Abstract
Patients immigrated or born to immigrated parents constitute an increasing proportion of TB in Germany. This study aimed to assess the clinical presentation and outcome of immigrated TB-monoinfected (TB/HIV–) patients versus TB patients with an HIV coinfection (TB/HIV+). Ninety-three patients (36 boys/men, 57 girls/women; age range, 2–59 years) were investigated of whom 47 were TB/HIV+ and 46 TB/HIV–. TB/HIV+ patients more frequently had an African background (76.6% [36/47], P <0.001), TB/HIV– patients (63.0% [29/46]) more often had an East-European or Asian background (P <0.001). Most common symptoms included lymphadenopathy (37.6% [35/93]) and weight loss (35.5% [33/93]). In TB/HIV+ patients acute presentation was more frequent including fever (57.4% [27/47]), gastrointestinal (44.7% [21/47]), and respiratory symptoms (36.2% [17/47]). TB skin test was highly positive in all performed cases (20/20), IGRA was positive in 98.2% (55/56) cases applied. Pulmonary involvement was relatively rare (51/93 [54.8%]), especially in TB/HIV– patients (36.10% [17/46]) vs. TB/HIV+ (70.23% [36/47]; P <0.001). An infectious etiology was suspected in only 43.5% (20/46) TB/HIV– vs. 72.3% (34/47) TB/HIV+ patients (P ⩽0.005); a malignancy was suspected in (21/46 [45.7%] TB/HIV– patients vs. 12/47 [25.5%] TB/HIV+; P ⩽0.043). The diagnostic delay between first presentation to a doctor and TB diagnosis was long in all cases, especially in TB/HIV– patients (range, 0–336 weeks; median, 8 weeks vs. TB/HIV+ patients; range, 0–288 weeks; median, 0 weeks; P <0.05). TB, especially in young immigrated HIV– patients frequently presents as an atypic extrapulmonary disease, which may end up in a delayed diagnosis of up to several years.
Keywords
Introduction
Germany is a low-incidence country for tuberculosis (TB). The number of cases and incidence rates decreased continuously until 2008. Since that time the count stagnates or rises slightly. 1 From 2009 the percentage of cases in children with a migration background raised on a low level until 2013 and is now stagnant. 1 As HIV infection spreads, the number of HIV-TB co-infection is increasing worldwide.2,3 Due to this fact, we carefully anticipate that the number of patients in Germany with an atypical TB manifestation will increase. 4 Therefore the aim of this comparative study was to determine how TB epidemiologically and clinically presents in this patient population and to find out how TB mono-infections differ from TB-HIV co-infections.
Subjects, materials, and methods
A total of 93 patients (36 boys/men, 57 girls/women; age range, 2–59 years; median age, 33 years) were included in a retrospective study for the time period 1994–2013. Forty-six of the 93 patients had TB/HIV–, 47/93 had TB/HIV+.
Patients were included in the study after informed and signed written consent. Patients were assured that they could always stop participating in the study without any disadvantage. The data were anonymized for analysis.
The study was approved by the Ethical Committee of the University of Düsseldorf.
For diagnostic purposes all facilities available in a University Hospital were taken advantage of, including the expertise of experienced infectious disease specialists, multi-disciplinary cooperation, and a wide range of current diagnostic methods. Therapy followed the guidelines of the German Central Committee against Tuberculosis 5 and the guidelines of the German-Austrian AIDS Society.5–7
Results
Patients
The patient groups were comparable for age and sex, apart from the fact that no children or adolescents were included in the TB/HIV+ patient group and that, among adults, the proportion of women in the TB/HIV– patient cohort was significantly higher compared to the TB/HIV+ patient cohort (Table 1).
Age and gender of TB-mono-(TB/HIV–) vs. TB-HIV-co-infected (TB/HIV+).

F, female; M, male; n.d., not done (number of cases was too small for statistical analysis); n.s., not significant.
