Abstract
The genus Pestivirus is composed of 4 important pathogens of livestock: Bovine viral diarrhea virus 1 and 2 (BVDV-1 and BVDV-2), Classical swine fever virus (CSFV), and Border disease virus of sheep (BDV). BVDV are major pathogens of cattle, and infection results in significant economic loss worldwide. A new putative pestivirus species, tentatively called “HoBi-like,” “BVDV-3,” or “atypical pestiviruses,” was first identified in Europe in fetal bovine serum (FBS) imported from Brazil. HoBi-like viruses are related to BVDV at the genetic and antigenic levels. Further, the disease caused by these new viruses resembles clinical presentations historically associated with BVDV infection, including growth retardation, reduced milk production, respiratory disease, reduced reproductive performance, and increased mortality among young stock. Current BVDV diagnostic tests may fail to detect HoBi-like viruses or to differentiate between BVDV and HoBi-like viruses. Further, commercial tests for BVDV exposure, based on serological response, do not reliably detect HoBi-like virus exposure, and cross protection against HoBi-like viruses conferred by current BVDV vaccines is likely limited. As many HoBi-like viruses, characterized to date, were isolated from FBS originating from Brazil, it is assumed that the agent is probably widespread in Brazilian herds. Nevertheless, reports of natural infection in Southeast Asia and Europe demonstrate that these viruses are not restricted to South America. Increased demand for FBS has led to widespread distribution of FBS originating in HoBi-like virus endemic regions. The contamination of such FBS with HoBi-like viruses may lead to spread of this virus to other regions.
Keywords
Overview of bovine pestiviruses
Pestivirus is a genus within the family Flaviviridae, composed of 4 recognized species, Bovine viral diarrhea virus 1 and 2 (BVDV-1 and BVDV-2), Classical swine fever virus (CSFV), and Border disease virus (BDV). 32 Pestivirus infections in cattle, noticeably BVDV-1 and BVDV-2, represent major concerns worldwide, leading to significant economic losses. 15 The disease is associated with a wide range of clinical manifestations. Calves infected with typically virulent BVDV generally display mild upper respiratory signs, a transient decrease in circulating white blood cells, and a low-grade, short-term fever. However, while a minority in nature, BVDV strains with enhanced virulence do exist. Infection with an enhanced virulence BVDV strain may result in severe respiratory disease, gastroenteric disorders, hemorrhagic syndrome, and pneumonia.1,4-6 Regardless, the most relevant economic losses associated with BVDV infection seem to be due to reproductive disorders. 16 The infection of pregnant cows during the first trimester of gestation with noncytopathic (ncp) BVDV strains, belonging to either BVDV-1 or BVDV-2 species, may lead to failure of fertilization, return to estrus, abortion, congenital malformations, stillbirths, or the birth of persistently infected (PI) animals. While some PI calves may have congenital malformations, others appear clinically normal. Persistently infected animals are negative for BVDV antibodies, prior to ingestion of colostrum, but are positive for virus antigen in nearly all tissues. Such PI animals shed the virus to the environment continuously over the course of their lifetimes.1,4 Thus, the identification and elimination of PI animals and the adoption of biosecurity measures that prevent the introduction of PI animals into herds are necessary actions in controlling and eradicating BVDV from cattle herds. Inactivated and modified live vaccines are used worldwide to prevent acute disease and fetal infections.16,21 Vaccination reduces the production of PI animals at the herd level but cannot be relied upon to prevent all fetal infections. Thus, vaccination alone cannot eliminate BVDV and must be combined with identification and elimination of PI animals and the adoption of biosecurity in control and eradication strategies.
The pestivirus genome is composed of a positive single-stranded RNA molecule approximately 12.3 kb in length, containing a single open reading frame (ORF) flanked by 2 untranslated regions (5′- and 3′-UTR). The long ORF encodes a single polyprotein that is co- and posttranslationally processed into 12 viral mature polypeptides in the following order: Npro-C-Erns-E1-E2-p7-NS2/NS3-NS4A-NS4B-NS5A-NS5B. 32 The 5′-UTR region is highly conserved among pestiviruses, and comparison of 5′-UTR sequences is widely accepted for phylogenetic analysis as are comparison of sequences coding for the viral proteins Npro and E2.14,24,26,28 Due to the nature of the single-stranded genome, these viruses display high mutation rates, which in some cases may lead to the emergence of “new” virus lineages. Whereas the correlation of high mutation rates with emergence of new lineages has not been proven, the rise of new pestiviruses species has been described.18,28,35
In addition to the recognized species, 4 additional Pestivirus species have been proposed but remain officially unrecognized (Fig. 1). These putative species include Giraffe virus, associated with an outbreak of mucosal-like disease in giraffes in the Nanyuki District of Kenya 32 ; Pronghorn virus, isolated from a blind pronghorn antelope in the United States 35 ; Bungowannah virus, detected in pigs following an outbreak of stillbirths and neonatal death in Australia 18 ; and a group of viruses variously referred to as HoBi-like, BVDV-3, or atypical pestiviruses,20,24,28 which are the subject of the current review.
