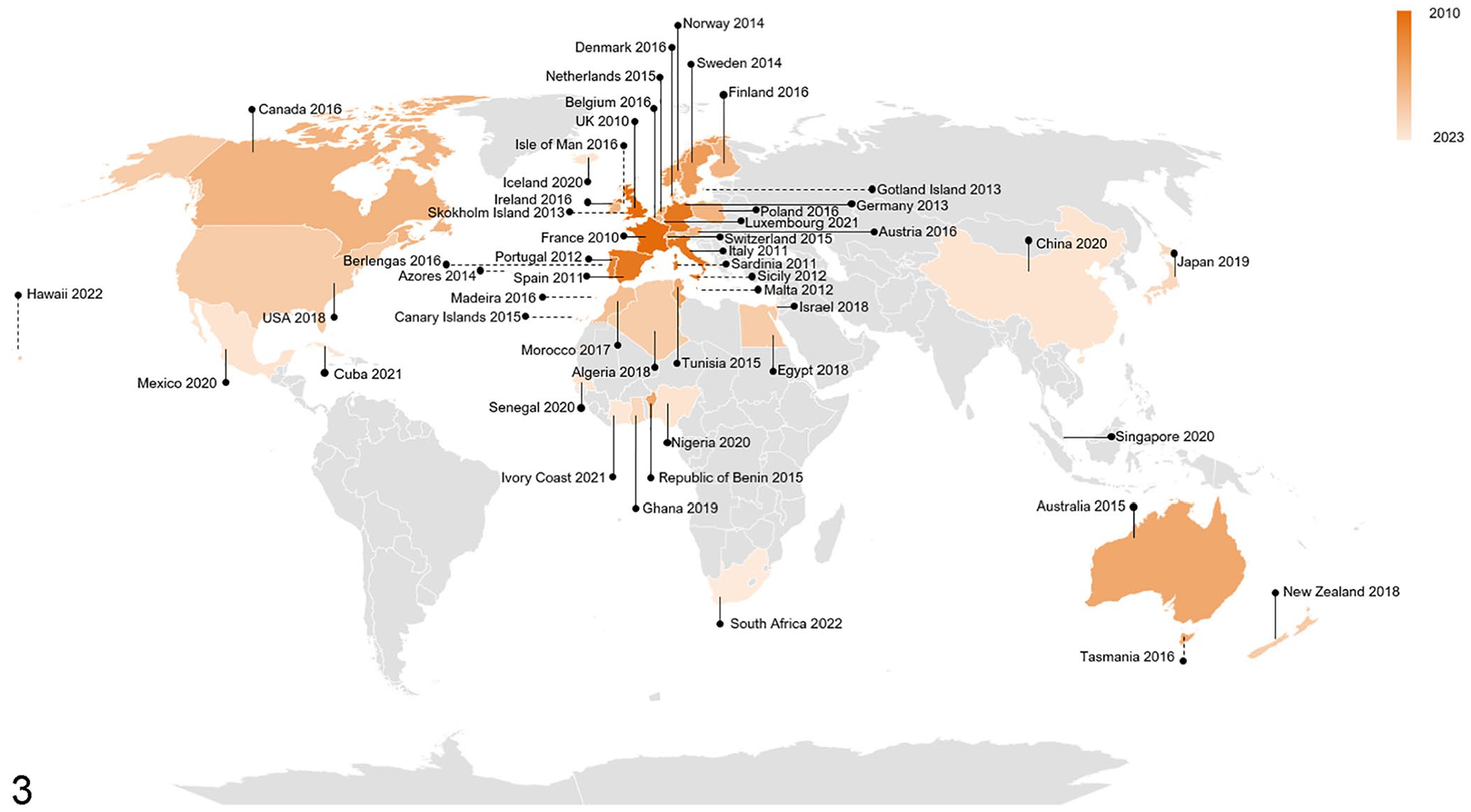
World map of rabbit hemorrhagic disease virus 2 detections reported between 2010 and 2023 in different countries (solid lines) and islands (dashed lines). Countries and islands with reported detections are in orange; earlier detections are darker, and the latest detections are lighter. Countries with no reported detections are in gray. References: Algeria, 152 Australia, 76 Austria, 19 Azores, 60 Belgium, 84 Benin, 176 Berlengas, 3 Canada, 13 Canary Islands, 121 China, 179 Cuba, 181 Denmark, 182 Egypt, 67 Finland, 92 France, 105 Germany, 70 Ghana, 12 Gotland, 130 Hawaii, 157 Iceland, 183 Ireland, 149 Isle of Man, 149 Israel, 184 Italy, 186 Ivory Coast (RHDV2 was most likely present at least since 2016 187 ), 188 Japan, 96 Luxembourg (RHDV2 was most likely present at least since 2015–2017 120 ), 189 Madeira, 43 Malta, 169 Mexico, 190 Morocco, 114 Netherlands, 63 New Zealand (RHDV2 was most likely present at least since 2017 124 ), 191 Nigeria, 55 Norway, 192 Poland, 69 Portugal, 7 Sardinia, 142 Senegal, 193 Sicily, 34 Singapore, 163 Skokholm, 156 South Africa, 195 Spain, 51 Sweden, 130 Switzerland, 9 Tasmania, 116 Tunisia, 144 United Kingdom, 171 United States. 196 Map developed using Bing technology and adapted.
