Abstract
T-cell–rich, large B-cell lymphoma (TCRLBCL) is the most commonly diagnosed type of lymphoma in horses. Here we describe the clinical signs, neuropathology, immunohistochemistry (IHC), and PCR for antigen receptor rearrangement (PARR) analysis results of a TCRLBCL in the brain of an 8-y-old male Quarter Horse that was euthanized after acute anorexia, tremors, head pressing, falling, blindness, incoordination, and seizures. Autopsy revealed a firm, smooth, pale-yellow mass that expanded both lateral ventricles and the adjacent subcortical white matter. Histologically, the mass consisted of a densely cellular neoplasm composed of large, CD79+ neoplastic B-lymphocytes admixed with sheets of small, CD3+ reactive T-lymphocytes, Iba1+ histiocytes, MUM1+ plasma cells, and rare eosinophils supported by a fine fibrovascular stroma. Formalin-fixed, paraffin-embedded tissue scrolls were retrieved and subjected to PARR analysis, which revealed a clonal reaction in the immunoglobulin gene and a polyclonal reaction for the T-lymphocyte receptor gene, consistent with a neoplastic B-lymphocyte and reactive T-lymphocyte proliferation. The diagnosis of TCRLBCL was suspected histologically and confirmed based on IHC and PARR analysis.
Lymphoma is the most commonly diagnosed hematopoietic neoplasm of horses worldwide, with an overall incidence of 1–3% of all equine neoplasms. 12 The age of affected patients varies, but most cases occur in adult horses. 2 No sex or breed predisposition has been reported to date.2,12 The majority of equine lymphomas are multicentric, but cutaneous and gastrointestinal lymphomas are also common.2,12
According to the veterinary adaptation of the World Health Organization (WHO) classification system for human lymphoma, most equine lymphomas are T-cell–rich, large B-cell lymphomas (TCRLBCL) and typically occur as subcutaneous nodules.2,4,9,10 TCRLBCL is characterized by large neoplastic B-lymphocytes that have irregular nuclei with coarse chromatin and prominent nucleoli admixed with sheets of small, reactive T-lymphocytes and histiocytes supported by a fibrous connective stroma. 4 Although TCRLBCL is a slowly progressive lymphoma in cats and dogs, that feature has not been confirmed in horses. 2 As with other animal species, equine CNS lymphomas are rare and can occur as part of multicentric lymphoma or as primary telencephalic or spinal lymphoma.2,6,7 According to our search of PubMed, CAB Direct, Web of Science, Scopus, and Google, 3 cases of TCRLBCL with CNS and peripheral nervous system (PNS) involvement have been reported in horses.6,15
Although tumor morphology and immunohistochemistry (IHC) typically suffice for a diagnosis of TCRLBCL, their utility may be limited because of the mixed lymphocytic population characteristic of these neoplasms. 11 In such cases, clonality detection by PCR should be indicated to confirm the diagnosis. 14 Here we describe the clinical signs, neuropathology, and IHC features of a case of TCRLBCL in the brain of a horse that was confirmed using PCR for antigen receptor rearrangement (PARR) analysis. Although the use of PARR has been described as an adjunct diagnostic test for lymphoma in horses and a donkey, the methods have not been described in detail in the literature.1,5,8
An 8-y-old male Quarter Horse developed anorexia of acute onset. A few hours later, the horse ran uncontrollably into a fence and was placed in a stall overnight for observation. The patient subsequently developed tremors, head pressing against the wall, and constant falling. During physical examination the next day, the horse was blind and had incoordination and seizures. Euthanasia was elected because of the poor prognosis and suspicion of an infectious disease (particularly rabies).