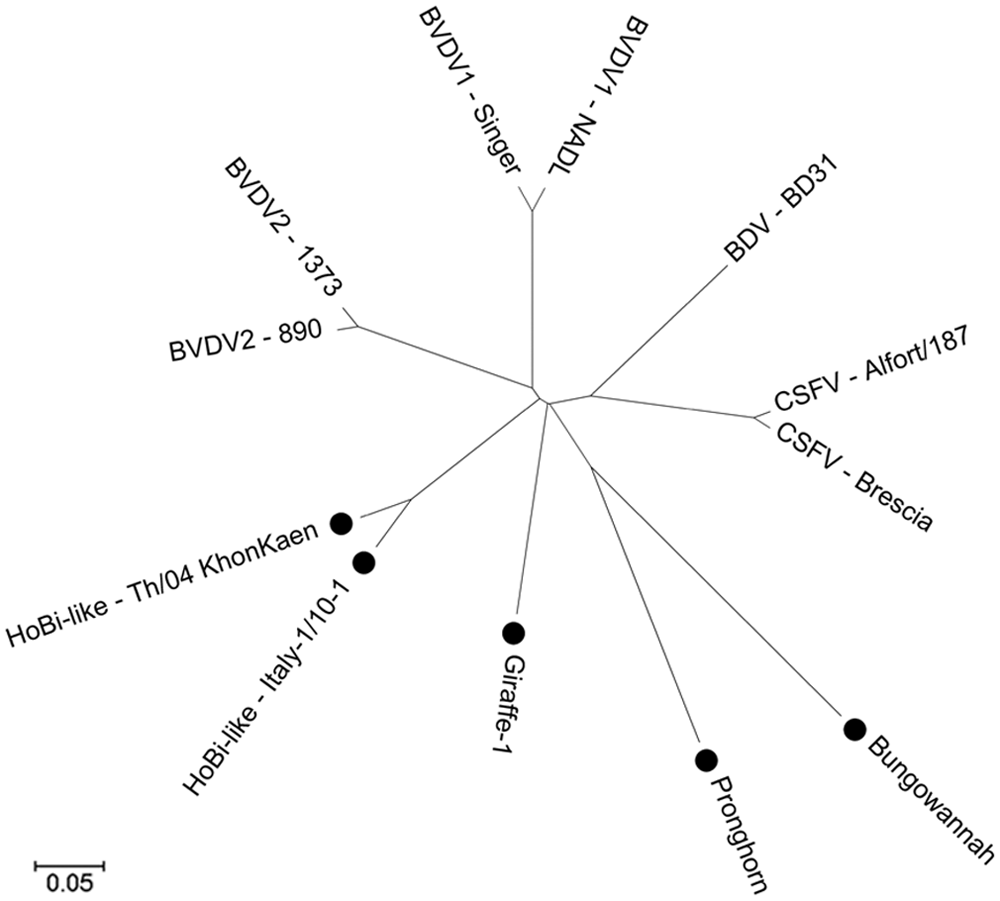
Phylogenetic tree of Pestivirus genus. Official and putative species represented. The isolates in the 4 putative species are market with “•”. Analyses were conducted in MEGA5, 31 using the neighbor-joining method; distances were computed using the Poisson correction method. Region analyzed consisted of the amino acids from the polypeptides Npro; C; Erns; E1 and E2.
The putative species, referred to hereafter as HoBi-like viruses, which was first described by researchers in Europe in 2004, was isolated from a batch of fetal bovine serum (FBS) imported from Brazil. 30 Since that first description, several reports of genetically similar agents contaminating FBS and cell lines have been published.25,28,30,36,37 Unpublished data indicates that the more than 30% of FBS batches originated from South America that are tested in Europe are contaminated with HoBi-like viruses (Martin Beer, personal communication, 2012), leading to the hypothesis that these viruses are probably widespread in South American cattle, especially in Brazilian herds. However, HoBi-like viruses have also been detected contaminating FBS that is processed and/or packaged in Europe, originating from Australia, Canada, Mexico, and United States. 37 Detection of HoBi-like viruses was also reported in FBS originating, processed, and packaged in Australia, 36 although this finding remains controversial considering the lack of reports indicating the circulation of these viruses in that country. Natural infection in cattle with HoBi-like viruses has being reported in Southeast Asia, 29 Italy,8,9,11 and Brazil3,7,30 (Fig. 2).
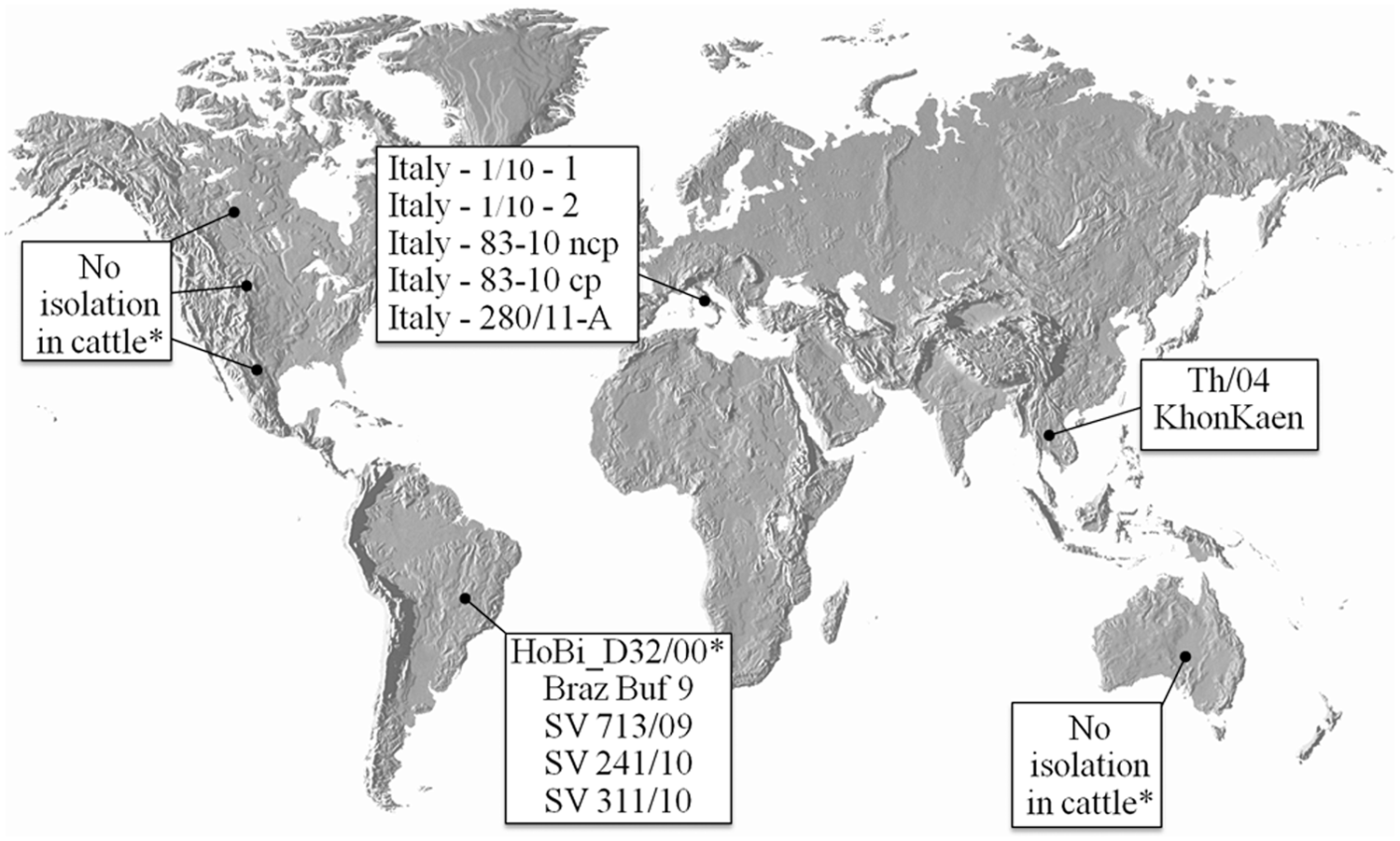
Distribution of HoBi-like viruses isolated to date. *Identification of HoBi-like viruses in fetal bovine serum batches claimed to be originated in these countries. cp = cytopathic; ncp = noncytopathic.
