Abstract
A pastured 2-y-old cross-breed bull developed brainstem encephalitis (rhombencephalitis); Listeria monocytogenes was isolated from the brain. In the brainstem, there was perivascular cuffing, multiple microabscesses, and positive immunostaining for L. monocytogenes. Samples of bovine feces, water, feedstuffs, milking parlor soil, and bulk tank milk were collected from the dairy farm. Seven isolates of the genus Listeria were obtained, 6 of L. innocua and 1 of L. monocytogenes, which was found in the pasture where the bull grazed. Both isolates belonged to serotype 4b and were positive for internalins A, C, and J. According to the DNA fragment patterns of pulsed-field gel electrophoresis, the isolates were closely related. The source of infection was the pasture, implying that listeriosis should not be discounted in cases with compatible clinical signs but the absence of silage feeding.
Listeria are gram-positive, facultative, anaerobic coccobacilli that do not form spores. 25 At least 15 species have been identified, 3 and the genus is widely distributed in the environment, including soil, water, sewage, decaying vegetation, feed, and human and animal feces.3,14,16,25 In humans, Listeria monocytogenes is the primary cause of listeriosis, although L. innocua infection has also been reported.7,25 L. monocytogenes, L. ivanovii, and L. innocua have been isolated as causes of animal listeriosis.22,25,27 Raw materials may contribute to environmental strains colonizing food processing plants, serving as a source for postprocessing contamination of food and a risk to human health.2,10
The most common clinical presentation of listeriosis in ruminants is the nervous form (brainstem encephalitis, rhombencephalitis). 20 Feed, silage in particular, is the main source of infection in ruminants.8,9,18 Several authors have reported fecal shedding of Listeria spp. in healthy cattle, as well as presence of bacteria in the dairy farm environment, including in feedstuff, bedding, soil, manure, drinking water, bulk tank milk, bird feces, and insects.12,17,19,24 We investigated the presence of L. monocytogenes in a dairy farm herd and environment with a confirmed case of bovine listeriosis (rhombencephalitis) to locate the source(s) of infection(s) and to characterize the isolates obtained.
The Northwest Regional Laboratory of DILAVE “Miguel C. Rubino” received the head of a 2-y-old cross-breed bull (Bonsmara × Red Poll) that displayed neurologic signs of circling and depression. The bull had not been treated, and the clinical signs evolved for 3 d until the bull died. It was located on a dairy farm in Canelones, Uruguay, grazing with 3 other cross-breed bulls (Bonsmara × Red Poll) and 60 Jersey dry cows (“study herd”) on 30 hectares of a ryegrass and oat pasture. The dairy farm did not use manure or sewage sludge for pasture fertilization. After the bull died, the herd was moved to another paddock, and no more animals had clinical signs. In the laboratory, half of the animal’s brain was utilized for microbial isolation, and the remainder was used for pathology studies.
Nine days later, the dairy farm was visited to collect samples for bacterial culture. Based on reports of healthy carriers of L. monocytogenes,12,16,19 fecal samples were collected from 30 randomly selected animals. Fifteen samples were taken from dry cows where the clinical case was detected and 15 from the lactating cow herd. Three pools of feces were analyzed from each herd. Samples of drinking water from water troughs at the entrance to the milking parlor and the paddock where the bull was housed, milking parlor soil, bulk tank milk, and feedstuffs from both herds were also collected (Table 1). All samples were collected in labeled sterile containers, and transported in coolers until processed.
Feedstuffs collected the day of farm visit.

Paddock where the bull with clinical signs grazed and died.
Paddock where the study herd was grazing the day of the visit.
Paddock where the lactating cow herd was grazing the day of the visit.
Ration fed during milking in the milking parlor.
