Abstract
Sixteen isolates of gram-positive, coccoid bacteria were obtained from clinical cases of diverse conditions in cattle and identified as Streptococcus suis using 16S ribosomal DNA gene sequencing and other bacterial identification methods. None of the isolates could be assigned to any of the known S. suis capsular types. Virulence-associated gene profiling that targeted muramidase-released protein, extracellular protein factor, suilysin, 89-kb pathogenicity island, and arginine deiminase (arcA) genes were negative except for 1 isolate that was arcA positive. The arcA-positive isolate caused severe widespread lesions, including multiorgan suppurative and hemorrhagic inflammation in the meninges, lung, liver, spleen, lymph nodes, and serosae of heart and intestines. The other isolates were primarily associated with meningitis, bronchopneumonia, and multifocal acute necrotizing hepatitis. The isolates differed from each other by 4–6 fragments when examined by pulsed-field gel electrophoresis, indicating they are possibly related. The isolates were susceptible to ampicillin, penicillin, and tiamulin. Resistance was noted to sulfadimethoxine (93%), oxytetracycline (86%), chlortetracycline (86%), neomycin (67%), tilmicosin (47%), clindamycin (47%), enrofloxacin (33%), gentamicin (13%), florfenicol (7%), trimethoprim–sulfamethoxazole (7%), and spectinomycin (53%). Multi-drug resistance (defined as resistance to at least 1 agent in 3 or more antimicrobial classes) was detected in 67% of the isolates. The pathology observations provide evidence that S. suis may be an important pathogen of bovine calves. S. suis is an agent that clinical bacteriology laboratories should consider when dealing with cases involving cattle.
Introduction
Streptococcus suis is a bacterial pathogen mainly known to affect swine, causing fever, meningitis, septicemia, arthritis, polyserositis, anorexia, endocarditis, and sudden death. 29 The organism has increasingly been implicated in human infections and outbreaks, thus making it an important emerging zoonotic agent. 5 Human infections are often reportedly the result of occupational exposure to pigs or pork products, or consumption of undercooked pork.10,16 However, a case of human infection that was associated with cattle has been reported. 18 The isolation of S. suis from dogs, cats, ruminants, and horses has also been reported previously,17,22,29 indicating that S. suis has a broad host range.
To date, 35 capsular serotypes of S. suis have been described (serotypes 1/2 and 1–34). 29 Although serotype 2 is the most frequently isolated serotype and also the serotype most often associated with disease, serotypes 1, 7, 9, and 14 can also cause disease.29,35 The pathogenic potential of S. suis strains varies. Pathogenic strains possess virulence-associated proteins such as muramidase-released protein, extracellular protein factor, and suilysin; these proteins are absent in nonpathogenic strains.29,33,35 Consequently, the proteins are often used as virulence markers to distinguish pathogenic versus nonpathogenic strains.25,28 However, some strains of S. suis that lack these proteins are also pathogenic, suggesting the existence of other virulence factors.27,29 Other reported factors that may play some role in pathogenicity and survival under stress or acid conditions are the capsule and the enzyme arginine deiminase.3,14 We report herein the isolation and partial characterization of alpha-hemolytic streptococci recovered from clinical cases of diverse conditions in cattle.
Materials and methods
Animals and clinical history
Samples from 15 animals that originated from various locations in Wisconsin were submitted for diagnostic workup to the Wisconsin Veterinary Diagnostic Laboratory (WVDL; Madison, Wisconsin) between December 2008 and December 2010. Two of the animals (a dead calf and an aborted fetus) were autopsied at the WVDL. The other samples were obtained from live (swab, milk, transtracheal aspirate) or autopsied animals submitted by field veterinarians (Table 1).
Clinical history, sample sources, and microbiologic and histopathologic findings associated with S. suis.
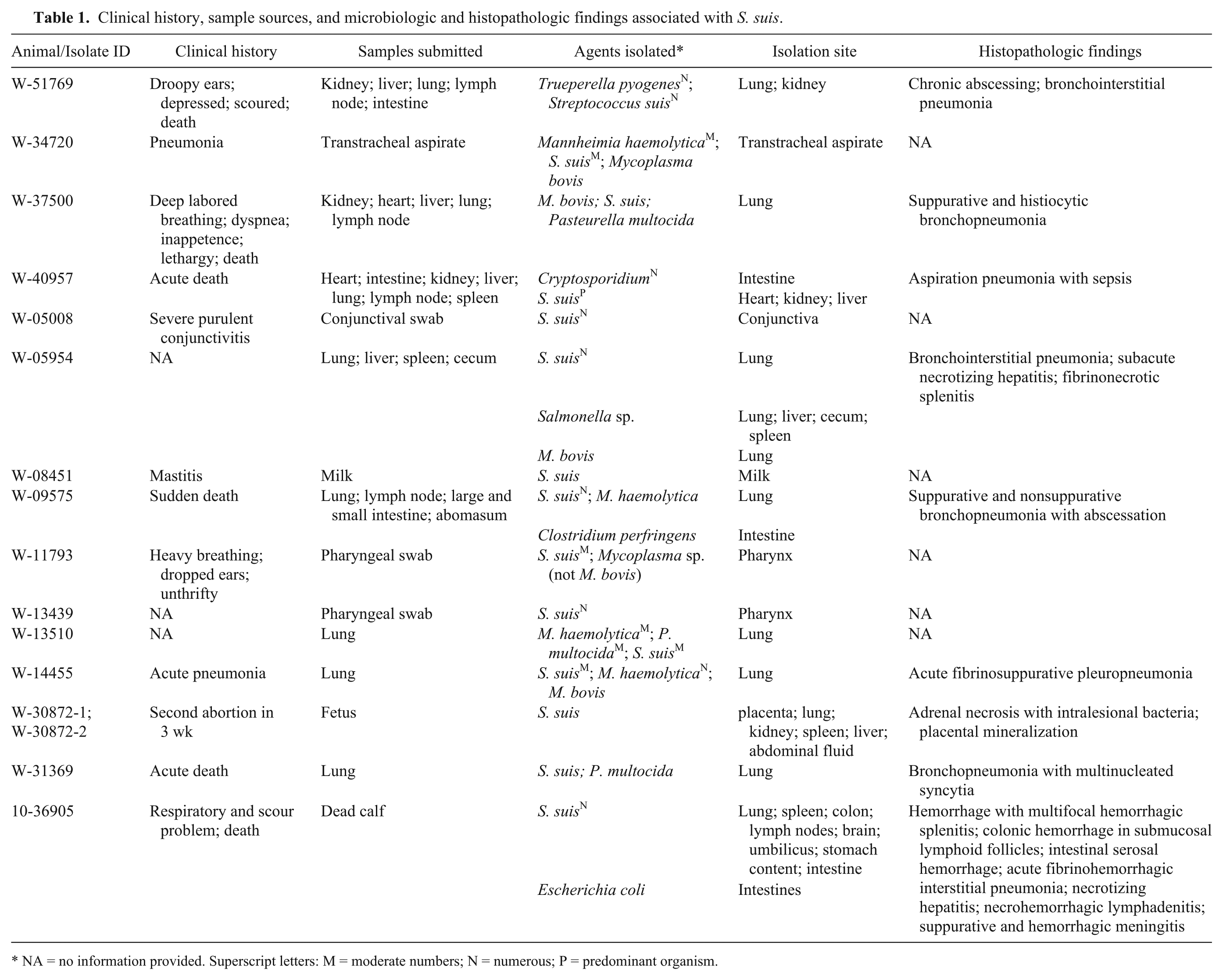
NA = no information provided. Superscript letters: M = moderate numbers; N = numerous; P = predominant organism.
Pathology
Animals were autopsied, and tissue specimens (Table 1) were fixed in 10% buffered (pH 7.2) formalin solution and processed for histologic examination. 2 Fresh samples (Table 1) were collected for bacteriologic, virologic, and parasitologic examination.
Bacteriology, virology, and parasitology