In contrast with the apparent widespread distribution of HoBi-like viruses, the other putative pestivirus species mentioned above were isolated from a limited number of animals residing in single geographic regions (Table 1). Therefore, HoBi-like viruses seem to pose the most threat for domestic livestock production of any of the “new” pestiviruses that have emerged recently (between years 2000–2012). The name “HoBi-like viruses” is derived from the first isolate called HoBi_D32/00. 28 Some authors also refer to these viruses as BVDV-3 24 due to the similarity of clinical presentation in cattle following infection with BVDV and HoBi-like viruses. There is resistance to declaring HoBi-like viruses a third species of BVDV because these viruses are genetically distant from BVDV-1 and BVDV-2, and diagnostics and vaccines that work well to detect and control the 2 species of BVDV are less efficacious for HoBi-like viruses.2,28 The term “atypical pestiviruses” has also been used8,23; however, this term could also be applied to any of the 3 other putative pestivirus species. To date, the International Committee on Viral Taxonomy has not arrived at a decision to recognize HoBi-like viruses as an official species within the pestivirus genus or declared an official name.
Worldwide distribution and control of pestiviruses.
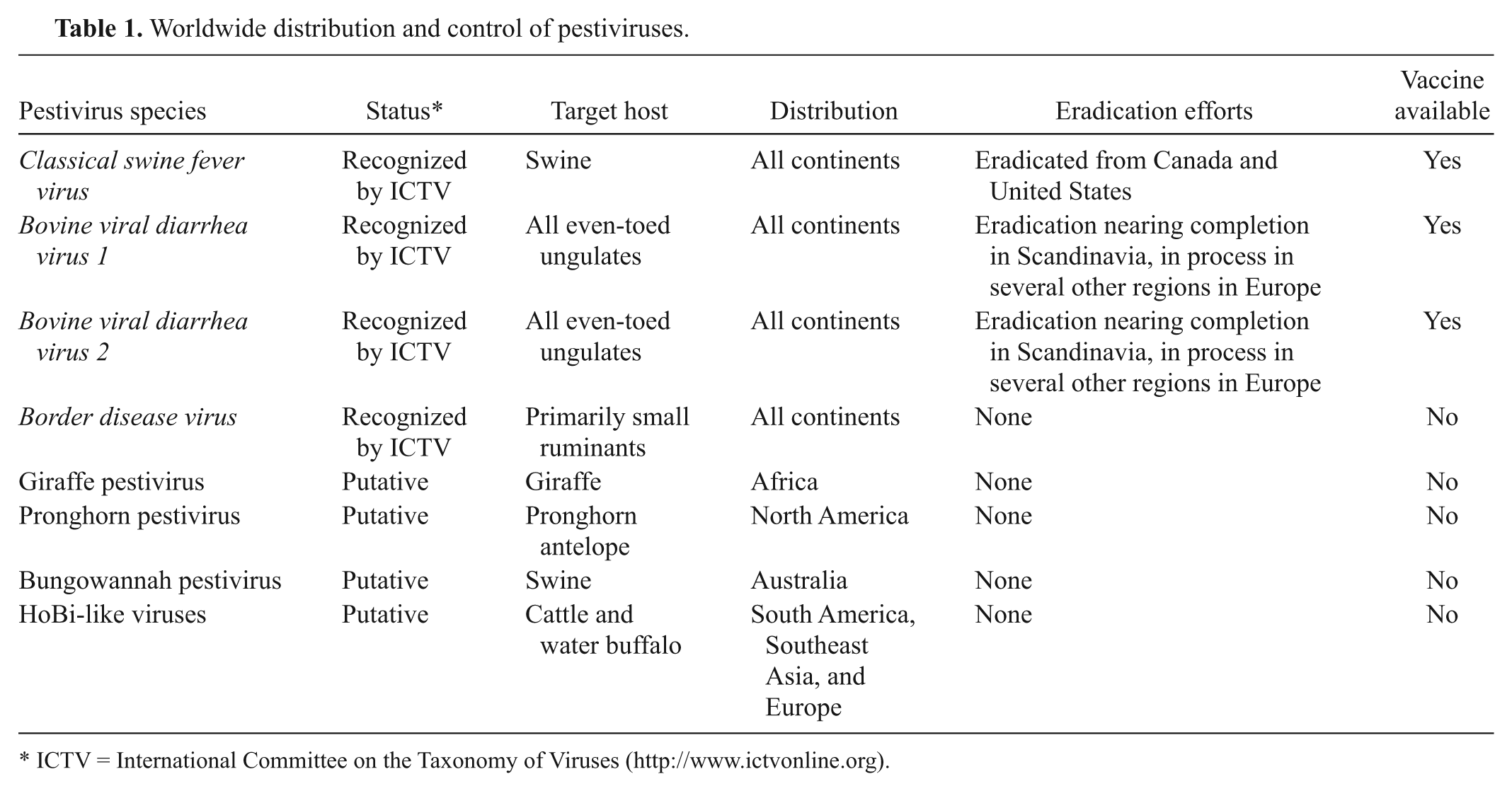
ICTV = International Committee on the Taxonomy of Viruses (http://www.ictvonline.org).
