Abstract
C-reactive protein (CRP) is a major, acute-phase protein in dogs; however, there is a need for automated assays to ensure in-time patient monitoring. Three automated immunoturbidimetric assays (Randox, Thermo, and Wako) developed for human beings were evaluated for their ability to detect canine CRP, including method validation, evaluation of diagnostic use, and establishment of exploratory reference intervals. Sera from 36 healthy dogs and 82 diseased dogs were included for method comparison with the enzyme-linked immunosorbent assay (ELISA; Tridelta) serving as the reference method. A nonparametric estimate of the 1-sided 95% reference interval was established (
Introduction
C-reactive protein (CRP) is well known as a major, acute-phase reactant in dogs and is part of the increasing field of research in acute-phase proteins. 5 Infection, inflammation, or tissue damage lead to an increased formation of proinflammatory cytokines, such as interleukins (IL-6 or IL-1), and subsequent synthesis of acute-phase proteins, mainly in the liver. 12,46 C-reactive protein is an opsonin and is, therefore, a member of the classical complement pathway of the host. 33 It induces a protective effect by several potential mechanisms including down-regulation of the inflammatory response, opsonization of apoptotic or necrotic cells, 32,34 and binding to bacterial proteins. 37 In dogs, increased CRP concentrations have been observed in a wide variety of different diseases and conditions, including infectious diseases, 24,26,27,28,35,43,44,49,50 immune-mediated diseases, 1,15,23 neoplasia, 31,36,38,47 and inflammatory bowel disease, 29 as well as after surgery 8,11,21 or after experimental injury. 2
Over the years, many assays have been developed for the detection of canine CRP. These have included immunodiffusion assays, 52 a time-resolved immunofluorimetric assay, 39 capillary tests, and slide reversepassive latex agglutination tests. 20,30,45 The test described by McGrotty is available as a rapid in-house test for general veterinary practice. Currently, a canine-specific enzyme-linked immunosorbent assay (ELISA) a is frequently used as a substantially validated reference method 18 ; however, ELISAs are labor intensive if performed without an automated ELISA processing system. Additionally, high between-run imprecision restricts the use of this ELISA. 18,25 To avoid these disadvantages and to ensure prompt determination of acute-phase protein concentrations, rapid, automated detection methods for human specimens have been developed for use with biochemical analyzers. In contrast to human CRP, canine CRP is glycosylated in 2 of 5 subunits 4 that make the molecules heterogeneous and that may impair their detection using human CRP tests. Thus, some human tests failed to detect canine CRP. 10 Few studies have reported the use of automated immunoturbidimetric assays for CRP determination in dogs, including 2 human tests obtained from Bayer b , 16 and Olympus, c , 3 as well as 2 canine-specific tests. 9,10 Use of a species-specific test would be preferable, but unfortunately, one of the tests 10 is no longer marketed commercially and the other 9 is not commercially available.
The Bayer CRP turbidimetric immunoassay b (hereafter, Bayer assay) for human beings has been proven to reliably detect canine CRP, 16 but Bayer no longer produces this assay. The same antibody, however, is used in the Randox CRP test d (Randox Laboratories Ltd., personal communication, 2010). In 2008, the Olympus human immunoturbidimetric assay c was used to detect CRP in dogs with hyperadrenocorticism. 3 Although excellent correlation to the canine-specific ELISA a and acceptable intra-assay and interassay coefficients of variation (CV) of <10% were reported for the Olympus assay, 3 full assay validation was not detailed by the authors because that was not the primary aim of their investigation.
In the current study, 3 automated immunoturbidimetry CRP assays d–f originally developed for use with human specimens were evaluated for use with canine serum samples. Test evaluation included method validation, assessment of clinical validity (i.e., the capacity of the tests to differentiate between dogs suffering from various diseases and healthy dogs), and establishment of exploratory reference intervals for the tests.
Materials and methods
Study design
The study was carried out between August 2007 and April 2008. Three automated CRP assays developed for human beings were evaluated in the present study. The first assay from Randox Laboratories d (hereafter, Randox assay) consisted of a high-linearity test using an unspecified-anti-human CRP antibody. The second assay from Thermo Clinical Labsystems e (hereafter, Thermo assay) used a goat–anti-human CRP antibody. The third test kit from Wako Chemicals f (hereafter, Wako assay) used an ovine–anti-CRP antibody. The manufacturers of the 3 tests did not clarify whether a monoclonal or a polyclonal antibody was used. All 3 CRP assays were run on an automated biochemical analyzer g according to the manufacturers' recommendations and were calibrated with test-specific calibration material. Because no test-specific material for internal quality control was available for the Wako assay, the Randox control was used.
The study was divided into 3 parts, beginning with method validation. Method validation included evaluation of linearity and assay precision and comparison of the new methods to the reference method (canine CRP ELISA a ), which was previously validated. 18 In the second part of the study, clinical validity was tested by comparing CRP concentrations of dogs with different diseases to those obtained in healthy dogs. In the third part of the study, exploratory reference intervals were established if all previous examinations were acceptable.
