Abstract
Urine biomarkers are sensitive indicators of early-stage renal injury, consequently, research in this area is expanding in both human and veterinary medicine. However, studies investigating the impact of preanalytical factors, such as storage conditions, on urine biomarker concentrations are largely lacking in veterinary medicine. Therefore, we evaluated the stability of several renal injury biomarkers in canine urine after storage for 4 y at −72°C. Urine samples were collected from 26 dogs: 18 dogs with babesiosis and 8 healthy dogs. Concentrations of urine immunoglobulin G (uIgG), urine C-reactive protein (uCRP), and urine retinol-binding protein (uRBP) were measured, using validated commercial immunoassays, at the start of the study and 4 y later. To investigate the effect of long-term storage, absolute and relative differences between both measurements were compared. Additionally, dogs with babesiosis were compared with the healthy controls at both time points. Storage caused significant absolute and relative decreases in concentrations of all 3 biomarkers. Significant differences between dogs with babesiosis and healthy dogs were found in uIgG and uRBP at both times; however, the difference in uCRP between both groups lost significance after storage. Because the main goal of these urine biomarkers is to detect early-stage renal injury, the statistically significant decrease in their concentrations will be clinically relevant when a mild degree of renal injury is present. Our data indicate that the investigated urine biomarkers show significant decay after 4 y of storage at −72°C, adversely affecting their diagnostic utility.
The use of urine biomarkers to detect renal injury at an early stage is gaining interest in both human and veterinary medicine,3,8,13,14,24 given that identification of early renal injury allows earlier therapeutic intervention. 17 The latter is hampered by the traditionally used diagnostic markers of decreased kidney function, serum creatinine and urea, as their concentration will only increase above the upper limit of the reference interval when >75% of nephrons are lost, making them insensitive markers for the detection of early-stage renal injury.2,6 In contrast, urine biomarkers are generally accepted to be sensitive indicators of glomerular or tubular renal injury, and additionally have the capacity to quantify as well as localize this damage.3,17 In research settings, enzyme-linked immunosorbent assays (ELISAs) are often used to detect and quantify biomarker proteins. Immunoassays of several glomerular and tubular biomarkers of renal injury, including immunoglobulin G (IgG) and C-reactive protein (CRP), respectively, as well as retinol-binding protein (RBP; Immunology Consultants Laboratory, Portland, OR), have been previously validated in our laboratory for use in canine urine. 14
Consideration of preanalytical factors, such as sample handling and storage conditions, is important in order to perform reliable biomarker research. Because of prospective sampling, shipping, and performing analyses in batches, immediate analysis of fresh urine samples is impractical in most research settings. Therefore, storage of frozen samples for long periods (several months to years) is common. However, studies evaluating the stability of urine renal injury biomarkers during long-term storage are very scarce in veterinary medicine. Even in human medicine, relatively few studies have evaluated the impact of preanalytical factors on urine proteins.4,7 To our knowledge, only a single urine biomarker study, investigating the stability of RBP, albumin, and