Half of the brain, 25 g of feces or other solid samples, and 500 mL of previously filtered water were processed as described. 19 Listeria spp. enrichment broth a (225 mL) was added to each culture, and the cultures were incubated at 30°C for 24 h. Aseptically, 100 µL of broth was transferred to 10 mL of another Listeria spp. enrichment broth b and incubated for 24 h at 30°C. Then, 100 µL was streaked onto modified Oxford medium (MOX) c and incubated at 35°C for 48 h. The milk was processed as described previously. 24 All colonies that exhibited a Listeria phenotype in MOX (black colonies with esculin hydrolysis) were tested as described. 24 Through multiplex polymerase chain reaction (PCR), the main serotypes of L. monocytogenes (1/2a, 1/2b, 1/2c, and 4b) were determined. 4 For species confirmation, the presence of internalin A (inlA) and virulence factors internalins C and J (inlC and inlJ) were determined using multiplex PCR. 15 Isolates were subtyped through pulsed-field gel electrophoresis (PFGE), according to the standardized CDC PulseNet protocol (http://goo.gl/nQ6DJQ) using AscI and ApaI restriction enzymes. DNA fragment patterns were visually analyzed and compared using predescribed criteria. 23
The other half of the brain was fixed in 10% buffered formalin for 48 h. Samples were routinely embedded in paraffin, sectioned, and then stained with hematoxylin and eosin. Brainstem sections were processed for the detection of L. monocytogenes for immunohistochemistry (IHC) using a specific antibody d at a dilution of 1/200. 5