Method validation
The 15 consecutive observations from serum samples with similar high, medium, and low CRP levels were included for assessment of interassay repeatability. Serum samples with 3 different CRP levels (high: >45 mg/l; medium: ∼15 mg/l; low: ∼8 mg/l) were prepared with specimens obtained from 1 healthy and 4 diseased dogs; aliquots were frozen at −80°C until analysis. Only vials needed for each analytical run were thawed to avoid repetitive freeze–thaw cycles, thus minimizing a source of potential variation. For each level, standard deviation (SD) was calculated as the square route of the estimated variance components. In the next step, CV was calculated as (SD/ global mean) × 100 to evaluate imprecision of the assays.
Clinical validation
Eighty-two dogs presented to the Small Animal Clinic (Faculty of Veterinary Sciences, Justus-Liebig-University, Giessen, Germany) with different diseases were included in the study to evaluate CRP concentrations in various illnesses. Depending on the clinical signs, various diagnostic tests and procedures were performed to establish the final diagnosis. These included biochemical profiles, radiography, ultrasonography, endocrine tests, microbial tests, bronchoalveolar lavage, analysis of cerebrospinal fluid, cytology, and biopsy. Diseased dogs were assigned to the following groups based on their final diagnoses: neoplasia (
Automated CRP assays were performed on the day of sampling; subsequently, the serum samples were frozen at −80°C until ELISA analysis. The dogs included for calculation of reference intervals served as controls.
Establishment of reference intervals
Thirty-six clinically healthy, adult (>1 year old) dogs were sampled to establish reference intervals. The dogs comprised the following breeds: 8 Beagles, 3 German Shepherd Dogs, 2 Rottweilers, 2 Australian Shepherd Dogs, 2 Great Danes, 2 German Wirehaired Pointers, 2 Golden Retrievers, 1 Bernese Mountain Dog, 1 Saint Bernard, 1 Austrian black and tan hound (Austrian brandlbracke), 1 Flat-Coated Retriever, 1 Kuvasz, 1 Labrador Retriever, 1 Newfoundland, 1 German spaniel, 1 Weimaraner, and 5 mixed-breed dogs. Females were slightly overrepresented with 22 out of 36 dogs (15 intact, 7 spayed), whereas 14 out of 36 were males (9 intact, 5 neutered). The median age of the dogs was 2 years (range: 1–8 years). Dogs were regularly vaccinated and dewormed blood donors, staff-owned dogs, or healthy dogs presented for routine radiologic examination to screen for hereditary hip or elbow dysplasia at the Clinic for Small Animals (Faculty of Veterinary Medicine, Justus-Liebig University, Giessen, Germany). Special care was taken in the selection of the healthy dogs for study. All dogs were clinically healthy and had no recent history of acute illness. As CRP is a major acute-phase protein, which increases in a wide variability of acute inflammatory diseases, traditional markers of inflammation (i.e., white blood cell count [WBC], erythrocyte sedimentation rate [ESR], and fibrinogen) were determined to exclude dogs with underlying inflammation that was not detected by routine clinical examination. Therefore, inclusion criteria for study were an unremarkable physical examination; lack of increase in WBC, ESR, and fibrinogen concentration; and a hematologic profile within reference range. Erythrocyte sedimentation rate was performed via the Westergren methodology. j An automated hematology analyzer k was used for hematological analysis. Fibrinogen concentration was determined by the Clauss method, using a coagulometer. l
Statistical analysis
All data were evaluated for normal distribution using the Anderson–Darling test. m
The 95% 1-sided reference interval was calculated using commercial software.
m
Possible differences between females and males in the reference range population were investigated using the Mann–Whitney
Results
Method validation
All 3 CRP tests showed excellent linearity (
Sample volume of the sera available for the method comparison experiment was too low to perform duplicate measurements. After the ELISA analysis, 24 sera had to be excluded from the statistical analysis because results exceeded the upper limit of linearity of the ELISA (>15 mg/l). Because of the low sample volume, dilution of the samples and reanalysis with the ELISA was not possible. Thus, 94 samples were finally included in the statistical analysis. There was no significant difference between the median results obtained with the Randox assay and the ELISA (
The Bland–Altman difference plot revealed a mean proportional bias of 0.28 mg/l for the Randox assay, −0.59 mg/l for the Thermo assay, and 0.61 mg/l for the Wako assay. For all tests, the 95% limits of agreement were large, with a range of −12.64 to 13.21 mg/l (Randox vs. ELISA), −9.96 to 8.79 mg/l (Thermo), and −11.67 to 12.90 mg/l (Wako).
