Abstract
Bovine leukosis associated with infection with the delta retrovirus
Bovine leukosis is an endemic disease in both dairy and beef herds throughout the United States. The causative virus is a delta retrovirus known as
The majority of states in the United States are currently accredited to be free of bovine tuberculosis, with the exception of Michigan, Minnesota, New Mexico, and California. In 1994, infection with
An 8-year-old, female Holstein was presented to the Diagnostic Center for Population and Animal Health (DCPAH; Lansing, MI) in 2006 as part of a screening program for bovine tuberculosis. Since this cattle herd was located in a bovine tuberculosis endemic area, the cow had received annual skin tuberculosis tests. In October 2006, this cow reacted to its annual skin test and several weeks later was confirmed as suspect on its gamma interferon assay. The commercial whole-blood gamma interferon assay uses the average optical density values of 2 enzymelinked immunosorbent assay (ELISA) plate wells inoculated with plasma from bovine purified protein derivative (PPD)-stimulated blood, minus the average optical density values of 2 wells inoculated with plasma from avian PPD-stimulated blood. Results of less than 0.1 are considered negative; results greater than 0.1 are considered positive. In the state of Michigan, these gamma interferon-positive reactions are considered “suspects” because only postmortem examination with positive polymerase chain reaction (PCR) for
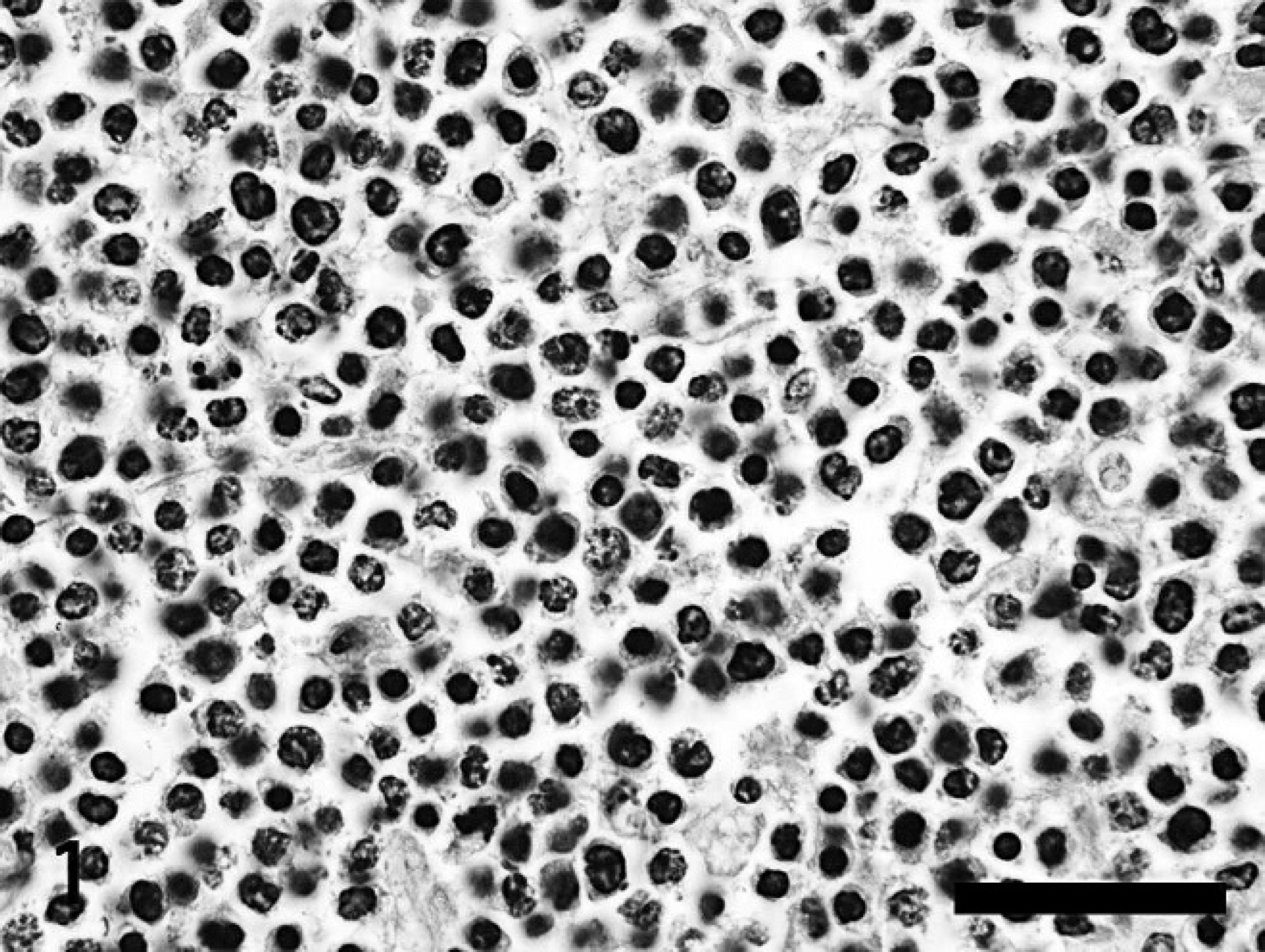
Lymph node, 8-year-old female Holstein. The architecture of the lymph node is replaced by monotonous sheets of medium-sized neoplastic lymphocytes supported by a rare fine trabeculae of fibrovascular stroma. There are numerous apoptotic bodies and karyorrhectic cell debris admixed with neoplastic lymphocytes. Hematoxylin and eosin. Bar = 100 μm.
