Abstract
Methicillin-resistant staphylococci (MRS) were isolated from nasal swabs of 56 of 159 (35.2%; 95% confidence interval [CI]: 27.9–43.2%) healthy horses. Two nasal swabs were collected from each horse; 43 of 159 (27%; 95% CI: 20.5–34.8%) of the cohort were colonized by MRS strains in 1 nostril, while in the remaining 13 of 159 (8.2%; 95% CI: 4.6–13.9%), different or identical MRS strains were isolated in both nostrils. Of the 29 humans in close contact with the horses tested, 4 (13.8%; 95% CI: 4.5–32.6%) were found to be carriers of MRS. All isolates were coagulase negative with the exception of 2 coagulase-positive MRS strains, Staphylococcus aureus and Staphylococcus pseudintermedius, both isolated from horses. To assay the methicillin resistance, a susceptibility test to oxacillin with standardized disk diffusion method, a PBP-2a latex agglutination test, and a methicillin resistance gene (mecA) polymerase chain reaction assay were performed. Pulsed-field gel electrophoresis patterns of isolates from horses and humans in close contact with the horses revealed similarity. The results suggest evidence of transmission between animals, from animals to humans, and vice versa.
The incidence of methicillin-resistant Staphylococcus aureus (MRSA) in human and veterinary medicine has been increasing. 7,10,15,25 Methicillin-resistant staphylococci (MRS) are resistant to all penicillins, and the basis of this resistance is conferred by a penicillin-binding protein, PBP-2′ (or PBP-2a), which is absent in methicillin-susceptible strains. 6 PBP-2a is encoded by the methicillin resistance gene mecA, residing on a large mobile genetic element designated staphylococcal chromosome cassette mec (SCCmec). To date, 7 major variants of SCCmec, types I–VII, have been distinguished. 12,13 There is a limited amount of information available on MRS in animals compared with those in humans; however, dogs and other pets living in close contact with human MRSA carriers can become colonized with MRSA. 8,15,20 Failure to detect and treat these colonized pets can result in recurrent MRSA colonization or infection in humans. Therefore, it is important to identify the source of MRSA colonization in pets, which might be the cause of unexplained carriage or relapse of infection in humans. Indeed, antimicrobial therapy of health care workers and, at the same time, of MRSA carriers and any family members or pets that have been in contact with infected subjects can eliminate recurrent MRSA infections. 