Abstract
In late January 2013, 10 nonpregnant Lacaune dairy ewes raised under extensive husbandry management on a farm in Rio de Janeiro, Brazil, presented with the general clinical signs of lethargy, hyporexia, edema of the face, hyperemia of the exposed parts of the skin, mouth lesions, pyrexia, and lameness. Additionally, 2 pregnant ewes died suddenly after the onset of respiratory signs. The complete blood counts and biochemistry analyses showed neutrophilic leukocytosis with monocytosis and reactive lymphocytes, normocytic normochromic anemia and increased aspartate aminotransferase levels. Postmortem examination revealed erosions on the lingual mucosa, bilateral submandibular ganglia infarctions, yellow foamy fluid accumulation in the trachea and bronchial bifurcation, pulmonary congestion, and edema associated with hemorrhagic lesions on the pulmonary artery and heart. The clinical and pathological findings were suggestive of bluetongue. For a molecular and virological diagnosis, tissue samples were analyzed by Bluetongue virus–specific real-time reverse transcription polymerase chain reaction (qRT-PCR), and viral isolation was performed in embryonated chicken eggs. For viral typing, positive tissue and egg-isolated samples were analyzed by qRT-PCR using primers and probes specific for the structural VP2 gene in genome segment 2 of all 26 serotypes. There are still no contingency plans for responding to an outbreak of bluetongue disease in Brazil, and this episode emphasizes the need for continuing serological and entomological surveillance programs. Additionally, this report describes the isolation of Bluetongue virus serotype 4 in sheep in the Americas.
Keywords
Bluetongue is a viral disease of ruminants and camelids whose vector is the hematophagous Diptera order of the genus Culicoides. Bluetongue virus (BTV; family Reoviridae, subfamily Sedoreovirinae, genus Orbivirus) is characterized as a nonenveloped double-stranded RNA virus with icosahedral symmetry. There are at least 26 serotypes of BTV worldwide. 9 In South America, Central America, and the Caribbean, serotypes 1, 3, 4, 6, 8, 11, 12, 14, and 17 have been detected by serotype-specific antibodies, and serotypes 1, 3, 4, 6, 8, 12, and 17 have been detected by virus isolation. 6 In the United States, serotypes 1–3, 5, 6, 9–14, 17, 19, 22, and 24 have been identified. 11 Furthermore, the clinical presentation of bluetongue is a very rarely reported condition. In Brazil, serotype 12 has been isolated in asymptomatic cattle and symptomatic sheep and goats.1,2 Serotype 4 has been isolated in asymptomatic cattle in Brazil and Argentina.3,4 However, to the authors’ knowledge, serotype 4 had never been isolated in sheep in the Americas.
In late January 2013, 10 nonpregnant Lacaune dairy ewes raised under extensive husbandry management on a farm in Rio de Janeiro, Brazil, presented a history of apathy, inappetence, edema of the face (Fig. 1), arching of the back, and lameness. Additionally, 2 pregnant ewes in the last trimester of gestation died suddenly after the onset of respiratory signs.
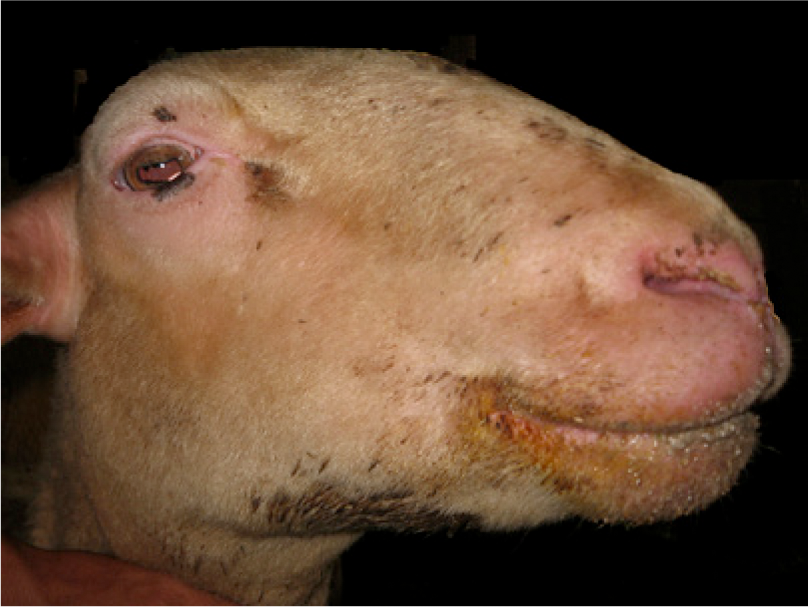
Sheep; edema of the face and hyperemia of the exposed parts of the skin.