Bacterial isolates were recovered from the samples after plating onto tryptic soy agar with 5% sheep blood a and incubation in a 5% CO2 atmosphere at 37°C for 24 h. Anaerobic bacteria were recovered by incubation in a commercial system. b The presence of Bovine coronavirus (BCoV), Bovine viral diarrhea virus (BVDV), Bovine respiratory syncytial virus (BRSV), Bovine herpesvirus 1 (BoHv-1), and bovine rotavirus in the samples was assessed directly using real-time polymerase chain reaction (PCR) assays following established and approved standard operating procedure used at the WVDL (unpublished). The presence of Neospora sp. was also determined by a PCR method. 7 The samples examined from animals that exhibited respiratory signs included lungs and pharyngeal swabs. Placenta, lung, kidney, spleen, liver, and abdominal fluid samples were used for an aborted fetus. Samples used for animals that exhibited enteric signs included intestine and feces. Detection of Cryptosporidium parvum was performed using a method described previously. 19 Twenty-two swine isolates of previously characterized S. suis belonging to serotypes 1/2, 1, and 2 24 were retrieved from our culture collection maintained at −80°C, grown aerobically as described above, and used to evaluate the performance of PCR primers derived from arcA sequences of S. suis from bovine and swine sources.
Bacterial identification
For the streptococcal organisms, a Gram stain and relevant biochemical tests were performed on the isolates following a standard procedure. 30 A commercial kit, c and 2 different identification systems,d,e were also used according to the manufacturer’s instructions. A S. suis–specific PCR assay that utilized previously published glutamate dehydrogenase (gdh)-based primers (JP4 and JP5) 23 in a monoplex format was used. In addition, a full-length nucleotide sequence determination and analysis of both strands of the 16S rRNA gene was also used. 1 Other bacteria were identified with a commercial system. e
DNA extraction
Genomic DNA was extracted from overnight cultures grown on tryptic soy agar with 5% sheep blood a using a commercial extraction kit f per the manufacturer’s instruction. The DNA concentration was determined spectrophotometrically. g
Serotyping and pathogenic character
To determine if any of the S. suis isolates belonged to serotypes 1/2, 1, 2, 7, or 9, ~100 ng of DNA from each isolate was used as template in a PCR with the serotype-specific primers in a multiplex format as reported previously. 23 Serotyping that covers all known S. suis capsular types was also performed at an accredited reference laboratory h using the standard agglutination test method. The pathogenic character of the S. suis isolates was evaluated using a PCR assay that targeted the genes for muramidase-released protein (mrp), extracellular protein factor (epf), suilysin (sly), an 89-kb candidate pathogenicity island, and arginine deiminase (arcA). 28 Isolates of Escherichia coli were analyzed for the presence of virulence factors using a PCR assay that targeted the genes encoding the heat-stable enterotoxin, intimin, shiga toxins 1 and 2, K99, and F41 fimbriae as described elsewhere. 11
Nucleotide sequence determination of arcA
A 118-bp amplicon of arcA of isolate 10-36905 from one of the calves was sequenced. i To extend the sequence beyond this region, an inverse PCR (IV-PCR) was performed with primers (arcAinvF2HaeIII: 5′-GTACAAACGGAATACCATTAC-3′ and arcAinvR2HaeIII: 5′-ACAACGACAACCCCTGGTG-3′; arcAinvF5EcoRV: 5′-GAAAAGACCATGGCTGGTATTC-3′ and arcAinv5REcoRV: 5′-AATTAGCTCTTTATTGTCCTC-3′; arcAinvF8HincII: 5′-GGTCGGGAACAATGGAACG-3′ and arcAinvR8HincII: 5′-AGCAGCTACAATATTTTCACC-3′) that were commercially synthesized. j Genomic DNA from isolate 10-36905 was digested with HaeIII, EcoRV, and HincII, k respectively, and religated prior to IV-PCR. The PCR products were cloned, sequenced using universal M13 forward and reverse primers, i and assembled in frame with commercial software. l
Amplification of arcA from bovine and swine isolates
The arcA sequences obtained in our study were aligned l with the sequences of arcA of S. suis from swine origin deposited in GenBank (accessions AF546864 and EU768886). Regions of sequence identity were identified and used for the design of PCR primers that would produce an amplicon size of 708 bp. The primer sequences are shown in Table 2 and assay parameters were as described previously. 23
Performance of PCR primers designed from arcA nucleotide sequences of Streptococcus suis isolates from swine and bovine origin, respectively.