27 In addition, mecA-harboring, coagulase-negative (CoN) staphylococci have been isolated from several domesticated and healthy animals. 14,32 Potential human-to-animal transmission of MRSA has already been reported. 1 Studies in the United Kingdom concluded that MRSA is present in the general horse population and may represent a reservoir of new or rare MRSA strains that could be transmitted to humans. 3 Furthermore, in Japan, mecA-positive CoN staphylococci were cultured from the skin and nares of healthy horses. 32,33 Recently, high prevalence of the CoN staphylococci was found in healthy horses in Europe. 4,28,26 However, no MRSA strains have been isolated. No cases of nasal colonization of horses with MRS have been described in Italy. The aim of the current study was to investigate the prevalence of MRS, both coagulase-negative and coagulase-positive, in healthy horses and humans working with horses in the Campania region of Italy. A total of 159 healthy horses, housed at 3 different locations (farms A–C in the Campania region of Italy), were used in the current study. Farm A is an in vitro fertilization center consisting of 32 mares aged 4–20 years, farm B is a farm for training horses with 50 horses and 17 mares over 15 years of age, and on farm C, 49 racing horses and 11 racing mares aged 3–8 years were analyzed. All samples were collected during summer to autumn 2006. The farms had no history of MRSA. A total of 29 people, including veterinarians and animal handlers screened voluntarily, were also tested for the presence of MRS. Legal consent was required and obtained. A cotton-tipped swab was collected from each nostril and kept at 4°C (not longer than 24 hr) in Amies medium until processing. Nasal swab samples were plated on mannitol salt agar and incubated aerobically at 37°C for 24–48 hr. From each nasal swab, morphologically different isolates were worked up, and staphylococcal isolates were identified also by Gram stain, catalase, and staphylocoagulase (tube coagulase) reactions. An oxacillin (methicillin) susceptibility test of all isolates was performed by oxacillin disk diffusion method in accordance with Clinical and Laboratory Standards Institute (M31-A2, 2002) guidelines. The S. aureus ATCC (American Type Culture Collection) 29213 (oxacillin susceptible) and S. aureus ATCC 43300 (oxacillin-resistant) strains were also tested as positive control (for comparison). Identification was confirmed by miniaturized biochemical tests, a and methicillin resistance was also confirmed by a positive latex agglutination test. b Stock cultures were stored at −70°C in microbank vials c for further analysis. Bacterial DNA was used to detect mecA gene by polymerase chain reaction (PCR) technique 32 and analyzed by pulsed-field gel electrophoresis (PFGE). 