Abstract
The emergence of a newly recognized group of pestiviruses in cattle, the HoBi-like viruses, requires an evaluation of the available diagnostic tools and vaccines. The present study compared antigenic characteristics of Bovine viral diarrhea virus 1 and 2 (BVDV-1, -2) strains and HoBi virus. This comparison was based on detection of HoBi virus and antibodies against it by commercial enzyme-linked immunosorbent assays (ELISAs) and the level of cross-neutralizing antibodies present in sera from animals vaccinated with BVDV. Reactivity with a panel of monoclonal antibodies (mAbs) revealed greater cross-reactivity between BVDV species (BVDV-1, -2) and HoBi epitopes within Erns and NS2/3 proteins than between epitopes located in the E2 glycoprotein. The results suggest that a diagnostic test designed to detect both BVDV species and HoBi could be based on Erns or NS2/3 epitopes, while variation among E2 epitopes could be exploited in tests for differentiation of pestivirus species. The threshold of detection of HoBi virus by an antigen-capture ELISA kit based on detection of Erns was statistically similar to that for BVDV. In contrast, 2 commercial ELISA kits designed to detect antibodies against BVDV missed 22.2% and 77.7%, respectively, of serum samples harboring HoBi virus-neutralizing antibodies. In addition, sera of calves vaccinated with BVDV-1 and -2 presented low neutralizing activity against HoBi virus. The results demonstrate that in spite of antigenic similarities, HoBi virus is antigenically distinct from both BVDV species. Detection and control of HoBi virus infections in cattle would thus require the development of new diagnostic reagents and reformulation of current vaccines.
Introduction
Pestiviruses are enveloped, positive-sense RNA viruses that are members of the family Flaviviridae. Currently, this genus is comprised of 4 recognized virus species: Bovine viral diarrhea virus 1 (BVDV-1) and 2 (BVDV-2), Border disease virus (BDV), and Classical swine fever virus (CSFV). 27 Pestiviruses of bovine origin, mainly represented by the 2 species of BVDV, are globally distributed and lead to major losses in dairy and beef herds. Respiratory, gastroenteric, and reproductive diseases are among the list of clinical consequences of BVDV infection in cattle. 2 A 2009 economic analysis revealed that economic losses could reach approximately $88/animal. 12
In addition to the recognized pestivirus species, several other putative species have been proposed. Such species include giraffe, based on the H138 virus, which was isolated from giraffe in Kenya 4 ; Pronghorn virus, isolated from a blind pronghorn antelope in the United States 29 ; Bungowannah virus, isolated from an Australian outbreak of myocarditis syndrome in swine 14 ; and the HoBi-like viruses. HoBi-like viruses are unique among the putative pestivirus species because they have been found on more than 1 continent, have been isolated from multiple animal species, and infection results in clinical presentations that cannot be distinguished from those associated with BVDV infection.7,8,22,25,26
The first isolate of HoBi virus (D32/00_HoBi) was described in Switzerland, contaminating a batch of fetal bovine serum (FBS) imported from Brazil. 22 Subsequently, other HoBi-like viruses were identified, including 2 isolates present in FBS batches from South America16,17; CH-Kaho/cont, a cell culture contaminant 26 ; Brz buf 9 found in buffalo in Brazil 26 ; Th/04_Khonkaen, found in serum of calves in Thailand 25 ; and 2 isolates identified in aborted fetuses in Brazil. 7 In Italy in 2010, a HoBi-like virus was isolated during an outbreak of severe respiratory disease in calves. 8
The genome of pestiviruses is a single-stranded RNA molecule approximately 12.3 Kb in length, containing a long open reading frame (ORF) flanked by 2 untranslated regions (5’ and 3’ UTRs). The long ORF encodes a single polyprotein that is co- and post-translationally processed into 12 viral mature polypeptides in the following order: Npro-C-Erns-E1-E2-p7-NS2/NS3-NS4A-NS4B-NS5A-NS5B. Based on the phylogenetic analysis of the 5’-UTR sequences, which is widely used for classification of genus members,3,11,19,22 viruses within the HoBi-like group share a high level of homology (>93%). In contrast, the similarity between these viruses and BVDV-1 and -2 is approximately 75% and 80%, respectively.15,17,25 Although this new pestivirus group has not been definitively classified, it has been proposed to compose a third BVDV genotype (BVDV-3), 15 or a fifth pestivirus species (HoBi-like),22,25 the latter term being used in the present article.
Control and/or eradication programs for BVDV developed by several countries are based on the identification and elimination of persistently infected (PI) animals, biosecurity measures to prevent the introduction of infected animals, and surveillance strategies to monitor for BVDV exposure. These programs may also incorporate vaccination, depending on the incidence of BVDV exposure in the region. 13 Reliable diagnostic tests, to monitor both the detection of PI animals and exposure to BVDV, are crucial for the success of control programs and for the maintenance of BVDV-free status. In this sense, several diagnostic tests, for detection of virus or specific antibodies, are available for BVDV. 13 Virus isolation (VI) is considered the most accurate virus detection technique. 9 However, if the samples are inappropriately conserved or autolyzed, enzyme-linked immunosorbent assays (ELISA) for antigen detection or RNA detection by reverse transcription polymerase chain reaction (RT-PCR) may yield higher sensitivity.6,21,23 For antibody detection, virus neutralization (VN) testing and ELISAs are the most commonly used techniques. Reports of the isolation of HoBi-like viruses contaminating cells and FBS batches from South America have caused concern. Fetal bovine serum is widely used to propagate tissue culture cells and for propagation of viral strains for vaccine production. As HoBi-like viruses have been isolated from FBS, the contamination of vaccines could result in the introduction of HoBi-like strains into new regions. Further, these uncharacterized pestiviruses may be a threat for BVDV control and eradication programs worldwide. As infection with HoBi-like viruses has been linked to reproductive and respiratory disease, the introduction of HoBi-like viruses into naïve cattle could have serious economic impact. 24 Thus, surveying for the presence of HoBi-like strains in animals, animal products, and biologics is important.