The migration background of 53/93 patients was Africa, of 34/93 Asia, and of 6/93 patients Eastern Europe. According to the epidemiology of HIV infection, the proportion of patients originating from Africa (36/47 [76.6%]; 30/36 patients coming from West or East Africa) was higher in the TB-HIV co-infected patient cohort P <0.001, while the proportion of TB/HIV–patients was higher in the Asian (29/46 [63.0%]; P <0.001) and Eastern European cohort (4/6 [66%]).
Symptoms
Most common symptoms in both groups were lymphadenopathy (35/93 [37.6%]) and weight loss (33/93 [35.5%]) (Table 2). TB/HIV+ usually presented with a more acute clinical febrile course (P <0.001), gastrointestinal symptoms (P <0.001), and with respiratory symptoms (P <0.004) compared to TB/HIV– patients. Of the TV/HIV+ patients, 39/47 (80.9%) patients had an advanced HIV infection, CDC stage C3, further 8/47 (17.0%) on a CDC stage C2.
Main symptoms of TB/HIV– vs. TB/HIV+ patients.

All cases, TB/HIV–, TB/HIV+: abdominal pain: 11, 1, 10; diarrhea: 5, 0, 5; dysphagia: 2, 1, 1; nausea: 4, 0, 4; hemorrhoids 1, 0, 1.
All cases, TB/HIV–, TB/HIV+: paralysis: 6, 4, 2; epilepsy: 3, 2, 1; headache: 3, 1, 2; vision disturbances: 2, 1, 1; deafness due to tuberculous otitis 1, 1, 0.
All cases, TB/HIV–, TB/HIV+: joint pain: 4, 0, 4; thoracic pain: 4, 1, 3; back pain: 3, 3, 0; pelvic pain: 1, 1, 0, pathological fracture: 1, 1, 0.
All cases, TB/HIV–, TB/HIV+: scrotal pain: 2, 2, 0; hematuria: 1, 1, 0; dysuria 1, 0, 1; primary infertility due to tuberculous endometritis: 1, 1, 0; hypermenorrhea: 1, 0, 1.
All cases, TB/HIV–, TB/HIV+: gingivitis: 2, 0, 2; parotitis: 1, 0, 1.
n.d., not done (number of cases was too small for statistical analysis); n.s., not significant.
Performance of diagnostic tests
TB/HIV– patients had less frequently abnormal inflammatory parameters such as:
C-reactive protein (CRP): TB/HIV– (26/46 [56.5%]) vs. TB/HIV+ (37/46 [80.4%]), P ⩽0.014, CRP turned out to be less sensitive inflammatory parameter for this chronic infection.
Accelerated erythrocyte sedimentation rate (ESR): TB/HIV– (29/37 [78.4%]) vs. TB/HIV+ (9/9 [100.0%]).
Abnormal protein electrophoresis TB/HIV– (25/31 [80.6%]) vs. TB/HIV+ (31/31 [100.0%]), P ⩽0.01.
TB screening tests were used particularly in the TB/HIV– patients but seemed to be helpful in both patient groups, especially in children and adolescents. Intradermal tests (TB skin test [TST]) were highly positive (dermal infiltration >15 mm) in all 20 cases applied (TB/HIV– 14/14 [100.0%]; TB/HIV+ 6/6 [100.0%]). Interferon-gamma release assays (IGRAs) were positive in all performed cases (55/56 [98.2%]) with no differentiation between TB/HIV+ or TB/HIV– patients.
TB/HIV+ patients had more often passed a hepatitis B virus infection: anti-HBc was positive in 18/47 (38.3%) TB/HIV+ vs. 1/46 (17.2%) TB/HIV– patients (P <0.001). Furthermore three patients (1 TB/HIV–, 2 TB/HIV+) were also HCV co-infected as confirmed by PCR for HCV-RNA.
Microscopies of cultured specimens were positive in 87/93 (93.54%) cases: 84 for Mycobacterium (M.) tuberculosis and three for M. bovis.
