Abstract
A 10-year-old, female spayed Chihuahua dog was presented to the Veterinary Teaching Hospital at the University of Georgia for evaluation of seizures, abnormal mentation, and cervical pain of 2 months duration. On magnetic resonance imaging, there was generalized thickening of the meninges overlying the left cerebral hemisphere and along the falx cerebri. Despite symptomatic treatment, the dog remained neurologically affected. Consequently, the owners elected euthanasia. On gross examination, the meninges covering the left cerebral hemisphere were severely thickened and firmly adhered to the calvaria. On transverse section, the white matter of the left cerebral hemisphere was swollen, enlarged, and extended across the midline with resultant compression of the right cerebral hemisphere. Cytologic evaluation of an impression smear of the thickened meninges showed numerous large, spindloid to polygonal cells with abundant, amphophilic, vacuolated cytoplasm, present either in clusters or in individual cells. Histopathologic evaluation of the meninges revealed a poorly circumscribed and infiltrative, moderately cellular neoplasm, composed of vacuolated, spindloid to polygonal cells with marked anisocytosis and anisokaryosis, arranged in sheets, and occasionally separated by thick bands of connective tissue. Immunohistochemistry for vimentin revealed diffuse cytoplasmic staining of the neoplastic cells. Although the periodic acid–Schiff reaction was negative, ultrastructural findings showed numerous vesicles that were empty or that contained membranous or electron-dense material. Based on gross, microscopic, immunohistochemical, and ultrastructural changes, the meningeal neoplasm was diagnosed as an atypical granular cell tumor.
Granular cell tumors (GCT) are rare in human beings and animals, and most commonly occur in the dog, horse, cat, rat, and ferret, and has also been reported in the cockatiel.13,16,17,19,21,23,28,29 In dogs, GCTs have been reported in the lung, mediastinum, diaphragm, heart, lymph node, brain, spinal nerve root, skin, vocal cord, and trachea.9,16,17,21 Granular cell tumors in the horse have only been reported in the lung,18,19,25 whereas in the cat, the tumors have been reported in the tonsil, tongue, vulva, cerebrum, and digits.13,19,28 There was 1 report of periocular tissue involvement in cockatiels. 19 In the central nervous system, GCTs are rare in animals; however, the tumors have been reported in rats,15,17,29 dogs,17,22,24 one ferret, 23 and one cat. 13 Although there is minimal variation in cell morphology by light microscopy, the histogenesis of GCT remains controversial. Multiple embryological origins of this tumor have been suggested.1,2,7 The current report describes a case of an atypical meningeal GCT in a dog with clinical, histological, histochemical, immunohistochemical, and ultrastructural findings.
A 10-year-old, female spayed Chihuahua dog was presented to the Veterinary Teaching Hospital at the University of Georgia (VTH-UGA; Athens, GA) for evaluation of seizures, abnormal mentation, and cervical pain of 2 months duration. Radiographs of the vertebral column taken at the onset of signs were normal. The dog was treated with carprofen and methocarbamol. Approximately 1 month later, the dog experienced a generalized seizure. Over the subsequent month, the dog became lethargic, began pacing, and had seizures with increasing frequency. Consequently, the dog was referred to the VTH-UGA. At the time of admission, physical examination was normal. Neurologically, the dog had a dull mentation and cervical pain. With the exception of hypermetria of the right pelvic limb, the dog had a normal gait but walked compulsively. Postural reactions and spinal reflexes were normal. Cranial nerve examination revealed an absent menace response, blindness, and miosis in the right eye. The palpebral and pupillary light reflexes were normal in both eyes. The remainder of the neurological examination was normal. Based on the neurological examination and history of seizures, the neuroanatomic diagnosis was consistent with a left-sided prosencephalic lesion. Although difficult to reconcile with a prosencephalic lesion, anisocoria may be observed secondary to release of a prosencephalic inhibitory upper motor neuron. 6 Likewise, cervical pain was thought to be secondary to a prosencephalic lesion. 5 Alternatively, multifocal disease with lesions involving prosencephalon or reticular activating system; the optic tract, lateral geniculate nucleus, optic radiation, or occipital lobe of the cerebrum; fibers providing sympathetic input to the right eye; and cervical spinal cord also could account for altered mental state, blindness, anisocoria, and cervical pain, respectively. Differential diagnosis included inflammatory noninfectious disease (granulomatous meningoencephalomyelitis or necrotizing meningoencephalitis), infectious disease, and neoplasia (primary or metastatic).