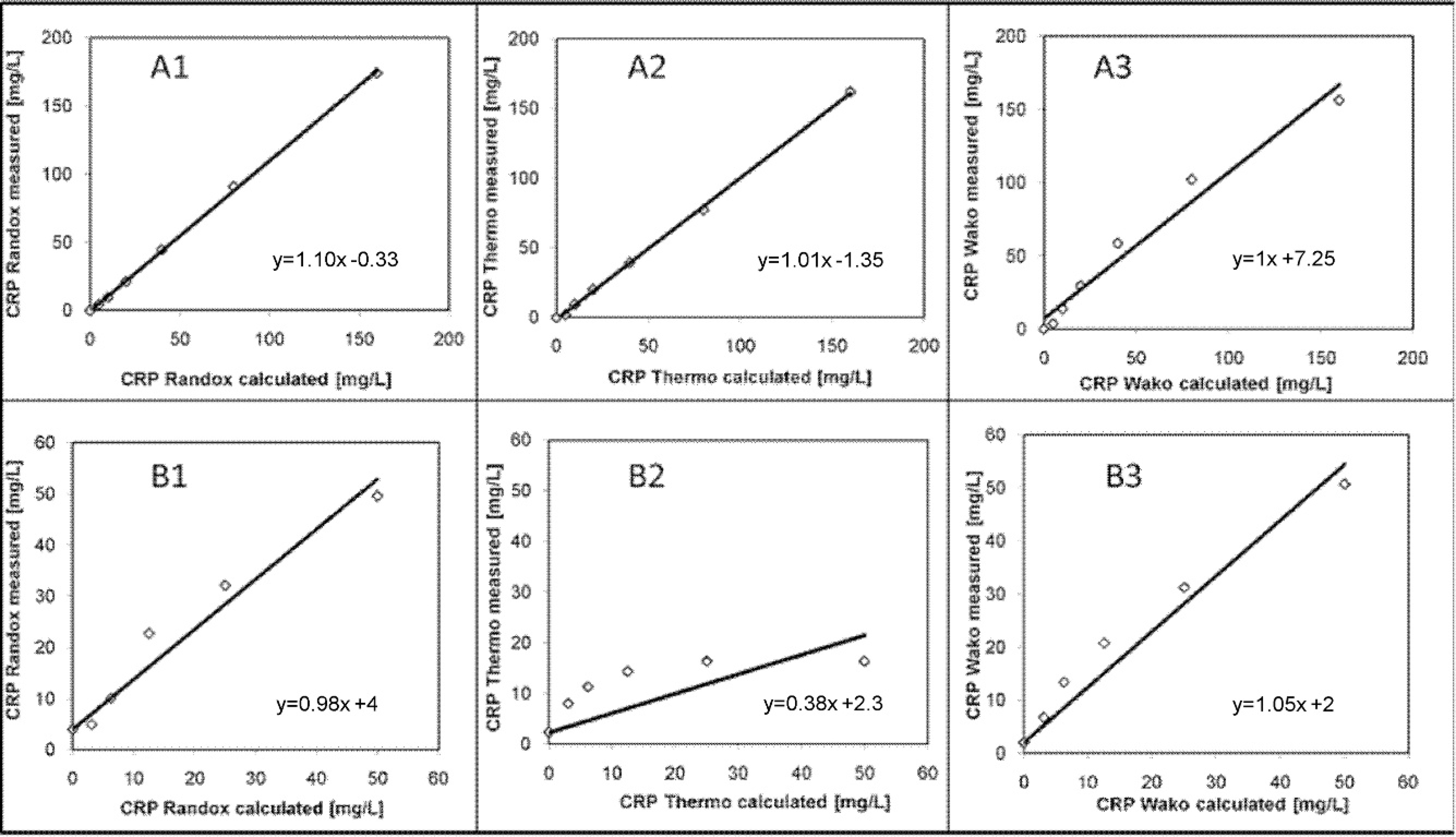
Linearity range of 3 immunoturbidimetric assays originally developed for human beings determined by serial dilution of a human calibration standard with a C-reactive protein (CRP) concentration of 153 mg/l (
Clinical validation
Data from the diseased dogs were not normally distributed. The 2-way ANOVA revealed a highly significant impact of the factor
Intra-assay repeatability of 3 automated immunoturbidimetric assays (Randox, Thermo, and Wako) originally developed for human beings for detection of a high (>45 mg/l), medium (∼15 mg/l), and low (∼8 mg/l) concentrations of canine C-reactive protein.* †