Listeria monocytogenes was isolated from the bull brain (Table 2). On histologic examination, the brainstem had multiple perivascular lymphohistiocytic cuffs, multiple microabscesses, and malacic foci (rhombencephalitis). There was also diffuse, mild, suppurative meningitis. IHC staining was positive for L. monocytogenes in microabscesses in the brainstem (Fig. 1).
Isolation of Listeria spp. in bovine and environmental samples collected from the farm.
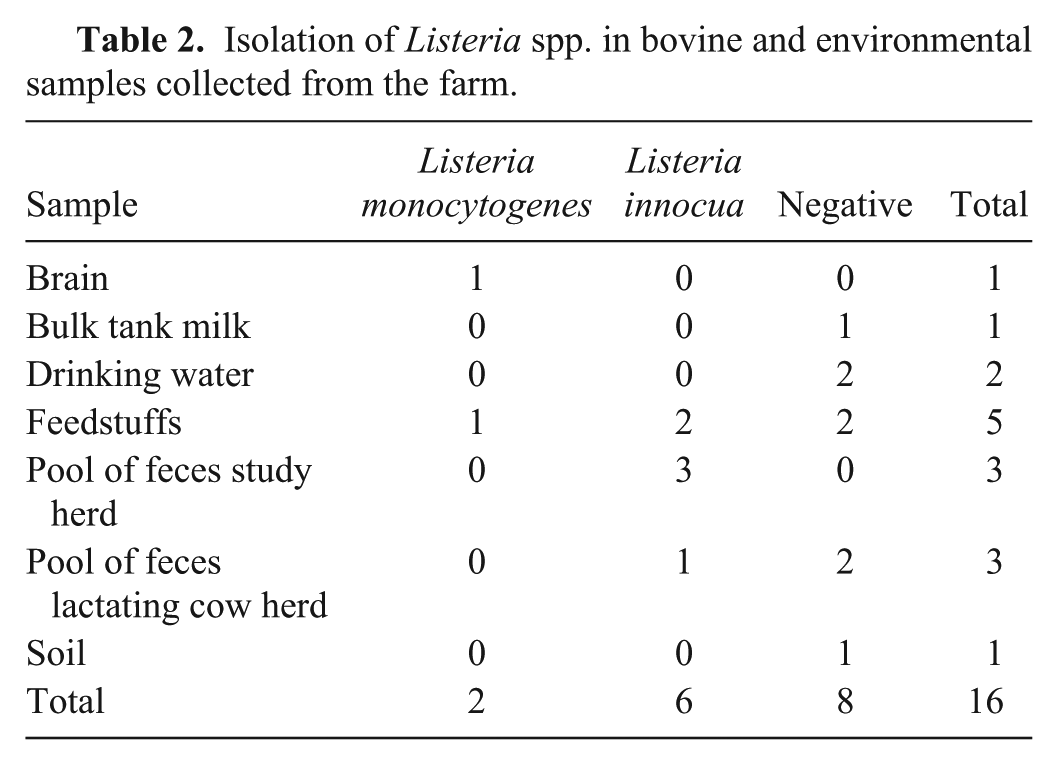
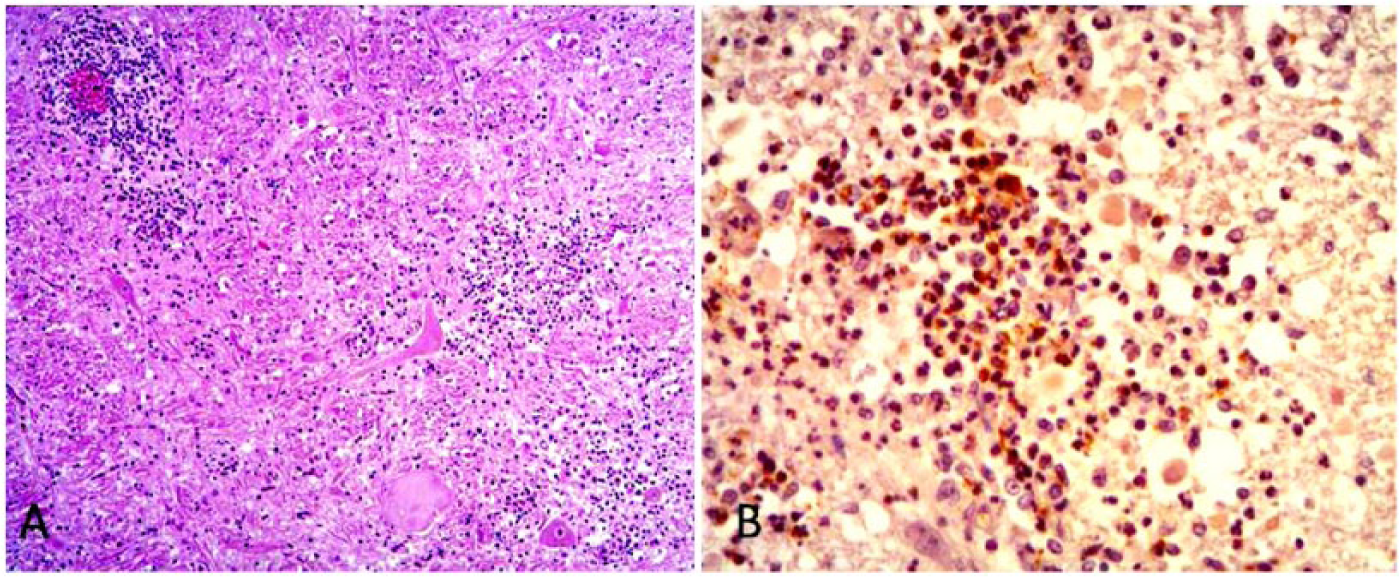
Brainstem encephalitis caused by Listeria monocytogenes in a bull.
Seven isolates of the genus Listeria were obtained from the dairy farm environment and cattle samples. Six of them correspond to L. innocua and 1 to L. monocytogenes (Table 2). The latter was isolated from the pasture (ryegrass and oat) where the bull with nervous symptoms was housed. Four L. innocua isolates were from pooled feces, 3 from the study herd, and 1 from the lactating cows. The other 2 L. innocua isolates were from the pastures where the 2 herds grazed the day of the visit (oat in both cases). Samples of silage, soil, bulk tank milk, and water tested negative (Table 2).
Both L. monocytogenes isolates obtained from the brain and the pasture were typified as serotype 4b. Additionally, both tested positive for the presence of genes inlA, inlC, and inlJ. According to the DNA fragment patterns of the PFGE, the isolate from the brain was classified as closely related genetically and epidemiologically to the L. monocytogenes isolate from the pasture where the bull grazed (Fig. 2).