The origin/emergence of HoBi-like viruses is unknown. One hypothesis is that the viruses originated in South America and were introduced to other countries and continents through contaminated biological products, such as FBS and vaccines. 29 Another explanation is that the emergence of HoBi-like viruses in cattle is the result of a host species jump in which these viruses crossed from water buffalo to cattle. This hypothesis would explain the presence of the viruses in regions raising significant water buffalo populations, such as Brazil and Thailand. The HoBi-like isolate BrazBuf9 30 was identified in the late 1990s in an outbreak with death of water buffalos in Brazil, yet the outbreak was not officially reported (Claudio Wageck Canal, personal communication, 2011).
Based on phylogenetic analysis, all isolates of HoBi-like viruses identified to date are highly similar and cluster together. Keeping in mind the highly genetic variability of pestiviruses, it could be hypothesized that HoBi-like viruses were originated in South America and the subsequent spread to other regions has been a fairly recent event, in evolutionary terms. On the other hand, the divergence of the isolate from Southeast Asia (Th/04_KhonKaen) from other HoBi-like viruses might also indicate an independent evolution of at least 2 groups of HoBi-like viruses (Fig. 3).

Representation of several HoBi-like pestiviruses based on the 5′–untranslated region (5′-UTR). Sequences are identified by GenBank accession number (available at http://www.ncbi.nlm.nih.gov/pubmed/). “▲” was used to mark the prototypes of 2 possible HoBi-like virus lineages (HoBi_D32/00 and Th/04_Khonkaen). Evolutionary analyses were conducted in MEGA5, 31 using the neighbor-joining method. Distances were computed using the Kimura 2-parameter method.
As stated previously, natural HoBi-like virus infections have been reported in South America, Southeast Asia, and Italy. No isolations of these viruses have been reported from cattle residing in the rest of Europe, North America, Africa, India, and Australia, indicating that herds in these regions remain serologically naïve and vulnerable to infection. Spread of HoBi-like viruses into these regions could have profound effects on cattle production worldwide. The recent introduction of the Schmallenberg virus into Europe illustrates how rapidly an introduced pathogen can spread in naïve herds. The veterinary community was not alerted to the initial introduction of Schmallenberg virus because the clinical presentation following acute infection was mild. However, the Schmallenberg virus, like BVDV and HoBi-like viruses, is able to cross the placenta and infect the fetus. The full impact of the Schmallenberg introduction was not realized until congenitally malformed offspring were born following infection during the early stages of pregnancy. The introduction of HoBi-like viruses would have an even greater impact because HoBi-like viruses can establish persistent infections in offspring following in utero infection. Further, BVDV diagnostic tests may fail to detect and/or differentiate these agents. Thus, detection of HoBi-like viruses may be overlooked. Improved surveillance for these viruses requires the attention of veterinary diagnostic laboratories, institutions involved with commerce of biological products, and governmental agencies.
In herds in Brazil, Italy, and Thailand, where these viruses may already be endemic, infection of cattle with HoBi-like viruses may contribute to economic losses associated with clinical disease, reduction in productive parameters, and perhaps immunosuppression. HoBi-virus–positive status for endemic countries may also become an issue for international trade of animals and products with countries that consider themselves free of HoBi-like virus.
Clinical presentation of HoBi-like viruses
Natural cases
The first evidence of natural infection with a HoBi-like virus was found in a water buffalo of Brazilian origin 30 in the late 1990s, yet the outbreak was not officially reported (Claudio Wageck Canal, personal communication, 2011; Table 2). In 2006, HoBi-like virus in 2 aborted fetuses (dated 2002 and 2004) from southeastern Brazil was described. 7 In the same country, in 2011, the isolation and sequencing of the 5′-UTR region of 3 HoBi-like isolates (SV713/09, SV241/10, SV311/10) was reported. 3 The isolate SV713/09 was identified in a commercial semen sample of a bull, following several descriptions of blind newborn calves in herds using semen from this bull. Subsequent diagnostic tests ruling out the presence of other agents evidenced the association of the HoBi-like virus with these events. Isolates SV241/10 and SV311/10 were detected in buffy coats from animals of herds with history of reproductive failure in Rio Grande do Sul State, southern Brazil, although further clinical and epidemiological information of the outbreaks are missing. 3 Also in Brazil, in the midwestern region, a case of acute gastroenteric disease leading to death of a calf, confirmed the presence of a HoBi-like virus in the calf spleen by reverse transcription polymerase chain reaction (RT-PCR) and nucleotide sequencing (Rodrigues WB, Otonel RAA, Fritzen JTT, et al.: 2011, Natural infection of calf with an atypical bovine pestivirus (BVDV-3). In: Proceedings of XXII National Meeting of Virology & VI Mercosur Meeting of Virology. Virus Rev Res 16(Suppl 1):74. Atibaia, Brazil).
Brief description of HoBi-like viruses detected to date (by sampling date).*

NI = not informed.
Claudio Wageck Canal, personal communication, 2012.
Rodrigues WB, Otonel, RAA., Fritzen, JTT, et al.: 2011. Natural infection of calf with an atypical bovine pestivirus (BVDV-3). In: Proceedings of XXII National Meeting of Virology & VI Mercosur Meeting of Virology. Virus Rev Res 16(Suppl 1):74. Atibaia, Brazil.