PCR was applied in 70 cases. The result was negative in 28 cases and positive in 42 cases. Positive results were confirmed by culture in all cases due to the fact that PCR was used in several specimens where PCR is not licensed (e.g. endometrial material). Even when analyzing specimens for whom PCR is licensed, sensitivity was not higher (26/42 [61.90%]; TB/HIV+ 22/29 [75.87] vs. TB/HIV–: 4/13 (30.77%]). However, specificity of PCR was 100%. Sensitivity of PCR was relatively low compared to culture (42/68 [61.77%]; TB/HIV+ 30/38 [78.95%] vs. TB/HIV– 12/30 [40.00%]; P <0.001). When 42 respiratory specimens including gastric aspirates were analyzed for which PCR is licensed
Imaging and histopathology findings
Among all patients, lung was comparatively rarely involved 51/93 (54.8%), applied particular to the TB/HIV– patients. The majority of all patients were radiologically, sputum-microscopy and sputum-cultural negative (TB/HIV– 29/46 [63.0%] vs. TB/HIV+ 13/47 [27.7%], P <0.001 [Table 3]). The histopathological picture showed in all of the 72 performed cases typical granulomas with necrosis, Langerhans giant, and epithelioid cells.
Organ involvement in TB/HIV– vs. TB/HIV+ patients.

Total: microscopy-positive: 20/51 (39.22%); microscopy-negative-culture-positive 22/51 (43.14%); all sputum tests negative: 9/51(17.65%). TB/HIV–: microscopy-positive: 6/17 (35.29%); microscopy-negative-culture-positive 9/17 (52.94%); all sputum tests negative: 2/17(11.76%). TB/HIV+: microscopy-positive: 14/34 (41.17%); microscopy-negative-culture positive 13/34 (38.23%); all sputum tests negative: 7/34(20.59%).
n.d., not done (number of cases too small for statistical analysis); n.s., not significant.
Delay of diagnosis
Symptomatic patients had their first doctoral visit after the beginning of symptoms after very variable time intervals in the range of 0–240 weeks (median, 4 weeks) with no differentiation between TB/HIV+ and TB/HIV–.
Surprisingly, TB was diagnosed in TB/HIV+ patients earlier (range, 0–288 weeks; median, 0 weeks) than in TB/HIV– patients (range, 0–336 weeks; median, 8 weeks; P <0.05).
Order of diagnosis of TB and HIV infection
While the diagnosis of TB rarely led to the diagnosis of an HIV co-infection (4/47 [8.5%]), a known HIV diagnosis in most cases led to the diagnosis of TB co-infection (32/47 cases [70.2%]; P <0.001). In addition, 11 of 47 cases (23.4%) with TB/HIV co-infection were detected at the same time.
Suspected diagnoses at presentation
An infectious etiology of patients’ symptoms was initially suspected in only 54/93 (58.6%) of all included patients, certainly more often in TB/HIV+ patients (34/47 [72.3% ] vs. 20/46 [43.48%] TB/HIV-; P ⩽0.005 [Table 4]) On the contrary, a malignancy was suspected more frequently in TB/HIV– patients (21/46 [45.7%] vs. TB/HIV+ 12/47 [25.5%]; P ⩽0.043; all TB patients: 33/93 [35.5%]).
First clinical suspicion in Tbc/HIV– vs. TB/HIV+ patients.
