Abstract
Samples of the liver, telencephalon, brainstem, and cerebellum were obtained from 22 bovids suffering from spontaneous or experimental acute toxic liver disease. Perreyia flavipes larvae, and leaves of Cestrum corymbosum, Cestrum intermedium, Dodonaea viscosa, Trema micrantha, and Xanthium cavanillesii were the causal agents in the disorders studied. Hematoxylin and eosin and periodic acid–Schiff staining, as well as anti-S100 protein (anti-S100), anti–glial fibrillary acidic protein (anti-GFAP), and anti-vimentin immunostaining were used to evaluate the brain sections. Astrocytic changes were observed in all samples and were characterized by swollen vesicular nuclei in gray (Alzheimer type II astrocytes) and white matter; and by abundant eosinophilic or vacuolated cytoplasm with pyknotic nuclei in the white matter. These changes were evidenced by anti-S100 and anti-GFAP immunostaining. Our study demonstrates major changes in astrocytes of cattle that died with neurologic clinical signs as the result of acute toxic liver disease.
Introduction
The consumption of various poisonous plants and the Hymenoptera Perreyia flavipes larvae have caused neurologic disorders secondary to hepatotoxic insults in cattle from Brazil4,12,13,17,20 and other countries.5,9,19 Status spongiosus and isolated or grouped astrocytes with large vesicular nuclei, termed Alzheimer type II astrocytes, have been described as microscopic changes in hepatic encephalopathy.15,22 Status spongiosus is characterized by microcavitations that are primarily evident in myelinated tracts of the cerebral white matter and in the white–gray matter interface of the telencephalic cortex. Such changes are attributed to intramyelinic edema, which induces myelin sheath vacuolation.1,15 In horses, hepatic encephalopathy is usually associated with Alzheimer type II astrocytes in gray matter. However, in other species, including cattle, both changes may occur, but status spongiosus has been the lesion described most frequently. 15
Astrocytes are among the primary targets of central nervous system (CNS) insults, most likely because of their important role in fluid and electrolyte balance.2,11,14,15 The CNS depends on ammonia removal via glutamine synthesis within astrocytes. 7 Hyperammonemia is the main factor in the pathogenesis of hepatic encephalopathy. Ammonia formed from the bacterial degradation of amines, amino acids, purines, and urea in the gastrointestinal tract of healthy animals is biotransformed into urea by the hepatic citrulline–arginine–ornithine–urea cycle. In cases of diffuse hepatopathy, such hepatic routes are impaired, and hyperammonemia occurs. 22
We describe the histologic changes and the immunohistochemical characterization of lesions using anti–glial fibrillary acidic protein (anti-GFAP), anti-S100 protein (anti-S100), and anti-vimentin antibodies on brain sections from cattle poisoned by spontaneous or experimental ingestion of P. flavipes larvae and leaves of the plants Cestrum corymbosum, C. intermedium, Dodonaea viscosa, Trema micrantha, and Xanthium cavanillesii.
Materials and methods
Archival (1998–2012) paraffin-embedded samples of the telencephalon, brainstem, cerebellum, and liver from 22 cattle that died with acute toxic hepatopathy were selected, sectioned at 3 µm, and stained with hematoxylin and eosin and periodic acid–Schiff. Immunostaining with anti-GFAP (polyclonal rabbit anti-GFAP antibody, Z0334, Dako Denmark, Glostrup, Denmark), anti-S100 protein (polyclonal rabbit anti-S100 protein antibody, Z0311, Dako Denmark), and anti-vimentin (monoclonal mouse anti-vimentin antibody, Invitrogen, Carlsbad, CA) antibodies was also performed, using the biotin–peroxidase–streptavidin method (LSAB+ system–HRP, K0690, Dako Denmark), with DAB (Dako Denmark) or NovaRed (Vector Red S-K 4800, Vector Laboratories, Burlingame, CA) as chromogen (Table 1). Additional information was retrieved from the files. The brain specimens from 11 adult bovids that died from various causes without neurologic clinical signs or hepatic and/or brain changes served as negative controls.
Antigen retrieval, dilutions, and origin of the primary antibodies used in the immunostaining protocols applied to the central nervous system sections from cattle affected by hepatic encephalopathy.*

DAB = 3,3′-diaminobenzidine; GFAP = glial fibrillary acidic protein; LSAB-HRP = biotin–peroxidase–streptavidin.
Performed in an electric pressure cooker.
