Abstract
While Mannheimia haemolytica and Mannheimia glucosida have been recognized as causes of intramammary infection in sheep, there has been no investigation of the epidemiology of the strains involved. Pulsed field gel electrophoresis was used to study the molecular epidemiology of isolates of these 2 species associated with ovine mastitis. Ten distinct strains were recognized among 12 M. haemolytica isolates, and 7 distinct strains among 13 M. glucosida isolates. The results demonstrate a high diversity of isolates with the ability to cause ovine mastitis. However, the presence of some identical isolates may suggest the possibility of horizontal transmission of these species in some flocks, possibly through lamb sucking, and/or differences in the capacity of some isolates to cause mastitis in sheep.
Mannheimia haemolytica is one of the most common causes of mastitis in sheep. 12 Mastitis is an important disease of sheep in both dairy and non-dairy production systems, with significant effects on flock economy and animal welfare. Mastitis causes reduced milk production, changes in milk composition, lower weight gain in lambs, and reduced longevity of ewes, because of mortality due to peracute mastitis, early culling due to chronic subclinical disease, or inability to raise lambs. 13
A number of Poll Dorset breeders in Australia have suggested that clinical mastitis is one of the most significant disease problems they face, with peracute and gangrenous mastitis a common occurrence during lactation (Barber S, Allen J, Mansell P, Browning G: 2006, Mastitis in the ewe. In: Proceedings of the Australian Sheep Veterinarians 2006 Conferences, pp. 127–132. Australian Sheep Veterinarians, Wagga Wagga and Hobart). A survey of Poll Dorset breeders has suggested a prevalence of mastitis in individual flocks of up to 20% (Barber et al.: 2006). The genus Mannheimia contains several species from the family Pasteurellaceae that have been reclassified based on phenotypic and genetic characteristics. 2 In addition to the 5 named species within this genus, M. haemolytica, M. glucosida, M. ruminalis, M. varigena, and M. granulomatis, there are several unnamed taxa that are distinct from the named species.1,2 A new species, M. caviae, from epidemic conjunctivitis and otitis media in guinea pigs, was reported in 2011. 4
Mannheimia haemolytica is regarded as the most important species in the genus, as it is the major organism involved in bovine respiratory disease and can cause pneumonia, septicemia, and mastitis in sheep.3,10 Mannheimia glucosida is a heterogeneous species that has been isolated from pneumonia 1 and mastitis in sheep, 12 and the nasal cavity of healthy sheep.1,15 The author’s laboratory (Asia-Pacific Centre for Animal Health and School of Veterinary Science, The University of Melbourne, Parkville, Victoria, Australia) has recently demonstrated the significance of M. glucosida as a cause of ovine mastitis for the first time since definition of the species. 12
A variety of typing techniques have been used to study the epidemiology of M. haemolytica associated with pneumonia in ruminants,5,9 but molecular epidemiological approaches have not been used to investigate relationships between Mannheimia species isolated from ovine mastitis. Thus, the current study aimed to investigate the molecular epidemiology of M. haemolytica and M. glucosida in ovine mastitis using pulsed field gel electrophoresis (PFGE) to study the similarity of isolates obtained from different flocks.
Twelve M. haemolytica and 13 M. glucosida isolates from cases of mastitis were included in the present study. The isolates represent all of the disease-associated isolates of family Pasteurellaceae from 8 different Poll Dorset flocks, designated A–H, and 1 mixed-breed flock, designated I. Milk samples were collected from any ewe with clinical mastitis during lactation or at weaning in all flocks (A–H) during 2006, with samples also collected from flock A in 2007–2009. Clinical samples were collected in flock I at weaning in 2006. The flocks ranged in size from less than 100 ewes to up to 500 ewes, and the mixed-breed flock had approximately 2,000 ewes. A review of the transfer of breeding sheep between the Poll Dorset farms in the current study was conducted using the registered flock books for the years 2004–2006 to assess the likelihood of transfer of Mannheimia species between flocks by breeding stock (2004 Flock register for Poll Dorset sheep in Australia, vol. 50; 2005 Flock register for Poll Dorset sheep in Australia, vol. 51; 2006 Flock register for Poll Dorset sheep in Australia, vol. 52. Available at: http://www.polldorset.org.au/Main.asp?_=Flock%20Register&d=/edit/flock_register/&W=F&X=R).