Europe
The first detections of RHDV2 occurred almost simultaneously in different parts of Europe at the beginning of the first decade of the 2000s, and the virus is thought to have emerged on that continent. The first reports of confirmed RHDV2 detections were from northwestern and central France in April–May 2010, with rapid spread in domestic and wild European rabbit populations.103,105 At the same time as RHDV2 was being detected in several countries in continental Europe, the authors of a 2015 study reported that the virus was also present in 2 locations in England since at least May 2010, based on detections in archived samples; these represented the first confirmed detections in the entire United Kingdom. 171 Given the geographic distance between the detections and the low level of RHD surveillance in England at the time, the authors suggested that RHDV2 may have been circulating in Great Britain before that year. 171 The virus continued to circulate in Great Britain and was also detected in Scotland and Wales in following years.20,171
RHDV2 was first detected in domestic and wild rabbits in the northeast of Spain in May 2011.33,51 However, mortality events associated with a disease compatible with RHD (although not confirmed by laboratory testing) were observed earlier that year in farmed rabbits vaccinated against RHDV in eastern Spain (regions of Catalonia and Valencia), suggesting that the incursion of RHDV2 into Spain may have occurred earlier. 73 Almost simultaneously, in July 2011, RHDV2 was also detected in a rabbitry in northeastern Italy, and cases were also observed that year on the Italian island of Sardinia.103,142,186 Later, in 2012, reports of RHDV2 emerged on the Italian island of Sicily 34 and possibly on Malta (A. Lavazza and L. Capucci, unpublished results mentioned in a 2017 study 169 ).
RHDV2 was detected in wild rabbits from the north and south of mainland Portugal between November 2012 and February 2013, 7 in wild rabbits from the Swedish island of Gotland in May 2013, 130 and in domestic rabbits in Germany in 2013. 70 Cases were detected in the uninhabited island of Skokholm, near the Welsh Coast, in 2013.156,171 Further analyses of wild rabbits revealed the circulation of RHDV2 in the Portuguese Azores islands in 2014, probably due to a previous virus introduction from southern Portugal.59,60 RHDV2 was detected in domestic rabbits in Norway in 2014. 192
In 2015, RHDV2 was detected in domestic rabbits in Switzerland and the Netherlands.9,63 In 2016, detections were reported in domestic rabbits in Austria, 19 Belgium, 84 and Denmark, 182 and in domestic, wild, and/or feral domestic rabbits in Finland, 92 the Republic of Ireland, 149 Poland, 69 the Isle of Man, 149 and the Portuguese archipelagos of Berlengas and Madeira.3,43 Phylogenetic analyses of the Madeira viruses again suggested a possible introduction from the south of mainland Portugal. Detections of RHDV2 were first reported to WOAH in domestic rabbits in Iceland and Luxembourg in 2020 and 2021,183,189 respectively; however, a 2018 study mentioned detections of RHDV2 in samples of domestic rabbits from Luxembourg collected between 2015 and 2017. 120 RHDV2 was most likely detected in Croatia in 2023 180 ; the latter country has not reported the virus type yet, but per the mentioned WOAH report, the detection was in a European brown hare, suggesting that it was RHDV2 rather than RHDV.
The broader host range of RHDV2 was first recognized in Europe when the virus was detected in association with disease in hares from several countries (see also section on
In countries in which RHDV2 was detected, there was rapid viral spread, and this new virus quickly replaced the classic RHDV strains that had been circulating. The spread of RHDV2 and the replacement of RHDV strains was faster in areas in which wild rabbit populations were abundant and the competitive advantages of RHDV2 over RHDV were enhanced,33,103 so that RHDV strains were almost completely replaced in <2 y after emergence in countries such as France, 103 Spain, 50 and Portugal. 112 However, in other countries, such as Germany and Poland, RHDV continued to co-circulate with RHDV2 for longer times.87,160
Shortly after its emergence, recombinant RHDV2 strains were detected in the Iberian Peninsula, 113 France, 104 and the Portuguese archipelagos of Azores and Madeira,10,111 infecting rabbits and also hares.104,168 Initially, most of the recombination events identified involved the junction between the genes encoding for VP60 (structural proteins) and RdRp (nonstructural proteins); RHDV2 (genotype GI.2) was the donor of the structural proteins, whereas the nonstructural proteins originated from either a pathogenic RHDV (genotype GI.1) or a nonpathogenic RCV (genotypes GI.3 or GI.4). RHDV2 recombinants encoding the nonstructural genes of EBHSV (genotype GII.1) were also reported to infect hares in Germany. 160 Furthermore, a second recombination breakpoint was detected in Portugal at the junction of the 2 genes encoding the nonstructural proteins p16 and p23, allowing the existence of triple recombinants in which RHDV2 was the donor of the structural part, a nonpathogenic lagovirus was the parental donor of p16, and RHDV genotype GI.1b was the donor of the rest of the nonstructural part. 155 The latter illustrates the high potential of lagoviruses to increase genetic diversity, leading to the emergence of new pathogenic strains. There is strong evidence that recombination contributed to the emergence of RHDV2 as a pathogenic virus in Europe, as phylogenetic and recombination analyses have shown that the first RHDV2 strains, previously considered as non-recombinants, arose from a recombination event between the GI.3 and GI.2 genotypes. 6
Detection methods evolved rapidly in response to the emergence of RHDV2 in Europe. Although it was initially reported that the virus did not agglutinate human RBCs of type O or A,
51
further studies demonstrated that RHDV2 efficiently agglutinated RBCs of type O at titers similar to those seen with most classic RHDVs, and hemagglutination (HA) was therefore often used to screen for RHDV2.