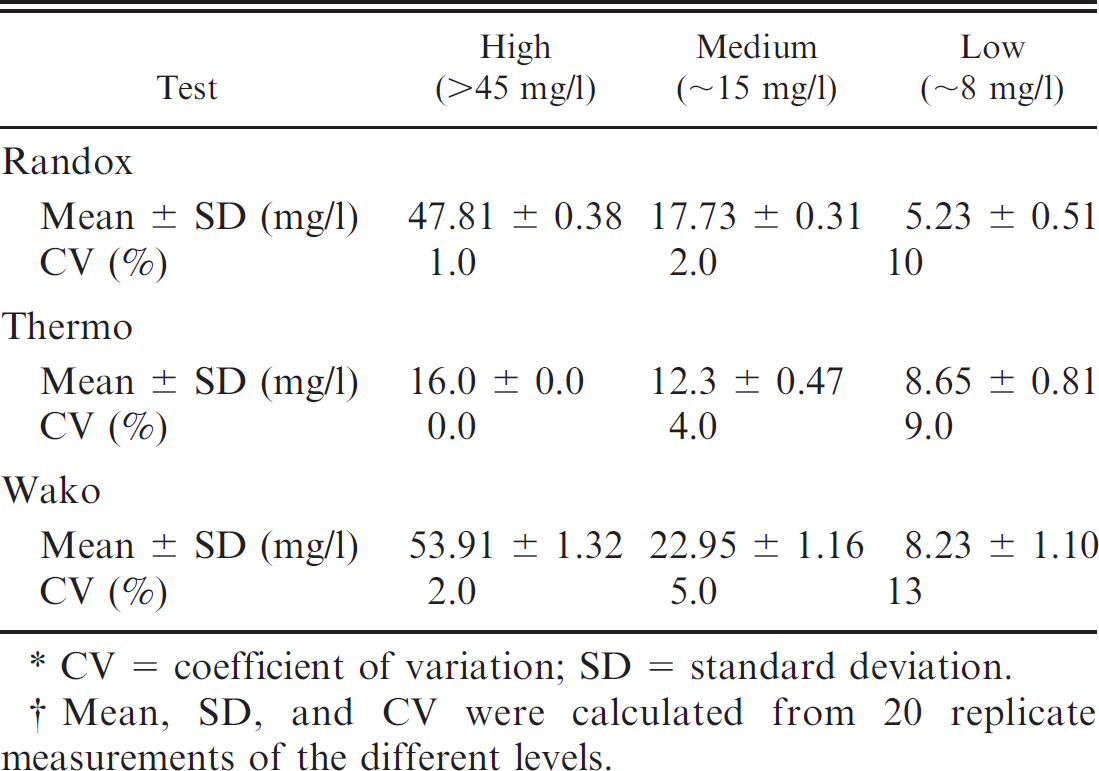
CV = coefficient of variation; SD = standard deviation.
Mean, SD, and CV were calculated from 20 replicate measurements of the different levels.
Interassay repeatability of 3 automated immunoturbidimetric assays (Randox, Thermo, and Wako) originally developed for human beings for detection of a high (>45 mg/l), medium (∼15 mg/l), and low (∼8 mg/l) concentrations of canine C-reactive protein.* †
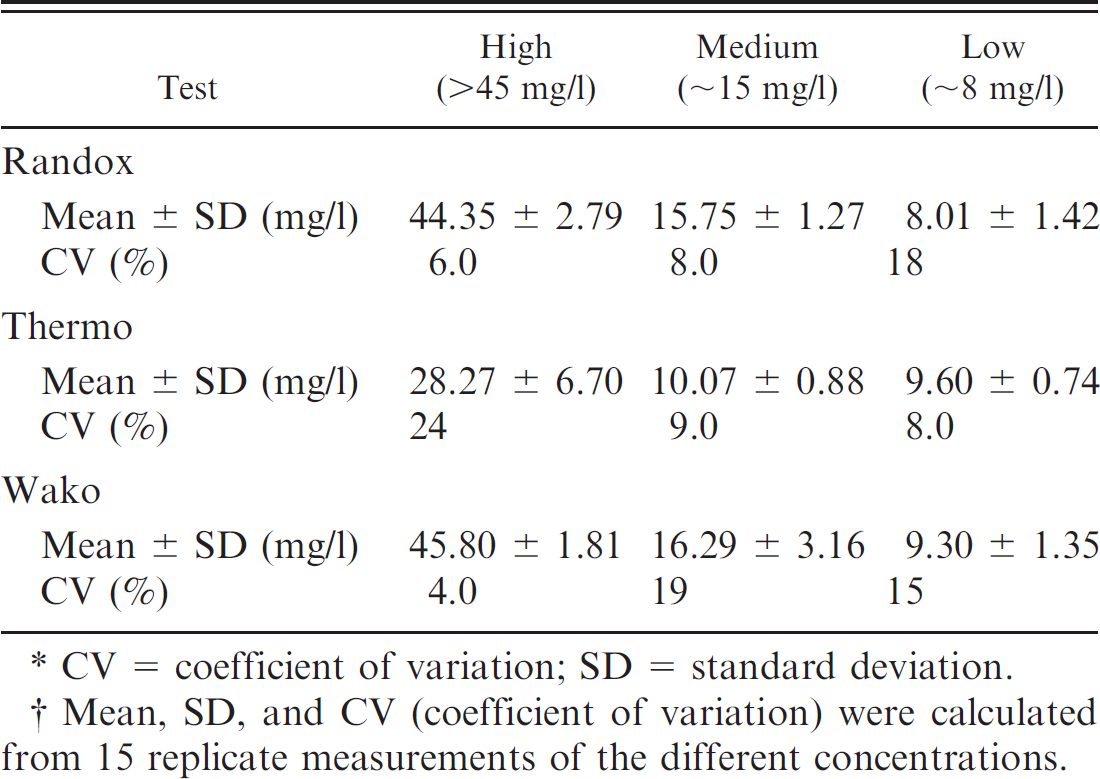
CV = coefficient of variation; SD = standard deviation.