Primer sequences derived from arcA of S. suis of swine origin.
Primer sequences derived from arcA of S. suis of bovine calf origin.
Pulsed-field gel electrophoresis
Typing by pulsed-field gel electrophoresis (PFGE) using SmaI restriction endonuclease was performed as described previously, 20 with some modifications. Briefly, bacterial cells were harvested into Tris–EDTA buffer (10 mM Tris–HCl and 1 mM EDTA, pH 8.0) after 24 h growth on tryptic soy agar with 5% sheep blood a at 37°C in a 5% CO2 incubator and adjusted to an absorbance of ~1.0 at 610 nm wavelength using a spectrophotometer. g The digested DNA was electrophoresed m in 1% agarose gel n with 0.5% Tris–borate–EDTA as the running buffer at 14°C with 2.2 s initial switch time; 68.8 s final switch time; 6 V/cm field strength; 120° included angle for 18 h. The PFGE patterns were analyzed with commercial software. o The TIFF images were normalized by aligning the peaks of the size standard strain (Salmonella enterica subsp. enterica serovar Braenderup strain H9812) with the database global standard. Matching of the PFGE patterns by dendrogram analysis using the unweighted pair group method with averages 8 was performed using the Dice coefficient with a 1.0–1.5% tolerance window. For data analysis, the recommended criteria for interpreting bacterial DNA restriction patterns produced by PFGE was used. 31 Briefly, when isolates had the same numbers of fragments that were the same size, they were categorized as indistinguishable and considered to be the same strain. Isolates that had 1 or 2 fragment differences were considered to be closely related and isolates with 4–6 fragment differences were considered to be possibly related. When ≥7 fragment differences existed between isolates, the isolates were considered to be different.
Antimicrobial susceptibility
Antimicrobial susceptibilities were determined by a microdilution method using a commercially prepared, dehydrated 96-well microtiter minimum inhibitory concentration (MIC) panel (BOPO6F) g for veterinary bacteria. Staphylococcus aureus ATCC 29213, Pseudomonas aeruginosa ATCC 27853, E. coli ATCC 25922, and Enterococcus faecalis ATCC 29212 were used as quality control strains following the manufacturer’s instructions g and Clinical and Laboratory Standards Institute (CLSI) guidelines. 6 The quality control strains and S. suis organisms were grown for 18 h on tryptic soy agar with 5% sheep blood a and checked for purity before use. Colonies were picked and cells were standardized in Mueller–Hinton broth to a McFarland standard of 0.5 (1.5 × 108 colony-forming units/mL) using a nephelometer. g Fifty microliters of the standardized bacteria suspension were dispensed into the wells of a 96-well microtiter plate containing different concentrations of test antimicrobials. Plates were incubated aerobically for 18 h in the commercial system. g Control wells without antimicrobial were also inoculated. Results were interpreted as sensitive and resistant using the automated built-in MIC breakpoint values in the system. g The antimicrobial agents used and their respective dilution ranges were as follows: ampicillin, 0.25–16 μg/mL; ceftiofur, 0.25–8 μg/mL; chlortetracycline, 0.5–8 μg/mL; clindamycin, 0.25–16 μg/mL; danofloxacin, 0.12–1 μg/mL; enrofloxacin, 0.12–2 μg/mL; florfenicol, 0.25–8 μg/mL; gentamicin, 1–16 μg/mL; neomycin, 4–32 μg/mL; oxytetracycline, 0.5–8 μg/mL; penicillin, 0.12–8 μg/mL; spectinomycin, 8–64 μg/mL; sulfadimethoxine, 256 μg/mL; tiamulin, 1–32 μg/mL; tilmicosin, 4–64 μg/mL; trimethoprim–sulfamethoxazole, 2–38 μg/mL; tulathromycin, 1–64 μg/mL; and tylosin tartrate, 0.5–4 μg/mL.