Results
All 22 bovids poisoned by spontaneous or experimental ingestion of P. flavipes larvae or leaves of the plants C. corymbosum, C. intermedium, D. viscosa, T. micrantha, and X. cavanillesii showed neurologic signs, characterized by tremors, ataxia, difficulty or reluctance to walk, head pressing, aggressive behavior, and terminal paddling movements. Hepatic changes (i.e., an enhanced lobular pattern) were present at autopsy in all cases included in our study. Gross changes linked to hepatic insufficiency were also noted, including scattered hemorrhages (n = 15), mild (n = 2) to moderate (n = 1) jaundice, and ascites (n = 5). Microscopic liver changes, characterized by marked necrosis of hepatocytes and hemorrhage (Fig. 1), were observed in all cases (Table 2). Many animals also had degeneration of hepatocytes at the periphery of necrotic areas. All affected cattle had histopathologic changes in the brain, primarily characterized by astrocytic alterations (Table 2).
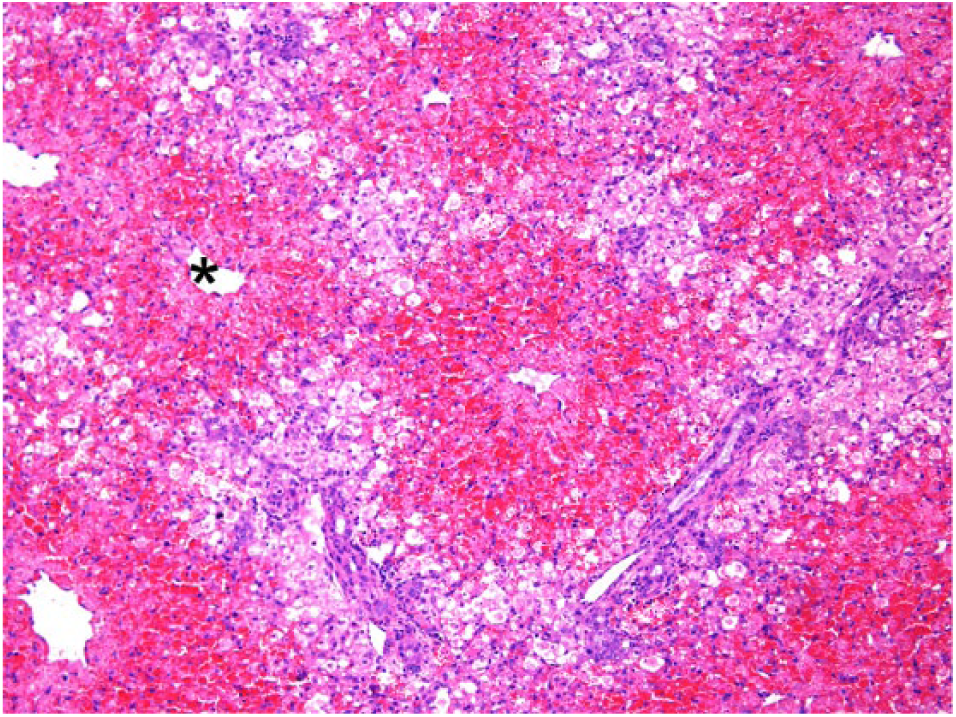
Diffuse, severe, coagulative necrosis of hepatocytes associated with hemorrhage in the liver of a bovid that died 106 h after ingestion of Trema micrantha leaves. Cytoplasmic vacuolation of the periportal hepatocytes can be observed with mild proliferation of the biliary epithelium; asterisk (*) identifies a centrilobular vein. Hematoxylin and eosin. 20×.