Mean, SD, and CV (coefficient of variation) were calculated from 15 replicate measurements of the different concentrations.
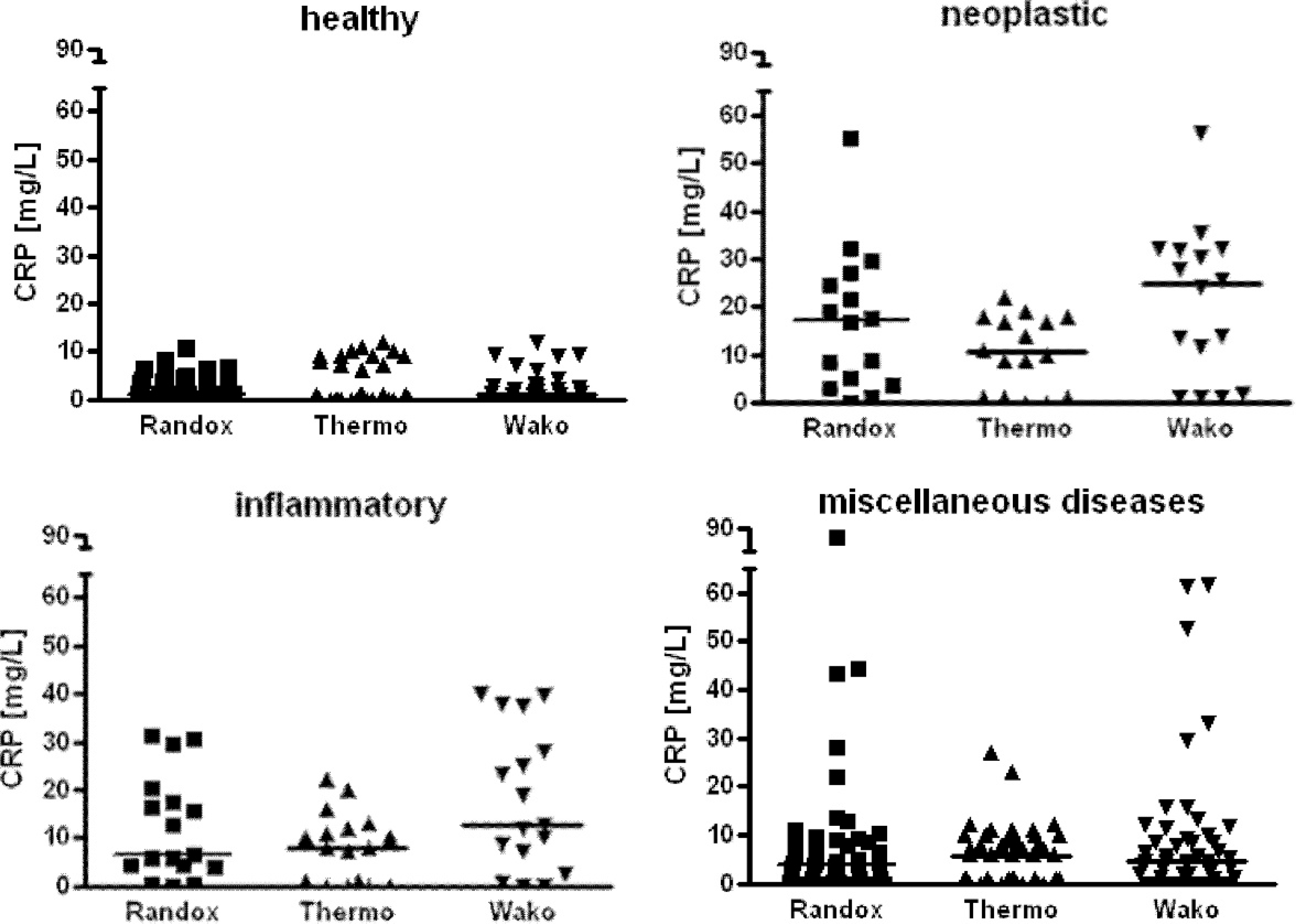
C-reactive protein (CRP) concentration measured with the Randox, Thermo, and Wako assays in healthy dogs (
Establishment of reference intervals
The Thermo assay was not included in the construction of reference intervals because data from the method validation were not acceptable. Data from the Randox and Wako assays showed nonnormal distribution even after logarithmic transformation. Descriptive statistics are shown in Figure 3. The upper reference limits were 6.8 mg/l CRP for the Randox assay and 9.9 mg/l CRP for the Wako assay. The Mann–Whitney
Median and range of the C-reactive protein (CRP) concentrations in healthy dogs and dogs with neoplastic, inflammatory, and miscellaneous diseases.*
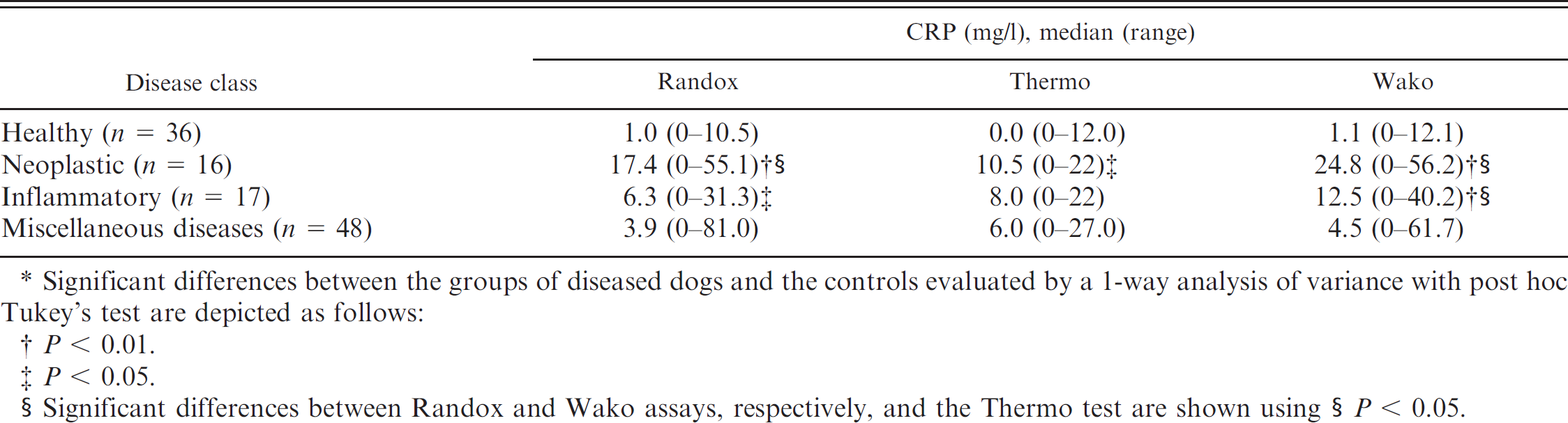
Significant differences between the groups of diseased dogs and the controls evaluated by a 1-way analysis of variance with post hoc Tukey's test are depicted as follows:
Significant differences between Randox and Wako assays, respectively, and the Thermo test are shown using §
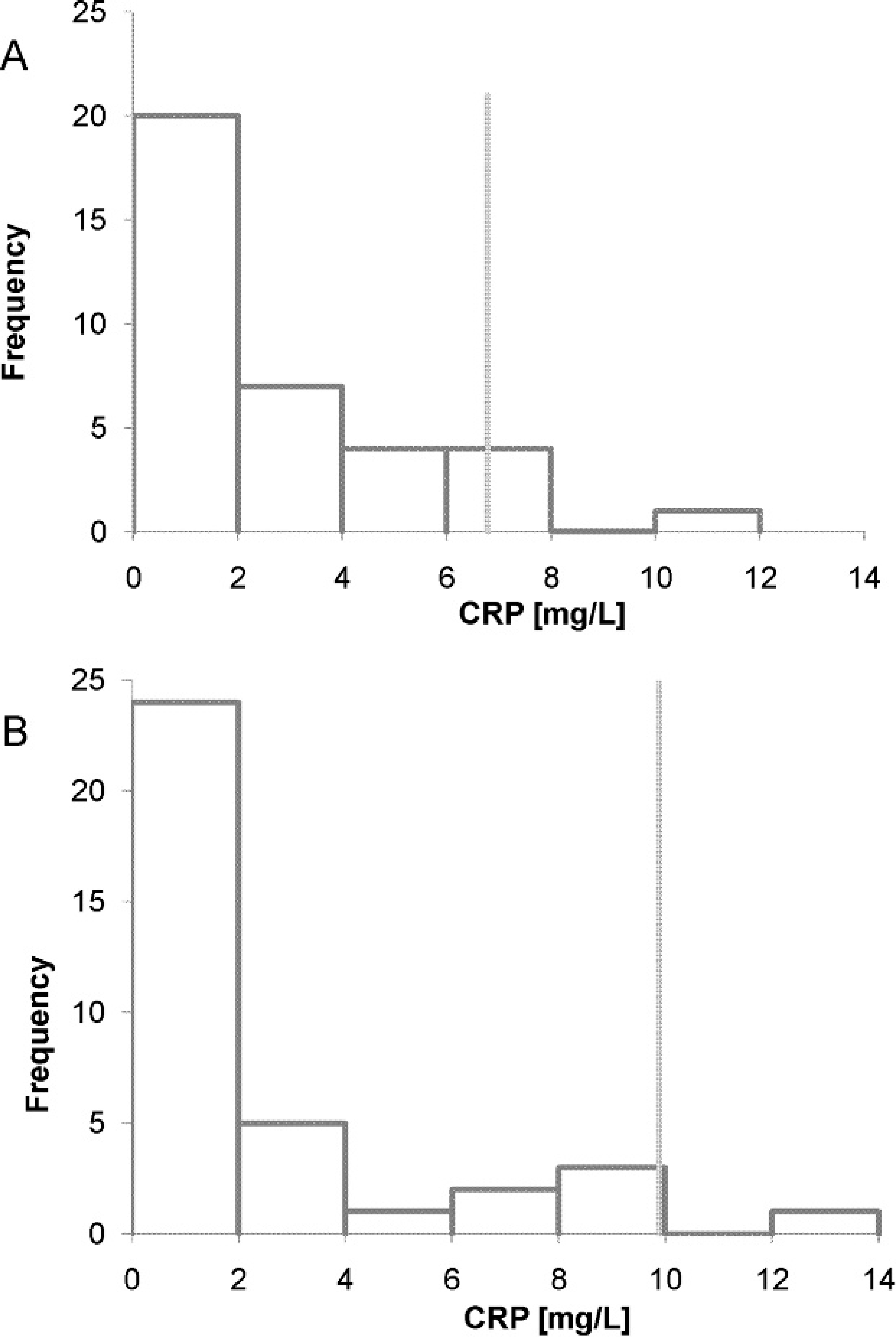
Frequency histograms demonstrating the distribution of data obtained for calculation of 95% 1-sided reference intervals (
Discussion
The Randox assay d (formerly Bayer b ; Randox Laboratories Ltd., personal communication, 2010) as well as a CRP test from Wako Chemicals f were able to detect canine CRP reliably. Linearity of the Randox and Wako assays was present over a range of 10–153 mg/l. In the present study, the limit of detection was not assessed because CRP concentrations below the reference interval are generally of no clinical relevance. Intra-assay CVs obtained for the Randox and Wako assays were comparable to the CVs reported in the veterinary literature