Results
Identification of bacteria
From the 15 cases submitted, 16 isolates that produced small, adherent, alpha-hemolytic colonies on sheep blood agar plates after incubation for 18–24 h were recovered. One case had 2 isolates. The colonies became beta-hemolytic when left at room temperature for an additional 3–5 d. The organisms were gram-positive cocci in chains, esculin positive, and bile-esculin and sodium hippurate negative by standard biochemical testing, suggesting a presumptive S. suis identification. The isolates were identified as S. suis by 3 commercial identification systems,c–e the S. suis–specific PCR, and full-length 16S ribosomal (r)DNA sequence–based analysis. The 2 molecular-based methods yielded the expected 688-bp (S. suis–specific PCR) and 1,517–1,529-bp (16S rDNA) products.4,26 No amplification product was detected in the negative control (data not shown). A BLAST search in GenBank revealed that the 16S rDNA sequences matched several S. suis sequences; accessions AF009508 and AB689682 were 99% identical to the isolates. The next closest match was S. bovis (96% sequence homology). Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry yielded identification scores between 1.7 and 2.0, indicating probable genus and species identification based on established criteria, e and gave S. suis as the best match. In 10 of the 15 cases described, S. suis was isolated along with known bacterial pathogens such as Mannheimia haemolytica, Mycoplasma bovis, and Pasteurella multocida. In 5 cases, S. suis was isolated alone (Table 1).
Serotyping and pathogenic character of isolates
The serotype-specific PCR assay for serotypes 1/2, 1, 2, 7, and 9 did not yield detectable product, and the standard agglutination test also yielded nontypeable results for all 16 S. suis isolates. Screening for S. suis virulence-associated genes (mrp, epf, sly, and the 89-kb candidate pathogenicity island) also produced negative results. Only 1 of the 16 isolates (isolate 10-36905) was positive by PCR for arcA and produced the expected 118-bp amplicon (Fig. 1). The identity of the amplicon was confirmed by sequencing (data not shown). The E. coli isolate was negative for all of the virulence genes screened (data not shown), suggesting possible postmortem contamination.
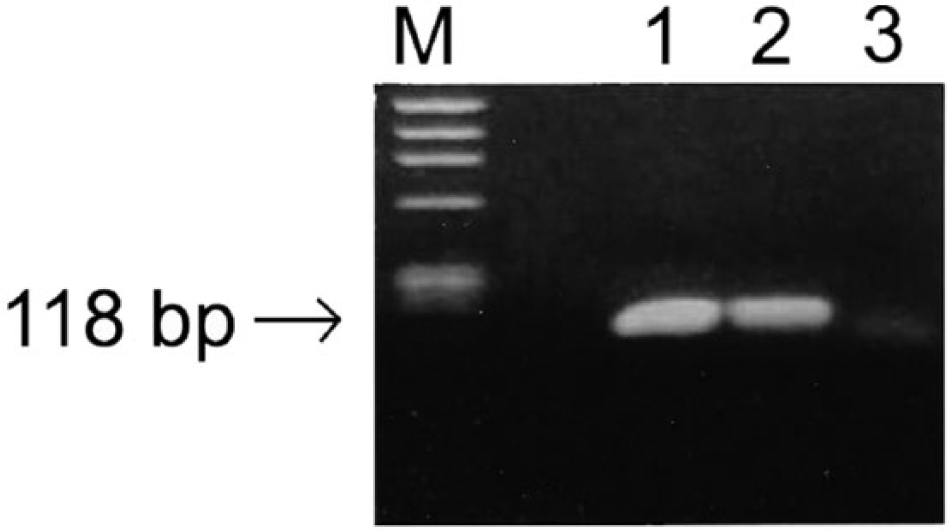
Ethidium bromide–stained agarose gel after electrophoresis of polymerase chain reaction products with arcA primers derived from the sequences of a swine isolate of Streptococcus suis producing the expected 118-bp product. Lane M: HaeIII-digested ØX174 DNA molecular weight standards; lane 1: S. suis from swine (strain 1933; positive control); lane 2: S. suis from bovine case (isolate 10-36905); lane 3: negative control (no template).
Analysis of arcA
The complete nucleotide sequence analysis of the arcA of isolate 10-36905 revealed that it contained an open reading frame (ORF) that started at the ATG in position 63 and terminated at the TAA in position 1292. The primary sequence of the protein translated from the ORF consisted of 409 amino acid residues. A search in GenBank revealed that the protein sequences shared identity at the amino acid level with various arginine deiminase sequences of S. suis swine isolates deposited in the database. Most significant identity (99%) was observed with sequences with accession WP_032512091 and WP_032535973. Other swine S. suis arginine deiminase sequences were 86–91% identical to the sequences of isolate 10-36905. At the nucleotide level, the bovine and swine isolates shared 80% sequence homology (data not shown). The GenBank accession for the arcA nucleotide sequence of isolate 10-36905 is KT429438.
Amplification of arcA of S. suis from swine and bovine origin
The PCR assay that utilized the primers designed from the arcA nucleotide sequence of a swine isolate 28 amplified efficiently the DNA of the 22 swine isolates tested, but only 1 of the 16 bovine isolates. All positive reactions yielded the expected product of 118 bp. In contrast, the PCR using the primers from the bovine arcA nucleotide sequences that were designed in our study following sequence alignment amplified efficiently the gene from 16 of the 16 bovine and 22 of the 22 swine isolates tested (Table 2) and yielded the expected 708-bp amplicon.