A complete blood count revealed a mature neutrophilia (12,180 cells/µl; reference [ref.] interval: 2,900–12,005 cells/µl). Serum biochemistry revealed increased alkaline phosphatase (131 IU/l; ref. interval 10–119 IU/l), hyperglycemia (123 mg/dl; ref. interval 66–109 mg/dl), and hyperchloremia (5.4 mmol/l; ref. interval 3.7–4.8 mmol/l). Magnetic resonance imaging (MRI) of the head was performed using a 3.0 tesla unit. a The following pulse sequences were obtained in the transverse and sagittal planes: T1-weighted fluid attenuated inversion recovery (T1W FLAIR), T2-weighted (T2W), T2W FLAIR, and T2*W. Additionally, T1W FLAIR images were obtained in the transverse, dorsal, and sagittal planes after intravenous administration of gadopentetate dimeglumine (0.1 mmol/kg). b
On MRI, there was generalized thickening of the meninges overlying the entire left cerebral hemisphere and along the falx cerebri (Fig. 1). The thickened meninges had irregular and poorly marginated borders. Similar, but less well-developed, thickening of the meninges was present overlying the dorsal and ventral aspects of the right cerebral hemisphere. In comparison to the cerebral gray matter, the thickened meninges were hyperintense on T2W, T2*W, and T2W FLAIR images and iso- to hypointense on T1W FLAIR images. After intravenous contrast administration, the meninges displayed heterogeneous enhancement. In addition to the meningeal thickening, there was marked enlargement of the white matter of the left cerebral hemisphere resulting in compression of the left lateral ventricle, evidenced by severe deviation of the falx cerebri and ventral longitudinal fissure to the right. There also was falcine herniation of the left cingulate gyrus, caudal tentorial herniation of the medial aspects of the occipital and temporal lobes, and herniation of the cerebellar vermis through the foramen magnum.