Cases of HoBi-like viruses infecting cattle in countries other than Brazil were reported for the first time in Thailand. 29 Between 2002 and 2004, an epidemiological study focusing on Bovine herpesvirus 1 and BVDV infections in 186 herds demonstrated that animals in 4 herds seroconverted to HoBi-like viruses, although no clinical signs were observed. Heat-inactivated serum samples were tested using antigen-capture enzyme-linked immunosorbent assay (ACE) for BVDV, and 1 positive calf serum was identified. 17 The virus was segregated to the HoBi-like group by molecular characterization. 29 A subsequent study involving transfection of viral RNA into bovine turbinate cells yielded the isolate called Th/04_Khonkaen. 22
In southern Italy, in 2009–2010, an outbreak of respiratory disease affecting 26 calves aged 6–7 months old was reported. The clinical signs included fever (39.4–40.1°C), cough, seromucoid nasal discharge, leukopenia, and accelerated pulse and breath. Most affected animals received support treatment and recovered within 2 weeks; however, the death of 2 animals was reported. Necropsy findings included severe tracheitis and bronchopneumonia involving the apical lung lobes. HoBi-like virus was detected by quantitative RT-PCR (qRT-PCR) in nasal discharge samples of 6 calves and in the lungs of the 2 dead animals. Virus isolation from lung samples was performed in Madin–Darby bovine kidney (MDBK) cells, and the viruses detected were named Italy-1/10-1 and Italy-1/10-2. 8 In the same herd, in June 2011, the occurrence of reproductive disorders was reported, including abortions in 8 cows between the fourth and sixth months of gestation. HoBi-like viruses were isolated from 2 fetuses. The nucleotide similarity of the E2 glycoprotein and the 5′-UTR region was higher than 99% with the strain Italy-1/10-1 that was isolated in the same herd during a previous outbreak of respiratory disease.8,9 Also in the same herd, the first cytopathic isolate of HoBi-like virus was identified in the lung of a heifer that died with respiratory disease. 11
Experimental infection
Experimental infections with HoBi-like viruses performed to date are summarized in Table 3. The first controlled study of acute infection used the first reported isolate of HoBi-like virus, HoBi_D32/00, which was involved the infection of only 2 calves and 2 pigs. The pigs seroconvert to the virus, although no shedding or clinical signs were observed. In contrast, in calves, the virus was detected in buffy coats at day postinfection (DPI) 5, and virus shedding was observed from DPI 3 to 6 in inoculated calves in addition to seroconversion. Mild increase in body temperature and slight leucopenia was also observed in the calves. 28
Concise description of clinical manifestation following experimental infection of calves with HoBi-like strains.*
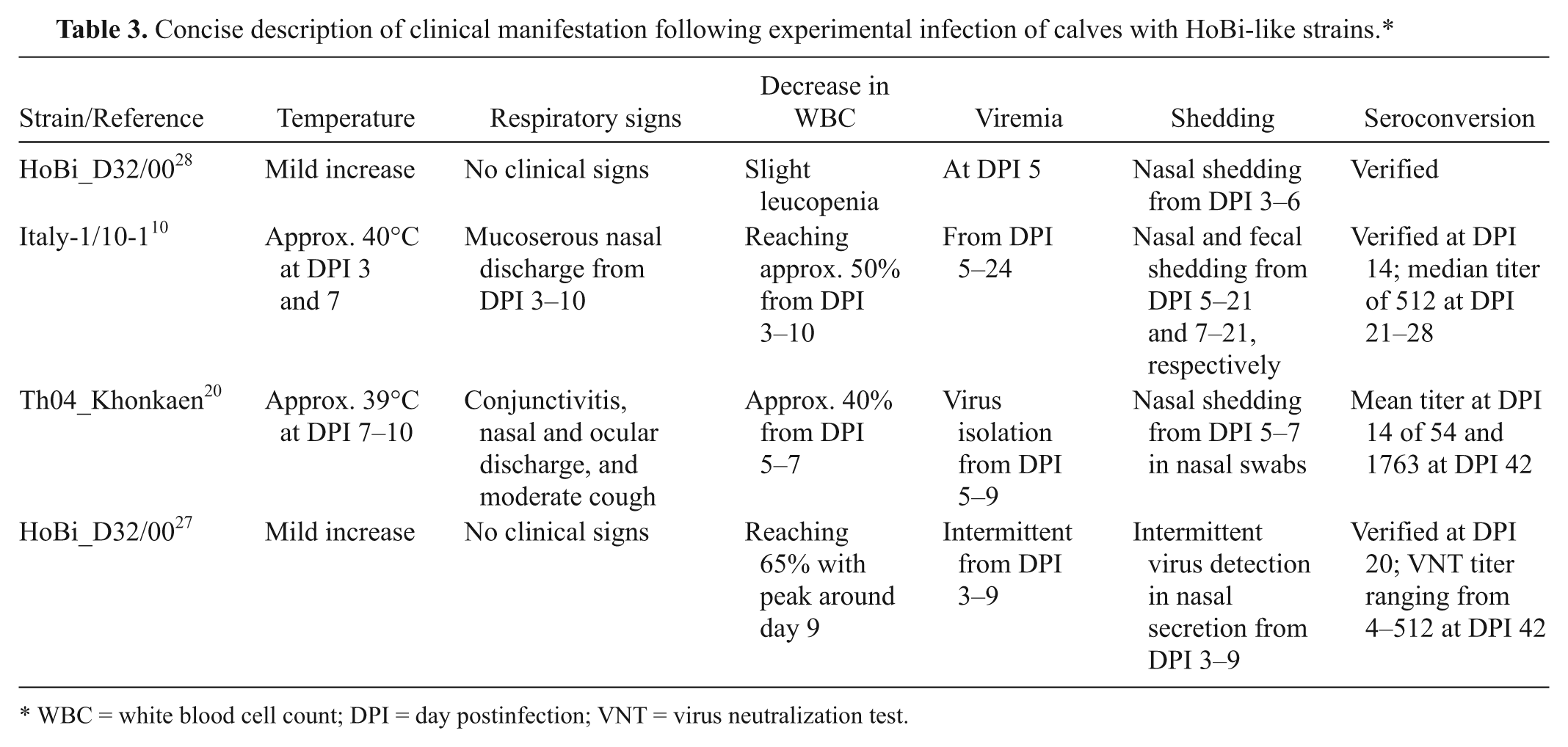
WBC = white blood cell count; DPI = day postinfection; VNT = virus neutralization test.