Pulsed-field gel electrophoresis
In general, the isolates produced 10–16 well-resolved fragments (Fig. 2A–C) except for isolate W-40957 (Fig. 2B, lane 8), which produced a smear and could not be subtyped regardless of efforts. Isolates W-30872-1 and W-30872-2 (Fig. 2C, lanes 13 and 14; Fig. 3), recovered from the placenta and intestine of the same animal were indistinguishable, suggesting that the tissues were likely infected by the same strain. All other isolates displayed distinct and unique profiles and had 4–6 fragment differences when compared with each other and with isolates W-30872-1 and W-30872-2, suggesting that they were possibly related (Figs. 2, 3). One isolate was recovered after the initial PFGE work was concluded and was not included with the earlier run. However, when tested, it produced 12 fragments and a different PFGE pattern to all other isolates, with 4–6 fragments that were different from the fragments produced by the other isolates (data not shown). No correlation was obvious between pulsotypes and isolate source (Fig. 3).
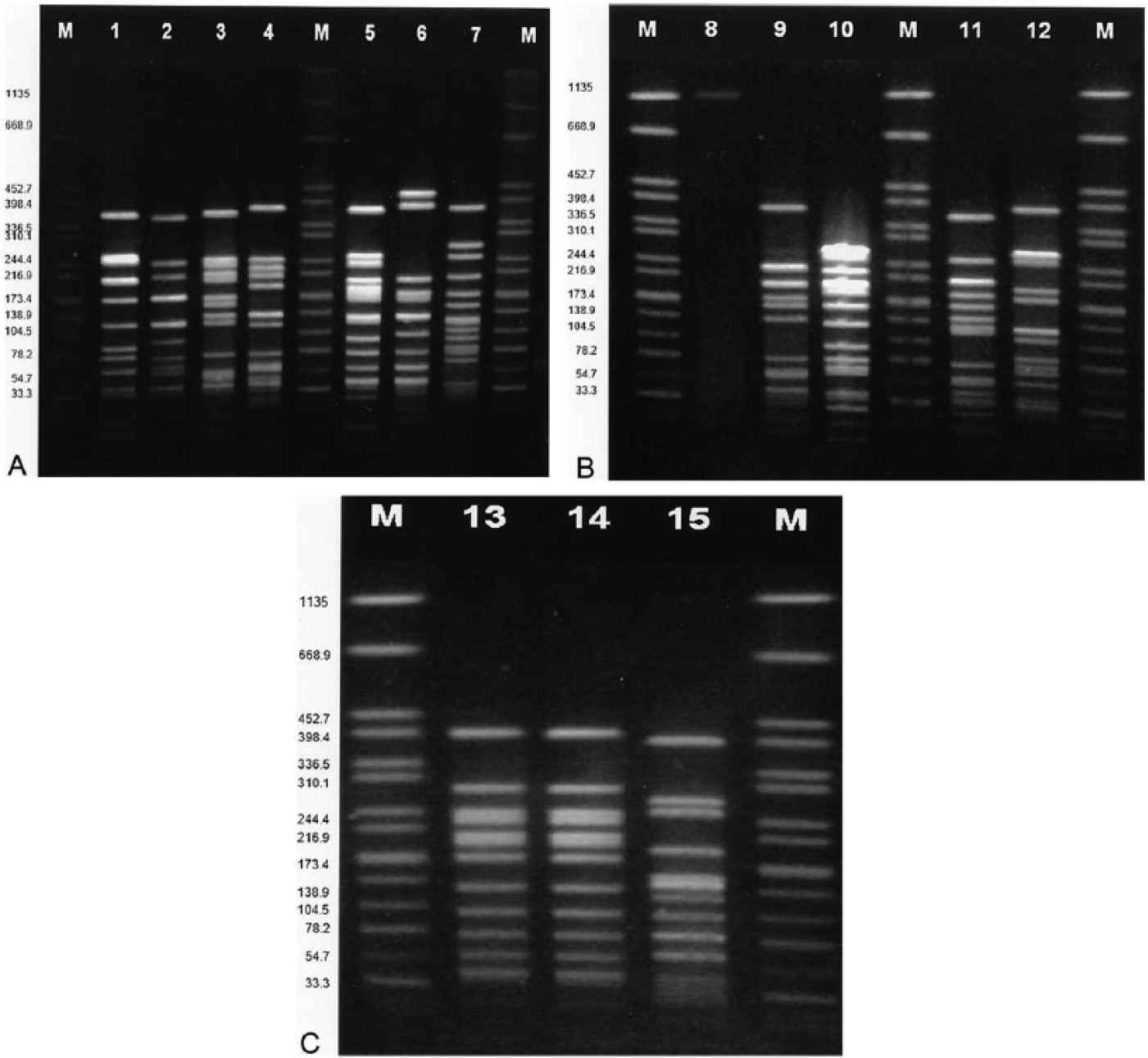
Pulsed-field gel electrophoresis separation of SmaI macro-restriction fragments of Streptococcus suis genomic DNA from the bovine clinical cases. Lane M: Salmonella enterica subsp. enterica serovar Braenderup H9812 digested with XbaI as reference size markers in kilobases; lane 1: W-13510 (lung); lane 2: W-05008 (eye); lane 3: W-37005-2 (lung); lane 4: W-34720 (tracheal wash); lane 5: W-14455 (lung); lane 6: W-11793-1 (pharyngeal swab); lane 7: W-0841-8 (milk); lane 8: W-40957 (lung); lane 9: W-51769 (lung); lane 10: W-13439 (pharyngeal swab); lane 11: W-09575 (lung); lane 12: 05954-6 (lung); lane 13: W-30872-1 (abdominal content); lane 14: W-30872-2 (placenta); lane 15: W-31369 (lung).