Poisoned bovids, disease duration, time elapsed between death and collection of samples for histologic evaluation, and histologic findings in the liver and brain.*
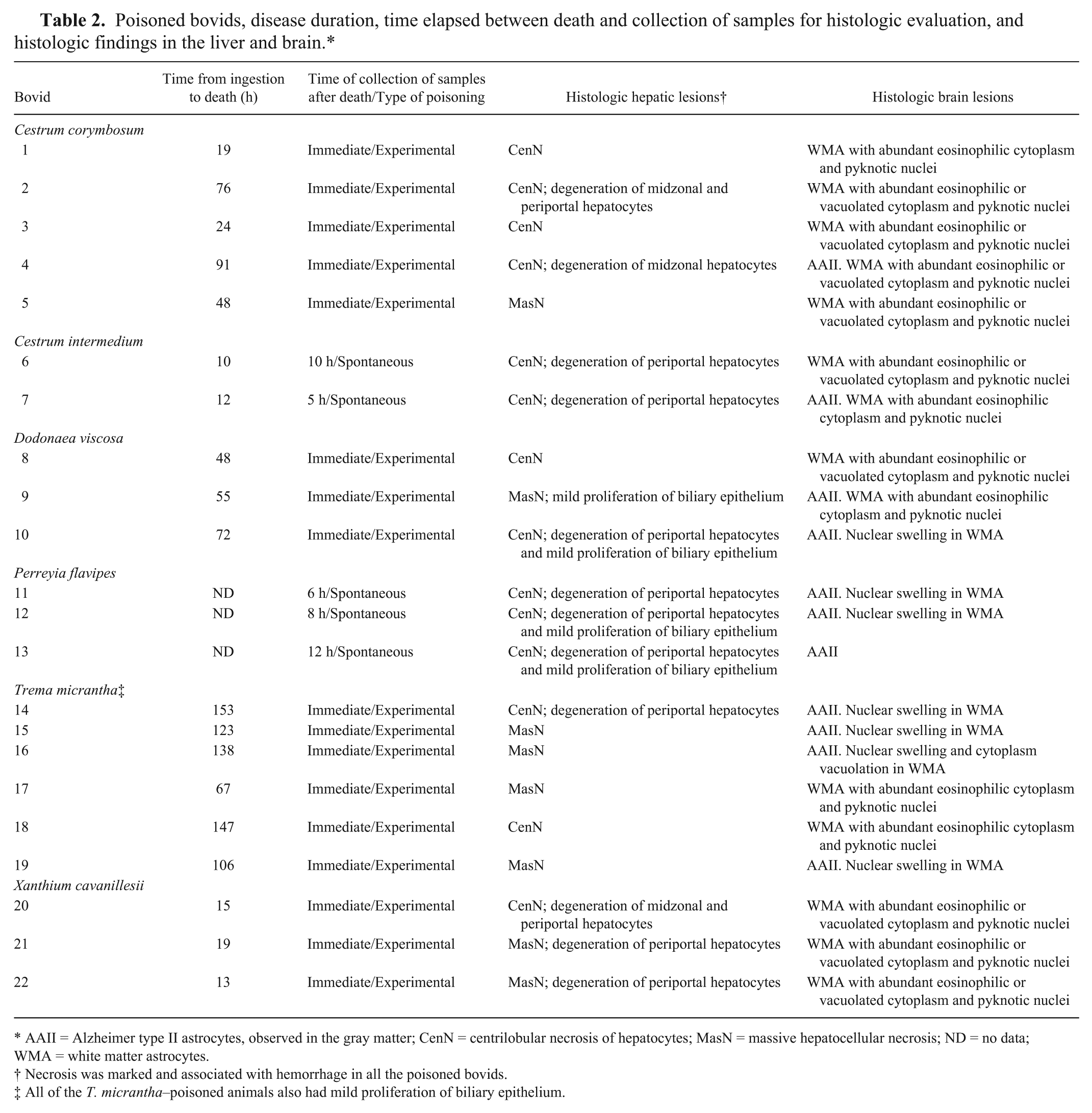
AAII = Alzheimer type II astrocytes, observed in the gray matter; CenN = centrilobular necrosis of hepatocytes; MasN = massive hepatocellular necrosis; ND = no data; WMA = white matter astrocytes.
Necrosis was marked and associated with hemorrhage in all the poisoned bovids.
All of the T. micrantha–poisoned animals also had mild proliferation of biliary epithelium.
Alzheimer type II astrocytes, observed in 11 of 22 cattle, were evident and occurred in the gray matter of the telencephalic cortex (Fig. 2), brainstem, and cerebellum (in which Bergmann astrocytes were especially affected). These 11 cases were mostly T. micrantha– (n = 4) and P. flavipes–poisoned (n = 3) cattle. Several of these cattle also showed various degrees of nuclear swelling in the white matter astrocytes. Astrocytic changes in samples from C. corymbosum–, C. intermedium–, X. cavanillesii–, and D. viscosa–poisoned cattle predominated in the white matter, which were vacuolated in 10 of 22 cattle, but the changes were mainly represented by abundant eosinophilic cytoplasm and pyknotic nuclei (n = 14; Fig. 3). Similar changes were observed in Bergmann astrocytes in the cerebellar gray matter. Within the telencephalon, these changes were prominent in the subcortical region. In the cerebellum, these changes were most frequent in the white matter from the folia, particularly in the cerebellar peduncles.