Using the virus strain (Italy-1/10-1) isolated from the outbreak of respiratory disease, a study comprising the inoculation of 3 groups of 7 animals was performed, as follows: six 1-month-old calves, five 1-month-old lambs, and two 1-month-old piglets. Clinical signs in calves included moderate hyperthermia, mucoserous nasal discharge, and lymphocyte count decreases. No increase in body temperature was observed in lambs, yet moderate to abundant nasal discharge persisted from DPI 4 to 16. Between DPI 5 and 10, occasional depletion in circulating lymphocytes, no higher than 60% compared to baseline values, was observed. Piglets did not display clinical and/or hematological signs, although seroconversion with median antibody titer of 4 was detected. Seroconversion in calves and lambs peaked at DPI 21, with median titer of 512 and 64, respectively. 10
In a study using the Thai HoBi-like strain, 3–5-month-old calves were inoculated with Th04_Khonkaen or with a highly virulent BVDV strain. The disease observed in animals inoculated with the HoBi-like strain was milder than that produced by the highly virulent BVDV strain. Animals presented limited to bilateral moderate conjunctivitis, watery-mucoid nasal and ocular discharge and cough. Depletion in the lymphocyte counts between DPI 2–5, returning to normal level at DPI 14 was also observed. A decrease in platelet counts was detected at DPI 7. The other hematological parameters remained within normal range. 20
The severity of clinical signs during the infection with HoBi-like viruses was compared to clinical signs following infection of cattle with strains of BVDV with typical and enhanced virulence. This study demonstrated that HoBi-like strains are transmissible between infected and sentinel calves, resulting in mild respiratory disease, transient pyrexia, and significant white blood cells depletion, which was similar to infection with a typically virulent BVDV strain. In the group of 6 calves infected with HoBi_D32/00 and 6 sentinel animals, 3 infected calves developed pyrexia. Viral RNA was detected in buffy coat and nasal swabs of infected and sentinel calves, and also, seroconversion was detected in all animals. During the experiment, at DPI 5, 1 animal developed a temperature of 39.4°C and was found dead the next day. At necropsy, no major lesions in the respiratory or gastroenteric tracts were observed. The virus was isolated from the buffy coat obtained from heart blood and RT-PCR. Sequencing confirmed HoBi_D32/00. 27
In summary, there is no experimental data indicating an adaptation of HoBi-like viruses in hosts other than ruminants, and all field isolations were from cattle or water buffalo. Pathogenesis studies of HoBi-like viruses in water buffalo may address the issue of HoBi-like virus origin. During natural and experimental infections, HoBi-like viruses seem to cause mild disease in cattle. While most descriptions of HoBi-like viruses in natural infections are related to reproductive disorders, most experimental studies center on reproducing acute disease in calves. Both field and experimental exposures resulted in clinical presentations similar to those observed following infection of animals with typical field strains of BVDV. The higher number of HoBi-like isolates detected in reproductive disorders compared to respiratory disease may reflect a greater impact as a reproductive pathogen or may merely reflect a high number of submissions from cow–calf operations. The increased concern and economic losses during outbreaks of abortions rather than in mild respiratory disease may play an important role and bias the detection of HoBi-like viruses under field conditions.
Diagnostic approaches to detecting HoBi-like viruses
Fetal bovine serum testing
Fetal bovine serum is widely used as a supplement in cell culture media and, inadvertently, many batches of this supplement are contaminated with pestiviruses. The production and packaging of FBS usually employs large pools of fetuses. Increasing the number of fetuses contributing to a commercial batch of FBS simultaneously increases the risk of including an infected fetus. Even low level contamination of FBS is harmful, as use of this supplement to grow cells can result in the infection of the cells and amplification of the virus. Therefore, high sensitivity tests are required. Antigen-capture enzyme-linked immunosorbent assay, RT-PCR, and qRT-PCR have been used for screening FBS for specific pathogens. The need for continuous development and evaluation of tests for broad spectrum pestivirus detection was further highlighted with the detection of HoBi-like viruses in FBS batches originating from regions with no reported case of HoBi-like virus infections in animals.36,37 It is desirable to have robust, sensitive tests for testing animals and animal products particularly as it applies to international trade.
There are several commercial tests available for detection of BVDV. Studies have been conducted to see if these tests can be used to detect HoBi-like viruses. A commercial kit was able to detect the isolate HoBi_D32/00 in supernatant of infected cells 2 and the isolate Th/04_Khonkaen in samples of experimentally infected animals and individual serum samples. 29 Interestingly, it was reported that the isolate IZSPLV_To was not detected by an ACE kit in a FBS batch. 25 In the current authors’ experience, more than 90% of FBS batches positive for pestivirus by molecular tests (RT-PCR) are not detected by ACE (personal observation). Therefore, FBS testing should not rely only on the use of ACE. However, ACE may be used with other diagnostic assays. The widely used pairs of primers HCV90-HCV368 26 and 324-326 34 were designed to detect a broad range of pestiviruses. The selection of the primer pair 324-326 for quality control of FBS has been described. 19 However, whereas this pair of primers is highly sensitive for BVDV and BDV strains, failure to detect HoBi-like viruses may occur. In order to address this issue, the current authors performed 10-fold dilutions of supernatant of bovine turbinate cells infected separately with pestivirus isolates, including HoBi_D32/00 and Italy-280/11A with titer from 103.7 to 10-0.7 TCID/50 µl. Supernatant of cells infected with the Brazilian HoBi-like strain SV713/09 were irradiated, and RNA was also tested. Reverse transcription PCR using the primer pairs HCV90-HCV368 and 324-326 was performed following conditions described previously.26,34 Both pan-pestivirus primer pairs tested displayed sensitivity of approximately 100.7 TCID/50 µl to BVDV strains while the sensitivity for HoBi-like strains was approximately 103.7 TCID/50 µl (Fig. 4). Tests were unable to detect virus RNA from irradiated supernatant of cells infected with the HoBi-like strain SV713/09 using these pan-pestiviruses primers.