The phenotypic characteristics, as well as sequences of 2 housekeeping genes, rpoB and rrnA, of all M. haemolytica and 9 of the M. glucosida isolates have been described previously. 12 There were 1–3 M. haemolytica isolates from each flock. All M. haemolytica isolates were associated with clinical mastitis and were identical in their phenotypic tests and rpoB gene sequences, but could be divided into 3 groups on the basis of 1 or 2 nucleotide differences in their rrnA genes. Eight of the 13 M. glucosida isolates were isolated from flock A. Eleven of 13 (85%) of the M. glucosida isolates were associated with clinical cases of mastitis. The M. glucosida isolates differed in both rpoB and rrnA gene sequences and could be divided into 3 groups based on phenotypic tests. All M. glucosida isolates were β-hemolytic on sheep blood agar (SBA), fermented D-sorbitol and D-xylose, and hydrolyzed salicin and esculin, and produced cytochrome c oxidase and β-glucosidase, but differed in their capacity to produce ornithine decarboxylase, and ferment L-arabinose and maltose. Details of the isolates are given in Table 1.
Mannheimia haemolytica and Mannheimia glucosida isolates from ovine mastitis.*
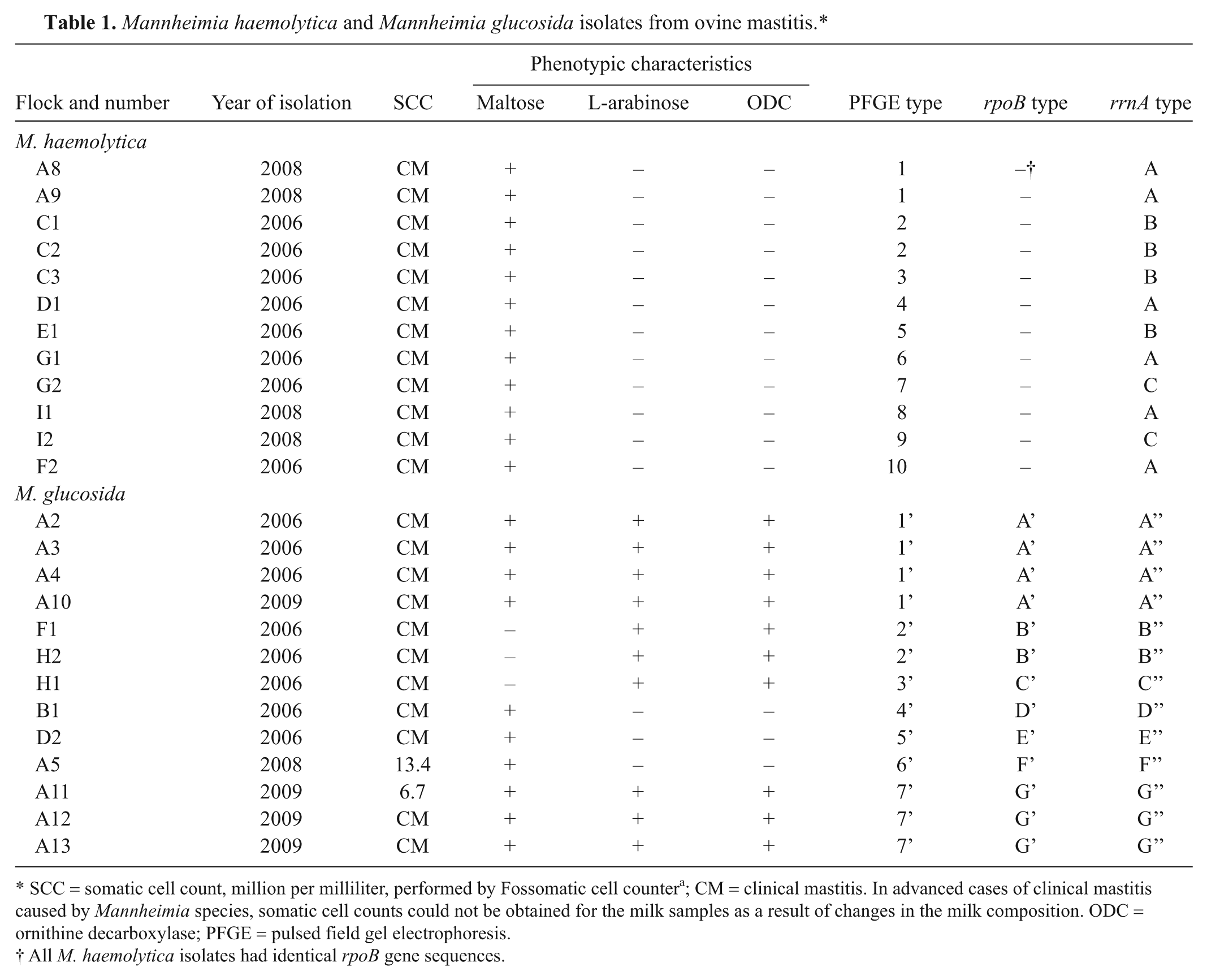
SCC = somatic cell count, million per milliliter, performed by Fossomatic cell counter a ; CM = clinical mastitis. In advanced cases of clinical mastitis caused by Mannheimia species, somatic cell counts could not be obtained for the milk samples as a result of changes in the milk composition. ODC = ornithine decarboxylase; PFGE = pulsed field gel electrophoresis.
All M. haemolytica isolates had identical rpoB gene sequences.
Genomic DNA of the isolates was prepared in 1% low melting agarose and digested using the restriction endonuclease SalI using a procedure described previously, 8 with slight modifications. The DNA fragments were separated in a 1% molecular biology grade agarose gel by PFGE as described previously. 11
Isolates that had the same PFGE patterns were designated identical or indistinguishable. The similarities between restriction endonuclease cleavage patterns of each strain were calculated using the Dice similarity index. 6