103
However, because HA cannot be used to differentiate between different lagoviruses, after the emergence of RHDV2, viral detection was mainly performed by amplification and sequencing of the partial or complete gene encoding VP60 (e.g.,50,103,105,130). Subsequently, given the different antigenic profile of RHDV2, differentiation from RHDV could also be performed using anti-RHDV monoclonal antibodies and a sandwich ELISA.103,142 Nevertheless, procedures based on RT-qPCR targeting a specific nucleotide RNA region located within the
Despite the undoubted usefulness of PCR-based methods, other less-sensitive detection tests have been developed in Europe, such as a sandwich ELISA for the detection of RHDV2 antigen in rabbit liver homogenates, 53 which allowed the detection of the virus with high specificity in laboratories without PCR equipment, or a qualitative immunochromatographic test developed to be used on-site, which allowed rapid and specific detection in the field (e.g., on farms). 52 Finally, serologic analyses to detect antibodies against RHDV2 were performed with a competitive ELISA already developed for RHDV strains and later used in epidemiologic surveys of RHDV2.28,103,169
Africa
Detections of RHDV2 were first reported in 2015 in 3 locations in Africa and close-by islands. The virus was detected in domestic rabbits in Tunisia; in domestic and wild European rabbits in Tenerife, an island of the Spanish Canary Islands archipelago; and in domestic rabbits in the Republic of Benin.22,121,144,176 In Tunisia and the Canary Islands, the virus was identified during routine surveillance of annual RHD outbreaks, and the specific date of introduction was unknown. Phylogenetic analysis showed that the Tunisian viruses originated from a single introduction of European strains, most consistent with strains from Italy, which was suspected to have occurred between 2012 and 2014, 144 whereas viruses from Tenerife had the highest nucleotide identity with Portuguese strains. 121
Subsequently, reports from Ivory Coast suggested that RHDV2 was present in that country in 2016, because both rabbits and hares were affected, but the virus type was not reported to be RHDV2 until 2021.114,187,188 In 2017, RHDV2 was first detected in Morocco in samples obtained from wild rabbits, and the strains identified were GI.1bP-GI.2 recombinants, as found in the Iberian Peninsula and in the Azores and Madeira archipelagos
114
; RHDV2 circulated widely in Morocco, at least up until 2021.
153
Subsequently, in 2018, the virus was detected in Egypt during regular surveillance for RHD outbreaks, so the date of the first introduction of the virus in that country is also unknown.
67
An August 2023 study reported that RHDV2 was circulating in the North of Algeria since at least 2018
152
; the viruses were most similar to the GI.3P-GI.2 recombinant and the earlier detections clustered with Tunisian viruses, whereas the most recent (2020–2021) samples included in the analysis were more similar to some North American viruses.
152
RHDV2 was first reported in commercial rabbit farms in Ghana in 2019,
12
and in domestic rabbits in Senegal and Nigeria in 2020.55,193 Phylogenetic analysis of
Oceania
Detections of RHDV2 in Oceania have been reported in Australia and New Zealand.175,191 The history of pathogenic lagoviruses in Australia is unique. Prior to the emergence of RHDV2, RHDV had circulated widely, mainly as a result of its use to control populations of invasive wild European rabbits. The Czech V351 (CAPM V-351) RHDV strain (a GI.1c virus) was released for this purpose in 1995 and was considered to be an ancestor of some circulating pathogenic lagoviruses.65,100,116 In 2017, the Korean K5 strain (a GI.1a virus) was also released as a biocontrol agent. 116 An incursion with active circulation of a different RHDV genotype GI.1a virus occurred in 2014. 118 Additionally, several variants of the nonpathogenic RCV (GI.4) were present in Australia prior to the release of RHDV.147,159
RHDV2 was first detected in a wild European rabbit in Australia in May 2015.76,175 Three dead wild rabbits were tested as part of surveillance of RHD outbreaks prior to the release of the K5/GI.1a in the Canberra region. While field RHDV was found in 2 carcasses, 1 animal tested positive for RHDV2. Subsequent phylogenetic analysis revealed that the virus was a GI.1bP-GI.2 recombinant.76,116 This virus was similar to some viruses previously circulating in mainland Portugal and the Azores.10,113 RHDV2 was also detected in Tasmania in 2016. 116 Retrospective serologic studies showed that RHDV2 had been circulating in Australia before its first detection, at least since spring 2014, 146 which was also supported by studies on the evolutionary history of Australian viruses and further serologic studies.116,138,158 RHDV2 had a significant impact on the wild rabbit populations between 2014 and 2018, with average population declines of up to 60% and more pronounced reductions in southern and western Australia. 146 Interestingly, only 18 months after its first confirmed detection, RHDV2 had replaced the circulating endemic RHDV viruses in Australia, and was also detected in domestic rabbits and European brown hares.78,116 Cases of RHDV2 infection continue to be detected in Australia; seroprevalence remained high until at least 2022, and it is thought to be the virus currently having the greatest impact on wild populations. 145 However, a low percentage of RHDV-seropositive animals are still being detected, suggesting co-circulation to some extent and ruling out total extinction of RHDV.145,146 In fact, the previous occurrence of multiple pathogenic and nonpathogenic lagoviruses in Australia has shaped the dynamics of RHDV2, fostering the occurrence of multiple recombinants, 117 and at least 7 different lagoviruses are believed to be circulating: RHDV genotype GI.1, RCV genotype GI.4, RHDV2 genotype GI.1bP-GI.2, RHDV-K5 genotype GI.1a, RHDV genotype GI.4eP-GI.1a, RHDV2 genotype GI.4cP-GI.2, and RHDV2 genotype GI.4eP-GI.2. 138
The use of different ELISA strategies has therefore been fundamental in shaping the Australian lagovirus landscape even before the emergence of RHDV2.145,147 However, various PCR approaches are the current method of choice and the test by which the occurrence of RHDV2 was initially confirmed in Australia. 76 A multiplex RT-PCR that was able to differentiate among all of the pathogenic lagoviruses circulating in Australia at the time was developed. 77 The assay was even able to identify mixed infections in individual animals. In addition, a SYBR green–based RT-qPCR was optimized to detect and quantify lagovirus genetic material in a single PCR reaction. 77 Monolayer cell cultures derived from European rabbit and European brown hare hepatobiliary organoids support the replication of several Australian RHDV2 (both rabbit and hare organoids) and RHDV (rabbit organoids only) 95 ; this provides a useful alternative to traditional viral culture systems, which have proven very challenging for lagoviruses.