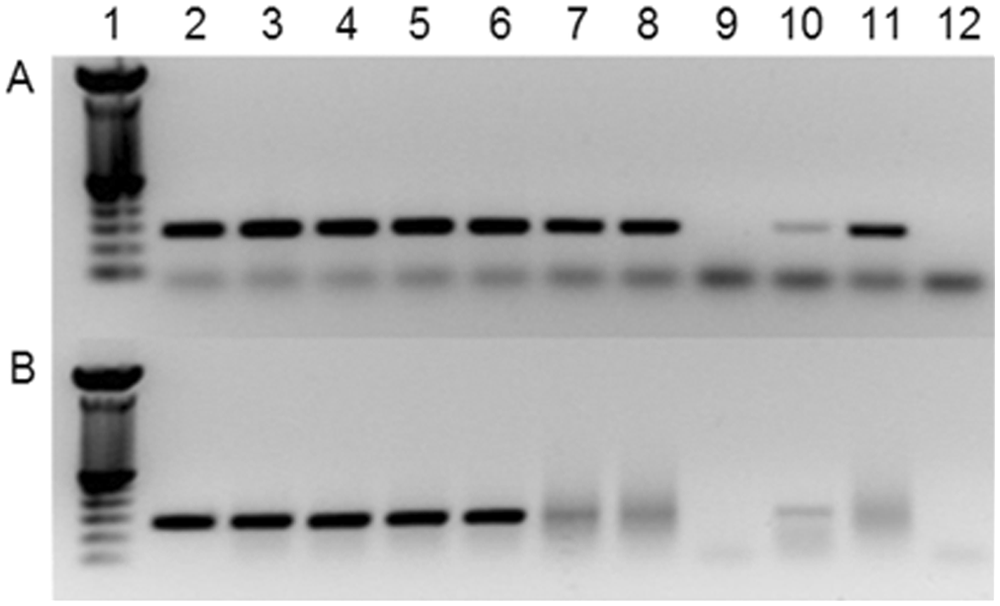
Reverse transcription polymerase chain reaction using the pan-pestivirus primers 324-326 (
To date, 2 specific RT-PCR reactions for HoBi-virus detection have been published. However, neither was validated under field diagnostic conditions. A qRT-PCR was successful in detection of HoBi-like viruses in FBS and in samples from calves acutely infected under experimental conditions.20,23,36 Cross-reaction with samples harboring high titers of BVDV-2 was observed. 12 The second reaction was a nested-RT-PCR. 12 Its application in screening large numbers of samples may be labor- and time consuming. Further, the increased number of steps in the reaction could lead to cross contamination. To date, there is no test fully validated to detect all bovine pestivirus species and/or to specifically detect HoBi-like viruses. Again, the high variability among pestiviruses indicates that laboratory diagnosis of these viruses should not rely on the use of a single test.
Virus detection in acutely and persistently infected animals
Commercial ACE kits are available and are designed to detect PI animals. Viral antigen may be detected in acutely infected animal samples such as buffy coats, nasal swabs, and tissues. These tests may also detect PI animals harboring HoBi-like viruses. However, studies validating the use of commercial BVDV tests to identify PI animals infected with HoBi-like viruses are still needed. These tests cannot differentiate between BVDV and HoBi-like viral infection.
The isolation and amplification of HoBi-like viruses in cell cultures seems to be easily performed using bovine turbinate cells and MDBK cells following standard protocols for BVDV isolation.2,9 Because most pestiviruses detected during acute and persistent infection are noncytopathic, virus identification requires the use of a posttest, usually RT-PCR, fluorescent antibody testing, or immunoperoxidase. The commercially available monoclonal antibody (mAb) 15C5 was shown to detect most BVDV isolates in immunoassays 13 and was also shown to detect HoBi-like viruses 2 (see reactivity of mAb with HoBi-like viruses and other pestivirus species in Table 4). Differentiation of animals acutely or persistently infected with HoBi-like viruses or BVDV can be performed by virus isolation followed by RT-PCR.2,12,23 Nucleotide sequencing of target sequences are required to confirm the agent identity. The use of commercial BVDV tests may be successful in some situations, although the differentiation between BVDV and HoBi-like viruses will require further testing such as RT-PCR and sequencing.