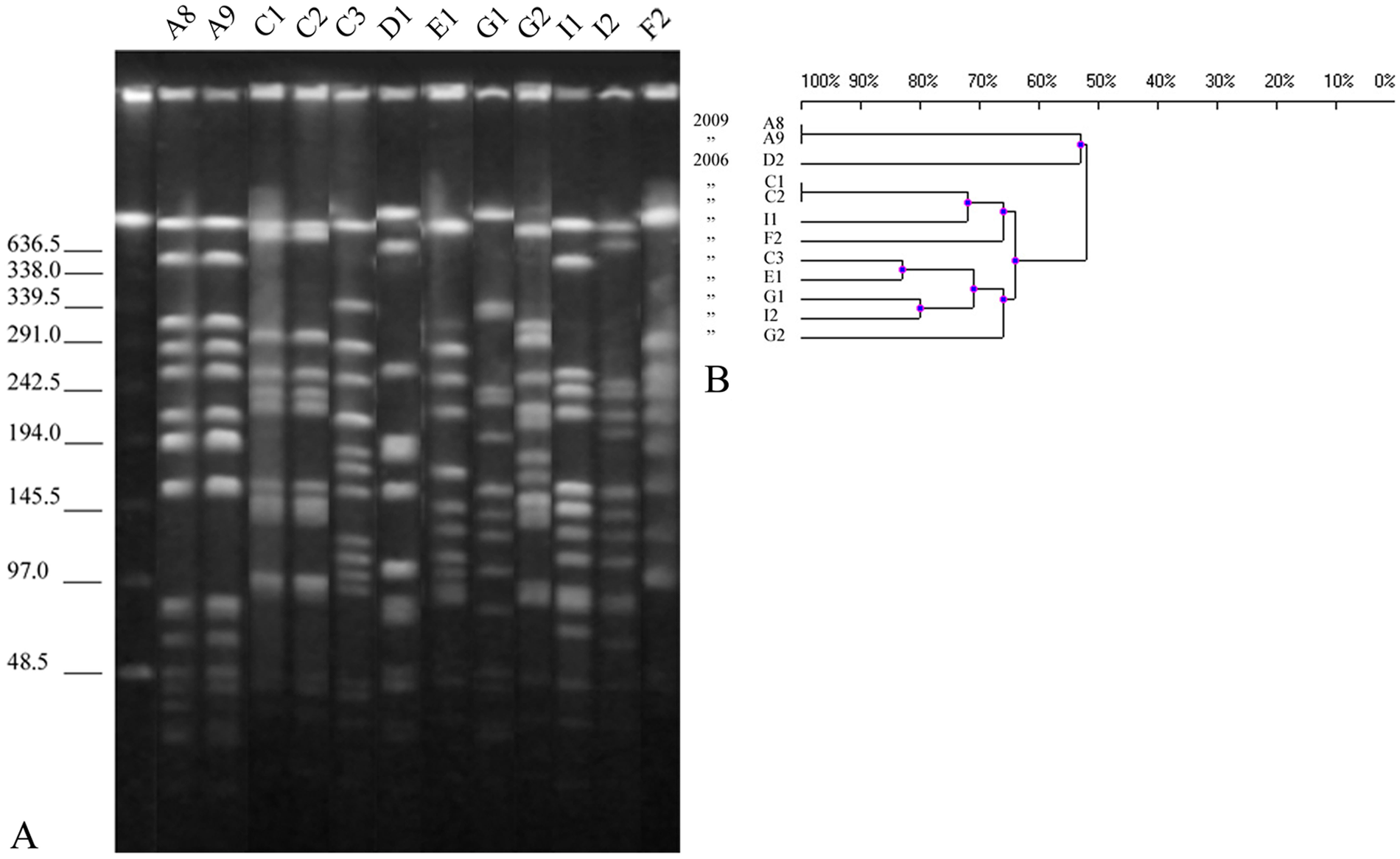
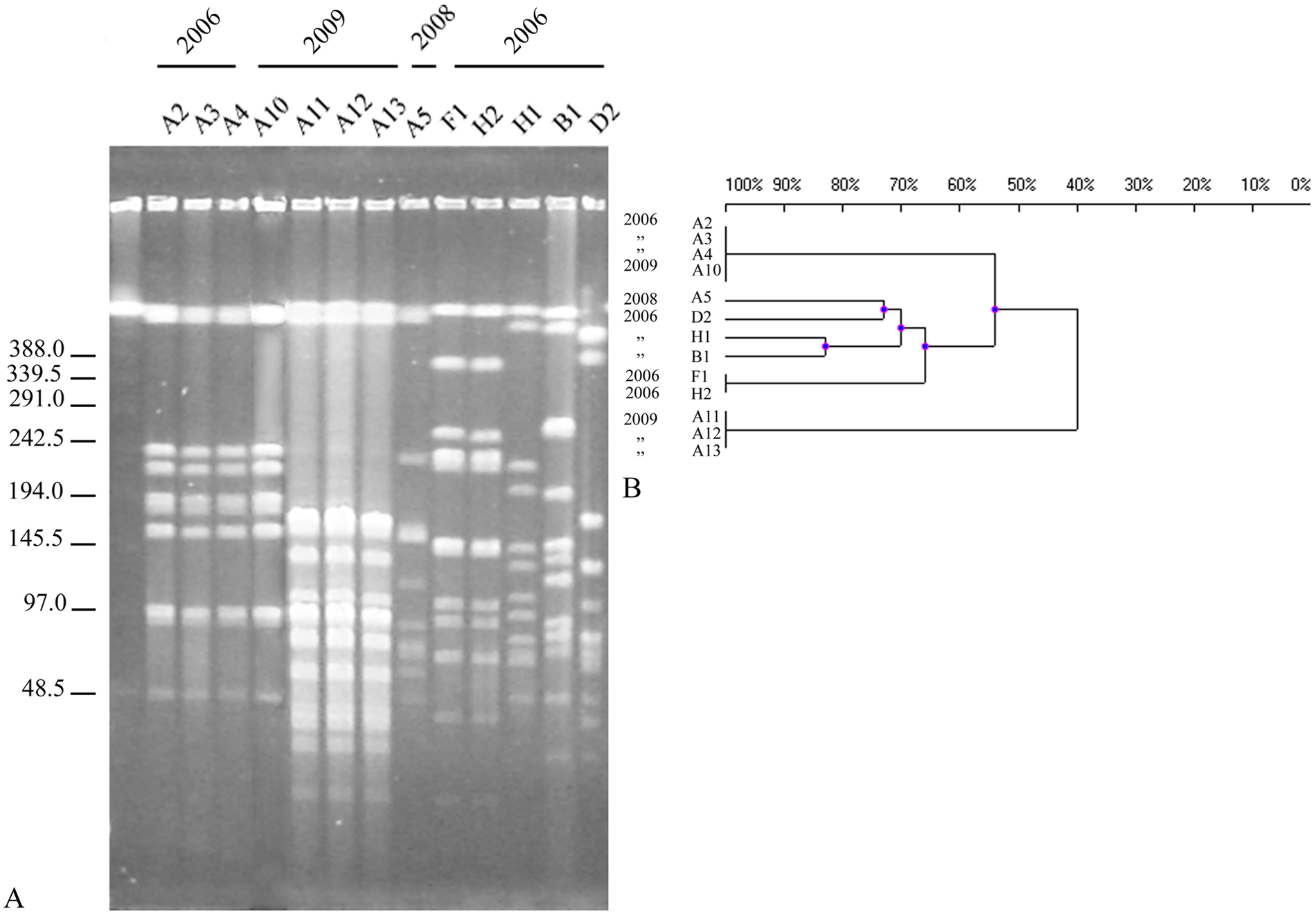
Pulsed field gel electrophoresis of M. haemolytica genomic DNA digested with SalI yielded 13 or 14 identifiable fragments, while digestion of genomic DNA of M. glucosida yielded 9–14 fragments (Fig. 1A and 2A, respectively). The reproducibility of the PFGE patterns was confirmed by growing and digesting 1 isolate of each species on 2 different occasions, M. haemolytica isolate G2 and M. glucosida isolate D2.
Pulsed field gel electrophoresis (
Pulsed field gel electrophoresis (
Ten distinct patterns were seen among the 12 M. haemolytica isolates. Mannheimia haemolytica isolates A8 and A9, and C1 and C2, pairs of isolates obtained at the same collection period from the same flocks, were identical in their endonuclease cleavage patterns. Isolate C3 was not related to the other 2 isolates from flock C.
Seven distinct patterns were seen among the 13 M. glucosida isolates. All 3 M. glucosida isolates obtained from flock A at different times in 2006 (A2, A3, and A4, with A3 and A4 obtained at the same time) had restriction endonuclease cleavage patterns identical to each other and to 1 of the 4 isolates obtained in 2009 (A10). Three of the 4 isolates (A11, A12, and A13) obtained in 2009 shared an identical restriction endonuclease cleavage pattern. Isolate A5, obtained in 2008, was not related to any of the other isolates associated with intramammary infection in this flock. Mannheimia glucosida isolates F1 and H2, obtained from different flocks, had identical patterns, while isolates H1 and H2, both obtained from the same flock at the same time from ewes with clinical mastitis, were not related.
The analysis of movement of sheep between properties revealed significant animal transfer onto the 8 farms in the current study. Sires from 27 other studs were purchased for use on these 8 Poll Dorset studs between 2004 and 2006. Ewes were purchased from 9 other studs over the same period, with an average of 8 ewes introduced into each stud per year. Four flocks (A, B, E, and H) had purchased a ram from a common stud, and flocks B and H yielded M. glucosida isolates that were more than 80% similar in the dendrogram (Fig. 2B).