The goal of the present study was to investigate the suitability of monoclonal antibodies (mAbs) and routinely used BVDV tests for the diagnosis of HoBi virus. The antigenic cross-reactivity between HoBi virus and other pestivirus species was also investigated in order to identify proteins with conserved epitopes and to evaluate levels of cross-protection resulting from vaccination with a commercial vaccine against BVDV.
Materials and methods
Viruses and cells
Eighteen noncytopathic isolates of pestiviruses a were used: BVDV-1a (VM, C24V, 7443), BVDV-1b (TGAN, NE, NY-1), BVDV-1c (AusB675, AusB730, AusB843), BVDV-2a (296nc, 890, 1373), BDV (Idaho207, CB5, BD31), and 3 isolates from putative new pestivirus species (HoBi, Pronghorn, Bungowannah).14,22,29 Viruses were propagated in bovine turbinate (BT) cells, a which are typically used between passages 7 and 12. Cells were grown in minimal essential medium, b supplemented with l-glutamine (final concentration 1.4 mM), gentamicin (final concentration 50 mg/l), and 10% FBS, tested free of BVDV and antibodies against BVDV, by RT-PCR and VN test, respectively.19,20 Cells were tested by RT-PCR and were found free of BVDV. 19 Cells and serum were also tested and found to be free of HoBi by RT-PCR. 19 The primers forward 5’-GGGTAGTC GTCAATGGTTCGA-3’ and reverse 5’-TAGCAGGTCTCTG CAACACCC-3’, predicted to amplify a fragment of the 5’-UTR of HoBi virus, were used. Cell culture monolayers were inoculated with the respective viruses when they were approximately 70% confluent. After inoculation, cultures were incubated at 37°C for 72–96 hr. Cultures were harvested by freezing at –20°C. After freeze–thaw cycle followed by centrifugation for 10 min at 1,000 × g, supernatants were collected, aliquoted, and stored at –80°C until use. The virus stocks were titrated in 96-well microtiter plates, and the titers were expressed as tissue culture infective doses (TCID). 18
Monoclonal antibody binding
A panel of 16 mAbsa,c with specificity for BVDV proteins (Erns, N2, or NS2/3) was used to compare antigenic similarity among pestivirus species. For this purpose, 18 pestivirus isolates (Table 1) were individually inoculated (final concentration 10 4 TCID/ml) in BT cells grown in 96-well plates and left to adsorb for 90 min. The first 2 columns of the plate were used as positive controls, inoculated with BVDV-1a (7443) and BVDV-2a (890), respectively. The last column was kept as negative control. Culture medium with 10% FBS was added, and plates were kept at 37°C for 96 hr. Cells were then