5 The epidemiological relationship between isolates from horses and humans was assessed using described criteria. 24 Statistical analysis was performed by 1-way analysis of variance with Bonferroni post-test using GraphPad InStat Version 3.00 for Windows 95. d
Methicillin-resistant staphylococcus strains isolated from horses and humans from farms A, B, and C.

Genetic characterization by pulsed-field gel electrophoresis (PFGE). *
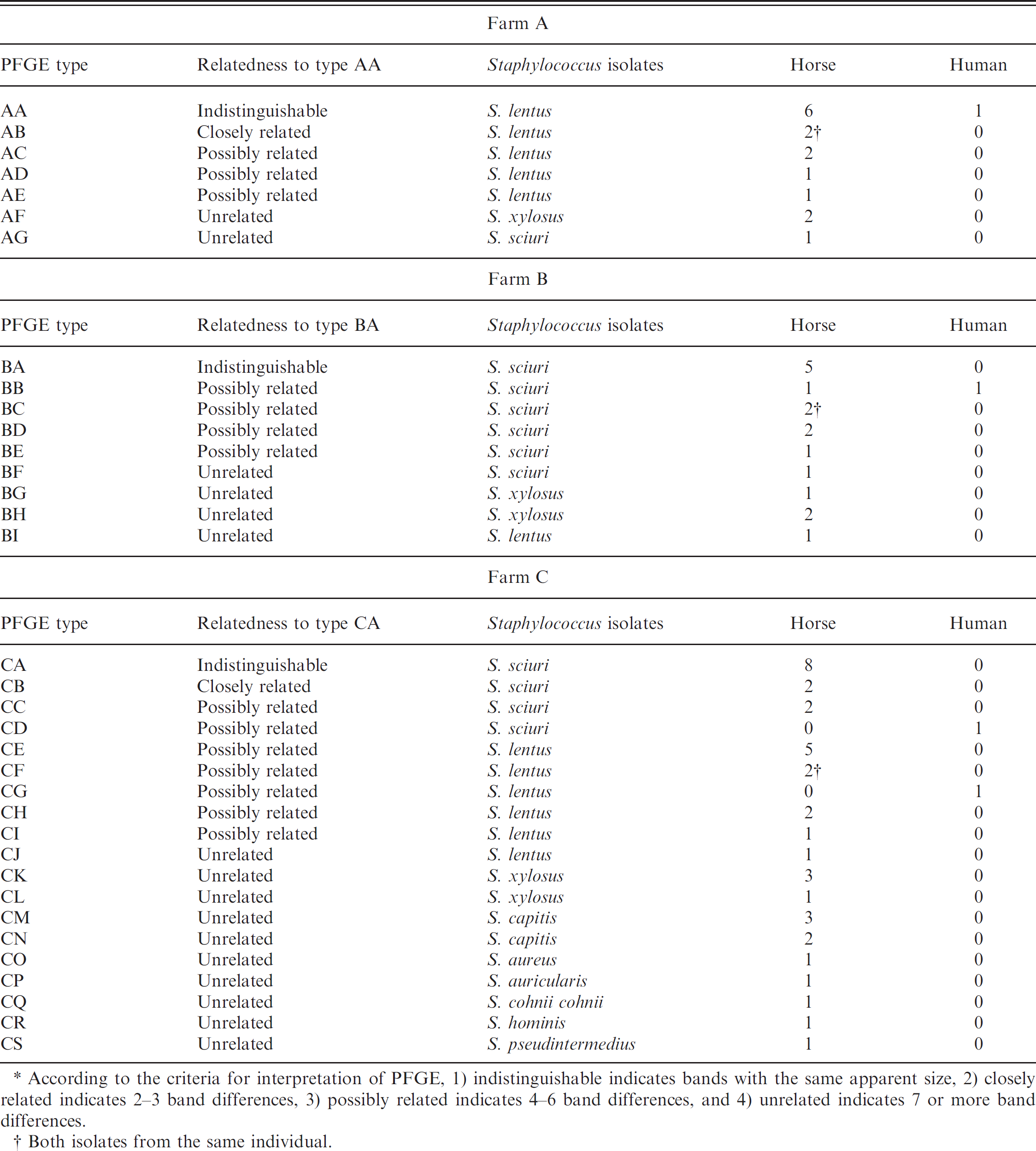
According to the criteria for interpretation of PFGE, 1) indistinguishable indicates bands with the same apparent size, 2) closely related indicates 2–3 band differences, 3) possibly related indicates 4–6 band differences, and 4) unrelated indicates 7 or more band differences.
Both isolates from the same individual.
From a total of 159 horses and 29 people, 56 and 4, respectively, had staphylococcus isolates showing resistance to methicillin. Forty-three of 159 (27%; 95% confidence interval [CI]: 20.5–34.8%) horses tested positive in only 1 nostril, while 13 of 159 (8.2%; 95% CI: 4.6–13.9%) horses presented different (7 horses) or identical (6 horses) MRS strains in both nostrils. For all isolates, the data obtained by oxacillin disk diffusion test was consistent with the mecA PCR results and PBP-2′ latex agglutination assay. The 159 horses were kept on 3 farms in the Campania region of Italy, and precisely 32, 67, and 60 horses were examined, respectively. The statistical analysis showed a significant difference (P < 0.001) in the prevalence of carriers (12, 12, and 37 carriers) and MRS isolates (15, 16, and 37) among the 3 examined farms, respectively. In Table 1, the identified species for each farm are described. In some cases, multiple staphylococcal species were isolated from the same carriers. Species identified as Staphylococcus lentus (12/15) and Staphylococcus sciuri (12/16) were mainly isolated from farm A and B, respectively, while in farm C, different MRS species other than the strain of MRSA were isolated. Of the 29 humans in close contact with the horses, 4 (13.8%; 95% CI: 4.5–32.6%) were found to be carriers of MRS; however, no MRSA was found in humans working with these horses. Sixty-eight