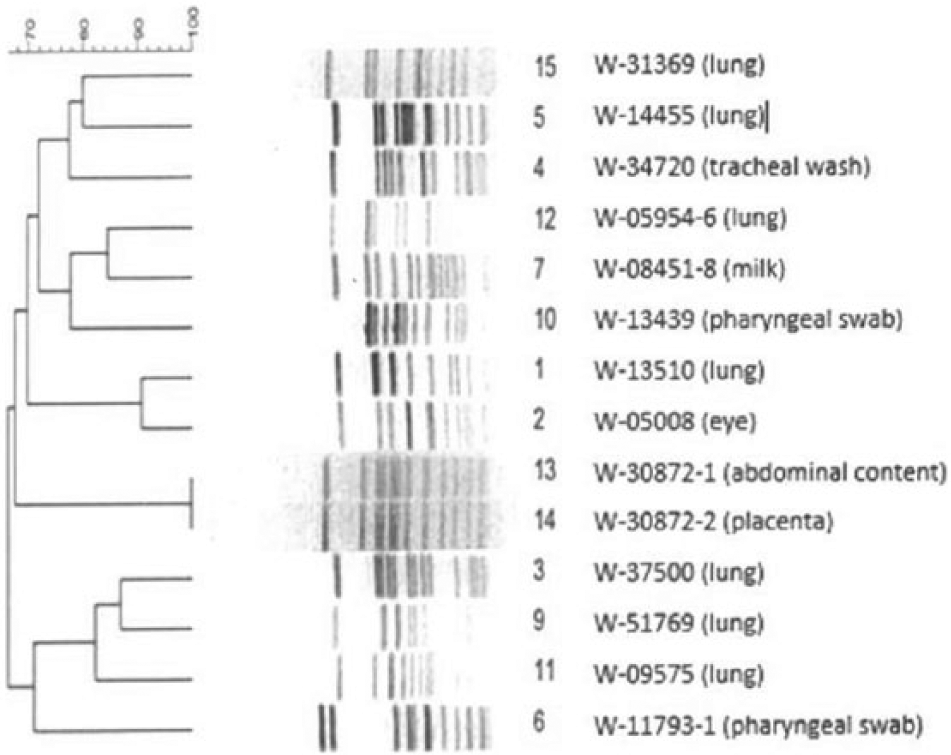
Dendrogram derived from pulsed-field gel electrophoresis profile of SmaI macro-restriction patterns. The numbers to the right side of the figure correspond to the lanes in Figure 2 followed by isolate designation and source.
Antimicrobial susceptibility
The quality control strains yielded acceptable quality check results. All S. suis isolates tested were susceptible to ampicillin, penicillin, and tiamulin, but they exhibited various levels of resistance to the other antimicrobials tested. Resistance to sulfadimethoxine (93%), oxytetracycline (86%), chlortetracycline (86%), neomycin (67%), tilmicosin (47%), clindamycin (47%), spectinomycin (53%), gentamicin (13%), and enrofloxacin (33%) was noted. Multiple drug resistance, defined as resistance to at least 1 agent in 3 or more antimicrobial classes, 21 was detected in 67% (10 of 15) of the isolates (Table 3).
Multiple drug resistance patterns found in the Streptococcus suis isolates.*
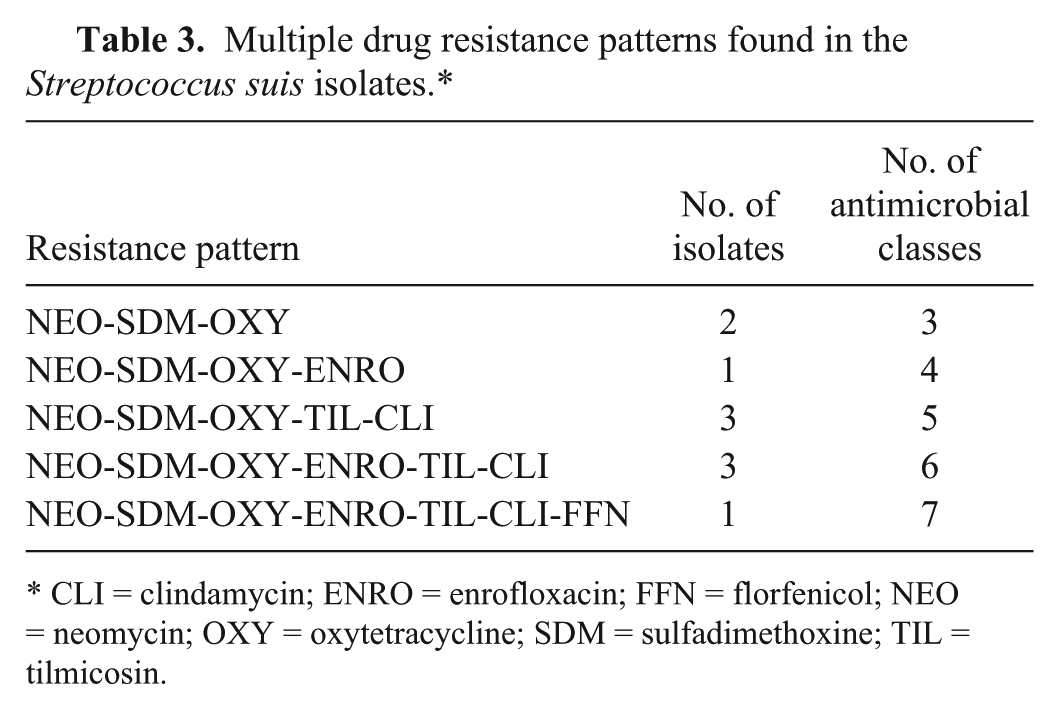
CLI = clindamycin; ENRO = enrofloxacin; FFN = florfenicol; NEO = neomycin; OXY = oxytetracycline; SDM = sulfadimethoxine; TIL = tilmicosin.