fixed and subjected to an immunoperoxidase test (IMPT) executed as previously described.1,20 Monoclonal Abs with availability of ascites fluids were diluted 1:1,000 in phosphate buffered saline (PBS) with bovine albumin (PBS-BSA 0.01%) and used as primary antibody. In the absence of ascites (Table 1), the supernatants of hybridoma cell cultures were diluted 1:2 in PBS-BSA 0.01%. Goat anti-mouse immunoglobulin G conjugated with horseradish peroxidase d was used as secondary antibody, and 3-amino-9-ethylcarbazole was used as the substrate. An IMPT for all mAbs was performed for each virus in 2 sets of plates in duplicate wells.
Reactivity of monoclonal antibodies with different pestivirus species virus isolates.*
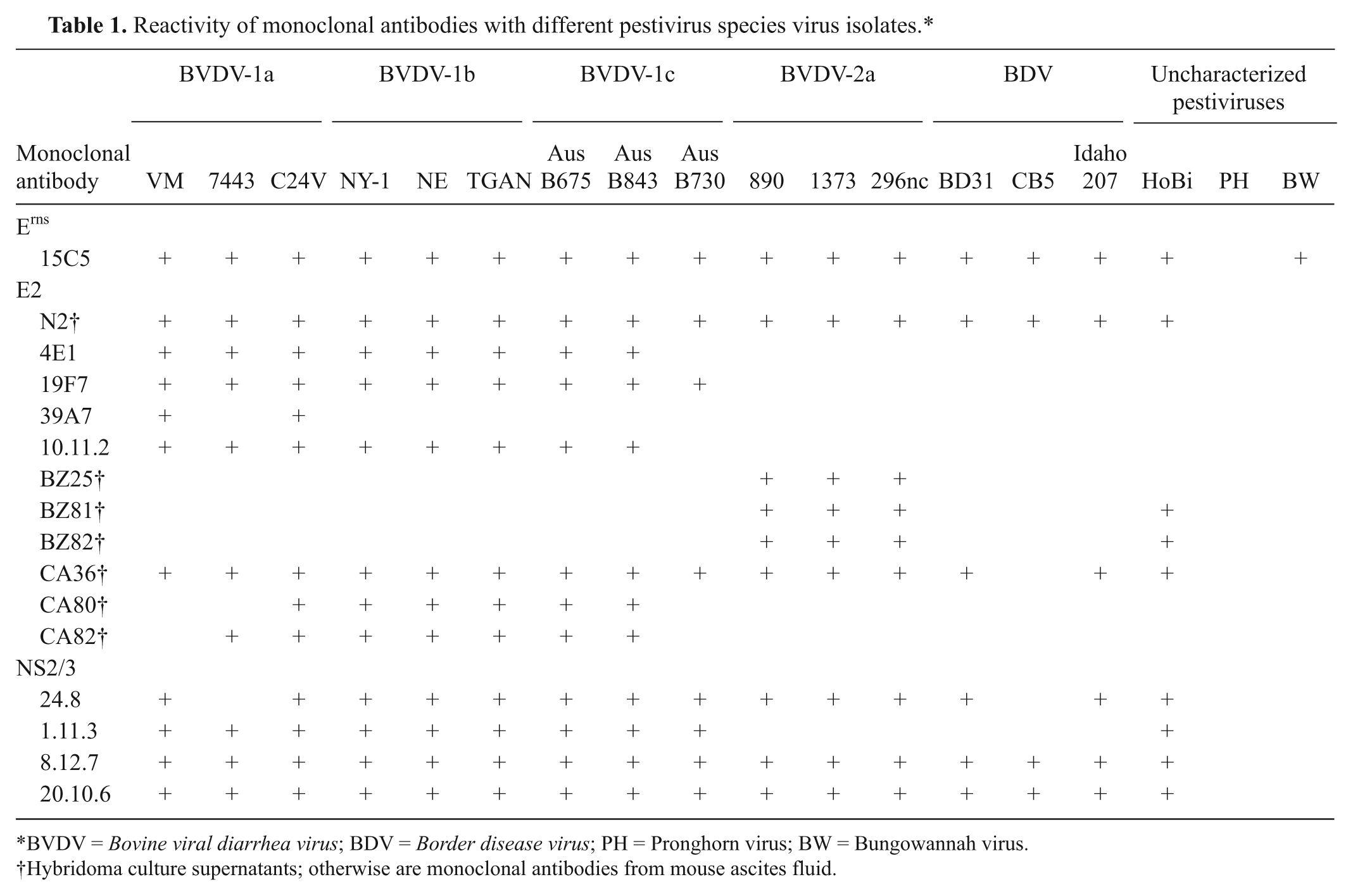
BVDV = Bovine viral diarrhea virus; BDV = Border disease virus; PH = Pronghorn virus; BW = Bungowannah virus.
Hybridoma culture supernatants; otherwise are monoclonal antibodies from mouse ascites fluid.
BVDV antigen-capture ELISA
To determine whether a commercially available BVDV antigen-capture ELISA (ACE) would be suitable for detection of genetically divergent pestiviruses, 10-fold dilutions of viral stocks in MEM were prepared. Three strains from each of the 5 genotypic groups (BVDV-1a–c, BVDV-2, BDV) from recognized pestivirus species and HoBi virus were diluted (10 6 –10 4 TCID). Due to the low initial virus titers, dilutions from 10 5 to 10 3 TCID were used for Bungowannah and Pronghorn viruses (Table 2). Detection of pestiviruses was performed using a commercially available BVDV ACE kit, e following the manufacturer’s instructions. The optical density (OD) value was used to calculate the sample to positive (S/P) ratio, where samples were considered positive with a value above 0.39. The S/P ratios for dilutions of each virus were used to calculate the R 2 and logarithmic regression, using a statistical software. f Based on the regression equation, the lowest titer with positive result for each virus was calculated, and the average of the lowest titer detected for each BVDV subgenotype and BDV species was determined. Then, means were submitted to Tukey–Kramer test, using statistical analysis software. g
Sensitivity of a Bovine viral diarrhea virus antigen-capture enzyme-linked immunosorbent assay kit for detection of pestiviruses in supernatant of infected bovine turbinate cells.*
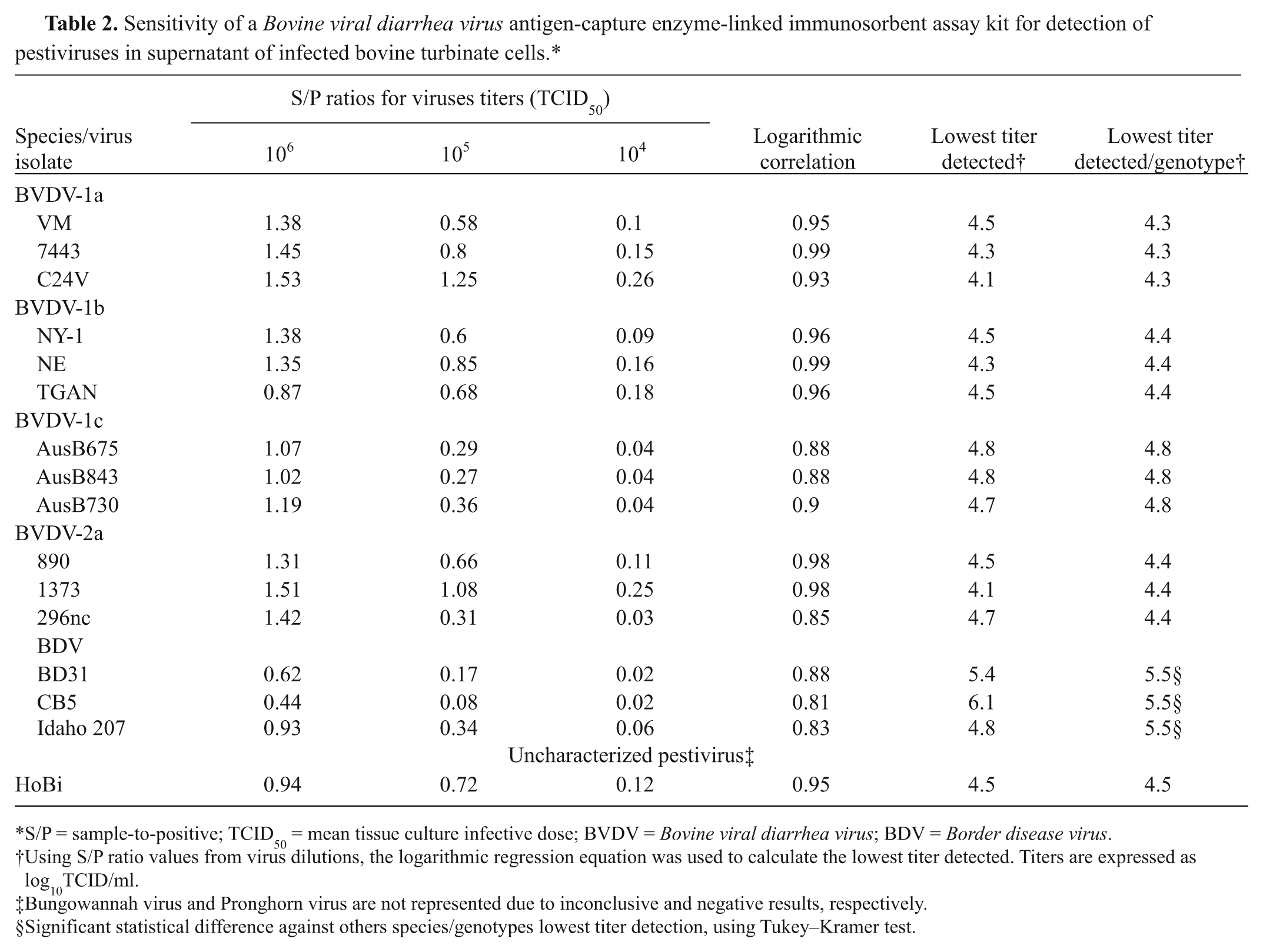
S/P = sample-to-positive; TCID50 = mean tissue culture infective dose; BVDV = Bovine viral diarrhea virus; BDV = Border disease virus.