DNA-based techniques are regarded as the most reliable tools for studying the epidemiology of M. haemolytica. 11 Pulsed field gel electrophoresis has been shown to be more discriminative than ribotyping or random amplified polymorphic DNA analysis for investigations of M. haemolytica isolates from cattle, sheep, goats, and chamois, with greater clonal correlation seen for the isolates associated with the same host within a geographical region than for those from different regions or hosts. 11 It has been suggested that selective pressure imposed by the different hosts may have resulted in genetic variation in this species. 11 A multilocus sequence typing (MLST) scheme has recently been developed for M. haemolytica and used to study the population structure of M. haemolytica isolates from 10 different countries obtained from cattle, sheep, goats, and chamois and to investigate the clonal relationships between these isolates and their common evolutionary origins. 14 Multilocus sequence typing data have demonstrated the high clonality of M. haemolytica isolates from different countries, and the greater sequence type diversity of M. haemolytica isolates from sheep than of those from cattle, as has been shown previously using other approaches. 14 While MLST has been commonly used to investigate clonal relationships between isolates from geographically and epidemiologically distinct sources, PFGE has been more commonly employed as a discriminative method for strain typing when investigating the relationships between more closely related isolates, such as those obtained from outbreaks of disease. 16
In a previous study, 2 ovine isolates belonging to serotype 11 of the former P. haemolytica, which has subsequently been classified as M. glucosida, were found to have identical PFGE patterns. 11 However, there has been no previous study of the molecular epidemiology of these 2 species in ovine mastitis.
Although some genomic instability has been seen in M. haemolytica after 36 passages in vitro, 17 at the lower passage levels used in the current study (≤10) such problems have not been reported. The reproducibility and stability of the PFGE patterns of the isolates of both species were confirmed by comparing the patterns obtained with different DNA preparations.
Mannheimia glucosida isolates A2–A4 from 2006 (but collected at different times throughout the year), and A10 from 2009, which had identical rpoB and rrnA gene sequences and phenotypic characteristics, also had identical PFGE patterns. The other 3 isolates from 2009, which shared an identical endonuclease cleavage pattern (but which was different from that of A2–A4), also had identical rpoB and rrnA gene sequences and phenotypic characteristics. Isolates F1 and H2, obtained from different flocks with no record of contact, were also identical to each other in all analyses. Although these 2 isolates had the same phenotypic characteristics as a second isolate from flock H (isolate H1), it did not have the same rpoB or rrnA sequence or PFGE pattern. Isolates A5, B1, and D2, which had identical phenotypic characteristics, but differed in their rpoB and rrnA sequences, also had distinct PFGE patterns. These results indicated that there was agreement between the restriction endonuclease cleavage patterns of M. glucosida isolates and their rpoB and rrnA sequences, and all M. glucosida isolates with identical PFGE patterns shared the same phenotypic characteristics, while M. haemolytica isolates with identical phenotypic characteristics and rpoB gene sequences could have different endonuclease cleavage patterns.
The sharing of PFGE patterns by isolates of M. haemolytica (A8 and A9; C1 and C2) and M. glucosida (A2, A3 and A4; A11, A12, and A13) within a flock suggests the possibility of horizontal transmission. Results of the current study also indicate that some strains appear to have been persistently associated with mastitis in flock A over a 3-year period.
The analysis of movement of stud Poll Dorset sheep between farms revealed significant animal transfer into the 8 properties in the present study. However, there was not enough evidence of clonal relationships between isolates on these farms to suggest transmission between farms.
Data from the current study suggests horizontal transmission of M. haemolytica and M. glucosida and greater persistence of specific strains of M. glucosida (such as strains belonging to pulsotypes 1’ and 7’) compared to other strains of M. glucosida associated with ovine mastitis. Lamb suckling has been suggested to be the major route of transmission of M. haemolytica into the udder from the nasopharynx of the lamb. 7 Stray lambs may have a role in transmission between ewes. There was more evidence for the possibility of horizontal transmission of M. glucosida than for M. haemolytica. Investigation of larger numbers of Mannheimia species isolates from a single flock may assist in understanding the significance of horizontal transmission in disease caused by these organisms.
Footnotes
a.
Fossomatic™ FC, FOSS, Hilleroed, Denmark.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