The situation in New Zealand was somewhat similar to that in Australia. Prior to the RHDV2 incursion, the biocontrol agent CAPM V-351 RHDV was detected in 1997, possibly as a result of an illegal introduction by farmers, 135 and further releases were made in the following years. The K5 RHDV strain was also released in 2018 for a similar purpose. The nonpathogenic RCV (GI.4) was also present in New Zealand before 1997. 131 RHDV2 was first detected in a wild rabbit collected from the South Island in May 2018, but analysis of samples from rabbits collected the previous year were also positive.124,191
Asia
The first reports of RHDV2 occurrence in Asia came from Israel in 2018. 184 The same country reported cases to WOAH in 2020 and 2021. 185 The only publicly available reports from Israel referred to detections in domestic rabbits. Reports of RHDV2 occurrence in East Asia appeared in 2019–2020, including detections in Japan and China.96,179 Maritime Southeast Asia has also been affected, with detections in Singapore in 2020. 194
Since 2019, RHDV2 has been detected in several prefectures in Japan.71,96,162,165 In May 2019, all 10 rabbits in an exhibition facility from Ehime Prefecture died with gross and microscopic lesions compatible with RHD; viral particles were visualized by electron microscopy and the diagnosis was confirmed by RT-PCR followed by partial
In April–May 2020, large outbreaks of RHD due to RHDV2 were reported in farmed rabbits in Sichuan Province, China and later spread to other provinces.44,89,108 These were the first reports of RHDV2 detection in the country. Mortality was high, ranging from ~43% to >70%, and affected also young individuals and rabbits that had been vaccinated against RHDV. The Sichuan viruses were most closely related to detections from the Netherlands in 2016 and Germany in 2017, and were GI.1aP-GI.2 recombinants.44,108,143 Based on the phylogenetic similarities with these European strains and the reliance of the Chinese rabbit industry on imports from European countries, it was suggested that some of these introductions into China may have originated from rabbit batches or imported semen purchased in Europe.44,108,143 A 2023 study reported that a novel recombinant, distinct from the original Chinese strain, was detected between 2020 and 2021 in at least 3 different provinces (Sichuan, Jiangsu, Guangxi) separated by 1,000–1,500 km. 88 The novel recombinant had structural genes of the RHDV2 strain first detected in China and a nonstructural component of an unclassified lagovirus genotype similar to a virus that had been identified in healthy wild rabbits in the country.81,88 This recombinant induced lower mortality and had a longer replication time than the RHDV2 genotype GI.1aP-GI.2 detected in some of the first Sichuan cases. 88
In September 2020, Singapore reported their first documented cases of RHDV2. 163 All cases occurred in domestic pet rabbits and involved at least 3 households from an urban area; 8 of 11 exposed rabbits died. Rabbits from the different households had had some degree of contact with each other, and intra- and inter-household transmission, including contact at a veterinary clinic, were highlighted as important modes of dissemination in this outbreak.109,163 The source of the outbreak could not be determined, and introduction via infected rabbits or contaminated feed was initially considered unlikely. 109 No further cases were reported that year, and the outbreak was declared resolved in December 2020. 109 Subsequent WGS and recombination and phylogenetic analyses revealed that the Singapore 2020 virus was a GI.4cP-GI.2 recombinant highly homologous (~99.2%) to certain Australian viruses circulating in rabbit and hare populations since 201799,117; therefore, it was suggested that the origin of the Singapore 2020 cases might have been an introduction of a virus that originated in Australia, but the means by which this occurred were unknown. 99
The Americas
As of September 2023, RHDV2 detections in the Americas have been limited to North America, specifically to Canada, 177 the United States, 196 Mexico, 190 and Cuba. 181 Interestingly, no detections have been reported from South America. This region remains as one of the largest officially unaffected areas in the world, and continued surveillance is warranted in the current context of rapid viral spread.
RHDV2 was first detected in North America in August 2016 in a 4-mo-old rabbit from a hobby farm in Mont-Joli, Quebec, Canada; the farm had received rabbits from another farm that was also presumably affected by RHD.13,177 Mortality in the affected premises was 72–89%, and several surviving rabbits were seropositive by ELISA. Sequences from this strain of RHDV2 were most similar to a virus collected in 2011 in Navarra, Spain. 13 Following this outbreak, RHDV2 was not detected again in Canada until 2018. 178 In December 2017, a significant mortality event was reported in feral rabbits from a private property south of Nanaimo, British Columbia but was not further investigated; high mortality was then reported in a population of feral domestic rabbits from the Vancouver Island University campus in early 2018. 13 Additional cases in domestic rabbits were reported in mainland British Columbia through the summer of 2018.13,85 The strain of RHDV2 detected in British Columbia in 2018 shares only ~93% sequence homology with the strain detected in Quebec in 2016 and is thought to represent a separate incursion. 13 In 2019, additional detections were reported in the region; of note, there was one case involving pet rabbits in a downtown Vancouver apartment that shares only ~97% identity at the nucleotide level with the 2018 British Columbia index case and is thought to represent a separate incursion into the region. 13 Between 2020–2023, RHDV2 was detected several times in feral and captive domestic rabbits in other Canadian provinces in addition to British Columbia and Quebec, including Ontario and Alberta. 72
RHDV2 was first detected in the United States in 2018, affecting pet rabbits in Ohio, 196 and, shortly thereafter, in feral domestic rabbits in Washington State in 2019. 172 The sequences of the viruses from Ohio and Washington State were both similar to a strain detected in British Columbia in 2018. 166 In March 2020, RHDV2 was reported in domestic rabbits from a veterinary clinic in New York City, likely representing an introduction different from the Ohio and Washington State cases.134,166
In March 2020, RHDV2 was detected in domestic rabbits in New Mexico.