strains from horses and 4 strains from humans identified as MRS were analyzed by PCR to test for the presence of the mecA gene. PCR results confirmed the presence of the mecA gene in all 72 phenotypically defined MRS strains. The PFGE allows the identification of different strains according to the DNA pattern. The results are summarized in Table 2. Seven S. lentus isolates (6 horses, Fig. 1 lanes 1–6; 1 human, Fig. 1 lane 7) from farm A shared a PFGE pattern designated AA. The identical PFGE pattern suggested a common origin and a possible transmission from horse to human or vice versa. Two S. lentus strains isolated from the same horse showed an identical profile (AB), closely related to the AA pattern. Four S. lentus horse isolates had possibly related profiles to themselves and to the AA pattern (AC, AD, and AE). Remaining staphylococci horse isolates showed unrelated profiles designated AF (Fig. 1, lanes 8–9) and AG (Fig. 1, lane 10). The BA pattern from 5 indistinguishable horse-isolated S. sciuri (Fig. 1B, lanes 11–15) was considered as a reference profile in farm B. Staphylococcus sciuri isolated from a worker had the same PFGE pattern (BB; Fig. 1, lane 16) as one of the horses from the same farm (Fig. 1, lane 17). This result suggests interspecies transmission. Comparison of PFGE patterns of isolates from farm C showed an indistinguishable profile (CA) in 8 strains of S. sciuri isolated from horses. Other staphylococcal strains were generally different. Isolates from horses and from humans in close contact with the horses on farm C showed no correlation between their PFGE profiles (data not shown). At present, MRSA, methicillin-resistant Staphylococcus pseudintermedius (MRSI), and CoN MRS infections represent the majority of problems in the human and/or animal community. It has been reported that many S. epidermidis and other CoN staphylococci from human clinical isolates are resistant to methicillin. 2,18 An evolutionary theory proposed that S. aureus could have acquired mecA from S. sciuri, a species frequently isolated from animals, which harbors a close structural homologue of mec. 9 These bacteria can serve as reservoirs of resistance determinants in the community, which could lead to the emergence of novel MRSA or MRS strains. The staphylococcal isolates were tested for growth on mannitol salt agar, which was followed by other tests such as Gram stain, catalase, staphylocoagulase (tube coagulase) reactions, and miniaturized biochemical tests. a In most cases, the CoN staphylococci grew and produced yellow colonies on mannitol salt agar, so it was necessary to perform further investigations to distinguish S. aureus from other staphylococci. 