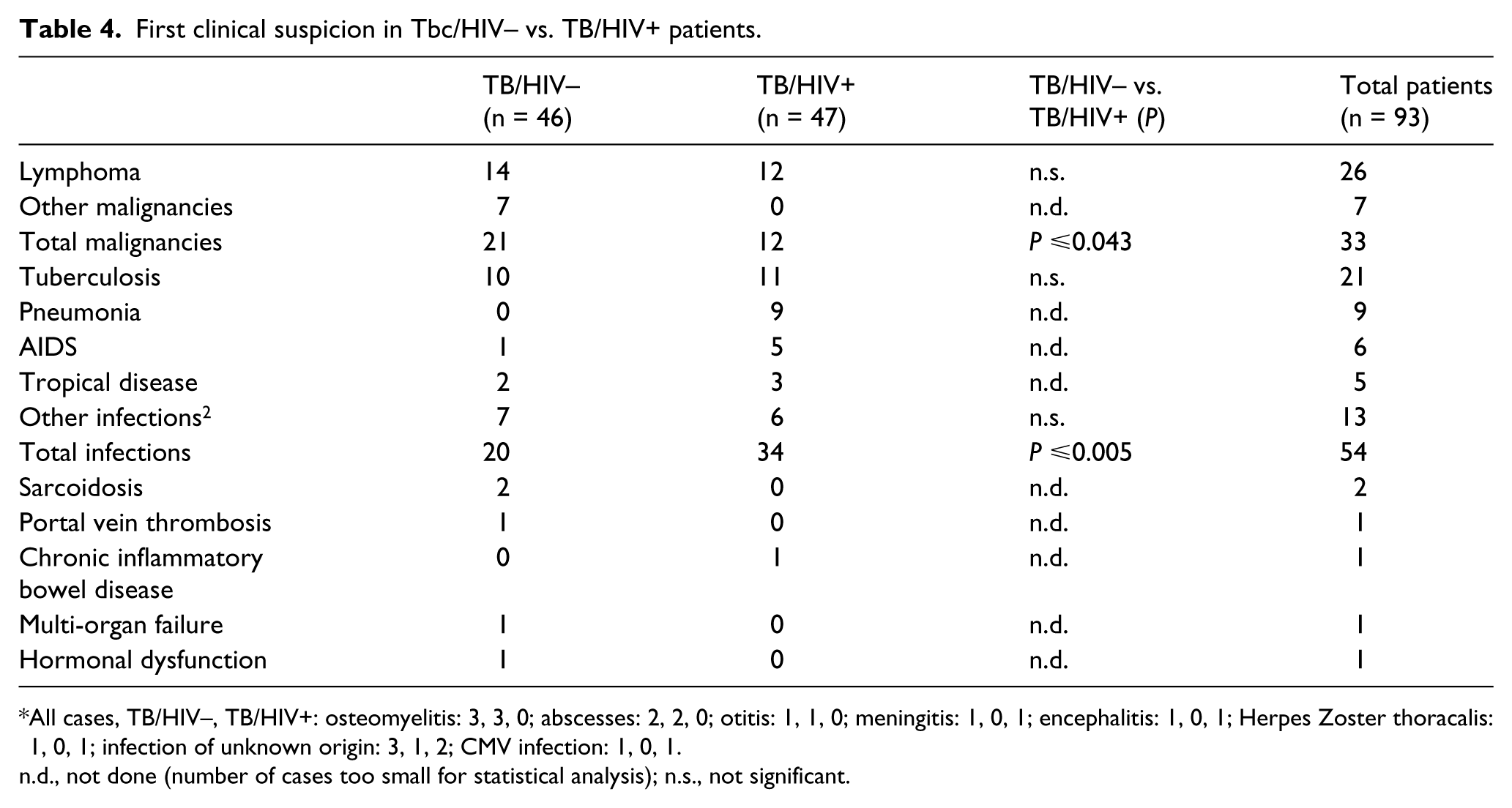
All cases, TB/HIV–, TB/HIV+: osteomyelitis: 3, 3, 0; abscesses: 2, 2, 0; otitis: 1, 1, 0; meningitis: 1, 0, 1; encephalitis: 1, 0, 1; Herpes Zoster thoracalis: 1, 0, 1; infection of unknown origin: 3, 1, 2; CMV infection: 1, 0, 1.
n.d., not done (number of cases too small for statistical analysis); n.s., not significant.