197
Since then, the virus has spread extensively throughout the southwestern United States and later to other areas of the country; RHDV2 is now considered to be endemic in most of the western United States, with sporadic detections reported in some eastern states.
197
The outbreak in the southwestern United States represents a new milestone in the global spread of RHDV2 as it is the first time that natural cases of RHD were reported in
The first public complete RHDV2 sequences from the United States were made available in a 2020 study 134 ; 3 sequences collected in Texas, New Mexico, and Arizona between March and April 2020, at the beginning of the southwest outbreak, had high nucleotide identity (almost 99.5%) and formed a cluster distinct from the New York 2020 detections. Sequences from California collected in late 2020 and early 2021 were highly homologous (99–99.4%) to the southwestern sequences reported in the aforementioned 2020 study.18,134 No comprehensive recombination studies have yet been published, but analyses of the topology of the phylogenetic trees of the structural and nonstructural genes of the 2020–2021 California sequences suggest that they were GI.3P-GI.2 recombinants. 18 Because the virus has circulated widely and no detailed phylogenetic and recombination studies have been published yet, it is possible that other recombinants are present in the country. Of the available sequences from North America reported before 2020, the aforementioned southwestern U.S. sequences were more similar to one of the 2018 detections in British Columbia13,18; however, at this stage, it cannot be excluded that some of the cases of the southwestern outbreak and further detections in other parts of the country represent different incursions. In fact, RHDV2 detections continue to date, and as of 2023 Sep 30, the virus has been reported in 29 states, including Hawaii (USDA-APHIS RHDV2 outbreak map: https://www.aphis.usda.gov/aphis/maps/animal-health/rhd [cited Oct 2023]). Kohl and collaborators of the “RHD Awareness Team” from the University of Georgia are performing an up-to-date follow-up of the cases in the United States, compiling information from the USDA-APHIS and other sources, and the results can be viewed at https://rhdv2.org/ (cited Oct 2023).
In April 2020, almost at the same time as the first cases of the southwestern U.S. outbreak were being notified, detections were also reported in several northern states of Mexico.
190
The virus has been reported in wild and domestic
Several detections of RHDV2 from Cuba have been reported to WOAH since 2021, mostly in farmed rabbits. 181 Further details of the spread and sequences are not available, but per the mentioned reports, multiple premises in different cities have been affected with up to thousands of animals being lost due to viral infection or culling.
During the recent outbreaks in the Americas, some novel detection approaches have been attempted. RT-qPCR from dried blood filter paper and ear punch samples obtained from cottontail rabbits (
Affected animal species
Oryctolagus
The first detections of RHDV2 were reported in domestic and wild European rabbits.103,105 The virus has circulated widely in this species, particularly in Europe and Oceania, and the vast majority of the experimental infections have been carried out using European rabbits. It is, therefore, the species in which the various aspects of the disease have been better characterized. As data for other species are much scarcer, disease features in European rabbits are often extrapolated to other
Mortality associated with RHDV2 in European rabbits varies among different reports of natural disease, and is generally 10–90%, or occasionally up to 100% in small, naïve domestic populations.18,75 In wild populations, high mortality has been reported, particularly when the virus first occurs in a geographic area, and this has been one of the reasons the European rabbit has been listed as “endangered” in its natural range.
94
Case fatality in experimental settings is also variable, 0–100%,75,88,164 and 42.6–48.2% in experimental settings with the highest number of animals used (
The clinical signs and pathology (Table 1) of RHDV2 infection in the European rabbit have been described in some detail in both natural and experimental cases.9,30,49,75,80,128,141,172 Major clinical signs include sudden death, fever, collapse, lethargy, seizures, icterus, oral bleeding, dyspnea, hypothermia, bradycardia, poor blood clotting, epistaxis, apathy, depression, anorexia, ataxia, and circling. Elevations in serum transaminases and alkaline phosphatase activities and bilirubin have been noted in pet rabbits24,141,163; however, one report mentioned significant decreases in aspartate aminotransferase and alanine aminotransferase activities in 2 pet rabbits, perhaps related to the short half-life of these enzymes in rabbits. 24 Peracute, acute, subacute, and chronic clinical presentations are described in individual animals. Sudden death without additional signs was frequently recorded in a survey of pet rabbits, 80 and it is thought to be the most common outcome in peracute disease. Subacute-to-chronic clinical courses were relatively common in the first reports of RHDV2. 103 Epistaxis or serosanguineous nasal discharge (Fig. 4) is considered a classic clinical sign of RHD; however, a large survey of commercially farmed rabbits found no clear association between serosanguineous nasal discharge and RHDV2 infection, 150 and therefore the absence of this finding should not preclude a presumptive diagnosis of RHD in cases of mass mortality.