An autopsy revealed a firm, smooth, pale-yellow, 3 × 2.5 × 6.5-cm mass that expanded both lateral ventricles and the adjacent subcortical white matter at the level of the right thalamus. Pathologic changes were not observed in the other organs. The main differential diagnoses for the solid intraventricular mass during autopsy included a cholesterol granuloma (or other types of granulomas), lymphoma, and, although rarely described in horses, a choroid plexus or ependymal neoplasm. 3 A cross-section of cerebellum and brainstem was submitted for fluorescent antibody testing (FAT) for rabies virus according to the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, GA, USA) procedures for rabies-suspect cases. The remaining brain tissue was fixed in 10% neutral-buffered formalin. Given the absence of gross changes in extraneural tissues, only brain was examined histologically. Sections of the intraventricular mass, as well as frontal, parietal, temporal, and occipital telencephalon, basal nuclei, thalamus, hippocampus, mesencephalon, cerebellum, and pons, were processed routinely for histology and stained with H&E.
FAT for rabies was negative. Histologically, the telencephalic mass consisted of a densely cellular neoplasm that occluded the lateral ventricles but also infiltrated the adjacent white matter (Fig. 1). Although the ventral ependymal lining could be visualized, most of the ependyma was effaced by the neoplasm. Neoplastic cells consisted