Therapy outcome
The tuberculostatic therapy of all TB patients was usually started immediately after sufficient material for culture was collected. The TB treatment was modified according to the drug-sensitivity profile of the causative Mycobacterium. Highly active antiretroviral therapy was deferred in newly detected HIV-co-infections according to the current guidelines in order to minimize the risk of negative drug interactions and severe immune reconstitution syndrome (IRIS).5–8
Of the TB/HIV+ patients 46 were infected with M. tuberculosis and one by M. bovis. For avoiding interactions between anti-TB and HAART tuberculostatic therapy was modified: in 33/47 cases (70.2%) RMP was replaced by rifabutin to avoid interactions with protease inhibitors. In 26/47 M. tuberculosis/HIV+ infections, sequential standard therapy had to be prolonged: in 14/26 cases each quadruple or double therapy courses required prolongation. In another 21/47 (44.7%) TB/HIV+ cases a modification of TB medication was required, including the use of SM, ciprofloxacin, capreomycin, PTH, clarithromycin, and moxifloxacin. In addition, the duration of the therapy had to be extended. In 4/21 TB/HIV+ patients multi-resistance was associated with a severe clinical course: in a Rwandan woman resistance to INH and SM required therapy including EMB, PZA, rifabutin, moxifloxacin, PTH, terizidone, and linezolid. Cerebral tuberculosis in one TB/HIV+ occurred simultaneously with an intracerebral B-cell lymphoma, requiring an extension of tuberculostatic therapy to over 12 months. A Laotian man had an INH and RMP-resistant TB requiring ciprofloxacin, EMB, PZA, PTH, rifabutin, and linezolid over a total period of 9.5 months. A South African woman had a XDR-TB with resistance to INH, PZA, RMP, and rifabutin. The poor response to therapy required repeated adaptions of tuberculostatics with addition of moxifloxacin, linezolid, capreomycin, terizidone, and prolonged treatment duration of more than 27 months.
Adverse events of TB therapy were observed more frequently in TB/HIV+ patients (17/47 [36.2%] vs. 4/46 [8.7%] TB/HIV–; P ⩽0.002) including liver toxicity, visual impairment, thrombocytopenia, pancytopenia, heart rhythm disorders, gastrointestinal, and central nervous system adverse events. A particularly serious course of adverse events to additive corticosteroid therapy occurred in a female HIV– fugitive from Sri Lanka with a cerebral TB. She suffered an aseptic necrosis of femoral and humeral heads, which were irreversible despite prostaglandin therapy and required repeated surgery. Later, the patient developed osseous cold abscesses with cutaneous fistulae, but was eventually cured.
The prognosis quoad vitam was worse (mortality of TB/HIV+ 4/47 [0.9%] vs. TB/HIV– 0/46 [0.0%]) due primarily to advanced HIV infection (3 patients) and toxic liver failure (1 patient).