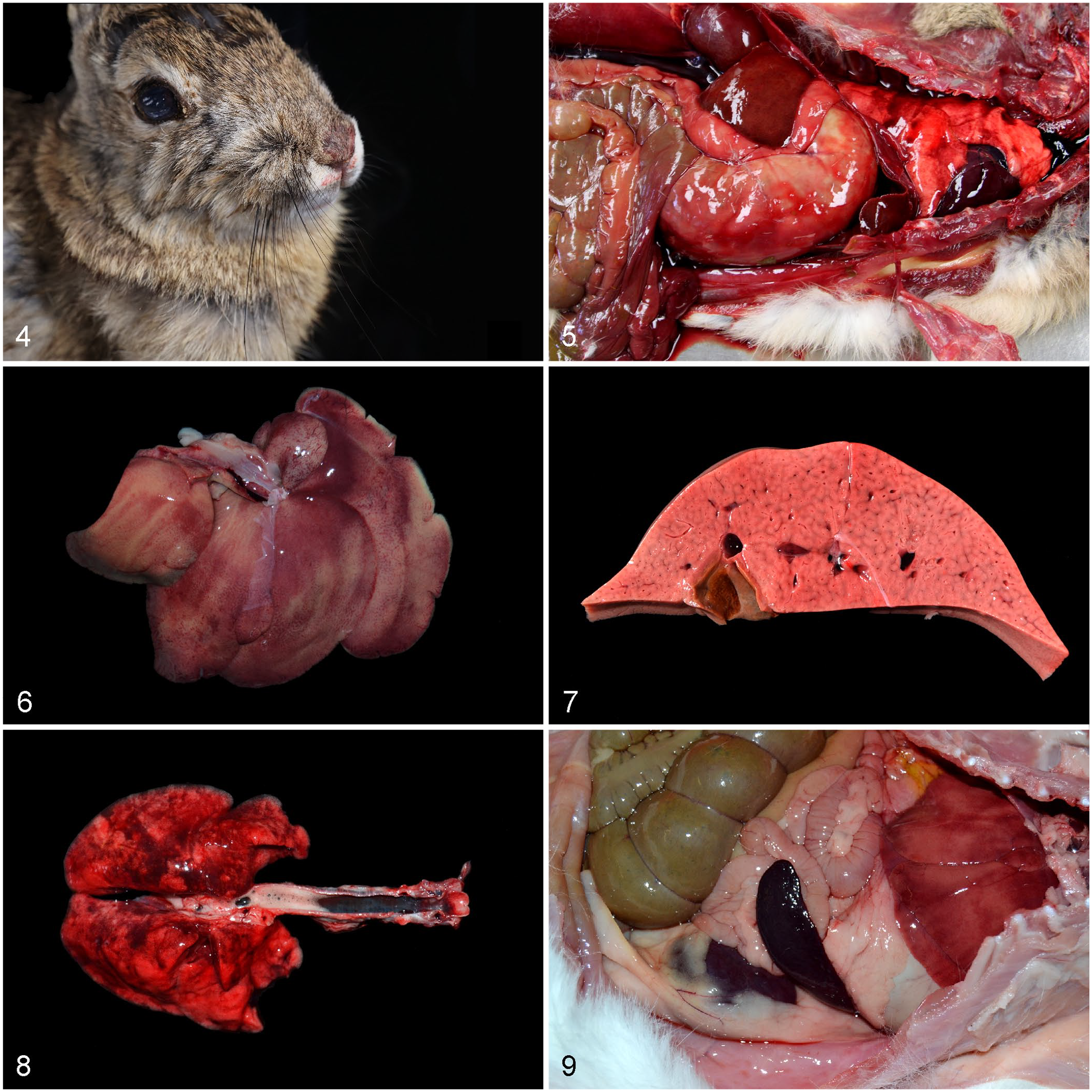
Gross lesions of rabbit hemorrhagic disease virus 2 infection.
On gross postmortem examination (Table 1; Figs. 4–9), a lobular or reticular pattern in the liver is frequently, albeit not always, observed, and overall enlargement, pallor, and friability usually affect this organ (Figs. 6, 7, 9). There is usually congestion and hemorrhage in several organs, being particularly severe in the liver and lungs (Figs. 5, 8). Dark-red discoloration of the tracheal mucosa has been noted as a distinctive gross change (Fig. 8). Serosanguineous fluid or blood in body cavities has also been described as a distinctive feature (Fig. 5). Less commonly, the spleen is enlarged (Fig. 9), and icterus is occasionally present. Importantly, gross lesions may be minimal to absent in some animals with RHD caused by RHDV2,9,80 and in wild and feral domestic rabbits, gross and microscopic lesions are usually obscured by postmortem decomposition or freeze–thaw artifacts. 172
Histologically (Table 1; Figs. 10–13), hepatic necrosis with hepatocellular dissociation and apoptosis, and a periportal-to-massive distribution, is the hallmark and consistent microscopic lesion (Fig. 10). This lesion usually has diagnostic value in cases with limited gross changes. However, hepatic necrosis with random rather than periportal distribution is occasionally observed, 172 which suggests that the pattern of hepatic necrosis should not always be considered as an unequivocally distinctive feature. Preparing overnight histologic sections may be effective in establishing a presumptive diagnosis of RHD while awaiting confirmatory PCR results (J. Asin, E.E. Henderson, and F.A. Uzal, personal observations). Hepatic necrosis is sometimes associated with congestion, hemorrhage, and sinusoidal thrombosis; mild-to-marked infiltration of heterophils and macrophages is frequently seen. The spleen may have deposition of hyaline eosinophilic material in the red pulp, marked hemorrhage and congestion, and lymphocytolysis in the white pulp (Fig. 11). Pulmonary congestion, edema, and hemorrhage are observed in most cases, usually with microvascular thrombosis (Fig. 12). Microthrombi in glomerular capillaries (Fig. 13) and acute tubular injury are occasionally seen in the kidneys. Microthrombosis can be observed in virtually any organ and is the result of disseminated intravascular coagulation and perhaps viral infection of endothelial cells (Table 1).80,137
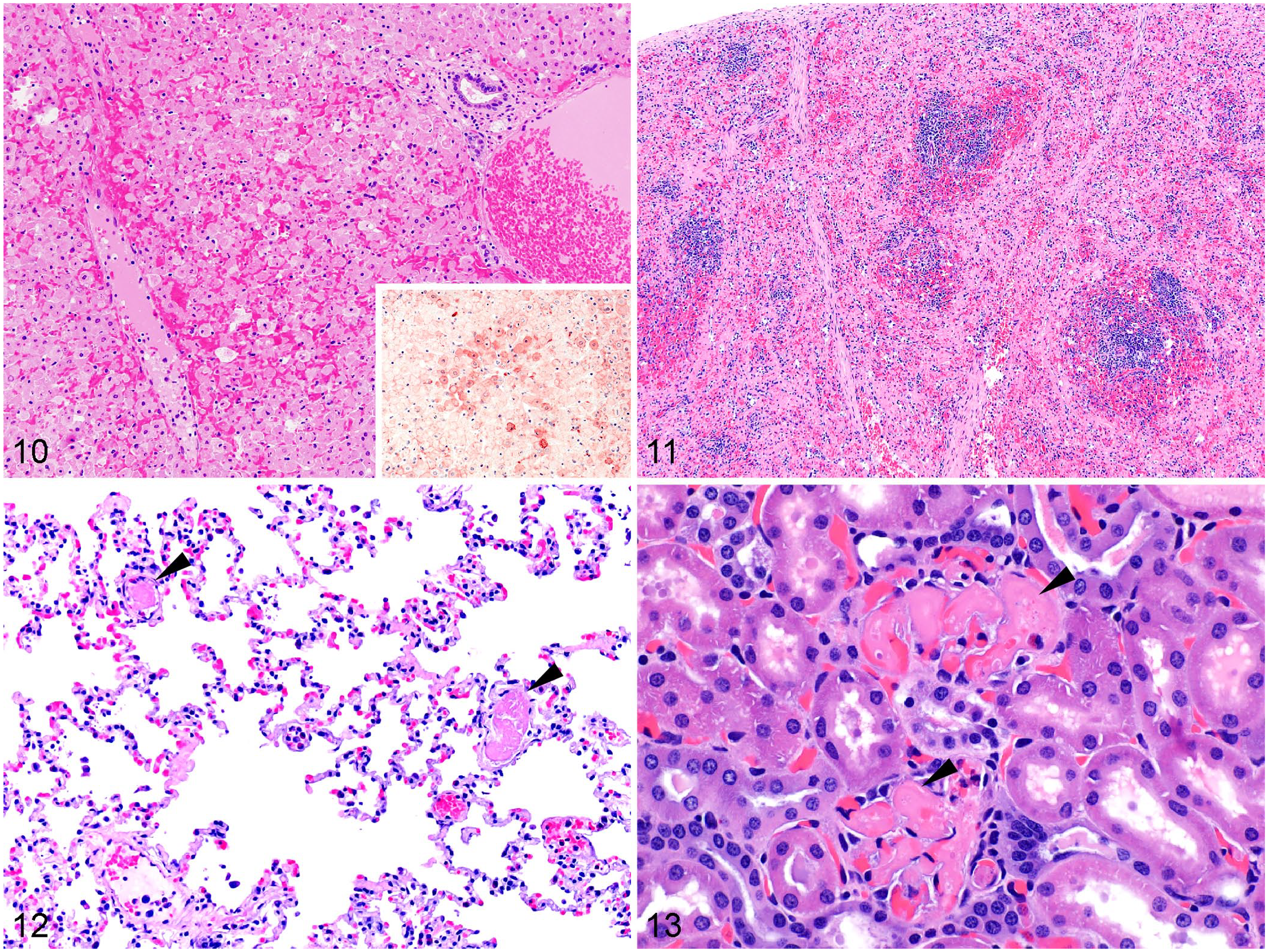
Microscopic lesions of rabbit hemorrhagic disease virus 2 infection. H&E.
Lepus
