Abstract
We evaluated whether dogs with severe brachycephalic obstructive airway syndrome (BOAS) developed a hypercoagulable state similar to people with obstructive sleep apnea. Five dogs with grade 3 BOAS were included as well as 5 healthy control Labrador Retrievers. Venous blood samples were collected from each dog for performance of thromboelastography and determination of hematocrit and platelet count. Groups were compared using a t-test, with p < 0.05 considered significant. Thromboelastography results identified that all BOAS dogs were hypercoagulable compared to the Labradors, having significantly shortened clotting time with increased angle, maximal amplitude, and clot rigidity. BOAS dogs also had evidence of delayed fibrinolysis. These results are consistent with, but more severe than, those previously documented in apparently healthy Bulldogs. Together, these findings support the presence of a hypercoagulable state in brachycephalic dogs, and suggest that this state is amplified by increasing severity of BOAS.
Brachycephaly, or a shortened facial structure, is a common canine condition that is magnified by selective breeding. Most dogs with this condition develop brachycephalic obstructive airway syndrome (BOAS), which predisposes them to chronic upper airway obstruction. 19 BOAS shares features of obstructive sleep apnea (OSA), 10 which is a condition frequently occurring in people characterized by episodes of total and/or partial upper airway collapse alternating with normal breathing during sleep. 13 This leads to chronic intermittent hypoxia, oxygen desaturation, sleep fragmentation, and arousal from sleep. 4 OSA is also associated with decreased survival as a result of development of cardiovascular and thromboembolic disorders.4,20
Human OSA and canine BOAS are very similar syndromes. In fact, the Bulldog has been used as a model of sleep-disordered breathing because of its disordered breathing and episodes of oxygen desaturation associated with abnormal upper airway anatomy. 10 In addition, brachycephalic dogs appear to have a higher risk of cardiovascular disease than dogs with normal facial conformation, and also have frequent snoring, excessive daytime sleepiness, exercise intolerance, and a perception among many clinicians of shortened lifespan compared with dogs of similar body weight.16,21
Over the last 20 y, OSA in people has been associated with hypercoagulability, and this appears to be an important factor linking OSA and cardiovascular complications. Specifically, one study demonstrated that patients with OSA had significantly shortened reaction (R) times on thromboelastography (TEG). 8 Increased nocturnal platelet activation and aggregation,3,6 and elevations in plasma tissue factor, von Willebrand factor, and fibrinogen concentrations have also been documented.7,12 In addition, TEG has been used to demonstrate hypercoagulability in a rodent model of OSA 15 and, most recently, in a population of clinically healthy Bulldogs. 11
TEG is a test of global hemostasis that has the ability to detect both hypocoagulability and hypercoagulability, and is performed as a simple, point-of-care test. TEG produces a standardized tracing, the components of which represent the different stages and quality of clot formation and fibrinolysis. 5 A hypercoagulable TEG tracing is represented by a short R time, short clotting time (K), steep angle, wide maximal amplitude (MA), and increased clot rigidity (G).5,9 Delayed fibrinolysis is identified through persistence of clot stability at 30 and 60 min (LY30, LY60). 9
BOAS is associated with a wide range of clinical signs, with all brachycephalic dogs being affected to some degree. Although TEG values have been reported in “healthy” Bulldogs, 11 they remain unreported in more severely affected Bulldogs and other brachycephalic breeds. Our study aimed to evaluate TEG in dogs with severe BOAS.
Dogs with severe BOAS (grade 3) admitted for surgical therapy to the intensive care unit of the Foster Hospital for Small Animals at the Cummings School of Veterinary Medicine at Tufts University (North Grafton, Massachusetts) were eligible for inclusion. Grading was in accordance with a previously published grading scheme, with grade 3 reflecting moderate to severe inspiratory effort and noise with moderate to severe dyspnea and exercise intolerance. 14 Exclusion criteria included conditions (e.g., neoplasia, hemolytic anemia or thrombocytopenia, hyperadrenocorticism, protein-losing enteropathy or nephropathy, liver disease, sepsis, trauma) or concurrent treatment with medications (e.g., non-steroidal anti-inflammatory drugs, long-term prednisone, heparin, clopidogrel) that could affect coagulation status. Control dogs consisted of healthy Labrador Retrievers (LAB), which is a common, non-brachycephalic breed. Dogs were assessed as healthy based on physical examination and laboratory testing (complete blood cell count, biochemistry, and prothrombin and activated partial thromboplastin times). The study was approved by the Clinical Science Review Committee, and owners provided informed consent.
Direct venipuncture was performed from the jugular vein, if tolerated, or a peripheral vein. A single venous blood sample was collected from each dog and placed into a 3.2% sodium citrate tube with a 9:1 blood-to-citrate ratio for evaluation of a kaolin-activated TEG. Hematocrit and platelet count were recorded. An adequate platelet count was defined as >180 × 109/L. All samples were maintained at room temperature for 30 min prior to analysis. TEG (TEG 5000, Haemonetics, Braintree, MA) parameters, including R, K, angle, MA, G, LY30, and LY60 values, were recorded for each dog. The values obtained for each TEG parameter were compared between BOAS and LAB using a t-test, with p < 0.05 considered significant.