of scattered large neoplastic lymphocytes, as well as admixed sheets and cords of reactive small lymphocytes and histiocytic cells, with fewer plasma cells and eosinophils supported by a fine fibrovascular stroma (Fig. 2). Neoplastic cells had scant eosinophilic cytoplasm with distinct margins and round-to-oval, large nuclei with coarse chromatin and 1 or 2 prominent nucleoli. Binucleate or multinucleate cells were distributed throughout (Figs. 3, 4). The mitotic count was 7 in 2.37 mm2 (10 FN22/40 × fields). Reactive lymphocytes had scant cytoplasm and small round nuclei with dense chromatin. The adjacent neuroparenchyma was compressed and edematous with areas of gliosis.
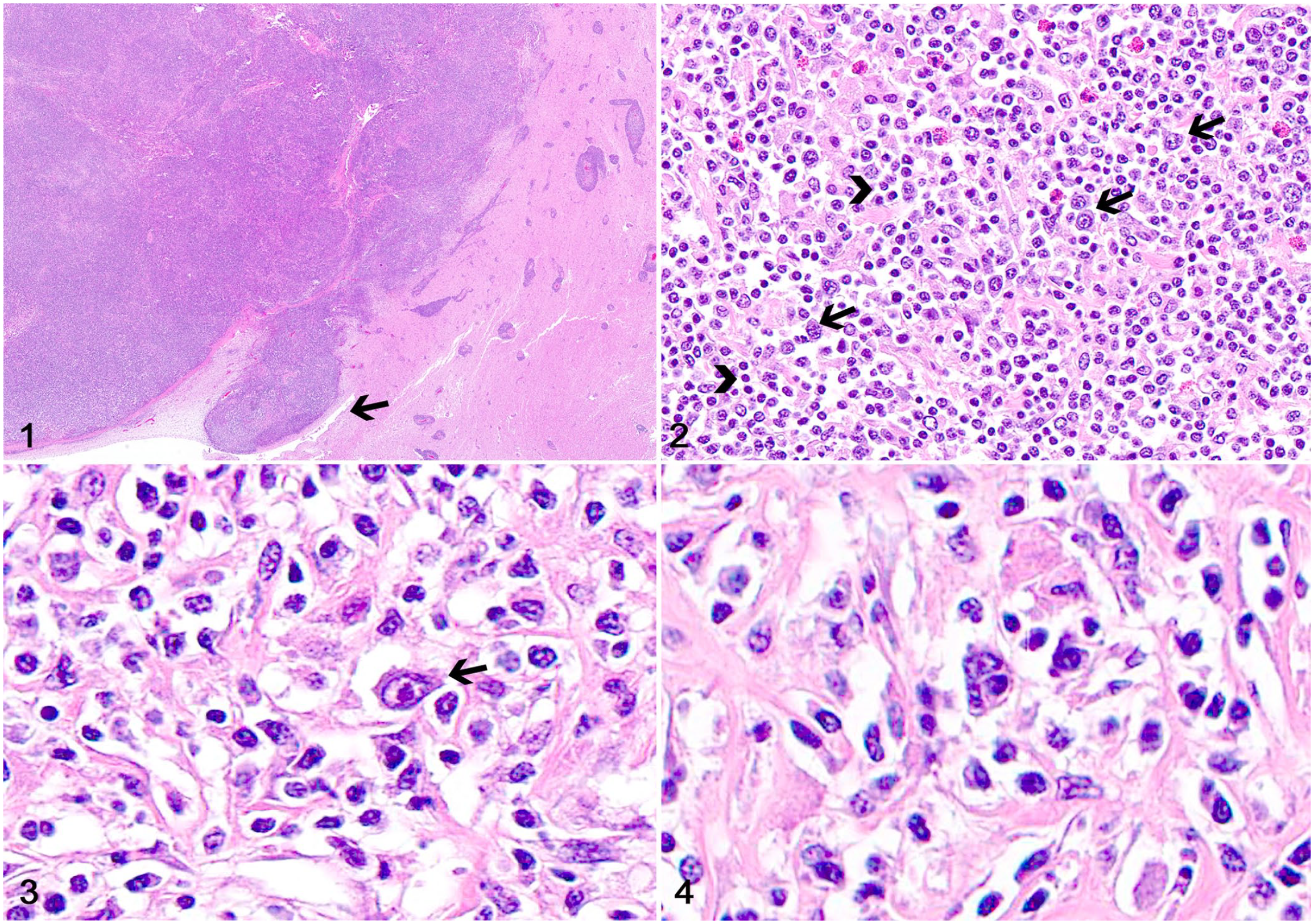
T-cell–rich, large B-cell lymphoma in the brain of a horse.
A TCRLBCL was suspected after histologic evaluation, and tissue sections were subjected to IHC for CD79, CD3, Iba1, and MUM1 for diagnostic support (Table 1). Large neoplastic lymphocytes had robust membranous immunolabeling for CD79 (Figs. 5, 6). Reactive intratumoral lymphocytes had strong membranous immunolabeling for CD3 (Fig. 7), reactive histiocytes had strong cytoplasmic immunolabeling for Iba1 (Fig. 8), and reactive plasma cells had strong nuclear immunolabeling for MUM1.
Details of immunohistochemical stains performed on sections of a T-cell–rich, large B-cell lymphoma in the brain of a horse.

NA = not applicable. In all stain reactions, the chromogen used was diaminobenzidine (DAB), and the autostainer was a Nemesis 3600 (Biocare).