16,22 The species identification by miniaturized biochemical tests a was accepted if the species identification probability was >80%. To evaluate the methicillin resistance of all staphylococcal isolates, PBP-2a latex agglutination test b and mecA PCR were performed. Methicillin-resistant staphylococci were isolated from nasal swabs of a total of 56 of 159 (35.2%; 95% CI: 27.9–43.2%) healthy horses. Results of the present study show a higher percentage (20.7%) of CoN MRS present in horse samples, in contrast to a very low frequency (0.6%) of coagulasepositive MRS horse isolates. A total of 68 mecA-positive isolates were obtained. The data are in agreement with a previous study, 4 in which the incidence of 22.5% CoN MRS was reported in a healthy horse population in Holland. Moreover, this study reported no coagulase-positive MRS strain from horses and only 1 from humans, whereas in our study, we isolated 2 coagulase-positive MRS strains from horses and no strain from humans.
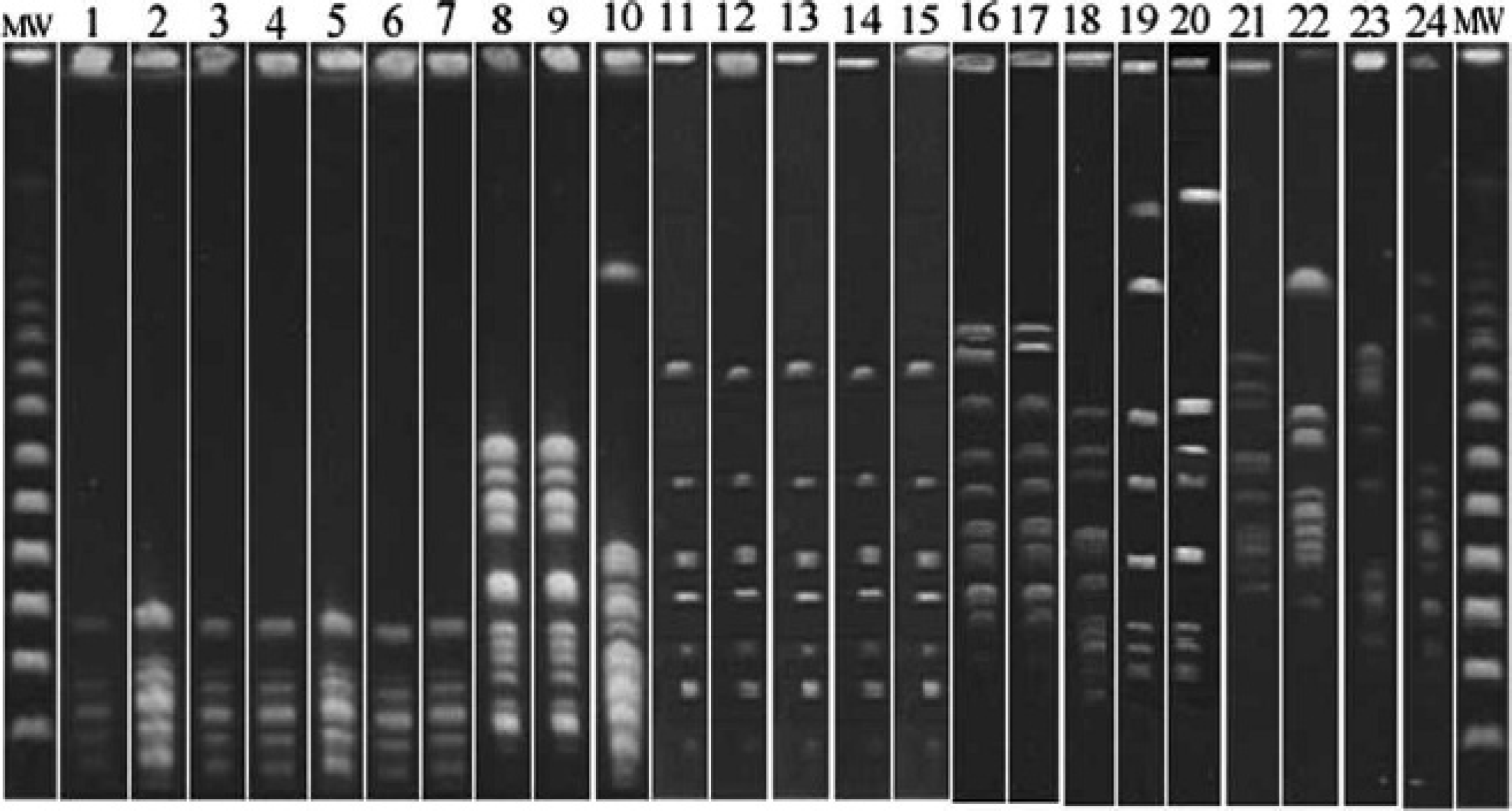
Pulsed-field gel electrophoresis (PFGE) profiles observed among methicillin-resistant staphylococci (MRS) isolated from horses and workers. Lanes come from different gels. The genomic DNA plugs were digested with SmaI endonuclease. MW = molecular weight. Lanes 1–6: indistinguishable PFGE patterns (AA) from horse Staphylococcus lentus isolated in farm A; lane 7: indistinguishable PFGE pattern (AA) from human S. lentus isolated in farm A; lanes 8–9: unrelated PFGE patterns (AF) from horse S. xylosus isolated in farm A; lane 10: unrelated PFGE pattern (AG) from horse S. sciuri isolated in farm A; lanes 11–15: indistinguishable PFGE patterns (BA) from horse S. sciuri isolated in farm B; lane 16: possibly related PFGE pattern (BB) from human S. sciuri isolated in farm B; lane 17: possibly related PFGE pattern (BB) from horse S. sciuri isolated in farm B; lanes 18–20: possibly related PFGE patterns (BC, BD, and BE) from horses S. sciuri isolated in farm B; lane 21: unrelated PFGE pattern (BF) from horse S. sciuri isolated in farm B; lane 22: unrelated PFGE pattern (BG) from horse S. xylosus isolated in farm B; lane 23: unrelated PFGE pattern (BH) from horse S. xylosus isolated in farm B; lane 24: unrelated PFGE pattern (BI) from horse S. lentus isolated in farm B.