Twenty-six dogs that were presented to the Onderstepoort Veterinary Academic Hospital, University of Pretoria, South Africa, were prospectively included after the approval of the Animal Use and Care Committee of the Faculty of Veterinary Science, University of Pretoria. The study group consisted of 18 dogs diagnosed with babesiosis, caused by
At admission, urine collection was performed by cystocentesis in all dogs. Routine urinalysis included a dipstick analysis (Combur 9 test, Roche Diagnostics, Mannheim, Germany), microscopic sediment analysis, evaluation of urine specific gravity by refractometry, urine protein-to-creatinine ratio, and bacterial culture. Quick centrifugation was performed after urine collection at 447 ×
SAS v. 6.4 (SAS Institute, Cary, NC) was used for data analysis. To investigate the effect of storage, the absolute difference between the measurements at T0 and T4 years was calculated, and we tested whether this difference was significantly different from zero using the Wilcoxon signed-rank test. The absolute difference was calculated twice, using biomarker concentrations that were unadjusted and adjusted to urine creatinine. The same statistical test was done for the relative difference between the measurements at T0 and T4 years (unadjusted to urine creatinine), which was defined as the absolute difference between the measurements at T0 and T4 years divided by the measurement at T0. Samples with a measurement of zero at T0 (i.e., biomarker concentration values that were either below detection limit or below quantification limit) were discarded because no decrease can be evaluated with such samples after storage. The results are also presented by Bland–Altman plots. To investigate whether the changes in urine biomarker concentrations after long-term storage would alter the study results of a previous publication, concluding that dogs with babesiosis had significantly higher uIgG, uCRP, and uRBP compared with healthy dogs,
5
dogs with babesiosis were compared with healthy control dogs separately for the results obtained at T0 and T4 years, using the Wilcoxon rank-sum test. These calculations were performed using biomarker concentrations adjusted to urine creatinine. Differences were considered statistically significant at
Absolute and relative differences between T0 and T4 years were statistically significant for all 3 measured biomarkers (Fig. 1). For the calculations unadjusted to urine creatinine, absolute uIgG decreased significantly from T0 to T4 years (
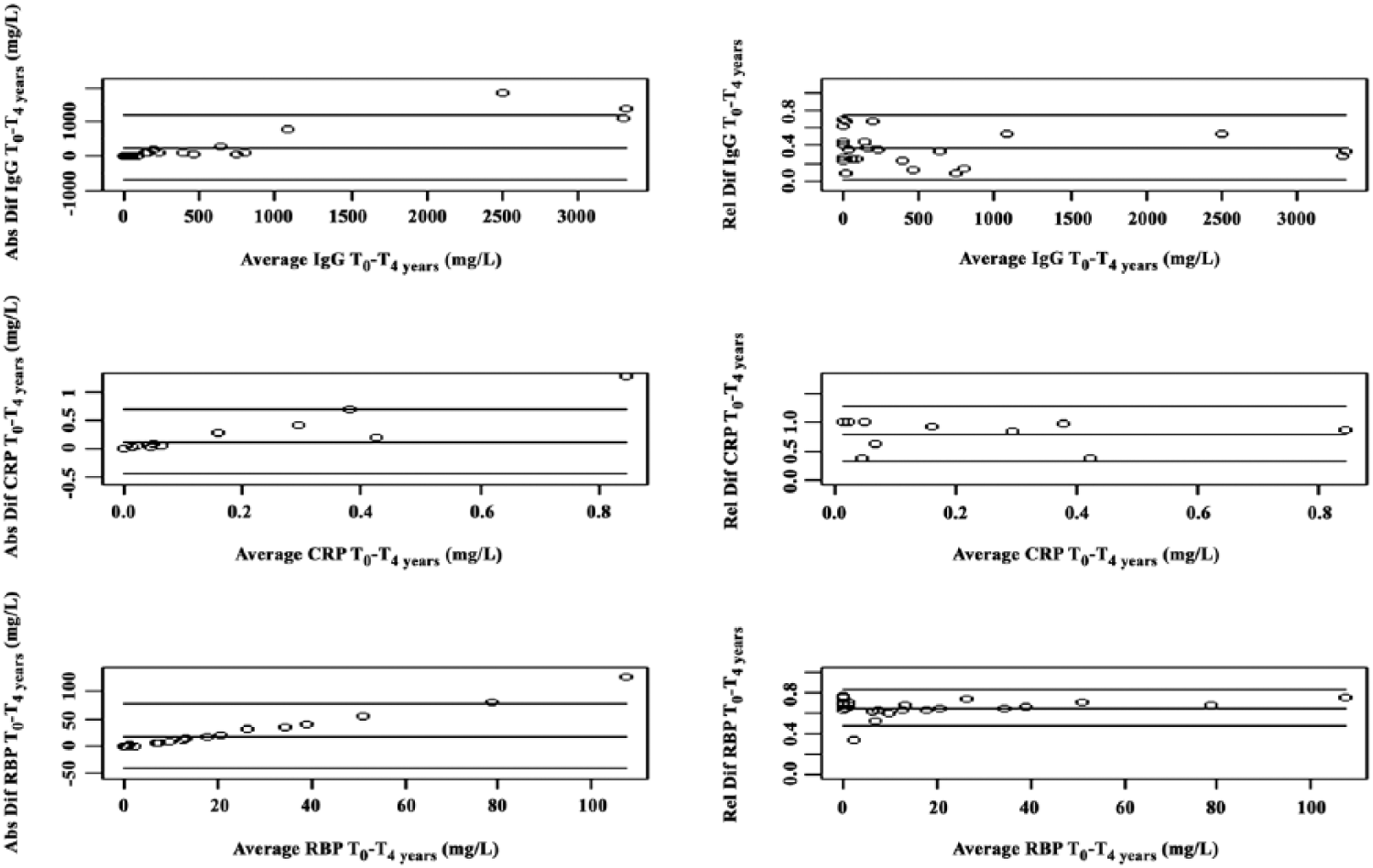
Absolute (Abs) and relative (Rel) differences (Dif) in urine biomarker concentrations immunoglobulin G (IgG), C-reactive protein (CRP), and retinol-binding protein (RBP), unadjusted to urine creatinine, after storage for 4 y at −72°C, presented by Bland–Altman plots. The middle horizontal lines represent the absolute and relative mean differences between T0 and T4 years. The upper and lower horizontal lines represent the 95% limits of agreement (mean difference ± 1.96 × standard deviation of the differences).