Following euthanasia at the DCPAH, a necropsy was preformed. All examined peripheral and visceral lymph nodes were markedly enlarged, with most ranging from 4 to 10 times the expected size. These lymph nodes were soft to fatty in texture, bulged on the cut surface, and varied from homogeneously pale white to mottled tan and dark red (Fig. 2). No distinction between cortical and medullary areas was apparent. In addition to being markedly enlarged, submandibular, mesenteric, middle mediastinal, and caudal mediastinal lymph nodes contained variably sized, often coalescing caseogranulomas. These ranged from 1 cm to 8 cm in diameter, were generally irregularly spherical, and were fairly circumscribed. Caseogranulomas were composed of a central necrotic cavity containing poorly organized, dense, friable pale yellow to tan, caseous debris and/or viscous, turbid, yellow fluid. Such central areas were generally surrounded by a 2–7-mm band of pale tan fibrosis that separated caseogranulomas from the surrounding lymphoid tissue.

Lymph node, 7-year-old female Holstein. This cow was a herd mate of the cow described in the current report that was infected with
Additional lesions included focally extensive marked thickening of the pericardial sac and the wall of the abomasum by soft, homogeneous pale white tissue. Such changes were poorly demarcated, with approximately 60% of the abomasal wall and 75% of the pericardial sac being affected. In the abomasum, normal architecture of all layers of the abomasal wall, including the mucosa, submucosa, and muscularis, was lost, and the thickness was expanded to 1.5–2.5 cm. Normal rugal folds in the mucosa were widely lost, and there were 5–7 focal, 1–7-mm, irregularly shaped, sharply demarcated, dark-red areas of superficial mucosal erosion.
A second 7-year-old cow from the same herd was presented 2 months later for necropsy with similar enlargement of multiple lymph nodes and thickening of the abomasum, but without the distinct caseogranulomas. This cow was diagnosed with lymphoma based on gross and histologic examination.
Samples of retropharyngeal, submandibular, parotid, tracheobronchial, mediastinal, hepatic, mesenteric, ileocecal, and supramammary lymph nodes from the first cow were fixed in 10% formalin for histopathology, and fresh samples were submitted to the National Veterinary Laboratory Services (NVSL; Ames, IA) for mycobacterial culture. 14 Formalin-fixed samples were routinely processed for histopathologic examination, sectioned at 5 μm, and stained with hematoxylin and eosin and Ziehl-Neelsen acid-fast stain. Additionally, immunohistochemistry (IHC) using anti-cluster of differentiation (CD)3, CD79a, B-lymphocyte antigen (BLA)36, and CD20 antibodies was preformed to identify T cells (CD3 positive) and B cells (CD79a, BLA36, and CD20 positive) using standard techniques. 16 A lymph node with lymphoma and several normal lymph nodes from cattle in previous diagnostic cases were used as controls for IHC. Immunohistochemistry on a bovine lymphoma control sample yielded strong positive perimembrane to stippled cytoplasmic labeling in neoplastic cells for CD79a and strong perimembrane immunoreactivity for CD20, indicating their applicability for use in IHC studies of lymphoma in cattle. BLA36 IHC also demonstrated strong positive perimembrane immunoreactivity in the control lymphoma sample, but IHC on several normal bovine lymph nodes labeled both lymphocytes in B-cell areas and histiocytes.