Furthermore, we found that species identified as S. lentus and S. sciuri were more frequent in farm A and B, respectively, while in farm C, different MRS species (S. lentus, S. xylosus, S. sciuri, S. aureus, S. capitis, S. cohnii cohnii, S. pseudintermedius, S. auricularis, and S. hominis) were identified in addition to the only MRSA strain. The current study provides the first evidence of methicillin-resistant S. capitis, S. cohnii cohnii, S. pseudintermedius, S. auricularis, and S. hominis in horses, which raises the possibility that horses might serve as natural reservoirs of unusual CoN MRS isolates that could spread to humans. The farm staphylococcal species predominance is an interesting finding. Another interesting finding is the significantly greater MRS prevalence found in the racing horses group (farm C) than the other groups, probably due a combination of several factors such as age, stress, environment, movements/activities, and frequent administration of antimicrobials. In addition, 7 S. lentus isolates, 6 equine and 1 human, from farm A shared the same PFGE pattern, whereas S. sciuri isolated from a worker had the same PFGE pattern as 1 of the horses from farm B. The results could suggest evidence of transmission between animals, from animals to humans, and vice versa. The evidence of animals as reservoirs of antimicrobial-resistant bacteria has been reported. 11 In addition, increasing bacterial resistance to various antimicrobials frequently used in veterinary practice has been observed, including some broad-spectrum drugs and preparations used in human medicine. 1,14,19 Since horses often are in close contact with their owners and farm staff members, the risk of transmission of these bacteria between animals and humans (or vice versa) must be taken into consideration. A putative risk of MRSA cross-transmission between humans and horses has been described. 21,26 In addition, a case of MRSA human-to-dog transmission, 27 as well as other cases of MRSA cross-transmission between domestic animals and humans, 17,23 has been reported. Instead, very little is known about CoN MRS transmission between animals and humans. Furthermore, whereas the role of nasal carriage of S. aureus infections in humans is known, 31 the significance of nasal carriage in animals is not clear. Moreover, the role of nasal carriage of CoN MRS in both humans and animals has not been elucidated. Racing and riding horses have frequent contact with humans, and it may be that highly resistant mecA-harboring staphylococci are transferred from humans to horses or vice versa, which may be the cause of positive results when PFGE patterns were compared among isolates within each farm. The presence of indistinguishable PFGE types of S. lentus and S. sciuri in farm A and B, respectively, suggests a horizontal transfer among horses and the possibility that the resident staphylococcal population spreads easily among the equine population of the same farm.
In conclusion, unlike CoN MRS, the prevalence of MRSA and MRSI in healthy horses of the Campania region of Italy appears to be low. However, in both cases, the horses have shown to be a potential reservoir of MRS for cross-transmission between humans and horses. Horses have frequent and close contact with humans and other animals of the same species, creating the potential for transmission of micro-organisms such as MRS. Although the results reported in the current study need further investigation, the data suggest that it is necessary to estimate the risk for the contamination of horses by MRSA, MRSI, and CoN MRS. Monitoring MRS in animals should be promoted in veterinary surveillance programs on antimicrobial resistance to elucidate the possible contribution of horses to the spread of MRS in the community. Moreover, continued surveillance is indicated because overall MRS, with their important determinants of colonizing factors, biomaterial adhesion factors, production of biofilm, and resistance to opsono-phagocytosis, may contribute to selection and dissemination of antimicrobial-resistant staphylococci, leading to a continuing source of infection in humans and animals alike. Particular attention needs to be given to controlling the spread of these potentially serious pathogens in the farm environment to limit the risk of zoonotic transmission. 29,30
Acknowledgements. The authors thank the participating veterinarians, particularly Dr. Raffaele Frontoso, and farmers for their constructive collaboration.
Footnotes
a.
API-ID 32 Staph system, bioMérieux SA, Marcy L'Etoile, France.
b.
PBP2' Test, Oxoid, Basingstoke, Hampshire, England.
c.
PRO-LAB Diagnostics, Richmond Hill, Ontario, Canada.
d.
GraphPad Software Inc., La Jolla, CA.