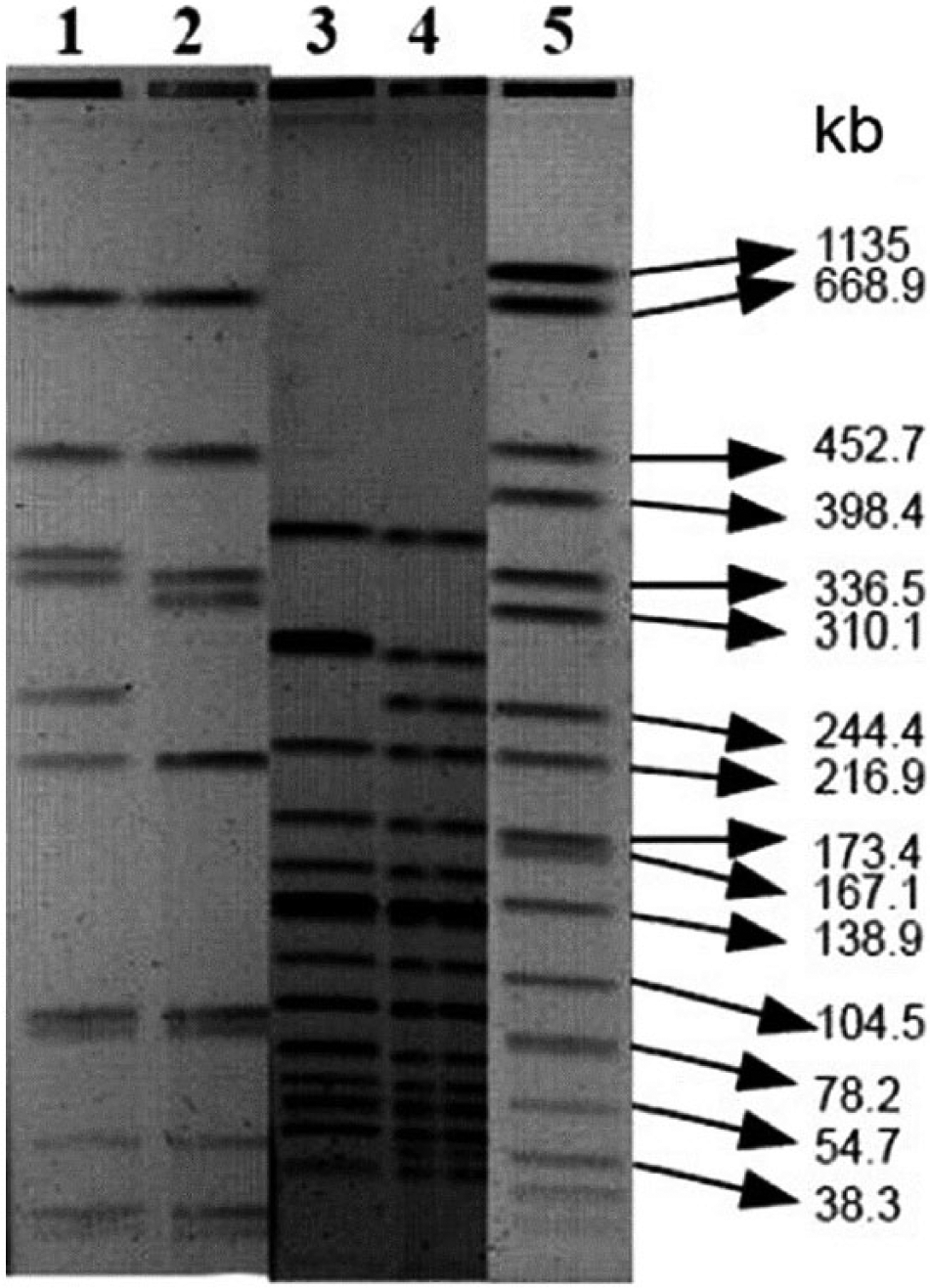
Listeria monocytogenes pulsed-field gel electrophoresis. Lane 1: brain isolate digested with AscI; lane 2: isolate from pasture digested with AscI; lane 3: isolate from bovine brain digested with ApaI; lane 4: isolate from pasture digested with ApaI; lane 5: standard Salmonella enterica subsp. enterica serovar Braenderup H9812. Comparing the isolates, the DNA had 2 different bands with AscI and 1 different band with ApaI.