Using S/P ratio values from virus dilutions, the logarithmic regression equation was used to calculate the lowest titer detected. Titers are expressed as log10TCID/ml.
Bungowannah virus and Pronghorn virus are not represented due to inconclusive and negative results, respectively.
Significant statistical difference against others species/genotypes lowest titer detection, using Tukey–Kramer test.
Neutralizing activity of HoBi virus antisera against other pestiviruses
Sera from 2 groups of calves infected with HoBi virus were used (Ridpath et al., manuscript in preparation). Briefly, the first group was composed of 1 calf inoculated intranasally with 2 ml of HoBi virus (10 5 TCID) and 3 calves kept in contact. In the second group, 2 calves were inoculated under the same conditions, and 3 animals were kept in contact (Table 3). The sera of animal no. 504 (group 1) and a pool from animals from group 2 (nos. 509, 510, and 513) from the day 42 post-inoculation were selected. These sera were tested by VN against viruses listed (Table 3), and the results were visualized through IMPT as previously described.1,20 Briefly, sera dilutions from 1:4 to 1:256 were incubated individually with 10 4 TCID of each virus for 90 min at 37°C in 5% with CO2. Suspensions of BT cells were added, and after incubation of 4 days at 37°C in 5% CO2, monolayers were fixed and subjected to IMPT. The mAb used for detection of Bungowannah antigens was 15C5 c ; the other viruses, with the exception of Pronghorn, were detected using antibody N2. a Neutralization of Pronghorn virus was verified by RT-PCR, due to the lack of specific mAbs. For RT-PCR testing, RNA was extracted from VN dilutions of each serum individually, using a commercial kit. h Reverse transcription PCR was performed using pan-pestivirus primers, following conditions described previously. 28 In the presence of a negative result, the serum dilution was considered positive for neutralizing activity. The VN titers were expressed as the reciprocal of the highest dilution that neutralized viral infectivity.
Reactivity of positive HoBi virus sera with different pestivirus species.*
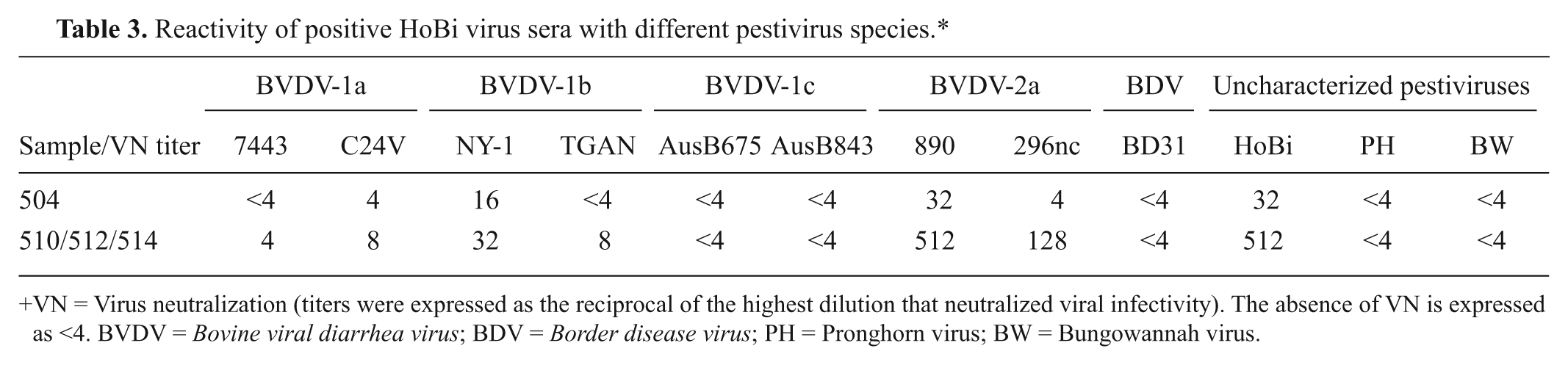
VN = Virus neutralization (titers were expressed as the reciprocal of the highest dilution that neutralized viral infectivity). The absence of VN is expressed as <4. BVDV = Bovine viral diarrhea virus; BDV = Border disease virus; PH = Pronghorn virus; BW = Bungowannah virus.