RHDV2 has been detected in a number of hare and jackrabbit species (Table 2).25,26,34,104,142,166,168,195 Most cases of RHDV2-associated mortality in hares coincided with concurrent outbreaks in rabbits, suggesting the role of rabbits as the primary source of infection to initiate outbreaks in hares. However, with the exception of some outbreaks such as the one that occurred in Cape hares in Sardinia, Italy, 142 in which a significant number of hares were affected, mortality is limited in the available reports and generally lower than in European rabbits, 34 suggesting the occurrence of spillover events from rabbits to hares favored by the high infection pressure during outbreaks in the former, but causing only sporadic infections in the latter. 169 Transmission between infected hares has been postulated but not definitively demonstrated experimentally.104,129 Interestingly, viruses collected from hares can also have high genetic variability. For instance, 2 RHDV2 genotype GII.1P-GI.2 recombinants were collected from European brown hares in Germany in 2014 and 2019, 160 and a GI.1bP-GI.2 recombinant was detected in the same species in Australia in 2016.78,116
Detections of rabbit hemorrhagic disease virus 2 in

Descriptions of lesions in hares and jackrabbits are limited and often based on single case reports of natural disease. Nevertheless, gross findings in hares and jackrabbits appear to be similar to those described in European rabbits with RHD and also in European brown hares with EBHS (Fig. 5).
119
Friable discolored livers with a prominent reticular pattern, congestion of viscera (particularly of the tracheal mucosa), splenomegaly, disseminated hemorrhages, and pulmonary edema have been described.17,25,26,34,97,101,129,142,168,169 Histologic findings are typically hepatic necrosis and apoptosis, usually with a massive distribution, hepatocellular fatty degeneration, and marked congestion and hemorrhage.129,142,169 Periportal-to-midzonal necrosis has been described in an Iberian hare.
168
Lesions in black-tailed jackrabbits in North America are similar to other members of the genus
Sylvilagus
The first detections of RHDV2 in rabbits of the genus
Descriptions of RHD lesions in rabbits of the genus
Viral antigen has been detected by IHC in the liver and other organs of eastern cottontails, desert cottontails, western brush rabbits, and riparian brush rabbits (Fig. 10).16,18,125 A mouse monoclonal antibody cocktail directed against several capsid epitopes (6G2, 3H6, 6D6) of pathogenic and nonpathogenic lagoviruses from the WOAH RHD Reference Laboratory (Brescia, Italy; provided by Drs. A. Lavazza and L. Capucci) was used in all cases.