The clinical findings and laboratory test results for the 10 ewes are summarized in Table 1. For symptomatic treatment, streptomycin (4 mg/kg) with penicillin (12.000 IU/kg) was administered intramuscularly every 24 hr for 3 days. After 1 week, only 1 ewe continued to exhibit prostration and hyporexia.
Clinical signs and laboratory tests from 10 nonpregnant ewes affected by bluetongue disease.*
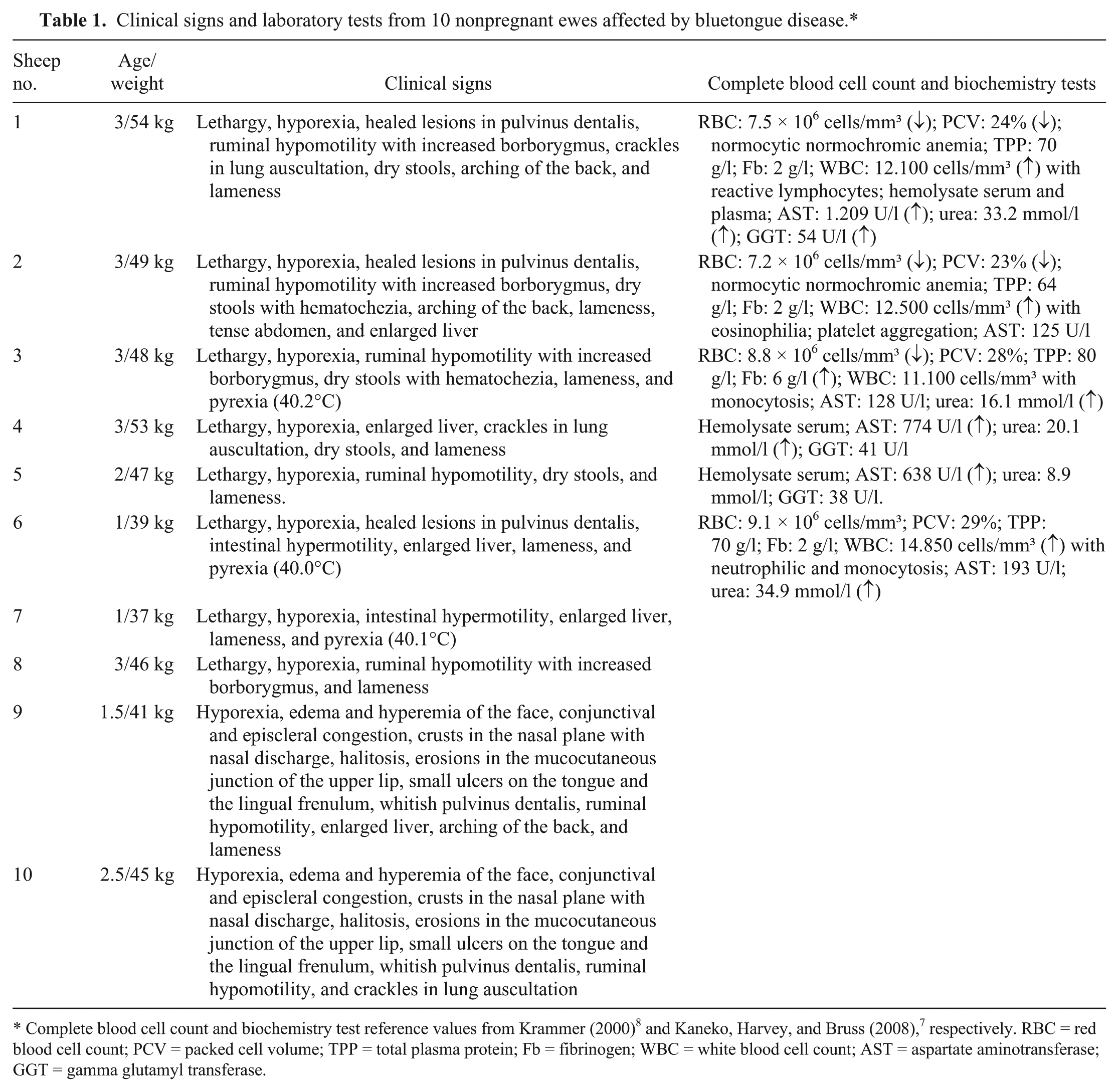
Complete blood cell count and biochemistry test reference values from Krammer (2000) 8 and Kaneko, Harvey, and Bruss (2008), 7 respectively. RBC = red blood cell count; PCV = packed cell volume; TPP = total plasma protein; Fb = fibrinogen; WBC = white blood cell count; AST = aspartate aminotransferase; GGT = gamma glutamyl transferase.