for canine specimens (Bayer assay b CVs: 5.2–10.8% 16 ; Olympus assay c CVs: <10% 3 ; ELISA a CVs: 6.9–10.1% 18 ; previously developed CRP assay CVs: <5% 9 ). In the current study, between-run CVs of the Randox and Wako assays were slightly higher than those reported previously for dogs (Bayer assay CVs: 3–10.2% 16 ; Olympus assay CVs: <10% 3 ; previously developed CRP assay CVs: 4.8–11% 9 ). However, the between-run CVs obtained in the current study were still markedly lower than those reported for the ELISA (7.5–29.0%). 18 It should be pointed out, however, that the interassay CVs of the Randox assay were nearly twice as high as those reported in the literature for the formerly known Bayer assay. Likely causes for the discrepancy between the previous results and the current findings include variability between different batches of the assay, the use of a different analyzer from the previous study, or changes of components of the test (e.g., the antibody) during the course of time. Furthermore, interassay imprecision of the tests evaluated herein was above the limits of the objective analytical performance standard for imprecision of CRP derived from data on biological variation of canine CRP. 19 Therefore, an assessment of reducing the imprecision of the test is warranted.
The Thermo assay evaluated herein failed to accurately detect canine CRP as reflected by a low linear range and a comparatively low correlation coefficient in the linearity study, as well as a high imprecision and inability to detect high canine CRP concentrations. The likely cause for this finding is the low cross-reactivity between the antibody used in the Thermo assay and the canine CRP molecule, which may be explained by the canine CRP is glycosylation in 2 of 5 subunits in contrast to the human unglycosylated molecule. 4