Detection of HoBi virus serologic response with BVDV antibody detection ELISA kits
To determine whether the ELISA kits designed to detect BVDV antibodies are adequate to detect antibodies to HoBi viruses, 2 groups of animals were inoculated with HoBi virus, keeping some animals as sentinels, for a total of 9 animals (Table 4). Sera obtained at the day of birth, and days 9, 18, and 42 post-inoculation were tested for antibodies by ELISA kits from 2 companies, ab-ELISA-1 i and ab-ELISA-2, j according to the manufacturers’ instructions. In parallel, VN testing using HoBi virus was performed on these samples (Table 4). The titers were expressed as the reciprocal of the highest dilution that neutralized viral infectivity.
Comparison of antibody detection enzyme-linked immunosorbent assay kits and virus neutralization test for HoBi virus antibodies in sera of experimentally infected calves, by days 18 and 42 post-infection.
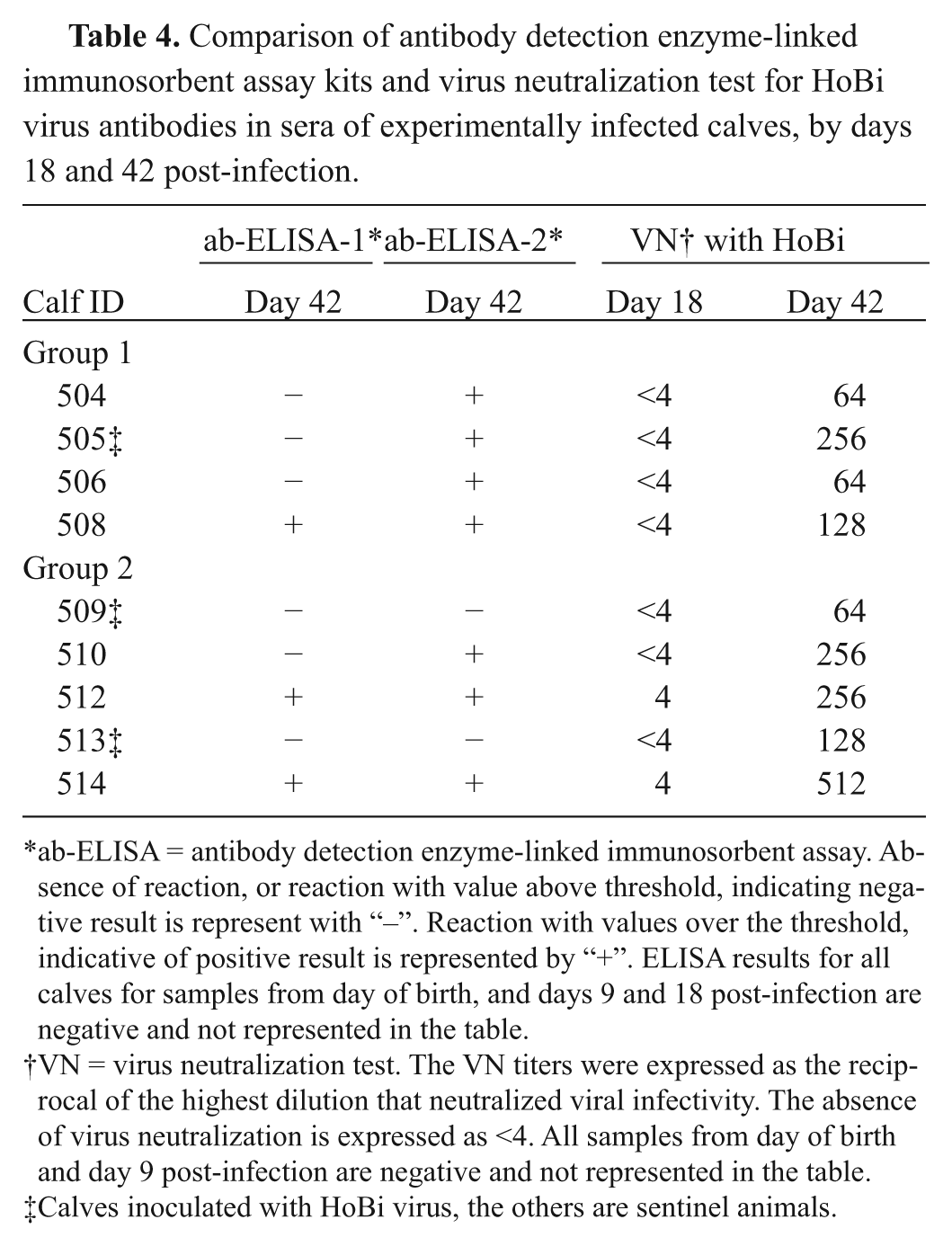
ab-ELISA = antibody detection enzyme-linked immunosorbent assay. Absence of reaction, or reaction with value above threshold, indicating negative result is represent with “–”. Reaction with values over the threshold, indicative of positive result is represented by “+”. ELISA results for all calves for samples from day of birth, and days 9 and 18 post-infection are negative and not represented in the table.
VN = virus neutralization test. The VN titers were expressed as the reciprocal of the highest dilution that neutralized viral infectivity. The absence of virus neutralization is expressed as <4. All samples from day of birth and day 9 post-infection are negative and not represented in the table.
Calves inoculated with HoBi virus, the others are sentinel animals.
Neutralizing antibody response to pestiviruses following BVDV vaccination
In order to analyze the cross-serological response between BVDV and uncharacterized pestiviruses, 50 calves were immunized twice, 30 days apart, with an inactivated vaccine k containing BVDV-1 and -2. After 8 weeks, sera was collected and tested by VN and IMPT against 13 pestiviruses (Table 5). The presence or absence of viral antigens in cells was used as indicator of absence or presence of neutralizing antibodies. In this way, IMPT was performed using mAbs 15C5 for Bungowannah and N2 for the other viruses. Pronghorn virus was detected by RT-PCR. The titers were expressed as the reciprocal of the highest dilution that neutralized viral infectivity.