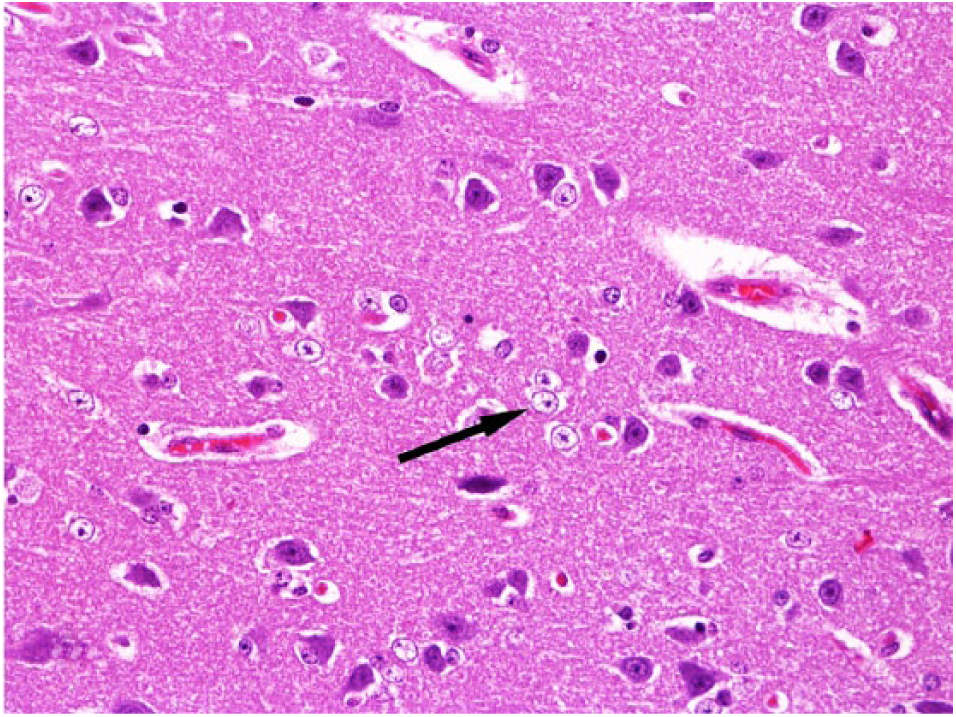
Alzheimer type II astrocytes (arrow) in the telencephalic cortex of a bovid poisoned by Trema micrantha. Hematoxylin and eosin. 40×.
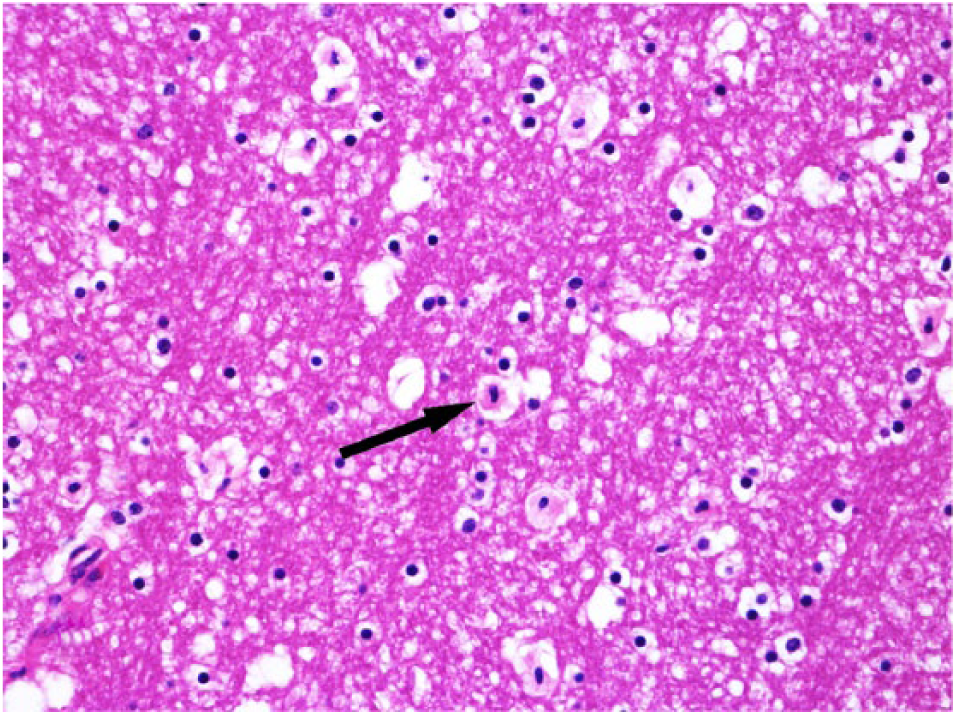
Astrocytes with swollen eosinophilic cytoplasm and pyknotic nuclei (arrow) in the cerebellar white matter of a bovid poisoned by Xanthium cavanillesii. Hematoxylin and esoin. 40×.