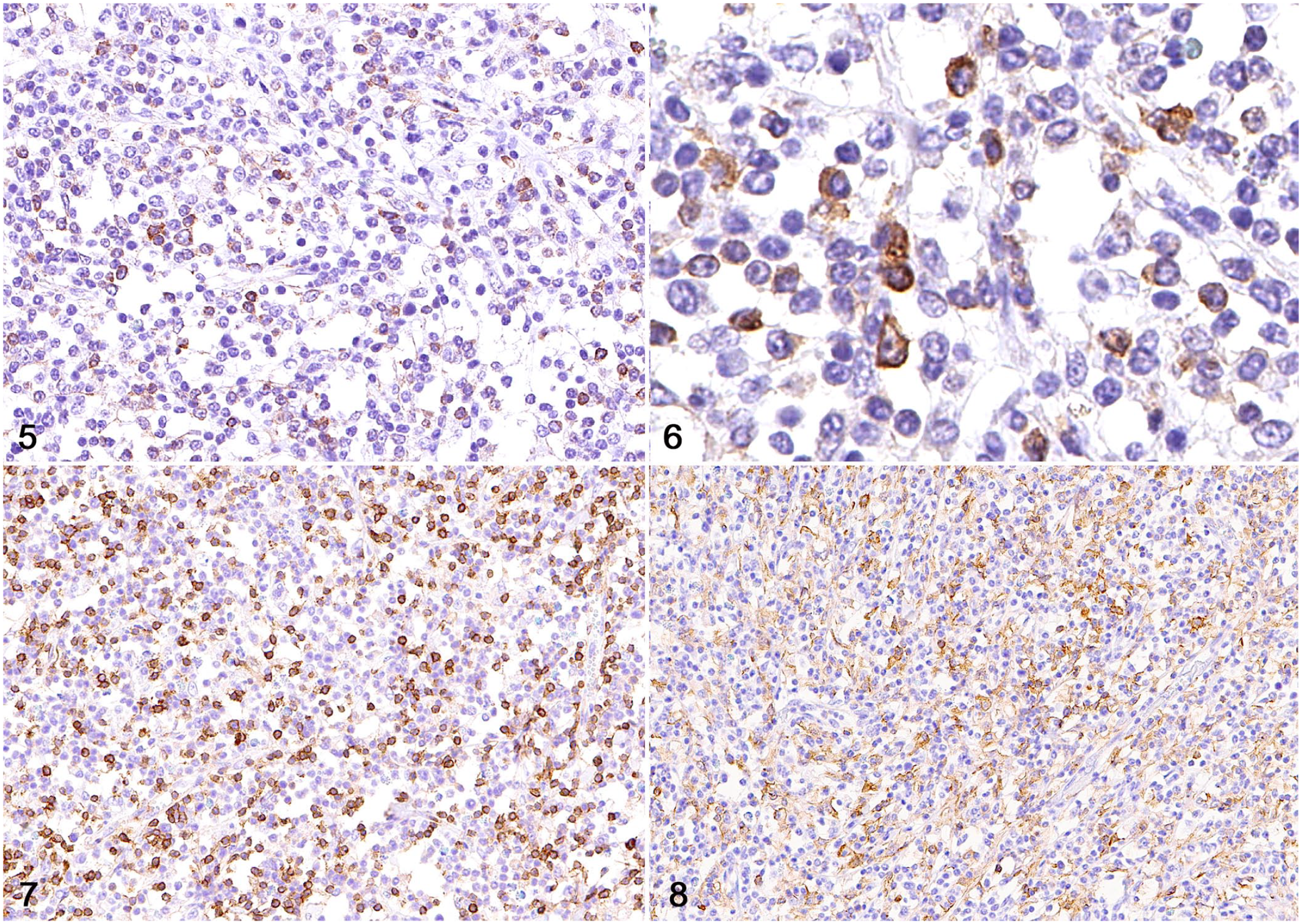
T-cell–rich, large B-cell lymphoma in the brain of a horse.
Formalin-fixed, paraffin-embedded (FFPE) tissue scrolls were retrieved and subjected to PARR analysis for diagnostic confirmation (Suppl. Table 1). For the equine PARR assay, primers were designed to amplify horse immunoglobulin (IG) and T-cell receptor gamma (TRG) gene rearrangements and were grouped into 2 primer mixes for PCR, one for amplifying IGH-VDJ, IGH-DJ, and TRG rearrangements (primer mix 1) and another for IGK-VJ, IGL-VJ, and kappa deleting element (KDE) rearrangements. Gene sequences were obtained from the International ImMunoGeneTics Repertoire (IMGT; www.imgt.org) and the Equus caballus genome (assembly EquCab3.0). Gene sequences for all relevant antigen receptor loci were available from IMGT except for TRG. To obtain these sequences, TRGV, TRGJ, and TRGC genes were retrieved from and mapped in EquCab3.0 using BLAST searches with a variety of mammalian variable, joining, and constant gene queries. Briefly, the locus, located on chromosome 4, contained 16 V-J-C cassettes and was flanked by the AMPH and STARD3NL genes on the 5′ and 3,′ respectively. These non-TRG genes flank all 11 mammalian TRG loci annotated in IMGT. Each cassette contained 1–6 TRGV genes, 1 or 2 TRGJ genes, and 1 TRGC gene. In total, there were 47 TRGV genes belonging to 5 subgroups and 14 TRGJ genes.