Virus neutralization test of sera from 50 calves vaccinated with Bovine viral diarrhea virus 1 and 2 strains, against different pestivirus species.*
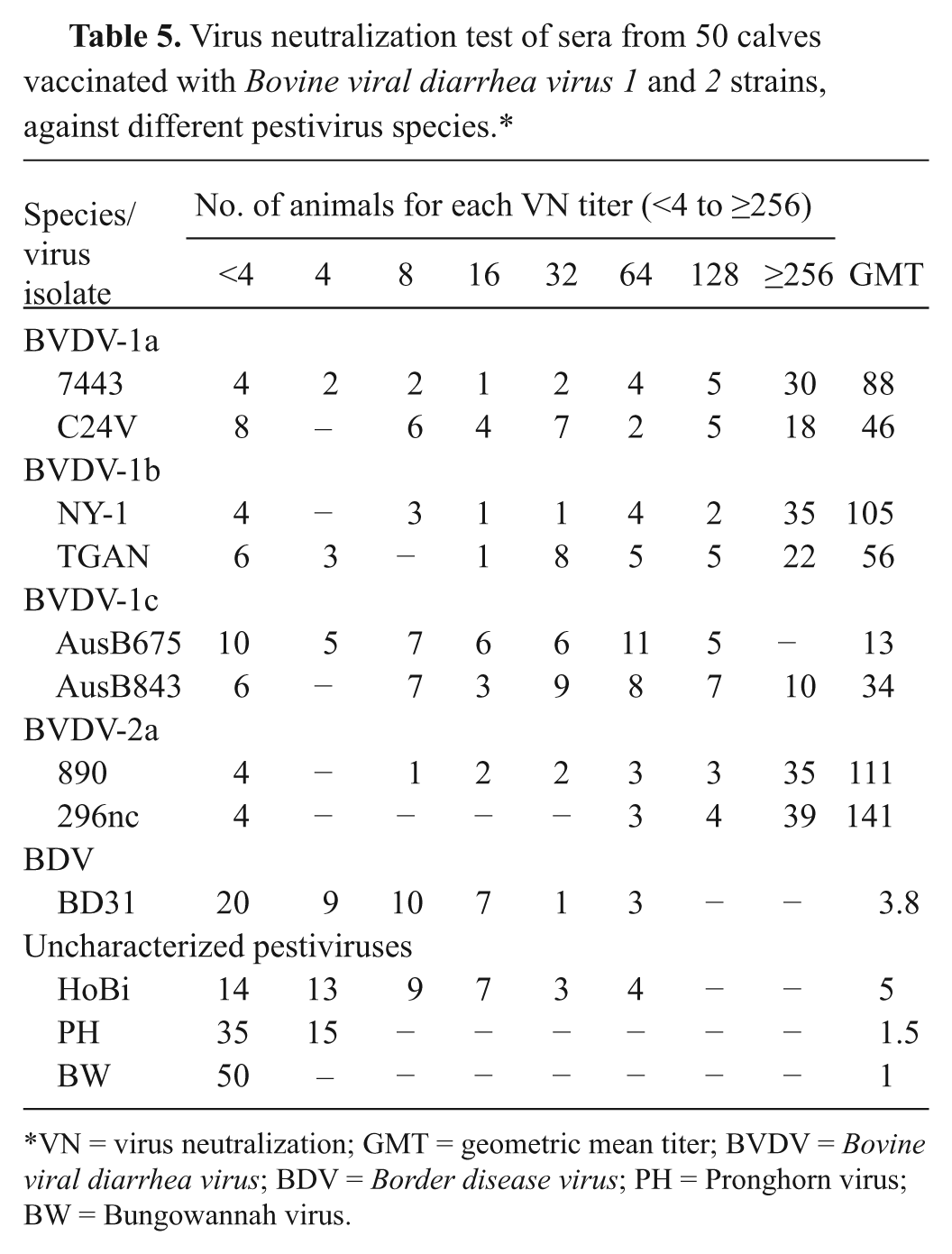
VN = virus neutralization; GMT = geometric mean titer; BVDV = Bovine viral diarrhea virus; BDV = Border disease virus; PH = Pronghorn virus; BW = Bungowannah virus.
Results
Monoclonal antibody binding
The use of a panel of mAbs demonstrated conserved epitopes shared among HoBi and BVDV-1 and -2 isolates (Table 1). Monoclonal Ab 15C5, specific for the pestivirus Erns glycoprotein, reacted with the highest number of isolates, failing to detect only Pronghorn virus. On the other hand, mAbs against E2 displayed a variable range of reactivity. In fact, only two E2 mAbs (N2 and CA36) reacted with both BVDV genotypes and HoBi virus. Seven mAbs bound only to isolates within BVDV genotypes, and 2 mAbs (BZ81 and BZ82) recognized BVDV-2 isolates and HoBi. However, mAbs against E2 failed to react with Bungowannah and Pronghorn viruses. The four NS2/3 mAbs used (20.10.6, 1.11.3, 8.12.7, and 24.8) were able to identify most pestivirus isolates, but among the uncharacterized pestiviruses used in the current study, only HoBi was detected by these mAbs. Monoclonal Ab 1.11.3 bound to BVDV-1a–c isolate and to HoBi virus. According to these results, an IMPT designed to detect a wide range of pestiviruses should include mAbs N2, CA36, 8.12.7, 20.10.6, and 15C5, which are able to recognize a wide range of isolates. In addition, differential binding by mAbs 1.11.3 and BZs 25, 81, and 82 may allow differentiation between HoBi and the 2 BVDV genotypes.
BVDV antigen-capture ELISA
The BVDV ACE kit was able to detect HoBi virus but failed to detect Pronghorn virus and yielded inconclusive results with Bungowannah virus. All strains of BVDV and BDV used in the test yielded positive results. Using logarithmic regression, the lowest HoBi titer detected by ACE was determined to be 104.5 TCID (Table 2). Analyses with Tukey–Kramer test using a statistics software g showed no significant difference between the threshold of HoBi virus detection and the average of the lowest titer detection of other BVDV genotypes. For these viruses, the threshold varied from 104.3 to 104.8 TCID. The only significant difference in the threshold of detection was verified with BDV, where the minimum titer detected was 105.4 TCID. The results show that ACE is adequate to detect HoBi virus but could not be used to differentiate between HoBi and BVDV-1 or -2 isolates.