40
Antigen and antibody ELISAs from the WOAH reference laboratory were used in liver homogenates and serum, respectively, from experimentally infected eastern cottontails.
125
In a study of California cases,
16
liver viral loads were significantly higher in a pool of carcasses of rabbits of the genus
Other lagomorph species
As the virus spreads around the globe, other lagomorph species restricted to specific geographic niches are being exposed. Mortality events were reported in the pygmy rabbit in Nevada (USA) in January–February 2022, and one rabbit tested positive by RT-qPCR.
47
Gross findings in affected rabbits included pulmonary congestion and hemorrhage and mild hepatic discoloration, but histopathology was not performed. Early 2023 detection reports to the WOAH include mortality in populations of red rock rabbits (syn. red rock hares;
Detection in previously unaffected lagomorph species is one of the main concerns of the current rapid global spread, particularly from a conservation standpoint. Concerns focus on unique lagomorph species such as pikas (
Non-lagomorph species
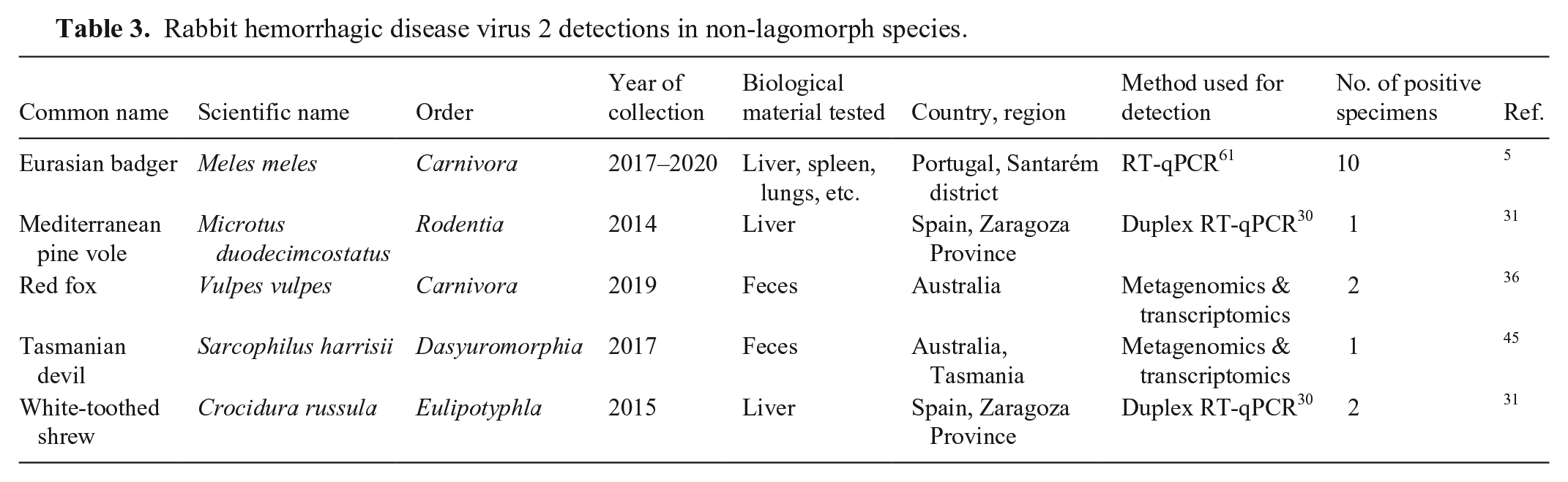
RHDV2 RNA has been sporadically detected in non-lagomorph species from at least 4 different orders (Table 3). These specimens included 1 Mediterranean pine vole (
Rabbit hemorrhagic disease virus 2 detections in non-lagomorph species.
Discussion and conclusions
Since its first reported detections in Europe in 2010, 103 the literature reviewed here suggests that RHDV2 has been more evolutionarily successful than classic RHDV strains. The detection of RHDV2 in a wider range of species is a significant improvement in its ability to survive, but nevertheless a serious concern, particularly from the standpoint of animal conservation and welfare.
RHDV2 continues its rapid global spread from its original wild and domestic rabbit host in Europe to several wild
The apparent speed at which RHDV2 is evolving is likely related to 2 well-documented recombination sites at the
The phylogenetic classification of lagoviruses is based on the
In this respect, the spread of the virus in North America since 2019–2020 represents an unprecedented situation. Prior to the incursion of RHDV2, there had been sporadic detections of classic RHDV and other pathogenic lagoviruses in Canada and the United States since at least 2000, but to our knowledge the virus never became endemic in those areas14,23,35,66; outbreaks of RHD caused by RHDV were also reported in domestic rabbits in Mexico in the late 1980s, but a successful control program led to viral eradication in the country.
74
However, RHDV2 has become endemic in those geographic locations in a short time, which is perhaps related to the susceptibility of some of the wild
Despite some subtleties, gross and microscopic lesions appear to be similar in every new
As with any virus that is genetically capable of rapidly evolving and adapting to new host species, vigilance in early detection and targeted efforts to manage the spread will be critical in preventing outbreaks and minimizing further dissemination. RHDV2 models a typical RNA virus that exploits all evolutionary survival strategies. The lessons learned from this virus can be applied to other viruses and should be reviewed by scientists and policy makers before large-scale interventions are planned.
Footnotes
Acknowledgements
Veterinarians, biologists, and other staff at the California Animal Health and Food Safety Laboratory System, the California Department of Food and Agriculture, and the California Department of Fish and Wildlife are acknowledged for submitting and processing some of the cases from the RHD outbreaks in California that were used as examples in our review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

