Abstract
Cell-mediated immunity (CMI) diagnostic tests, such as the gamma interferon enzyme-linked immunosorbent assay (IFN-γ ELISA) and the Johnin skin test, have the potential to detect animals infected with Mycobacterium avium subspecies paratuberculosis (MAP) early in the course of the disease. While these CMI tests tend to be relatively specific in noninfected flocks, in MAP-infected flocks, these tests often identify animals that cannot be confirmed infected by any other reference test, including necropsy and culture. The aim of this study was to determine if antigen exposure by inhalation or oral ingestion of killed MAP organisms would cause a detectable CMI response in sheep. Forty-eight lambs 4 months of age were randomly divided into a control group, an orally exposed group (dosed with 1 × 10 10 autoclaved MAP organisms 3 times), and an inhalation-exposed group (dosed once with 1 × 10 5 dead organisms). Lambs were skin tested and/or bled pre-exposure and 1, 2, 3, 4, and 12 months postexposure. No significant difference was seen with either the oral- or inhalation-exposed groups of lambs versus controls with either the IFN-γ ELISA or the skin test at any time pre- or postexposure. These results suggest that infection/invasion of MAP organisms must occur in order to have a positive skin test or IFN-γ ELISA beyond the false-positive rate. Simple exposure is not enough to elicit a detectable CMI response.
Mycobacterium avium subspecies paratuberculosis (MAP) causes Johne's disease, which is one of the major diseases that cause chronic, severe weight loss and eventual death in sheep and other ruminants. Strong interest has been expressed in developing diagnostic tests that detect animals in the early stages of disease, as current diagnostic tests (fecal culture and serology) usually detect animals only after they start to shed the organism. Because the cell-mediated immune (CMI) response predominates in the early stages of MAP infection, diagnostic tests based on CMI responses hold considerable promise. 9 Two such tests, the Johnin skin test and the gamma interferon enzyme-linked immunosorbent assay (IFN-γ ELISA) have proven to be relatively specific (>98%) in sheep from noninfected flocks, provided a specific and sensitive antigen is used. 7,8
However, when these CMI-based diagnostic tests are used in flocks or herds with infected animals and a reference test such as histology or tissue culture is used, specificity drops to below 90%. 3,4,8 Animals in these infected populations that have positive skin test responses or have high optical density (OD) IFN-γ ELISA readings but do not appear to be infected using the reference test are of particular interest and must be more clearly defined if these tests are to have clinical value in infected populations.
Three potential explanations for these CMI test-positive but reference test-negative sheep have been proposed in a previous article: 8 1) the reference test is not sensitive enough to identify all infected animals, 2) the animals were infected in the past but have recovered and harbor a residual immune response, and 3) animals were exposed to MAP antigens but infection did not occur. If the third hypothesis occurs, these tests will likely have limited use in infected populations due to environmental contamination. While all of these hypotheses need to be tested, the objective in this study was to evaluate the exposure-without-infection hypothesis. Specifically, the aim was to determine if antigen exposure by inhalation or oral ingestion of killed MAP organisms would cause a detectable CMI immune response.
A total of 48 white-faced cross-bred ewe lambs 4 months of age from the Iowa State University McNay research flock were randomly assigned to 1 of 3 groups. This flock of 300 ewes was managed as a typical Midwest commercial winter lambing flock and had been used as a noninfected flock in previous studies. 7,8 The flock was rigorously screened for Johne's disease in 1999–2001 as previously described. Since then, cull and thin animals have been screened via culture or necropsy and strict biosecurity practices have been in place. The flock has been closed to outside ewes for over 15 years, and only skin test-negative rams have been introduced.
The MAP strain used in this study was MAP ATCC strain 19698 (bovine strain). This strain was chosen because of ease of differentiating it from a wild-type sheep strain in the event the flock became infected with MAP after the study started. It is also the strain routinely used in the authors' laboratory to experimentally infect calves. 11,12 The bacteria were grown in Middlebrook 7H9 liquid media (pH 6.0) supplemented with 10% oleic acid albumin dextrose complex, a 0.05% Tween 80, a and 2 mg/ml of mycobactin J. b MAP cultures were grown to log phase at an OD 450 nm (OD450) of 0.4, at 37°C without shaking. Bacteria grown to an OD of 0.4 have been determined to be at a concentration between 1 × 107 and 1 × 108 by plating out serial 10-fold dilutions on Herrold's egg yolk media a (HEYM) and adjusted to obtain a final concentration of 1 × 1010 for the oral drench or 1 × 105 for the aerosolization. The bacteria were then autoclaved at 120°C at 17 psi for 30 minutes and then inoculated onto HEYM and monitored for growth for 8 weeks.
The first group of lambs was orally drenched 3 times, 1 week apart with 1 × 1010 of killed MAP organisms for a total of 3 × 1010. Group 2 lambs were forced to inhale 1 × 105 one time using a nebulizer and a mask as described elsewhere. 5 The last group was left as controls. The ewe lamb groups were housed separately in 20 m2 pens in an open-sided barn until 1 week after the final oral dose was given, and then the animals were housed together in a small 3-acre pasture. Lambs were skin tested using 0.1 ml of a 1 mg/ml purified protein derivative (PPDj) c in the axillary region and palpated for induration 72 hours later as described previously, 7 blood was collected in 10-ml heparinized blood tubes immediately before exposure, and the lambs were then bled once every 30 days and skin tested every 60 days for 4 months and then at 12 months. Protocols were approved by the Institutional Animal Care and Use Committee of Iowa State University.