Urine biomarker concentrations (expressed in mg/g after normalization) in dogs with babesiosis (
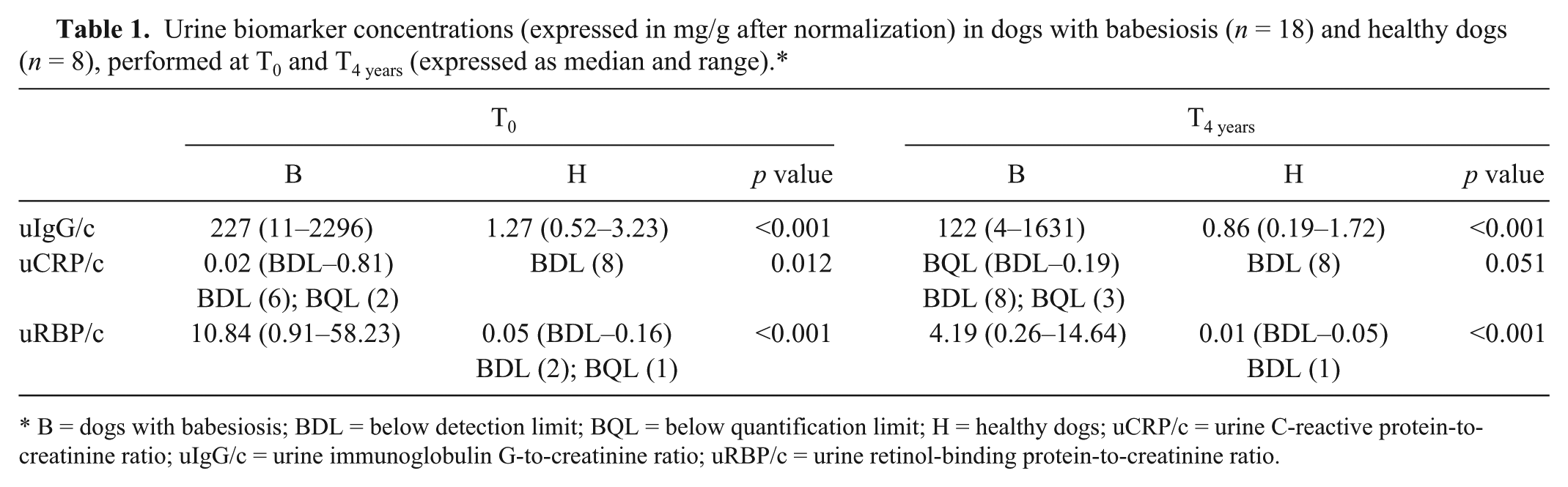
B = dogs with babesiosis; BDL = below detection limit; BQL = below quantification limit; H = healthy dogs; uCRP/c = urine C-reactive protein-to-creatinine ratio; uIgG/c = urine immunoglobulin G-to-creatinine ratio; uRBP/c = urine retinol-binding protein-to-creatinine ratio.