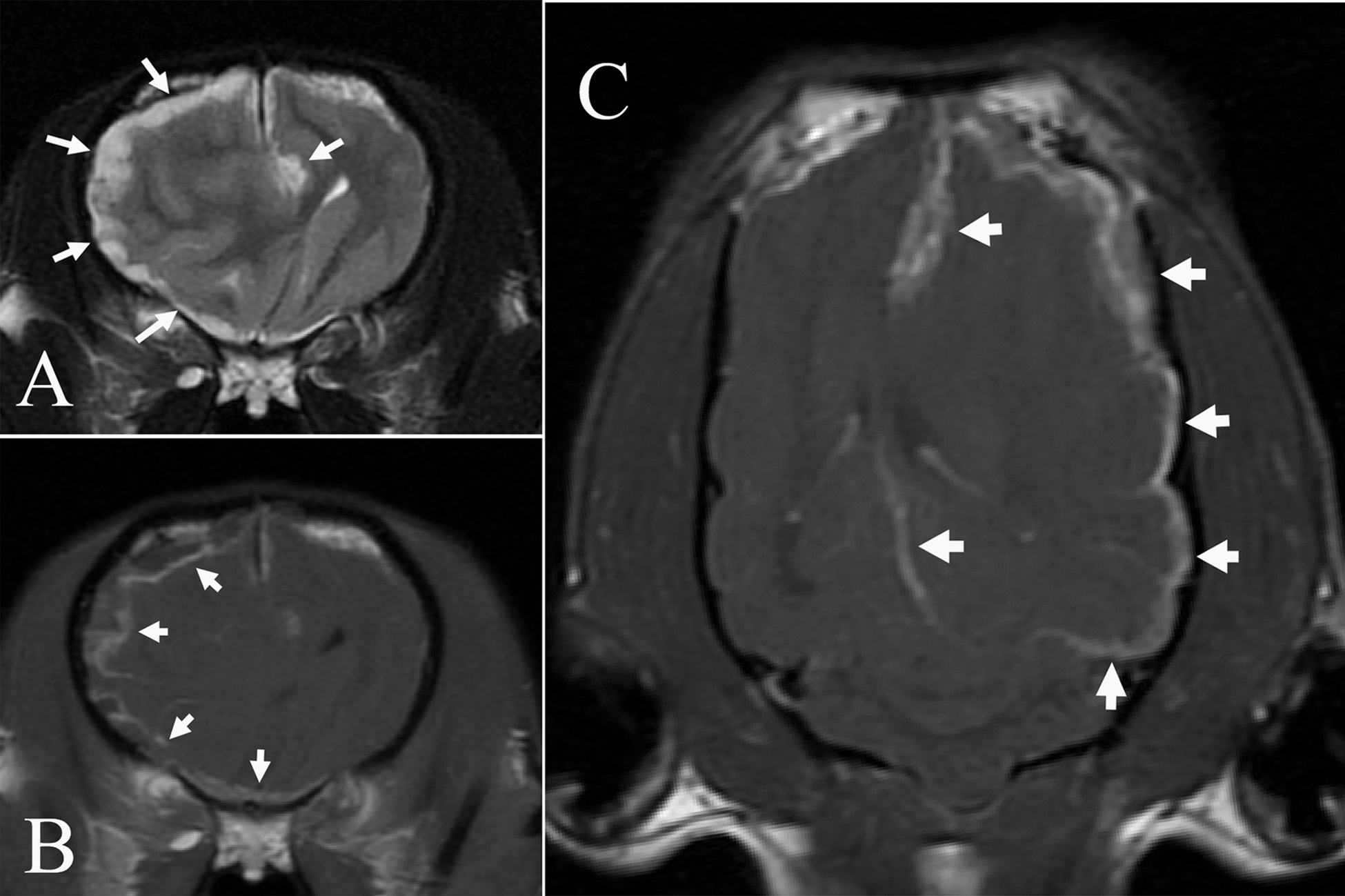
Magnetic resonance images obtained from the brain of a Chihuahua with a meningeal granular cell tumor.
Analysis of cerebrospinal fluid obtained from the cerebellomedullary cistern revealed albuminocytologic dissociation; the nucleated cell count was 5 cells/µl (ref. interval: 0–5 cells/µl) with a protein content of 50.6 mg/dl (ref. interval: <24 mg/dl). Serum latex cryptococcal antigen agglutination test was negative. Initially, the dog was treated with mannitol and prednisone. After 48 hr, cytosine arabinoside and mycophenolate was administered in addition to prednisone. Over the course of 5 days, the dog displayed minimal improvement in the neurological deficits. Based on the lack of response to therapy and guarded prognosis, the dog was euthanized at the owner’s request and submitted for complete necropsy to the Athens Veterinary Diagnostic Laboratory, University of Georgia (Athens, Georgia).
On gross and microscopic examination, abnormalities were limited to the meninges covering the brain and cranial cervical spinal cord, and underlying neuroparenchyma of the brain. The meninges covering the left cerebral hemisphere were white, firm, and thickened, and firmly adhered to the calvaria (Fig. 2A). The underlying cerebral gyri showed no gross abnormalities other than compression. Ventrally, the thickened meninges were also adhered to the sphenoid and occipital bones. On transverse section, the white matter of the left cerebral hemisphere was swollen, and the cerebrum was displaced to the right, compressing the right cerebral hemisphere (Fig. 2B). The occipital bone covering the cerebellum was thin, translucent, and fragile. A caudal portion of the vermis and the underlying medulla were herniated through the foramen magnum.