Rhombencephalitis is the most common form of listeriosis in ruminants. 20 Isolation of L. monocytogenes from the brain was associated with lesions in the brainstem and with positive immunostaining. Although bacterial isolation will most likely remain the definitive criterion for diagnosis of listeriosis, IHC is useful when only formalin-fixed brain is submitted. 5 There are no reports of cross-reaction between L. monocytogenes and L. innocua in IHC. 22 Therefore, these results determined the causative agent and guided the subsequent epidemiologic study.
In the pasture where the bull showed clinical signs and later died, a strain of L. monocytogenes with genes that encoded for virulence factors was isolated. 15 Moreover, PFGE showed that both isolates were closely related. 23 These results suggest that this case of listeriosis was related to pasture grazing and not to contaminated silage, as described in other reports.8,9,19,20 There have been reports from New Zealand of ovine listeriosis associated with pasture grazing. 6 Variation of the DNA fragment pattern between the 2 isolates could be explained by the fact that sample collection was performed 9 d after the diagnosis, and the L. monocytogenes strains could have experienced genetic modifications during that period.23,26 It has also been reported that a wide genetic diversity of L. monocytogenes exists in farm environments and that cattle are capable of carrying different strains in their gastrointestinal tract.1,9,19 Because none of the dairy farm pastures were fertilized with manure or sewage sludge, it is probable that fecal shedding of L. monocytogenes by cattle was the source of the contamination. 11
The serotype of both isolates was 4b, which has been responsible for human listeriosis outbreaks worldwide and the encephalitic form of listeriosis in ruminants.21,26 L. monocytogenes is an opportunistic pathogen, and animals that develop listeriosis often have physiological factors such as pregnancy, stress, or concomitant conditions that compromise the immune response.20,25 Given that only 1 of 64 animals was affected in this case study, it is likely that the bull had an associated condition that made it susceptible to infection. This could not be determined because no additional organs or sera were submitted for study.
Based on the number of animals sampled, the fact that no additional fecal carriers of L. monocytogenes were detected could be related to a prevalence of fecal shedders of <10% in the dairy farm herd. Other authors have reported a prevalence of shedders below 10%.12,16 The fact that samples were collected 9 d after the clinical case may affect this result because it has been demonstrated that L. monocytogenes fecal shedding is intermittent and does not last longer than 10 d postinfection.9,28
Four fecal pools were positive for L. innocua in the study herd and in the lactating cows. There have been reports of listeriosis caused by this species in ruminants and in humans.7,22,27
The bagged silage tested negative for Listeria spp.; it was well preserved and not spoiled. Listeria was not isolated from bulk tank milk, likely the result of prevention of fecal contamination of milk, and also to correct sanitization and good maintenance of the milking equipment.12,13 Similarly, Listeria was not isolated from the drinking water, likely because of the absence of fecal contamination. Reports have described feces as the source of water contamination.12,17 Soil at the entrance and exit of the milking parlor had no growth of Listeria. Moisture and the presence of organic material are necessary conditions for bacterial growth of this genus in the soil.11,14
Footnotes
Acknowledgements
We thank Valeria Braga and Victoria Vico of the Instituto de Higiene who processed cultures for PCR and PFGE, and Marcela Preliasco who performed the IHC staining.
Authors’ contributions
C Matto contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. G Varela, MI Mota, and E Gianneechini contributed to analysis and interpretation of data. R Rivero contributed to acquisition of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions related to the integrity or accuracy of any part of the work are appropriately investigated and resolved.
a.
Listeria UVM I broth, Oxoid, Basingstoke, United Kingdom.
b.
Listeria UVM II broth, Oxoid, Basingstoke, United Kingdom.
c.
Modified Oxford agar, Oxoid, Basingstoke, United Kingdom.
d.
Listeria O antisera polyserotypes 1 and 4, Difco Laboratories, Sparks, MD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Project CSIC-PLANISA “Fortalecimiento de la Investigación de Calidad en Salud Animal” for the Master Scientae degree, Facultad de Veterinaria, Universidad de la República, Uruguay.