Reactivity of monoclonal antibodies with different pestivirus species virus isolates.*
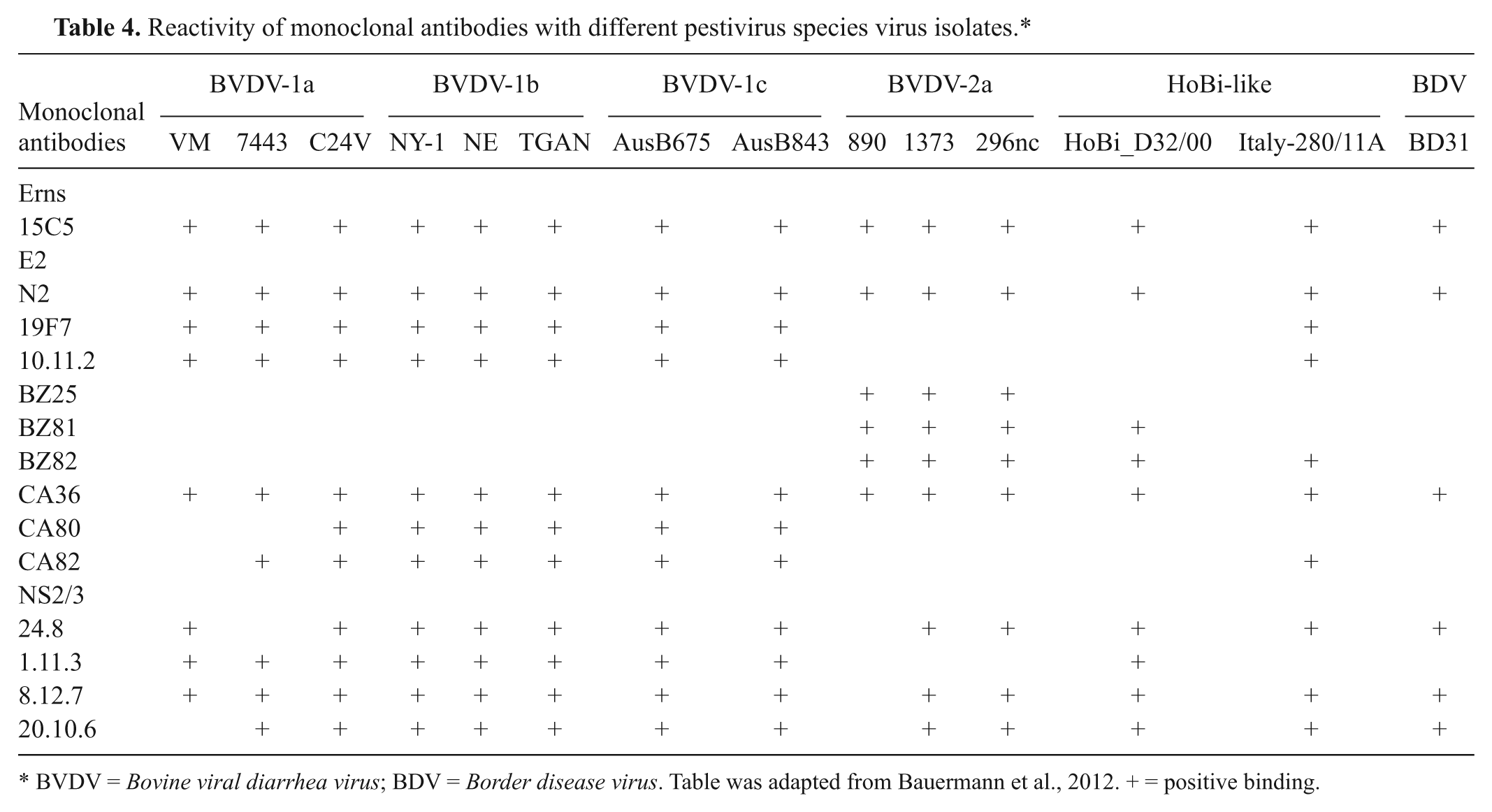
BVDV = Bovine viral diarrhea virus; BDV = Border disease virus. Table was adapted from Bauermann et al., 2012. + = positive binding.
Identification of exposed animals
Commercially available ELISA kits to detect BVDV antibodies (Ab-ELISA) were recently reported to yield false-negative results upon testing of serum samples of calves harboring moderate to high antibody titers against HoBi-like viruses.2,20 An additional concern is the time frame necessary for the development of detectable titers of antibodies by infected animals (i.e., up to 3–4 weeks).20,27 Also, differentiation as to whether the animal is harboring antibodies to BVDV, HoBi-like virus, or both is not possible with the current Ab-ELISA kits. Recently, 2012, a novel assay, called microsphere immunoassay, was used to detect antibodies against BVDV-1 and HoBi-like viruses based on binding to the glycoprotein Erns of a HoBi-like isolate. 33 Differentiation between BVDV-1 and HoBi-like immune response is not possible with this test in its present format.
Virus neutralization test (VNT) using HoBi-like strains can be used to detect exposure,2,20 and seems to be promising for differentiation between animals exposed to BVDV or HoBi-like viruses through the comparison of antibody titers against these viruses (unpublished data). It seems that cross-reaction between HoBi-like viruses and BVDV is limited. Therefore, serum samples harboring antibody titers to HoBi-virus 2- or more-fold higher than to BVDV strongly suggest exposure to HoBi-like viruses. Although VNT is time consuming and requires trained staff and laboratory structure for cell culture, to date, it seems the most suitable approach for detection and/or differentiation of animals exposed to BVDV and/or to HoBi-like viruses. Nonetheless, validation of this method is still required. At the herd level, comparative viral neutralizing titers between HoBi-like viruses and BVDV strains may indicate whether a herd was exposed to HoBi-like viruses. The detection of high titers to BVDV and/or HoBi-like viruses in nonvaccinated herds may suggest the presence of PI animals. Also, the presence of higher titers to HoBi-like viruses than BVDV species in herds vaccinated for BVDV is also an indicative factor of exposure to the emerging viruses because inactivated and modified live BVDV vaccines induce only limited cross-reaction against HoBi-like viruses. Regardless, the best diagnostic approach seems to be the search for PI animals by virus isolation followed by molecular characterization.
Conclusion
Although the origin remains unknown, data gathered to date indicates the widespread distribution of HoBi-like viruses in Brazilian cattle, and to a lesser extent, in other selected countries. The extent of economic losses due to HoBi-like virus infection and the prevalence of exposed animals are as yet unknown and, thus, are goals for future investigations. Further, there is a need for improved diagnostics, as the lack of accurate diagnostic tools for virus or antibody detection and the scarce information regarding the clinical presentation of the disease may lead to misdiagnosis at both the field and laboratory levels. To summarize, the clinical signs associated with HoBi-like infection may be undistinguishable from those observed in typical BVDV infections and, therefore, the impact of the disease may be underestimated due to misdiagnosis. The genetic and antigenic similarities among HoBi-like viruses and BVDV species may also promote a false idea that BVDV diagnostic tests are accurate enough to be used for HoBi-like virus detection. Veterinary diagnostic laboratories and biological product companies need to be aware that, whereas the diagnosis may be successfully performed in certain situations using BVDV tests, the percentage of misdiagnosis may be high, resulting in uncontrolled spread of the viruses and potentially leading to significant economic losses.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