A week later, 1 ewe aborted twins in the last trimester of gestation. The following clinical signs were observed for this female: hyporexia, prostration, head and neck edema, halitosis, ulcers inside the oral cavity, anorexia, and death. At necropsy, there were erosions on the lingual mucosa, bilateral submandibular ganglia infarctions, yellow foamy fluid accumulation in the trachea and bronchial bifurcation, pulmonary congestion, and edema associated with hemorrhagic lesions on the pulmonary artery and heart. Representative sections from different organs were fixed by immersion in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4 µm, routinely processed, and stained with hematoxylin and eosin. Histologic examination indicated severe and diffuse acute pulmonary edema, multifocal hemorrhages in the myocardium and cardiac papillary muscles, vasculitis and multifocal intramural hemorrhages of the pulmonary artery (Fig. 2), discrete focal ulcerative stomatitis, discrete nonpurulent rumenitis and reticulitis, nephrosis, and purulent ulcerative pododermatitis.
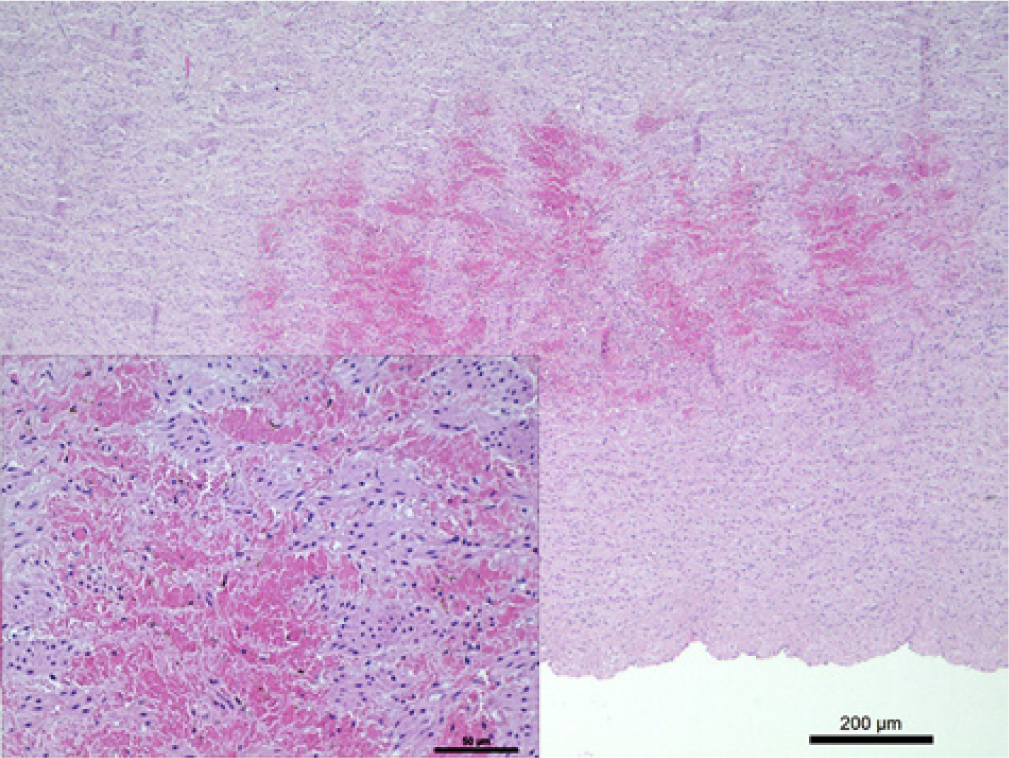
Sheep; heart; basis of pulmonary artery, with multifocal hemorrhaging in the intima and media. Hematoxylin and eosin (HE). Bar = 200 μm. Inset: sheets of erythrocytes between the tissue. HE. Bar = 50 μm.