Human immunoturbidimetric tests evaluated in the current study were able to detect a significant rise in median CRP values in dogs with neoplastic diseases. Given the results of the current study, the Randox and Wako assays were also able to differentiate between healthy dogs and dogs with inflammatory diseases, which is in accordance with results obtained for other CRP assays. 6,16,27,31,38 In the dogs evaluated in the present study, a marked overlap of CRP results was present in the different disease groups, which might be explained by patients being sampled in different stages of illness rather than in the acute phase of disease only. In the current study, results of the patients usually did not exceed a CRP concentration >50 mg/l. This is in contrast to the established canine ELISA detecting CRP concentrations of approximately 100 mg/l. 17,10 A higher range of CRP concentrations (up to ∼240 mg/l) has been reported in septic dogs using the human Olympus assay. 3 C-reactive protein concentrations up to 90 mg/l have been frequently observed in canine patients with infectious diseases when using the Bayer assay, which is still higher than the results obtained in the current study. 16 This discrepancy between results is surprising because the Randox and Bayer assays are identical (Randox Laboratories Ltd., personal communication, 2010). One explanation might be that because different patient groups were studied, absolute CRP concentrations were not directly comparable. Differences between the reagent batches used in the present study and the previous investigation might also contribute. A further possibility might be that antibodies or other components of the test kit have been changed by the manufacturer, although this is not the case according to the information provided by Randox. The presence of lower CRP results in human CRP tests than in assays specifically designed for use in canine specimens indicates a lower antibody-binding capacity.