Circulatory changes were also observed in the brain. Moderate-to-severe congestion was observed in the telencephalon, brainstem, and/or cerebellum. Furthermore, there were submeningeal and multifocal hemorrhages in the cerebellar gray matter, brainstem, and telencephalon in some cattle. Perivascular edema (n = 17) was also observed in the telencephalic gray matter, brainstem, and cerebellum, with perineural edema in the telencephalic cortex.
Anti-GFAP immunostaining did not mark the cellular bodies from Alzheimer type II astrocytes, but was observed in the cellular processes, which were thickened compared with the controls. Astrocytic processes were strongly labeled in the white matter, and demonstrated increased thicknesses and labeling of their terminal feet within the gray and white matter. The astrocytes with swollen cytoplasm and pyknotic nuclei, mainly observed in Cestrum spp.– and X. cavanillesii–poisoned bovids, were strongly labeled in the white matter from the telencephalon, brainstem, and cerebellum (Fig. 4) and in the cerebellar gray matter (Bergmann astrocytes).
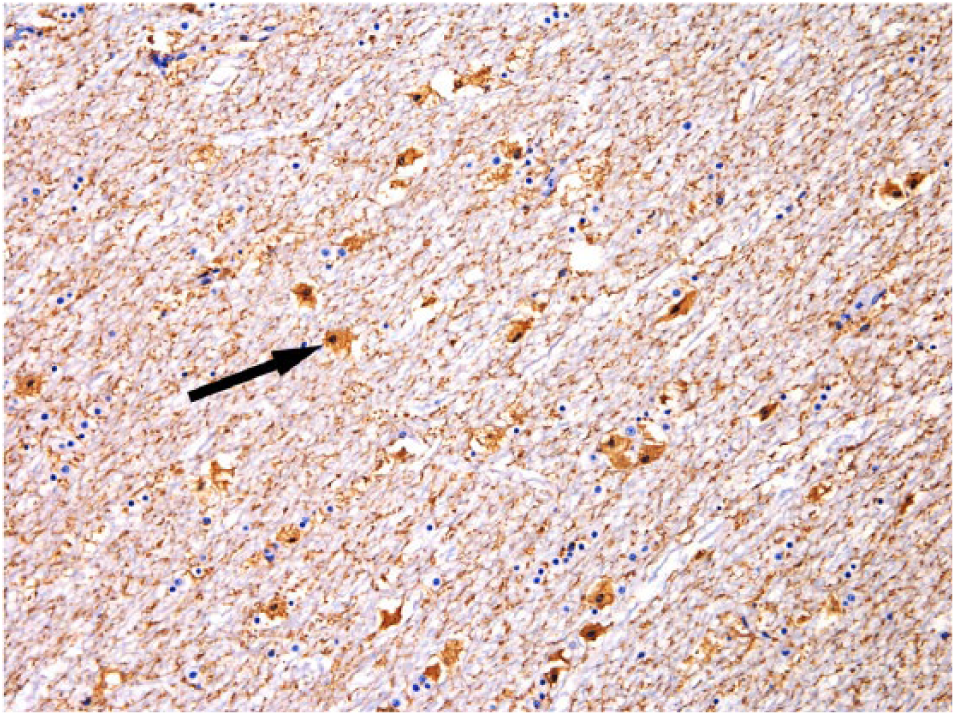
Anti–glial fibrillary acidic protein (anti-GFAP) immunolabeled astrocytes (arrow) with swollen cytoplasm and pyknotic nuclei in the cerebellar white matter of a bovid poisoned by Xanthium cavanillesii. Streptavidin–peroxidase, chromogen 3,3′-diaminobenzidine (DAB). 20×.
Anti-S100 immunostaining demonstrated the astrocytic changes in the gray matter (Alzheimer type II astrocytes; Fig. 5). In the white matter, the vacuolated astrocytes had peripheral labeling (Fig. 6), and the astrocytes with swollen cytoplasm were strongly marked (Fig. 7). In the control bovids, there was standard immunostaining of astrocytic cytoplasm and processes (Fig. 8). Anti-vimentin immuno-staining strongly labeled the vascular walls; however, there were no differences compared with control sections.