Neutralizing activity of HoBi virus antisera against pestiviruses
The neutralizing activity of HoBi virus antisera against BVDV-2a by VN was higher than against other pestiviruses (Table 3). The 2 serum samples tested (no. 504 and pool) displayed, respectively, the same neutralizing titer (32 and 512) against HoBi virus and the isolate 890 (BVDV-2a). The VN titers against BVDV-2a 296nc were 4 and 128, respectively. For isolates from subgenotypes of BVDV-1a–b, the titers ranged from <4 to 16 for calf no. 504 and from 4 to 32 for the pool. No neutralization was observed against BVDV-1c, BDV, and the other uncharacterized pestiviruses.
Detection of HoBi virus seroconversion using BVDV antibody detection ELISA kits
Positive reaction was only detected in sera collected at day 42 post-infection, by both kits. Ab-ELISA-1 detected 3 out of 9 positive samples; ab-ELISA-2 detected 7 out of 9 positive samples (Table 4). By VN, at day 42 post-infection, all samples were positive, with titers ranging from 64 to 512. Two samples (animal nos. 512 and 514) were positive earlier, at day 18 post-infection, with VN titer of 4.
Neutralizing antibodies to pestiviruses in sera of BVDV-vaccinated cattle
Eight weeks after vaccination of cattle with 2 doses of a vaccine containing inactivated BVDV-1 and -2 strains, sera from 50 animals were collected and tested by VN against 12 pestiviruses (BVDV1-a–c, BVDV-2, BDV, and atypical isolates). As expected, the highest neutralizing titers were observed against viruses from the homologous genotypes (Table 5). Geometric mean titer reached 141 and 111 for BVDV-2 strains 296nc and 890, respectively. For isolates of BVDV-1a–b, GMT ranged from 46 to 105. Lower GMT was found for BVDV-1c, with values of 34 and 13 for isolates AusB843 and AusB675, respectively. For BVD strain BD31, the GMT was 3.8 and 5.0 for HoBi virus. The neutralization for Pronghorn virus was 1.5; no neutralization was observed against Bungowannah virus.
Discussion
The emergence of novel bovine pestiviruses, including the HoBi-like viruses, and their potential introduction into naïve cattle populations represents a major concern for beef and dairy producers. The contamination of FBS with HoBi-like viruses may also pose a threat for the safety of biological products. Therefore, the availability of adequate reagents and suitable techniques to detect these agents is important for BVDV control programs and for surveillance of animals and animal products subject to international trade. In particular, HoBi virus poses the greatest threat among the uncharacterized pestiviruses, based on reports of persistent infection, apparent dissemination in different continents, and detection in commercial FBS batches.16,17,22,25,26
A panel of mAbs showed that HoBi virus was recognized by 4 mAbs directed to the glycoprotein E2, the major and most variable envelope BVDV glycoprotein, 5 with greater similarity with BVDV-2 (Table 1). A closer relationship between HoBi and BVDV-2 (than between HoBi and BVDV-1) was also observed in VN test performed in the course of the present study, in which serum against HoBi displayed higher neutralizing activity against BVDV-2 isolates than against BVDV-1 isolates (Table 3).
The ACE detected HoBi virus in culture supernatants with the same sensitivity as it detected both BVDV genotypes. On the other hand, inconclusive and negative results were obtained for Bungowannah and Pronghorn viruses, respectively (Table 2). The findings are not completely unexpected because phylogenetic analysis has shown that these viruses are more distant from the recognized pestivirus species than HoBi-like viruses.14,29 Monoclonal Ab 15C5, directed to a well conserved epitope within Erns, 5 was able to recognize Bungowannah virus in IMPT (Table 1). In contrast, ACE based on this mAb yielded inconclusive results. Probably, technical adjustments in the test would be able to increase the sensitivity. On the other hand, none of the mAbs tested bound to Pronghorn antigens. Analysis of Pronghorn Erns revealed an amino acid mutation at position 315 (data not shown); a change in this site has already been described as responsible for the failure of mAb 15c5 to recognize BVDV-2 isolate AU501. 10 In addition, mutations in the adjacent sites 314 and 316 were also found, which might explain the failure of mAb biding. A new version of this kit is now commercially available, in which a second mAb was added for detection of isolates with mutations similar to AU501. Thus, the new kit may be able to detect Pronghorn and Bungowannah viruses. Nevertheless, further studies are required to confirm this information. Although genetically more distantly related to BVDV than Pronghorn virus,14,15,29 Bungowannah virus was recognized by mAb 15C5 in IMPT. Interestingly, the Erns amino acid residue at position 315 is conserved in this virus (data not shown).
The high sensitivity of the ACE test, in which HoBi detection titer was comparable to those for BVDV genotypes, indicates that the ACE test may be adequate for HoBi detection. This finding agrees with another study that reported the detection of the HoBi-like Th/04_Khonkaen, using this kit. 25 In contrast, the HoBi-like isolate IZSPLV_To was not detected using the same test. 17 Further studies including a broader selection of HoBi-like isolates are required to determine whether this kit would be suitable for surveillance of HoBi-like viruses.