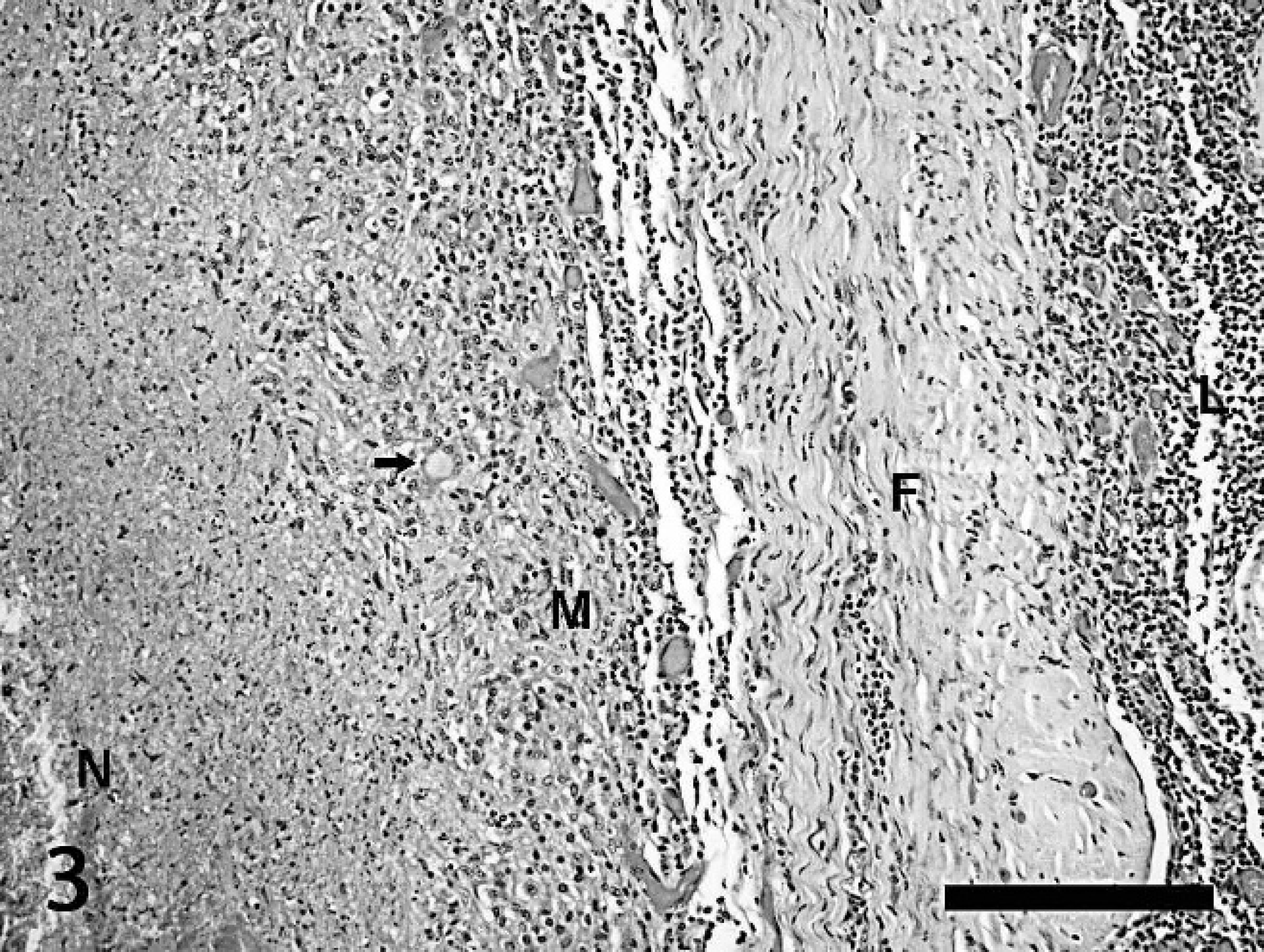
Lymph node, 8-year-old female Holstein. This image represents the wall of a caseogranuloma. To the left of the image is the necrotic core composed of dense amorphous accumulations of eosinophilic caseous debris (N). A zone of plump epithelioid macrophages and rims of lymphocytes (M) surrounds the necrotic center. There are occasional multinucleated giant cells, highlighted by an arrow, within this zone. Dense bands of fibrosis (F) envelop the caseogranuloma and separate it from surrounding monomorphic populations of neoplastic lymphocytes (L). Hematoxylin and eosin. Bar = 500 μm.