Virology and parasitology
The PCR assays were negative for the viruses targeted except that case W-13439 was positive for BCoV and case W-05954 was positive for both BRSV and BCoV. Except for case W-40957, no parasite was identified or detected in any of the other samples.
Pathology
Compared with other S. suis isolates examined in our study, isolate 10-36905 was associated with more acute and widespread disease (Table 1). Morphologically, the infected calf had petechial hemorrhages on the serosal surfaces of the intestine (Fig. 4), suggestive of disseminated intravascular coagulopathy secondary to sepsis. Mesenteric lymph nodes were enlarged, hemorrhagic, and necrotic. Severe acute suppurative and hemorrhagic meningitis was noted. Acute fibrinohemorrhagic interstitial pneumonia and extensive suppurative bronchopneumonia were observed. Multifocal acute necrotizing hepatitis, multifocal hemorrhagic splenitis, necrohemorrhagic lymphadenitis, and epicardial and endocardial hemorrhage were observed. In addition, there were severe depletion of bone marrow, and mild myopathy. Numerous colonies of S. suis (including isolate 10-36905) were grown from the brain, lungs, umbilicus, liver, spleen, stomach content, lymph nodes, and heart, suggesting bacteremia–septicemia. Final diagnosis was determined to be sepsis caused by S. suis.
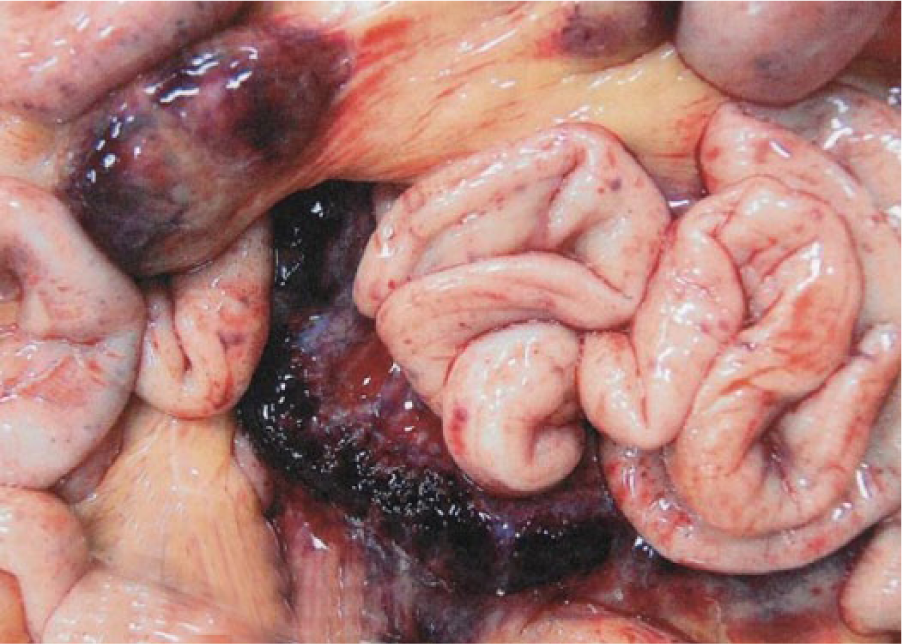
Jejunum and mesentery of a 5-d-old Holstein bull calf with enlarged, hemorrhagic, and necrotic lymph nodes. Also present are petechial hemorrhages on serosal surfaces of loops of bowel suggestive of disseminated intravascular coagulopathy secondary to sepsis.
One case that involved abortion (W-30872) was attributed to S. suis because the organism was isolated as a pure culture and in high numbers from the placenta and other parts of the aborted calf, and no other pathogen was recovered (Table 1). In 7 cases involving animals diagnosed with bronchopneumonia, S. suis was isolated from the lungs in moderate-to-high numbers with other pathogens such as P. multocida, M. haemolytica, or Trueperella pyogenes (Table 1).
Discussion
Streptococcus suis is a major swine pathogen, 29 but it has also been isolated from other animal species including dogs, cats, ruminants, and horses,17,22,29 indicating a diverse host range. In reports involving hosts other than swine, the isolates were not characterized to examine virulence potential or genetic relationships.
There is little to no information on the isolation and molecular characterization of S. suis from bovine clinical samples, probably because alpha-hemolytic streptococci are not routinely identified to species level by many laboratories given the inherent difficulty in this task. 15 Hence, S. suis isolates may not be recognized, and infections caused by this species may go unrecognized. In addition, S. suis exhibits variable biochemical characteristics,15,30 making it difficult to speciate accurately.