The evaluation of ELISA kits for BVDV antibodies demonstrated the need for specific diagnostic tests for HoBi-like viruses because they failed to detect a considerable number of samples harboring HoBi virus antibodies (Table 4). The VN test against HoBi virus showed that the samples missed by these kits had VN titers ranging from 64 to 256. None of the kits was able to detect the 2 Ab-positive samples collected on day 18 post-infection, while VN testing detected 2 positive samples with a titer of 4. Despite the fact that these tests rely on proteins, NS3 and Erns, which show relatively high homology among pestiviruses, the tests are not sensitive enough to detect antibody levels below 256. Following natural infections with BVDV strains, antibody titers are frequently below 256. Thus, this lack of sensitivity could lead to failure in detecting animals naturally exposed to HoBi viruses. Surveillance for exposure to HoBi-like viruses could begin by including HoBi-like viruses in routine VN tests. However, a comparison of VN test results using other isolates of HoBi-like viruses needs to be completed to determine the degree of antigenic variability among HoBi-like isolates.
Sera from animals receiving an inactivated BVDV-1 and -2 vaccine presented low neutralizing GMT against HoBi virus, comparable to GMTs to BDV strains (Table 5). These findings demonstrated that the serological cross-reactivity between these viruses is low, in spite of antigenic similarities with BVDV. Furthermore, neutralization against Pronghorn was even lower; and no neutralization was detected against Bungowannah. The relative antigenic divergence observed correlates with the phylogenetic distance between these groups of viruses. The sequence similarity between E2 protein of the reference strains NADL (BVDV-1) and 890 (BVDV-2) is approximately 70%, while the similarity of NADL strain with Bungowannah and Pronghorn viruses is around 48% and 56%, respectively. 14 Interestingly, animals infected with HoBi virus, and with high antibody titers to HoBi, showed moderate to high neutralizing titers to BVDV-2 isolates. In contrast, animals with high titers to BVDV-2 showed low to moderate neutralizing activity against HoBi. These findings agree, in part, with a previous study in which anti–BVDV-2 sera showed low neutralizing titer against HoBi, although higher than anti–BVDV-1 sera. 22 Modified live (MLV) BVDV vaccines tend to induce broader cross-reactivity with BVDV isolates, which may also be observed with HoBi. Further studies using sera of animals immunized with inactivated and MLV vaccines would be useful to compare the degree of cross-neutralizing activity in HoBi virus.
The emergence and spread of HoBi-like viruses in commercial dairy operations in Southeast Asia raised concerns about the efficacy of detection of HoBi-like viruses with diagnostic tests used for BVDV. One concern is determining whether current surveillance programs for BVDV can be adapted for surveillance for HoBi-like viruses. The other concern is that HoBi virus outbreaks may be misdiagnosed as BVDV outbreaks. Monoclonal Ab binding in the present study demonstrated that while HoBi virus shares epitopes with BVDV isolates, the binding pattern is distinct for the pestiviruses species. A conserved epitope in the Erns viral protein was confirmed by the ACE test, where HoBi was detected with the same efficiency as BVDV-1 and -2. Nevertheless, this region seems to be variable within HoBi-like viruses because the isolate IZSPLV_To was not detected in a previous study. 17 Although similarities were identified with mAb binding at the NS2/3 protein, ab-ELISA-1 based on NS3 only detected 33.3% of HoBi antibody sera. These results indicate the need to develop appropriate ELISAs for surveillance for exposure to this new virus group. Similarly, VN routine should include HoBi, since moderate levels of HoBi antibodies were missed in VN testing using the strains 7443 (BVDV-1a) and TGAN (BVDV-1b; Table 3). Also, specific vaccines for these viruses are needed because antibodies generated by animals vaccinated against BVDV-1 and/or -2 have displayed low cross-reactivity against HoBi virus.
The evolution/emergence of “new” pestiviruses is an unpredictable event. Therefore, the adoption of control and eradication programs for pestiviruses and the safety testing for biological products must be in constant evolution and should not rely on a single test. As the present study was performed using a single isolate of HoBi-like virus, additional studies involving a higher number of isolates are obviously required. Regardless, it is possible to conclude that the emergence and spread of HoBi-like viruses do represent a justifiable concern, as the traditional commercial serological diagnostic tests for BVDV may fail to detected animals harboring HoBi antibodies. Comparative quantitative VN using HoBi-like and BVDV strains may be the best option for increasing the sensitivity of pestivirus antibody detection and may contribute to the knowledge about the presence or absence of BVDV and HoBi-like virus in tested herds. Further studies are necessary to access the diagnostic impact in populations where both viruses are present. On the other hand, the antigenic similarities observed between HoBi and BVDV may pose difficulties for discrimination among pestivirus species.
Footnotes
Acknowledgements
The authors thank Kathryn Fulk, Patricia Federico, and Renae Lesan for technical support; Brian VanderLey for statistical analysis; and Edward J. Dubovi for kindly providing monoclonal antibodies. In addition, the authors thank the National Veterinary Services Laboratories’ Diagnostic Laboratory, USDA/APHIS, particularly Beverly Schmitt, Sabring Swenson, and Diane Rodman, for help in generating HoBi convalescent sera and for help in designing appropriate biosecurity measures for working with emerging pestivirus species; and Novartis Animal Health for providing antisera from vaccinated animals.
a.
National Veterinary Service Laboratories/APHIS/USDA, Ames, IA.
b.
F15 Eagle medium, Life Technologies Corp., Carlsbad, CA.
c.
Cornell University, Ithaca, NY.
d.
Cappel Laboratories Inc., Cochranville, PA.
e.
HerdChek BVD Antigen Test Kit, IDEXX Laboratories, Westbrook, ME.
f.
Excel 2007, Microsoft Corp., Redmond, WA.
g.
SAS Institute Inc., Cary, NC.
h.
RNeasy Mini Kit, Qiagen Inc., Valencia, CA.
i.
SVANOVIR BVDV p80-Ab ELISA kit, SVANOVA Biotech AB, Uppsala, Sweden.
j.
IDEXX BVDV Ab Test kit, IDEXX Laboratories, Westbrook, ME.
k.
Novartis, East Hanover, NJ.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Disclaimer: Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Portions of this work were supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