Statistically significant decreases in all 3 measured urine biomarker concentrations were observed after 4 y of −72°C storage. The absolute decrease was highest for uIgG, intermediate for uRBP, and lowest for uCRP. This ranking can at least partly be attributed to the inherent differences in absolute concentrations of each biomarker in dogs with babesiosis. However, when evaluating the relative decreases, these were ranked vice versa, being highest for uCRP, and lowest for uIgG. Finally, the variation of the latter difference was highest for uCRP, and lowest for uRBP. The Bland–Altman plots illustrate the consistent bias that exists between the measurements at T0 and T4 years (i.e., consistently lower values at T4 years). Additionally, no significant proportional bias could be identified for any of the biomarkers because similar absolute and relative mean differences were observed for low versus high average concentrations. The decrease, found for all biomarkers, cannot be explained by assay variation based on their previously described interassay CV, 14 and based on the consistent decrease of all measured values. Although the same conclusions were made for uIgG and uRBP at T0 and T4 years (i.e., significantly higher concentrations in dogs with babesiosis compared with healthy controls), conflicting conclusions were made for uCRP at T0 and T4 years. A loss in statistical significance occurred only for uCRP after long-term storage because the difference between the diseased and the healthy population at T0 was much smaller for uCRP compared with uIgG and uRBP. The higher relative decrease in uCRP compared with the other 2 biomarkers further contributes to the latter finding. In populations in which renal injury is subtle, the statistically significant decrease in concentrations of all 3 biomarkers after long-term storage will be clinically relevant. Therefore, because the main goal of urine biomarkers is to identify renal injury at an early stage, our data strongly indicate that long-term storage before analysis should be avoided. This is mainly relevant for biobanking and research settings, in which long-term storage is commonly performed.18,20
Of the 3 biomarkers evaluated, only uRBP has been previously tested for stability in canine urine. 22 In the latter study from our group, no significant changes in uRBP were found after storage for up to 1 y at −80°C. However, our results indicate that after much longer storage, stability of uRBP is severely affected. In humans, several studies investigated the stability of uIgG and uRBP during frozen storage.9,10,15,21,23 Results of these studies are apparently conflicting yet hard to compare given differences in storage conditions and analytical methods. Urine IgG, measured by ELISA, was not stable after 9 wk of −20°C storage in undiluted urine. 10 When measured by immunoturbidimetry, a significant decrease in uIgG was found after 6 mo of storage at −20°C. 23 Another study reported that uIgG, measured by nephelometry, decreased significantly during storage at −20°C, but remained stable during 2 y of −70°C storage. 9 When stored at −70°C for longer than 4 mo, uRBP, measured by ELISA, started to decrease significantly in one study. 15 However, another study reported no significant changes in uRBP, although also measured by ELISA, when stored at −70°C for 8 mo. 21
The main limitation of our study is the absence of biomarker analyses at intermediate time points to document the decay of these biomarkers over time in order to determine the maximum recommended storage time. Biomarker concentrations at T0 and T4 years were indexed to urine creatinine concentrations obtained at T0, assuming that urine creatinine remained stable during storage. Long-term stability studies showed that urine creatinine is very stable in human urine. 18 Although similar long-term studies have not been performed in canine urine, a previous study in dogs did show urine creatinine concentrations to be stable at −20°C for at least 3 mo. 19 Another limitation of our study is that an additional freeze–thaw cycle occurred at T4 years compared with T0, which might have influenced our results. One study evaluating the effect of multiple freeze–thaw cycles on several renal injury biomarkers in human urine 20 showed a statistically significant, but clinically insignificant, decrease in concentration of all biomarkers after 3 freeze–thaw cycles. The authors concluded that reusing urine samples undergoing up to 3 freeze–thaw cycles resulted in only a minimal decrease of the tested biomarkers, so retesting could be performed with excellent stability. 20 The effect of multiple freeze–thaw cycles was also assessed in a previous study on renal injury biomarkers in canine urine. 16 The study showed a statistically significant, but clinically insignificant, increase in urine NAG activity after 4 freeze–thaw cycles; urine neutrophil gelatinase-associated lipocalin concentrations (uNGAL) were not significantly affected by up to 4 freeze–thaw cycles. Although it should be emphasized that the effect of multiple freeze–thaw cycles has never been evaluated for canine uIgG, uCRP, and uRBP to our knowledge, we presume that 1 additional freeze–thaw cycle is highly unlikely to be a relevant confounding factor. Our results also only apply to urine samples stored at −72°C. At −20°C, long-term stability of urine proteins is expected to be shorter. Several human studies have demonstrated that urine proteins are significantly underestimated after freezing at −20°C.1,9,15,21 Measurement of urine proteins on specimens stored at −70°C is recommended whenever analysis of fresh urine samples is not feasible.9,12,21
Many studies investigating renal injury biomarkers in human and canine urine fail to mention the time interval between urine collection and biomarker analysis, although storage temperature is usually specified. Our results emphasize the importance of reporting these preanalytical factors.
Footnotes
Acknowledgements
We thank Kristel Demeyere and Jorien De Loor for assisting with the ELISA analyses.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