In 10 of the 15 cases described, S. suis was isolated with other known bacterial pathogens such as M. haemolytica, M. bovis, and P. multocida, making it difficult to determine the clinical importance of S. suis and the other bacterial species. In 3 cases, S. suis was isolated alone and was associated with mucosal surfaces (conjunctiva and pharynx) or body fluid (milk; Table 1). Thus, in these 13 cases, S. suis may be an opportunistic pathogen or even a commensal. Similar results and conclusions on the isolation of S. suis with other bacterial agents in other animal species have been reported previously.9,29 Further research that would provide a clear understanding of reoccurring multiple infections involving S. suis will be invaluable.
Attempts to serotype the isolates using a S. suis serotype–specific PCR assay for types 1/2, 1, 2, 7, and 9 developed for swine isolates 23 failed. Moreover, serotyping that covers all known S. suis capsular types using the standard agglutination test produced nontypeable results. We speculate that the primers used may have insufficient nucleotide sequence homology at the various primers binding sites. As well, the isolates may have exhibited some differences in antigenic makeup compared with the S. suis reference strains and such differences may be sufficient to have resulted in the failure to serotype them using specific antiserum raised against the reference strains. Serotyping can also yield negative results if the isolates fail to express target antigens in detectable amounts. 13 It is worth noting that many S. suis isolates are nontypeable. 15
The isolation of S. suis from lungs of young calves with pneumonia has been reported previously in Switzerland but the isolates were not characterized to determine their pathogenic potential, serotype, or relationship to one another or to swine isolates. 34 Because all of the isolates were negative for the virulence-linked genes tested, except 1 isolate that was positive for arcA, the results may indicate the existence of pathogenic factors other than the ones screened. Additionally, the possibility that nucleotide sequence differences in the PCR primers binding regions may produce false-negative results cannot be discounted. Finally, some of the isolates may simply be secondary invaders (Table 1).
A set of arcA primers has been successfully used for virulence-associated gene profiling 28 of S. suis of swine origin. For the primers to be broadly useful, they should amplify efficiently the arcA of S. suis when present, regardless of isolate source. Because the primers failed to amplify the arcA of 15 of the 16 bovine S. suis isolates tested (Table 2), the published arcA primers sequences from swine isolate are therefore not suitable for inclusion in a PCR assay for virulence gene profiling when screening S. suis isolates of bovine origin. Thus, the primers developed in our study may serve as an alternative.
We noted that although the isolates exhibited restriction fragment pattern differences, they were possibly related by the accepted criteria for PFGE pattern interpretation. 31 The observed restriction pattern differences were in agreement with previous reports on S. suis of swine origin. 24 Because a limited number of strains were used, a more comprehensive approach that significantly increases the sample size of S. suis of cattle origin will add strength to our observations. A better understanding of the population structure and genetics of S. suis from various animal species may provide insight into the development of control strategies such as improved identification of common genotypes that are associated with disease and protective antigens that can be used for the production of efficacious vaccines against S. suis. It may also enhance the development of accurate and sensitive molecular assays for the detection of pathogenic S. suis stains.
The susceptibility patterns of the S. suis isolates revealed that they were susceptible to ampicillin, penicillin, and tiamulin, but they exhibited variable levels of resistance to the other antimicrobials tested in our study. A literature review showed that the vast majority of S. suis isolates are susceptible to β-lactams, and the effective therapeutic use of penicillin has been reported in animals infected with S. suis.32,34 Because penicillin resistance has also been reported in a few cases,12,32,36 it is essential to perform antimicrobial susceptibility testing on S. suis isolates to monitor changes in resistance patterns.
Compared with available reports on S. suis of swine origin, our pathology findings in bovine calves were similar to those documented for swine, except that swine cases typically involve polyserositis. 29 Overall, the clinical signs and the lesions caused by S. suis are similar in pigs and calves.
Footnotes
Acknowledgements
We thank the bacteriology, virology, and pathology staff at the Wisconsin Veterinary Diagnostic Laboratory, and students Erik Rolland-Puente and Aisha Inuwa for technical support.
Authors’ contributions
O Okwumabua contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. H Peterson contributed to acquisition and interpretation of data. HM Hsu contributed to analysis and interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. P Bochsler contributed to acquisition and analysis of data and gave final approval. M Behr contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; and gave final approval. All authors agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Tryptic soy agar with 5% sheep blood, Becton Dickinson, Franklin Lakes, NJ.
b.
Mini mac DG250 system, Microbiology International, Frederick, MD.
c.
API 20 Strep kit, bioMérieux, Durham, NC.
d.
Omnilog GEN III system, Biolog, Hayward, CA.
e.
MALDI-TOF system, Biotyper, Bruker Daltonics, Billerica, MA.
f.
DNeasy blood and tissue kit, Qiagen, Valencia, CA.
g.
NanoDrop 2000 spectrophotometer, pCR2.1 cloning vector, GENESYS 2 spectrophotometer, Nephelometer, Sensititer, BOPO6F; Thermo Fisher Scientific, Waltham, MA.
h.
Veterinary Diagnostic Laboratory, Iowa State University, Ames, IA.
i.
Functional Biosciences sequencing facility, Functional Biosciences, Madison, WI.
j.
Primers, Integrated DNA Technologies, Coralville, IA.
k.
Restriction enzymes, Promega, Madison WI.
l.
MacVector version 14.5 sequence analysis software, MacVector, Cary, NC.
m.
CHEF-MAPPER II, Bio-Rad, Hercules, CA.
n.
Seakem-Gold agarose, Lonza Rockland, Rockland, ME.
o.
BioNumerics software version 5.1, Applied Maths, Austin, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