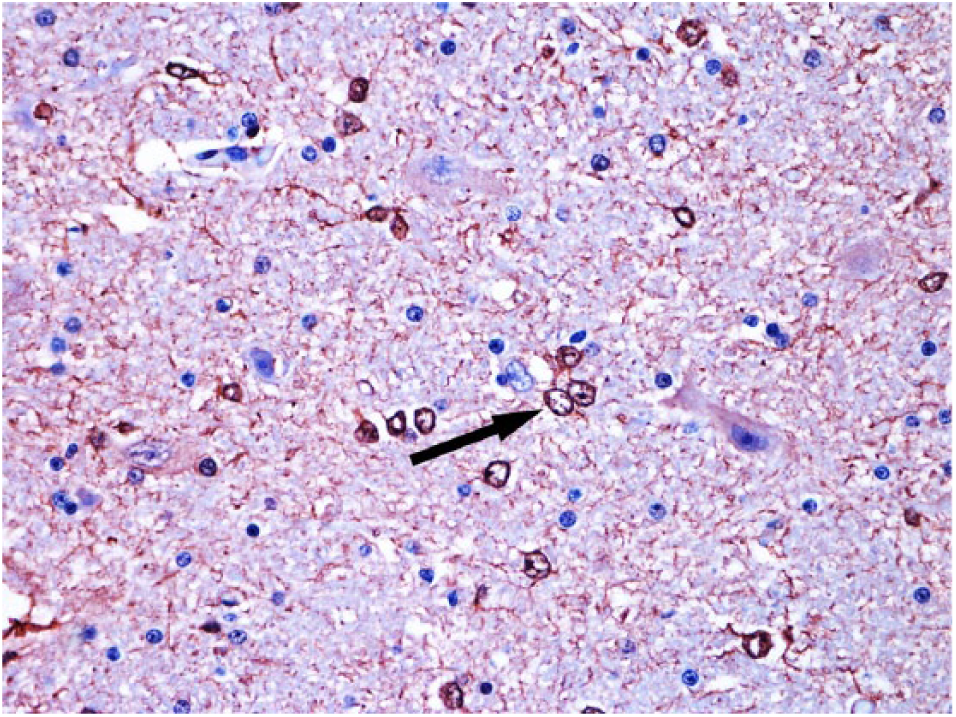
Alzheimer type II astrocytes (arrow) in anti-S100 immunolabeled telencephalic cortex of a bovid poisoned by Trema micrantha. Streptavidin–peroxidase, chromogen NovaRed. 40×.
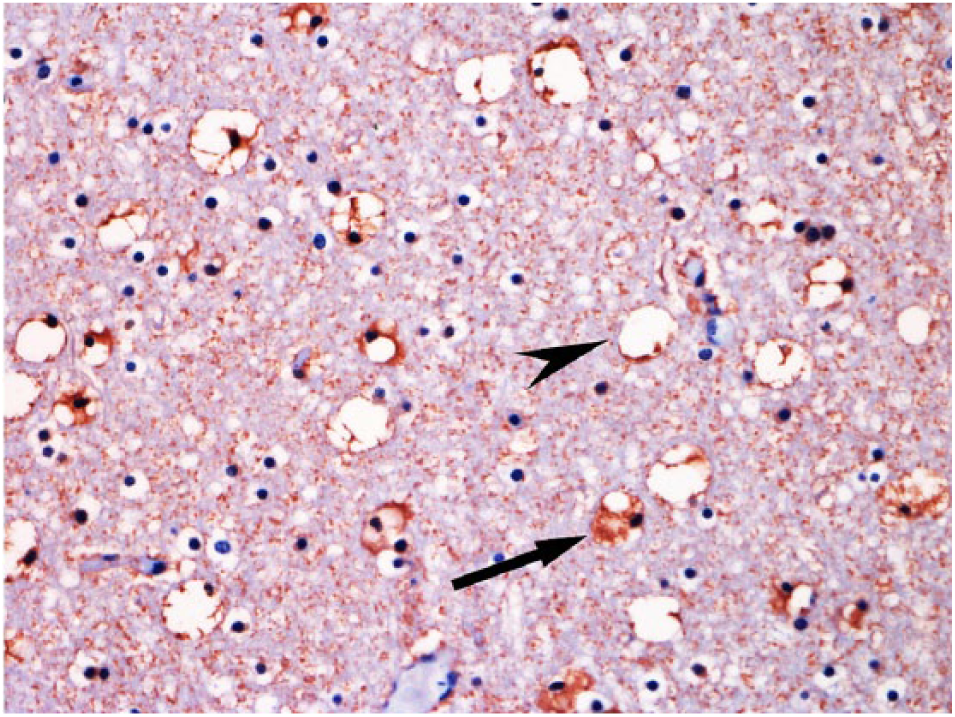
Astrocytes with enlarged cytoplasm (arrow) and vacuolation in the cerebellar white matter of a bovid poisoned by Xanthium cavanillesii. Note the vacuolar anti-S100 peripheral immunolabeling (arrowhead) in the vacuoles. Streptavidin–peroxidase, chromogen NovaRed. 40×.