To design PCR primers, the amassed variable and joining gene sequences were aligned, and primers were designed within short, conserved regions. Alignments were conducted with Geneious (www.geneious.com), and primer sequences were designed with the Primer3Plus web interface. 13 To perform the PCR assay, genomic DNA was extracted from FFPE scrolls prepared from the telencephalic mass (ReliaPrep FFPE gDNA miniprep system; Promega). One microliter of DNA was amplified (Multiplex PCR master mix kit; Qiagen) with 3.2 pmol of each primer in 20-μL reactions (1.6 pmol of VL10v2 and 6.4 pmol of HKdef1 and HKder1). The optional Q-Solution was used in both reactions at 0.5×. The cycling conditions were as follows: initial denaturation at 95°C for 15 min; then 40 cycles of 94°C for 30 s, 61°C for 30 s, and 72°C for 90 s; then a final extension of 72°C for 30 min. PCR products were resolved (3130xl system, Applied Biosystems; DS-33 dye set, Thermo Fisher). The PCR product was diluted 10-fold, and 1 µL was added to 14.5 µL of HiDi formamide and 1 µL of GS600LIZ size standard. Data analysis was carried out with GeneMarker (Soft Genetics). The PARR assay revealed clonally rearranged IGH-VDJ and IGK-VJ genes in the telencephalic mass DNA (Fig. 9) and polyclonal rearranged T-lymphocyte receptor gamma genes, lambda light chain genes, and KDE (not shown). These findings were consistent with a neoplastic B-lymphocyte population and a reactive T-lymphocyte population.
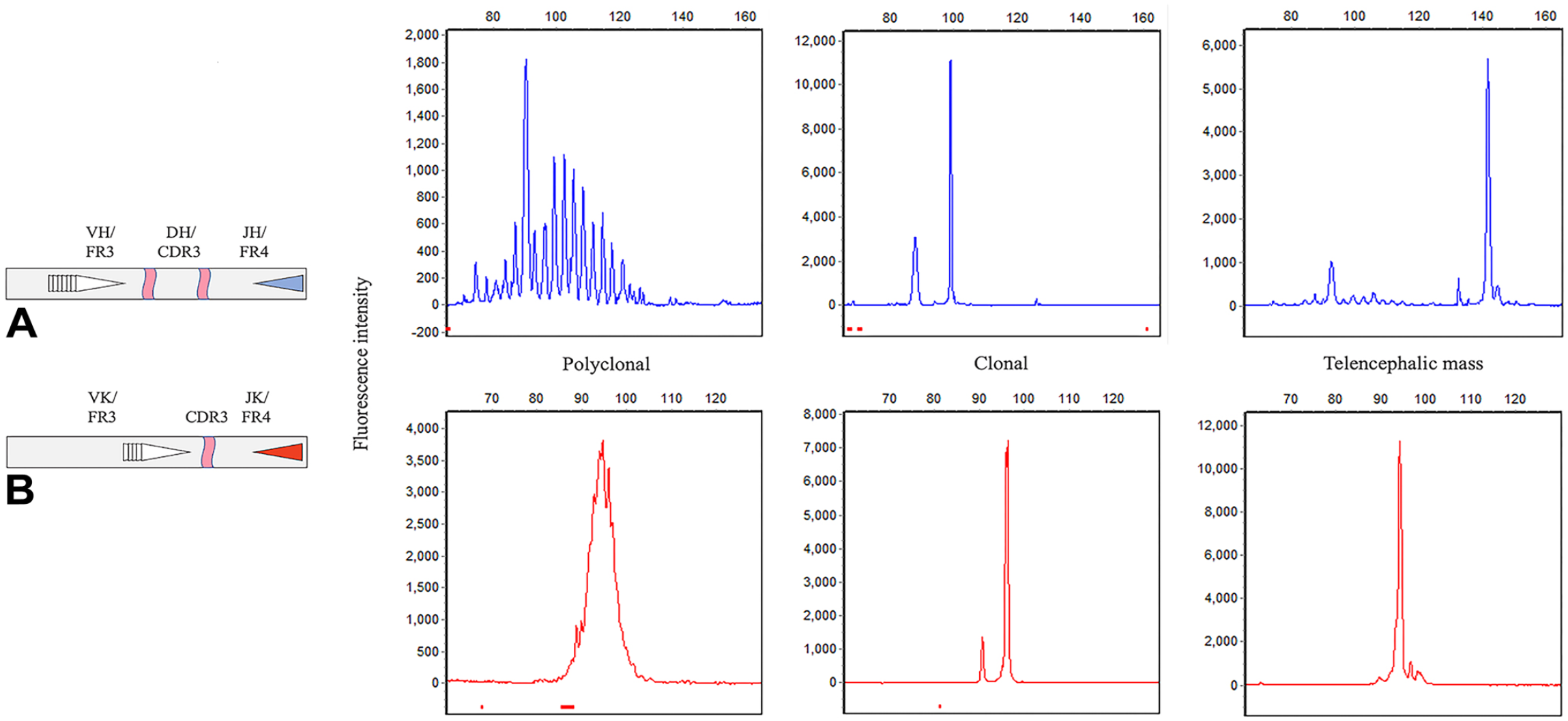
PARR analysis for IGH-VDJ and IGK-VJ rearrangements. IGH-DJ, TRG, IGL-VJ, and KDE rearrangements in the telencephalic mass DNA had polyclonal distributions (not shown).
The gross features of the telencephalic tumor in our case were similar to those reported in a multicentric TCRLBCL with CNS involvement and those typically described for equine lymphomas.6,12 It was not possible to determine whether our case was a primary telencephalic TCRLBCL or a systemic lymphoma with CNS involvement because only brain tissue samples were collected and examined histologically. However, the sudden onset of neurologic clinical signs and the absence of extraneural lesions during the autopsy suggest that the neoplasm was a primary CNS lymphoma. 