Five severely affected BOAS dogs were included in the study: 3 Pugs, 1 Pekingese, and 1 Bulldog. Four dogs were spayed females and 1 was a castrated male. The median age of BOAS dogs was 9 y (range: 5–11 y).
The mean hematocrit was 47 ± 2 L/L in the BOAS group and 42 ± 2.5 L/L in the LAB group (p = 0.02). Platelet counts, or estimates if platelets were clumped, were determined to be adequate in all dogs. TEG results are summarized in Table 1, and a representative TEG from each group is shown in Figure 1. All BOAS dogs were hypercoagulable based on increased clot strength (MA and G) and, compared to LAB dogs, had significantly shortened K times with increased angle, MA, and G. The BOAS dogs also had evidence of delayed fibrinolysis compared to LAB based on significantly lower LY60 values (p < 0.01).
Thromboelastography values from dogs with BOAS and LAB.
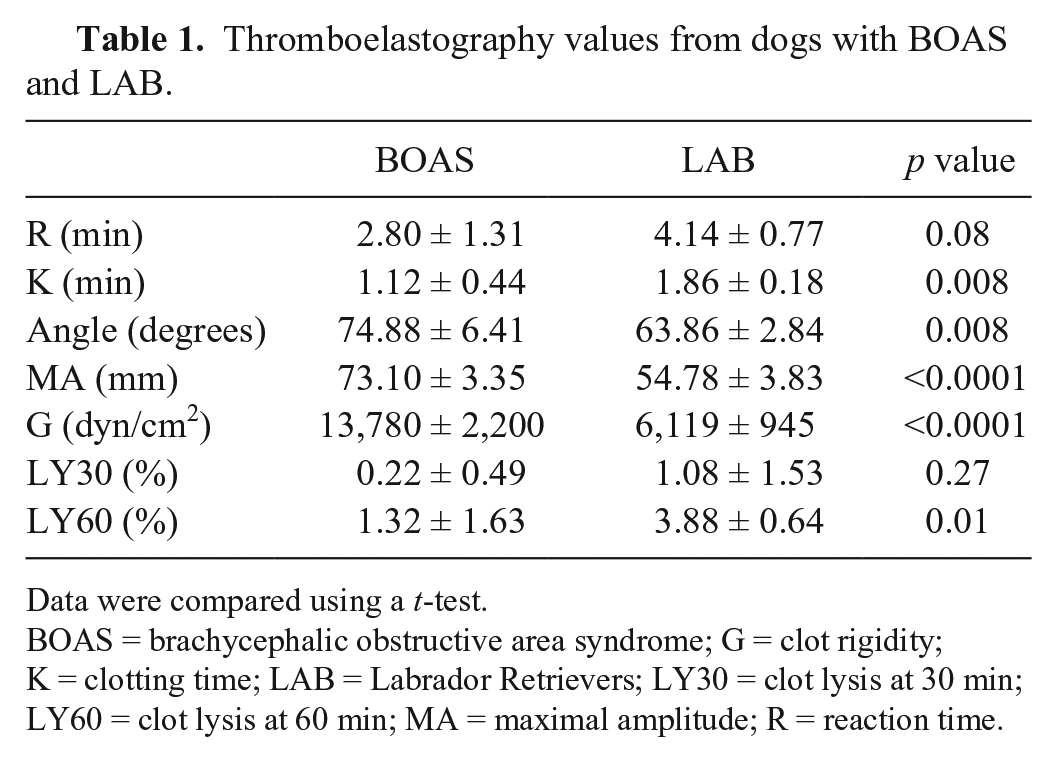
Data were compared using a t-test.
BOAS = brachycephalic obstructive area syndrome; G = clot rigidity; K = clotting time; LAB = Labrador Retrievers; LY30 = clot lysis at 30 min; LY60 = clot lysis at 60 min; MA = maximal amplitude; R = reaction time.