Discussion
The incidence of TB in Europe is decreasing but there are important disparities between countries and settings. 3 We assume that immigration of people and their families from TB-endemic regions to Germany will increase in future years. This anticipates an increase of both TB and HIV patients in immigrants to Germany.1,3,9–13 Furthermore, extrapulmonary TB, which appears to affect more often in female patients, might also increase. 14 Both infections, TB and HIV, need to be recognized early, to avoid further spread to the population and mismanagement of the individual patient. Our investigations show that frequently too long a time lapses between first symptoms, first visit of a doctor, and establishment of diagnosis. The suspicion and diagnosis of TB was particularly delayed in atypical extrapulmonary TB presentations of women and children compared to HIV+ associated TB cases. Extrapulmonary TB cases take up 21.6% of all cases observed in Germany. 1 It would be worthwhile to analyze in detail all notified cases to see which patient population was particularly affected and if diagnosis was particularly delayed. The results of our investigation suggest that this percentage may be higher especially in young female immigrants or children of immigrants although we used an especially restrictive definition of extrapulmonary TB. 14
Several medical and practical reasons may be responsible for the mentioned diagnostic delay: TB is a multifaceted infection, which is not simply detected by a single test. TST and IGRA only reveal if a patient has ever had a TB contact. This result can be unsatisfactory considering that one-quarter of the world’s population contains TB within themselves due to contact (and even higher population parts in endemic regions). Therefore TST and IGRA should not be seen as competing but as complementary diagnostic tests:4,14 IGRA offers the advantage of being more specific, especially excluding false positive reactions to BCG-vaccinations whereas TST offers the advantage of semi-quantitation and low cost.15–20 In particular, TST is useful applied to adult immigrants for highly endemic countries whereas IGRA is useful in children and adolescents born to immigrants from highly endemic countries that grew up in Germany or other low-risk countries. However, it has to be taken in account that children aged younger than 5 years IGRAs may yield false negative results.16–20 PCR is valuable because the result is achieved promptly contrary to culture. However, it must be considered that a negative result does not rule out TB. The reason for numerous false negative results of PCR also depend on how the specimen was obtained. 21 In our cohort, false negative results occurred particularly in TB/HIV– patients. The diagnosis of TB is often not straightforward and is therefore achieved in most cases by the combination of exposure, history, symptoms, organ manifestations, TST, IGRA, PCR, culture, and histopathology. Sometimes calculated therapy yields confirmation by clinical improvement within 2–3 weeks whereas confirmation by culture may take 6–8 weeks. To every TB patient HIV diagnostics should be offered since HIV co-infection may present particular problems.2,10,16 To our initial surprise TB was recognized earlier in HIV+ patients analogous to an observation confirmed by two recent studies from the UK.22,23 Explanations may be that in HIV+ patients infectious diseases are considered earlier, that the treating doctor is experienced with infectious diseases, that paucibacillary cases are less frequent, that consequently PCR less frequently yield false negative results, that the clinical course is more acute, and that pulmonary manifestations are more common.
Several practical logistic issues further hamper early diagnosis in Germany: TST need to be imported from abroad and TST and IGRA are not adequately refunded to the practitioner by most health insurance. 24 This means that general practitioners have to refer the suspected patients to the Public Health Office. Besides the logistic complications, many patients, especially illegal patients, might be reluctant to present at the Public Health Office. This latter assumption is corroborated by a recent Swedish study where legally immigrated students answered that they would defer HIV testing because of their fear of being expelled from the country. 25 Many of our patients are already afraid in the waiting room to be indicated as HIV+ or TB+ by some other attendant of our service. Patients with other cultural backgrounds frequently interpret the signature to be given for informed consent for HIV testing as the decline of responsibility by the doctor rather than as expressing a basic right of the patient. They doubt the maintenance of the professional secret of doctors and the German laws. These are also not always evident to the patients since TB legislation focuses on the protection of the public including the possibility of hospital conviction whereas HIV legislation focuses mainly on the rights of the individual patient. Another issue is that a general practitioner might fear a negative impact on his patient attendance when a patient with TB suspicion is sitting in the waiting room wearing a surgical mask. Other difficulties include the fact that, contrary to most European and Extra-European countries, in Germany a medical specialty for infectious diseases did not exist for decades and that especially young German doctors do not have sufficient opportunities to gain experience with the diagnosis of TB. 4
Conclusion
Considering the probably increasing immigration to Europe patients from TB-endemic countries in the future, doctors will increasingly face TB patients. Barriers for a prompt TB and HIV diagnosis have to be overcome to accelerate the diagnosis of TB and HIV and thus to decrease the frequency of misdiagnosis and spread of both infections to the population.
Footnotes
Acknowledgements
This work is a part of the medical thesis by Kerstin Ennemoser. We are indebted to Dr. Klaus Göbels, MPH, Head of the Public Health Advisory, Düsseldorf and Dr. Regina Mertens, DTM&H, Physician and General Practitioner, Bochum, for their valuable comments.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