15 Regardless of its neuroanatomic distribution, equine lymphoma with CNS involvement accounts for ~2% of all equine lymphomas. 2 TCRLBCL with CNS involvement are exceedingly rare.6,15 In addition, PNS lymphoma has been reported in horses, 6 and some of the clinical signs reported in our case could be attributed to infiltration of nerves by neoplastic cells. However, no nerve tissues were examined histologically, thus such infiltration could not be confirmed.
The histologic findings in our case were highly suggestive of TCRLBCL, 2 but initially raised the suspicion of an inflammatory lesion, as reported elsewhere. 11 Diagnostic confirmation was subsequently achieved by IHC and PARR. Our IHC highlighted neoplastic large CD79+ B-lymphocytes admixed with numerous small CD3+ T-lymphocytes and Iba1+ histiocytes, similar to what has been previously reported for TCRLBCL in horses.4,6,15 The PARR assay is routinely used as an adjunct diagnostic technique in canine and feline lymphoma, and 3 reports describe its application in equine lymphoma and leukemia, although the methods were not provided.1,5,8 Here we describe the use of the PARR assay for diagnostic confirmation of an equine lymphoma with detailed methods and utilized primers. Because the assay is newly developed, diagnostic sensitivity and specificity have not yet been determined. In our case, the PARR assay demonstrated clonal rearrangements of both the immunoglobulin heavy chain and immunoglobulin kappa light chain in support of the diagnosis of TCRLBCL.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387231164368 – Supplemental material for T-cell–rich, large B-cell lymphoma in the brain of a horse
Supplemental material, sj-pdf-1-vdi-10.1177_10406387231164368 for T-cell–rich, large B-cell lymphoma in the brain of a horse by Daniel R. Rissi, Anne C. Avery and Robert C. Burnett in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