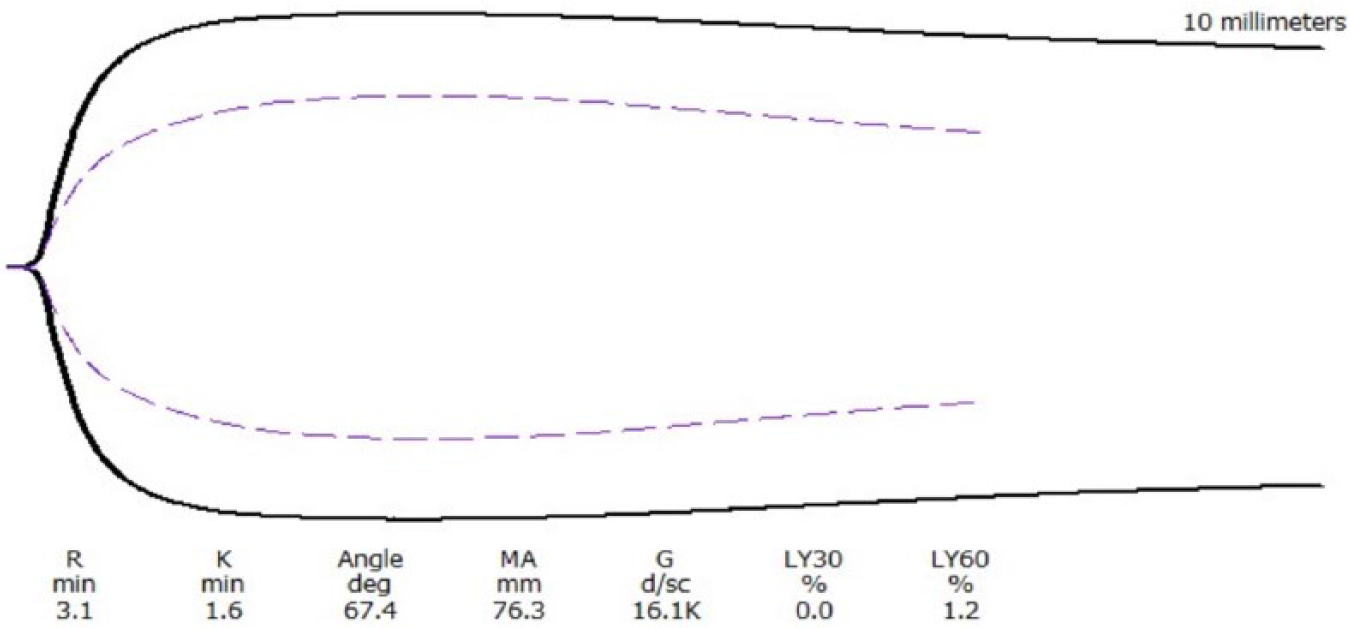
A representative thromboelastography (TEG) tracing from a brachycephalic dog (solid line) and from a normal Labrador Retriever (dotted line). G = clot rigidity; K = clotting time; LY30 = clot lysis at 30 min; LY60 = clot lysis at 60 min; MA = maximal amplitude; R = reaction time.
Our pilot study identified profound hypercoagulability and delayed fibrinolysis in severely affected BOAS dogs. A 2015 study also documented similar, although less pronounced, TEG changes in apparently healthy Bulldogs. 11 Although that study did not utilize the BOAS grading system, they included Bulldogs that were considered clinically normal and therefore may be presumed to have grade 0–1 BOAS, reflecting no audible noise, inspiratory effort, or dyspnea at rest. Together, these results support the presence of a hypercoagulable state in brachycephalic dogs that is potentially magnified with increasing severity of BOAS.
As in people with OSA, there are a number of factors that may contribute to the development of hypercoagulability in BOAS, including systemic inflammation, platelet activation, and delayed fibrinolysis. Systemic inflammation has been repeatedly documented in OSA and often resolves with therapy.1,2,23 Although little is known about the inflammatory effects of BOAS, increased levels of circulating pro-inflammatory cytokines (tumor necrosis factor–alpha [TNF-α], interleukin [IL]-10, IL-13, IL-17A) and nitric oxide were previously documented in a group of brachycephalic dogs. 17
Increased platelet activation has also been identified in people with OSA3,6,20 and, although it was not directly evaluated in our study, the documented increases in MA, K, and G do partly reflect platelet function. Thus, platelet activation may also contribute to hypercoagulability in BOAS.
Delayed fibrinolysis, as evidenced by increased plasminogen activator inhibitor type 1 (PAI-1) concentrations, has been reported in people with OSA,18,22 although TEG evaluation of fibrinolytic changes has not been described. Delayed fibrinolysis amplifies complications of thrombotic disease, and has been recognized as a risk factor for development of adverse cardiac events. 18 The magnitude of the change identified in the dogs in our study was small, and the clinical relevance of delayed fibrinolysis, especially of small magnitude, remains unknown. Further investigation of fibrinolysis in dogs with BOAS is warranted.
The hematocrit of the BOAS dogs in our study was higher than that of the LAB dogs. It is not clear if this is a reflection of chronic hypoxemia, or simply relative hemoconcentration associated with respiratory losses of body water. Healthy Bulldogs have also been reported to have higher hematocrits. 11
There are several limitations to our study. The number of animals reported is small, and represents only a subset of severely affected dogs. Given previous studies, it is likely that the “average” brachycephalic dog may be less severely affected. In addition, because the BOAS population in our study was not restricted to Bulldogs as was the 2015 study to which it was compared, 11 the possibility cannot be excluded that breed variations in coagulation contribute to the reported differences. Also, in the absence of a thorough diagnostic investigation, it is possible that unknown comorbidities were present. Finally, although Labrador Retrievers are a common dog breed that is unaffected by BOAS, it is possible that another group of dogs would have served as a better control.
Future directions include the continued evaluation of TEG and markers of inflammation in BOAS dogs, as well as performing analysis pre- and post-operatively to determine whether treatment is associated with normalization of clotting parameters as it is in people with OSA. Such studies should focus on a single dog breed with different grades of BOAS, or include various dog breeds grouped by BOAS grade, in order to confirm that coagulation status is associated with worsening grade.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded internally (Department of Clinical Sciences, Cummings School of Veterinary Medicine at Tufts University, North Grafton, MA).