The reference intervals established herein have been derived from a slightly lower number of individuals (36 instead of 40 dogs) than recommended previously.
14
Additionally, nonparametric statistics had to be used, which would have required a sample size of
The present study had several limitations. It had been recommended that canine-specific control material be used, but the material was not available in the authors' laboratory at the time of the experiment. Additionally, test-specific control material could not be used for the Wako assay. The Randox control was used instead. It was considered to be adequate because it fulfilled the defined criteria of a control, including an acceptable stated CRP concentration, and because it demonstrated the stability of the material over a certain period of time (e.g., 1 year). 48 Moreover, test principals of the Wako and Randox assays were equal, which made the use of the Randox control possible. A similar principle is also applied in external quality assessment, where control material from 1 company or reference laboratory is used for assays from different manufacturers. 47 A further limitation of the study is that the CRP concentration of the canine serum used for the linearity study should have been approximately the same as the CRP concentration of the human-calibration standard. However, serum with a similar high canine CRP level was not observed in the dogs evaluated in the current study, even in patients with severe underlying inflammatory diseases. A reason for this might be that, even in the Wako and Randox assays, the cross-reactivity between the antibodies and canine CRP was not as high as for human CRP.
For the ELISA performed in the method comparison experiment, sera had to be frozen and analyzed in a batch. According to the literature, CRP is stable for at least 4 months if frozen at −10°C. 42 In accordance with other studies, a negative influence from freezing on CRP stability was, therefore, considered un-likely. 16,17 Simultaneous measurements of the automated assays and the ELISA would have been optimal. However, this procedure was not performed in the current study because the preliminary results of the automated CRP assays were directly compared with the actual disease status of the patients to gain further experience with the different new tests, whereas the ELISA analyses had to be performed in a batch. The ELISA was performed by 2 examiners (SK and staff member); however, 1 person would have been optimal to keep interassay variation as low as possible. Interassay and intra-assay repeatability of the ELISA could have been further reduced by using an automated ELISA processing system, but an automated system was not available in the authors' laboratory. For interpretation of the ELISA results, sera with high CRP concentrations that exceeded the upper limit of the ELISA's linearity (>15 mg/l) could not be diluted and reanalyzed because of the low sample volume. Thus, no information on method agreement in higher concentrations can be given. Further studies assessing the linear relationship between the new automated tests and the ELISA as the gold standard are recommended, especially for higher CRP concentrations.
In summary, the current study revealed that automated CRP tests developed for human beings from Randox and Wako demonstrated cross-reactivity with canine CRP and, thus, could be used to detect an increase in canine CRP concentration from neoplasia or inflammation. Further studies are required to minimize the imprecision of the tests and to gain deeper insight into the accuracy of the assays before their routine use in veterinary labortories. The Thermo assay cannot be recommended for use with canine specimens because of a low cross-reactivity with canine CRP.
Acknowledgements
The authors wish to thank Dr. Klaus Failing, Institute for Biomathematics and Data Processing, Justus-Liebig-University, Giessen, Germany, for his kind advice in statistical questions. The authors affirm that they do not have any affiliations or financial involvement in any organization or entity with a direct financial interest in the tests discussed in this manuscript.
Footnotes
a.
Phase™ Range canine CRP, Tridelta Development Ltd., Maynooth, County Kildare, Ireland.
b.
Bayer CRP TIA, Bayer plc, Newbury, Berkshire, United Kingdom.
c.
CRP OSR 6147, Olympus Life and Material Science Europe GmbH, Lismeehan, O'Callaghan's Mills, Ireland.
d.
High Linearity CRP (catalog CP7950), Randox Laboratories Ltd., Crumlin, United Kingdom.
e.
Konelab™ CRP (catalog 981699), Thermo Clinical Labsystems Oy, Vantaa, Finland.
f.
CRP-HS (catalog 419-22007), Wako Chemicals GmbH, Neuss, Germany.
g.
Horiba ABX Pentra 400, Horiba ABX, Montpellier, France.
h.
CRP Plus Calibrator, Thermo Clinical Labsystems Oy, Vantaa, Finland.
i.
Dade Behring, Siemens Healthcare Diagnostics GmbH, Eschborn, Germany.
j.
Sediplus® BSG/ESG/VS-Westergren, Sarstedt, Nürnbrecht, Germany.
k.
ADVIA 2120, Siemens Healthcare Diagnostics GmbH, Eschborn, Germany.
l.
Amelung-Coagulometer KC4A, Heinrich Amelung GmbH, Lemgo, Germany.
m.
Analyse-it for Microsoft Excel (version 2.00), Analyse-it Software Ltd., Leeds, United Kingdom.
n.
BMDP Statistical Software 8.1., Statistical Solutions Ltd., Cork, Ireland.