The clinical and pathological findings were suggestive of bluetongue. For molecular and virological diagnosis, whole blood samples, along with dermal (mouth and coronary band of the hoof), lymphatic (submandibular, prescapular, and mediastinal lymph nodes), and visceral (liver, lung, and spleen) tissue samples originating from the dead ewe, were prepared in a 20% suspension of Eagle minimum essential medium with 2% antibiotics. These samples were screened by BTV-specific real-time reverse transcription polymerase chain reaction (qRT-PCR). 12 Viral genomes from these samples were extracted with a commercial reagent a according to the manufacturer’s recommendations. The viral RNA was amplified using primers and probes as previously described from the highly conserved region of the BTV nonstructural gene, NS3. 12 For viral typing, positive samples were analyzed by qRT-PCR using primers and probes specific for the structural VP2 gene in genome segment 2 for all 26 serotypes 13 ; these BTV serotype sequences were blasted (http://blast.ncbi.nlm.nih.gov/Blast.cgi), and the specificity of each 26 serotypes was confirmed. The amplification was optimized using a commercial kit b according to the manufacturer’s instructions. The sample was considered negative if the threshold cycle (Ct) was ≥ 40 amplification cycles. Quality control for the qRT-PCR process included both negative (nuclease-free water) and positive (BTV serotype 4 RNA) controls in each assay plate. The sensitivity of the test was 9.27 × 100 copies/ml. Using this technique, BTV-4 was identified. A final diagnosis was obtained by isolating the virus in embryonated chicken eggs (ECEs). Briefly, 0.1 ml of a 1:10 dilution of each sample was inoculated into the ECEs as previously described. 12 The viral isolates from these ECEs were also identified as BTV-4 using primers and probes specific for the structural VP2 gene in genome segment 2 for all 26 serotypes. 9 Serum samples from 97 sheep from the flock were tested for antibodies against BTV using the agar gel immunodiffusion method, 15 and 81.4% (79/97) were positive. In 77.1% (74/96) of the ovine serum samples, neutralizing antibodies against BTV-4 were detected, with titers ranging from 20 to 5,120. The same samples were also tested using the 3ABC enzyme-linked immunosorbent assay for foot-and-mouth disease 16 ; all samples gave negative results. The molecular and virological results corroborated the clinical signs and pathology that were consistent with cases of BTV infection described in the literature.1,10,14 The first report of BTV-4 outbreak, out of the African mainland, occurred in Cyprus in 1969. 13 Additionally, the first case where BTV-4 was isolated in the Americas was reported in 1980 in zebu cattle (imported from Brazil) in Florida, USA. 4 In 2002, in Argentina, BTV-4 was isolated from blood samples from asymptomatic cattle. 3 A phylogenetic analysis revealed a South American genetic identity for the Argentine sequences, indicating the independent evolution of this viral lineage. 5
There are still no contingency plans for responding to an outbreak of bluetongue disease in Brazil. Therefore, when vesicular disease is suspected, the Animal Defense Office initiates procedures developed against foot-and-mouth disease. After a negative differential diagnosis, additional relevant measures are taken according to the epidemiology of the disease involved. During the outbreak reported herein, the following measures were implemented inside the affected farm: 1) prohibition of movements of susceptible animals; 2) blood and serum collection every 2 months for qRT-PCR and agar gel immunodiffusion flock tests; and 3) the history of sheep movements was traced. The farm could only be released after the absence of clinical signs and 2 sequential negative qRT-PCR results from the flock. In the perifocal area, epidemiological and serological surveillance programs were instituted. Furthermore, a stamping-out policy is not justified in Brazil, and vaccination is still not allowed as a control measure.
In summary, the present study reports a BTV-4 outbreak in a dairy sheep flock in Brazil. This outbreak emphasizes the need for continued serological and entomological surveillance programs with an effective contingency plan for cases where infection is detected, particularly in light of BTV epidemiology and its impact on animal production.
Footnotes
Acknowledgements
The authors thank the anonymous reviewers from the Journal of Veterinary Diagnostic Investigation for their critical comments that reshaped the paper into its final version.
a.
TRIzol reagent, Life Technologies, Carlsbad, CA.
b.
LightCycler 480 RNA Master hydrolysis probes, Roche Diagnostics GmbH, Mannheim, Germany.
c.
PrioCHECK FMDV NS, Prionics, Schlieren-Zurich, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported by the Carlos Chagas Filho Foundation for Research Support in Rio de Janeiro (FAPERJ; E-26/102.963/2012) and Instituto Biologico. Felipe Z. Brandão is a National Council for Scientific and Technological Development (CNPq) fellow.