Whole blood for the IFN-γ ELISA was transported at ambient room temperature and stimulated within 8 hours as described previously. 8 Briefly, the whole blood was allocated into three 1-ml aliquots. One well was left as a negative control, 1 well was stimulated with 1 mg of Staphylococcus enterotoxin A (SEA) d as a positive control, and 1 well was stimulated with 10 μg of PPDj. The samples were incubated at 37°C for 24 hours, and the plasma was harvested. A commercial IFN-γ ELISA kit e was used according to manufacturer's instructions. Samples were considered valid if the OD of the SEA well was greater than 0.10. Results were reported subtracting the unstimulated well OD values from the wells stimulated with PPDj. The same PPDj that was used in the IFN-γ ELISA was used in the skin test.
For statistical analysis, IFN-γ ELISA results from the 3 groups were compared at each time point using analysis of variance in Stata 9. f The categoric skin test data were analyzed using the Fisher's exact test.
There were no differences in either the mean OD value of the IFN-γ ELISA or the skin test between the groups of lambs, either before or after exposure to MAP organisms. (Table 1). If a cut point at or above 0.2 was used in the IFN-γ, no lambs would have tested positive at any time throughout the experiment. A previous receiver operating characteristic analysis found that a cut point of 0.25, using this particular PPDj in the IFN-γ ELISA, was determined to have the best overall accuracy. 8
Because of the expense and lack of required facilities, a positive control group (sheep dosed with live organisms) was not used. While this was a significant study design flaw, other researchers have found that live oral inoculations in sheep of laboratory-propagated MAP organisms reliably produced CMI responses at lower doses than those used in this study before 4 months postinfection. 1,2,6 The authors were not aware of any MAP-infection studies using a nebulizer, so the inhalation dose 1 × 10 5 was similar to the high-dose regimen used in deer with Mycobacterium bovis, using the same apparatus, and it is unlikely that animals would naturally inhale a higher number of dead organisms. 5 A bovine-type strain was used in this study rather than a sheep strain. While there are some questions about the differences in strain pathogenicity, experimentally both strains caused similar early CMI responses when live organisms were used. 10
Average gamma interferon enzyme-linked immunosorbent assay (IFN-γ ELISA) optical density values and number of animals with positive skin test results in lambs pre-and postexposure to autoclaved Mycobacterium avium subspecies paratuberculosis.
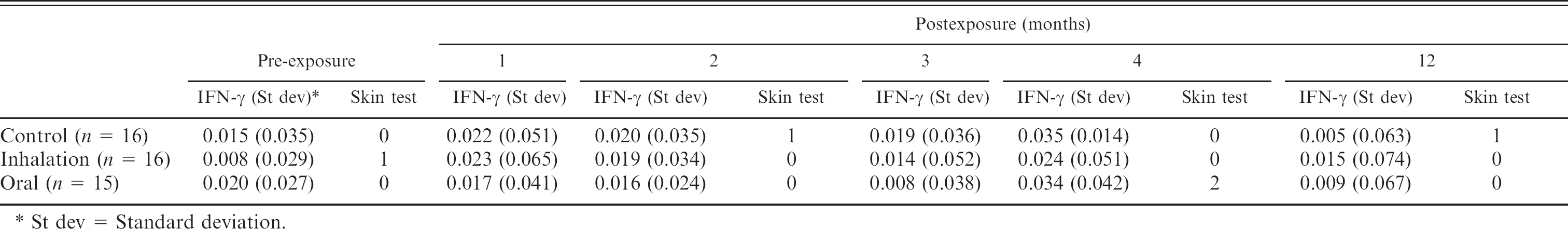
St dev = Standard deviation.
The authors have shown in previous articles that the skin test and IFN-γ ELISA can be specific in MAP-noninfected populations. This has not been true using postmortem testing of animals residing in infected populations as the reference test. While it is likely the reference tests lack sensitivity, animals in noninfected populations have no opportunity for environmental exposure to antigens of dead organisms. Because there is interest in using these tests in infected populations, the potential causes of positive CMI test results must be understood. The results of this current study suggest that in order for a detectable CMI response, as measured by the skin test or the IFN-γ ELISA, to occur against MAP, actual infection/invasion with MAP organisms rather than simple exposure must occur. Therefore, the assumption can be made that positive test results (beyond the false-positive rate) would be the result of infection, not just simple exposure to dead organisms, and the sheep are not identified as infected at necropsy because the reference tests are not sensitive enough or the sheep are no longer infected.
What is not known is if and how often animals recover from an infection with MAP organisms. If most animals that get infected become latently infected and harbor the organism, those animals are likely to be potential threats to the rest of the flock or herd. A producer could then potentially use CMI test results as a selection tool for replacement animals. On the other hand, if most infected animals recover, a single positive CMI test result may have little meaning. Indeed, it seems reasonable that in heavily contaminated environments, there can be nearly 100% infection rates, some animals may eliminate the organism, some may remain latently infected, and a small number of those may come down with clinical Johne's disease. Whatever the case, further understanding of the meaning of a positive CMI result is certainly needed for proper positive test interpretation. This would likely require longitudinal studies.
Footnotes
a.
Becton Dickinson Microbiology, Sparks, MD.
b.
Allied Monitor Inc., Fayette, MO.
c.
Johnin PPD 0201 lot 3, NVSL, Ames, IA.
d.
Toxin Technology, Sarasota, FL.
e.
Bovigam, Prionics, Omaha, NE.
f.
Stata Corp., College Station, TX.