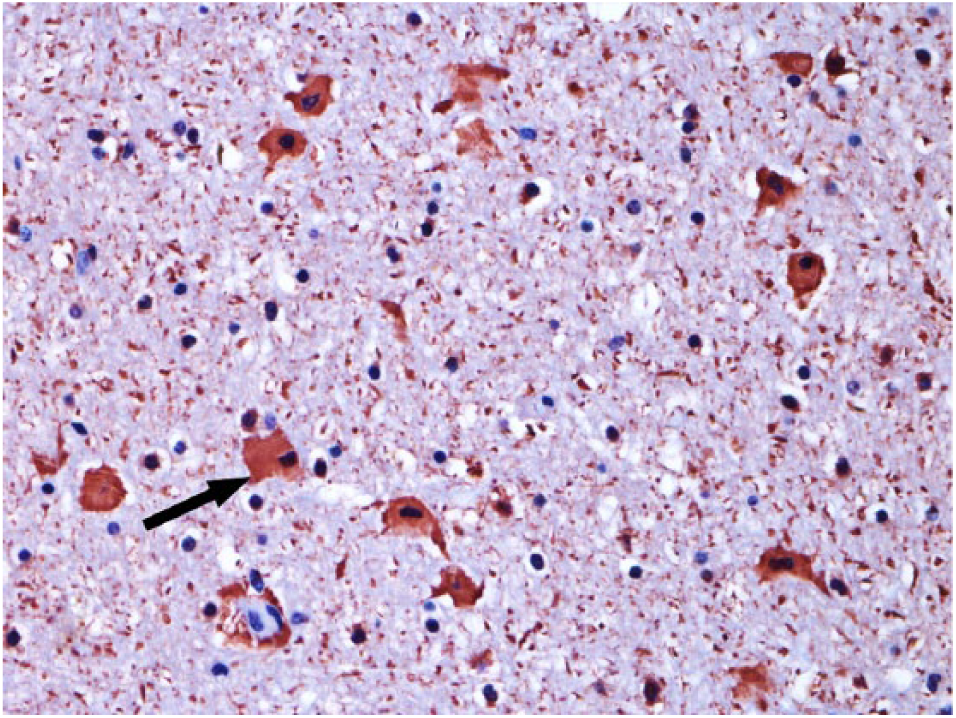
Anti-S100 immunolabeled astrocytes (arrow) with swollen cytoplasm and pyknotic nuclei in the cerebellar white matter of a bovid poisoned by Cestrum intermedium. Streptavidin–peroxidase, chromogen NovaRed. 40×.
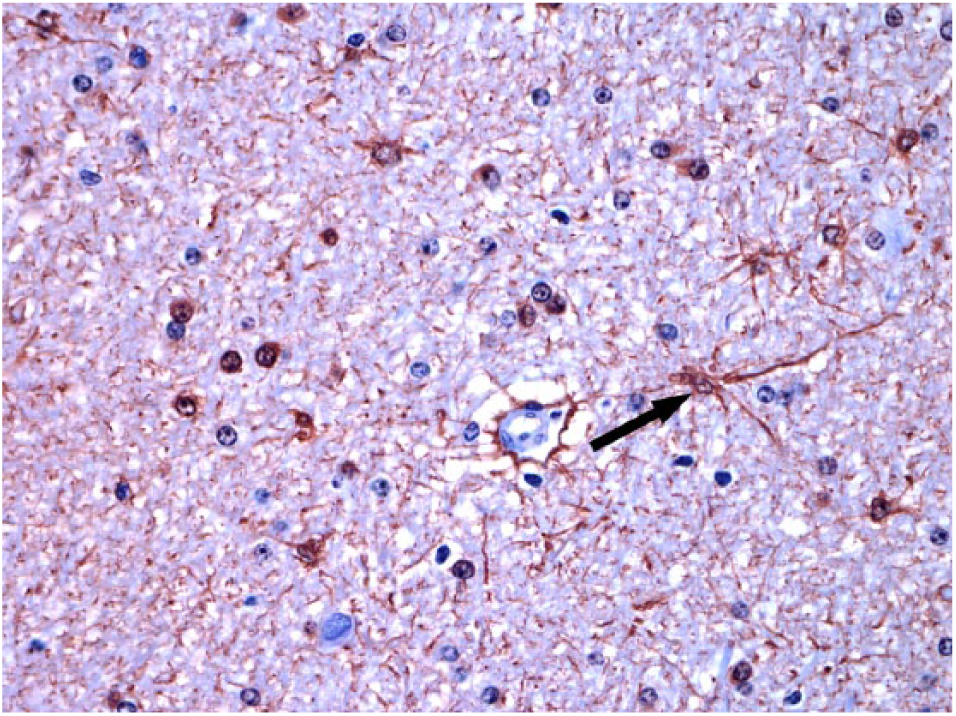
Anti-S100 immunolabeled astrocytes (arrow) in the cerebellar white matter of a control bovid, showing standard immunostaining of astrocytic cytoplasm and processes. Streptavidin–peroxidase, chromogen NovaRed. 40×.