Histologically, dense sheets of relatively monomorphic neoplastic medium- to large-sized lymphocytes obscured the normal tissue architecture of all lymph nodes (Fig. 1). Neoplastic cells had mild to moderate amounts of eosinophilic cytoplasm and distinct cell borders. Nuclei were round and occasionally indented, coarsely stippled to hyperchromatic, and often contained 1–2 distinct nucleoli. Anisokaryosis was moderate. Mitotic figures were common, with up to 6 observed per high-power field. Numerous shrunken, apoptotic bodies containing karyorrhectic debris and occasional plump histiocytes were distributed throughout sheets of neoplastic cells.
Sections of submandibular, mesenteric, and mediastinal lymph nodes contained well-demarcated, occasionally coalescing caseogranulomas in addition to the neoplastic lymphocytic population. Caseogranulomas were composed of central pools of densely eosinophilic, amorphous eosinophilic necrotic material that was often admixed with variable amounts of densely basophilic, granular, mineralized debris. Dense bands of epithelioid macrophages, few lymphocytes and plasma cells, and occasional multinucleated giant cells of both the foreign body and Langhan's type surrounded these central pools (Fig. 3). Caseogranulomas were surrounded by variable bands of dense mature fibrosis and granulation tissue. Acid-fast bacterial rods were rarely identified within multinucleated giant cells with Ziehl-Neelsen staining. These histologic findings associated with tuberculosis are similar to the other 100-plus tuberculosis-positive cattle previously examined by the authors, none of which had gross or histologic evidence of BLV.
Immunohistochemical staining was performed on sections of enlarged lymph node that did not contain caseogranulomas typical of tuberculosis. These lymph nodes contained dense sheets of neoplastic cells surrounded by compressed bands of normal tissue containing numerous small lymphocytes. Compressed normal small lymphocytes showed strong cytoplasmic immunoreactivity to CD3, consistent with a T-cell origin. Homogeneous sheets of overtly neoplastic cells were negative for CD3 and CD79a. Ninety-five percent of these neoplastic cells had strong perimembrane to coarsely stippled, intracytoplasmic immunoreactivity for BLA36. Scattered cells representing 10–15% of the neoplastic population had weak perimembrane immunoreactivity for CD20. Given this immunoreactivity profile, the neoplastic cells were most likely B cells in origin, which is consistent with previous reports of B-cell lymphoma associated with BLV infection. While BLA36 is not completely specific for B cells in cattle, the strong immunoreactivity of a high number of neoplastic cells for BLA36 and the scattered immunoreactivity for CD20 are indicative of a B-cell origin. The lack of labeling for CD79a and the weak immunoreactivity for CD20 may be due to loss of expression during neoplastic transformation or may be an artifactual consequence of fixation and processing.
Confirmation of BLV infection was by PCR on shavings of affected formalin-fixed, paraffin-embedded lymph nodes. DNA was extracted from formalin-fixed, paraffin-embedded tissue samples using a commercial kit, a following the manufacturer's protocols. DNA was tested by PCR using a previously published 5 primer set (forward primer 5′-TGGCTATCCTAAGATCTACTG-3′ and reverse primer 5′-AGAGGGAACCCAGTCACTGTT) that detects a 330-base pair env-gp51 region of the BLV viral genome. With this single primer set, the previously published protocol was carried out with a slight modification in reagents, using the Taq PCR master mix a with a final MgCl2 concentration of 4 mM in a 50-μl reaction volume. 8,9 The amplification products were extracted from the gel using a commercial extraction kit a and sequenced for confirmation.
The scarcity of identifiable acid-fast organisms in tissue sections is not unusual, as infections with
Infection with
In contrast to the paucity of reports of concurrent lymphoma and mycobacterial infections reported in veterinary species, there are numerous reports in humans. The vast majority of these involve nontuberculous mycobacteria, but occasional infections with
Whether such immunosuppression was involved in the coinfection of
In the case described in the current report, it is tempting to speculate that an underlying infection with BLV predisposed this animal to infection with
Footnotes
a.
DNeasy® Tissue Kit,