Discussion
In our study, all of the clinically examined bovids exhibited neurologic signs and brain histologic lesions, characterized by astrocytic changes. Alzheimer type II astrocytes were observed in the brains of T. micrantha– and P. flavipes–poisoned cattle. However, astrocytes with swollen cytoplasm and pyknotic nuclei, such as those observed in the white matter from 14 of the 22 cattle, have only recently been identified in brain sections from cattle suffering from acute toxic hepatopathy caused by Cestrum intermedium consumption. 21 These brain changes were observed in all of the C. corymbosum–, C. intermedium–, and X. cavanillesii–poisoned cattle, in which the mean time elapsed from plant ingestion to death was <35 h. Astrocytes with expanded cytoplasm and pyknotic nuclei were also the main finding in 2 additional plant poisoning cases (1 case of D. viscosa and 1 case of T. micrantha, in which the aforementioned period was <72 h). This observation indicates that the time elapsed between plant consumption and death is the main factor influencing the morphologic astrocytic presentations in acute toxic hepatopathy. In C. intermedium–poisoned cattle, 21 death occurred <24 h after the onset of clinical signs. Immediately after injury, astrocytes begin to synthesize large amounts of cytoplasmic GFAP, and then respond with morphologic changes characterized by expanded cytoplasm and thicker cellular processes. As early as 36–48 h after injury, anti-GFAP immunolabeling can be observed. The maximum reaction occurs at 72 h, and there is decreasing intensity over the following weeks and months. 16 In cultured rat astrocytes exposed to ammonia, a progressive increase in cellular volume occurs in 1–3 days after exposure. 8
Conversely, in the T. micrantha– and P. flavipes–poisoned cattle, astrocyte nuclear changes predominated, with Alzheimer type II astrocytes in the gray matter; nuclear swelling in astrocytes within the white matter was also observed. In T. micrantha poisoning, the mean time elapsed from plant consumption to death was 133 h. In spontaneous P. flavipes poisoning, no clinical signs were recorded; however, in an experimental P. flavipes poisoning of sheep, 12 the mean time from ingestion to death was 102 h.
The vacuolation observed in the same white matter areas of brains with the aforementioned astrocytic changes was associated with swelling, death, and disintegration of astrocytes. The vacuolation was evidenced by strong anti-S100 immunostaining at the periphery of the vacuoles. These vacuoles were round, well delimited, and different from those described in encephalopathy caused by chronic hepatopathy, termed status spongiosus.1,15
Cytoplasmic swelling of astrocytes has also been observed in human hypoglycemia. 6 Similarly, hypoglycemia has been considered in the pathogenesis of the neurologic disorders linked to acute diffuse hepatopathy in cattle.1,3,4,18 However, the association of hypoglycemia could not be made with the acute toxic hepatopathy cases in our study because only 4 of the 22 cattle were tested. Hypoglycemia was detected in 3 of those cases.
Anti-vimentin immunostaining was used to evaluate vascular walls and astrocytes because young and/or injured astrocytes may synthesize vimentin. 10 Strong labeling was observed in the vascular walls in all of the brain sections from poisoned and control cattle in our study. The absence of labeling in astrocytes indicates that astrocytic vimentin synthesis had not occurred. When considered with the astrocytic changes and the absence of brain vascular changes, the brain edema in cattle affected by acute toxic hepatopathy should be characterized as being of cytotoxic origin.
The identified astrocytic degenerative-necrotic changes indicate that astrocytes are important targets in hepatic encephalopathy resulting from acute toxic hepatopathy in cattle. The anti-GFAP and anti-S100 immunostaining techniques were useful tools for evaluating these astrocytic changes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil.