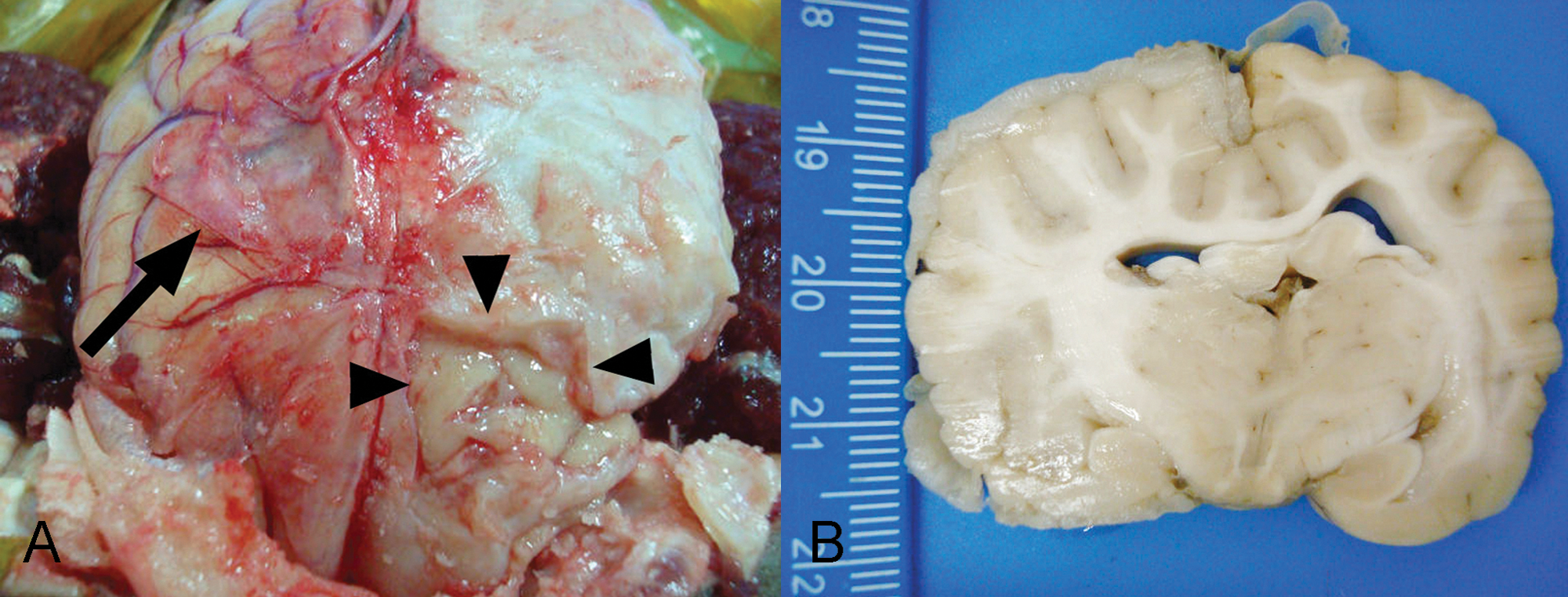
Brain, dog.
Ultimately, the neurological deficits were explained by the MRI and gross findings at necropsy. Clinically, the abnormal mental state, lack of menace response, and blindness in the right eye can be explained by the abnormalities involving the white matter of the left cerebrum observed on MRI. Specifically, such pathology would likely disrupt the left optic tract and optic radiations resulting in contralateral blindness evidenced by a lack of menace response. It is likely that the cervical pain reflected the meningeal involvement of the cervical spinal cord. However, the anisocoria remains difficult to reconcile.
Multiple impression smears were taken from the cut surface of the meninges and stained with Wright stain. The brain and spinal cord were fixed in 10% neutral buffered formalin for histopathology and immunohistochemistry. Histochemical stains for formalin-fixed tissues included hematoxylin and eosin, periodic acid–Schiff (PAS) with and without diastase, and oil red O. Immunohistochemical staining was performed using primary antibodies to vimentin, c pancytokeratin, c cytokeratin AE1/AE3, c glial fibrillary acidic protein (GFAP), c E-cadherin, d S100, e lysozyme, e and neuron-specific enolase (NSE). f To detect the primary antigens, specific biotinylated horse anti-mouse immunoglobulin G (IgG; vimentin, pancytokeratin, cytokeratins AE1 and AE3, GFAP, NSE, E-cadherin) g or goat anti-rabbit IgG (S100, lysozyme) g labeled with streptavidin–horseradish peroxidase were used and developed with diaminobenzidine chromogen. Appropriate positive control tissues were included in all staining procedures. Additional meningeal samples were fixed in 2.5% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4), for evaluation by transmission electron microscopy.
Cytologic evaluation of the impression smear taken from the cut surface of the meninges showed moderately to densely cellular specimens in a background of amphophilic amorphous material. There were numerous large, neoplastic, spindloid to polygonal cells, either present in clusters or as individual cells admixed with erythrocytes and lymphocytes in the background. (Fig. 3A). These neoplastic cells had distinct cytoplasmic borders and contained abundant amphophilic, granular cytoplasm, with round, central to peripheral nuclei, and 1 distinct magenta nucleolus. Anisocytosis was marked, and anisokaryosis was moderate. Mitotic figures were not observed. Additionally, the neoplastic cells did not stain using oil red O for lipid, and PAS for glycogen.
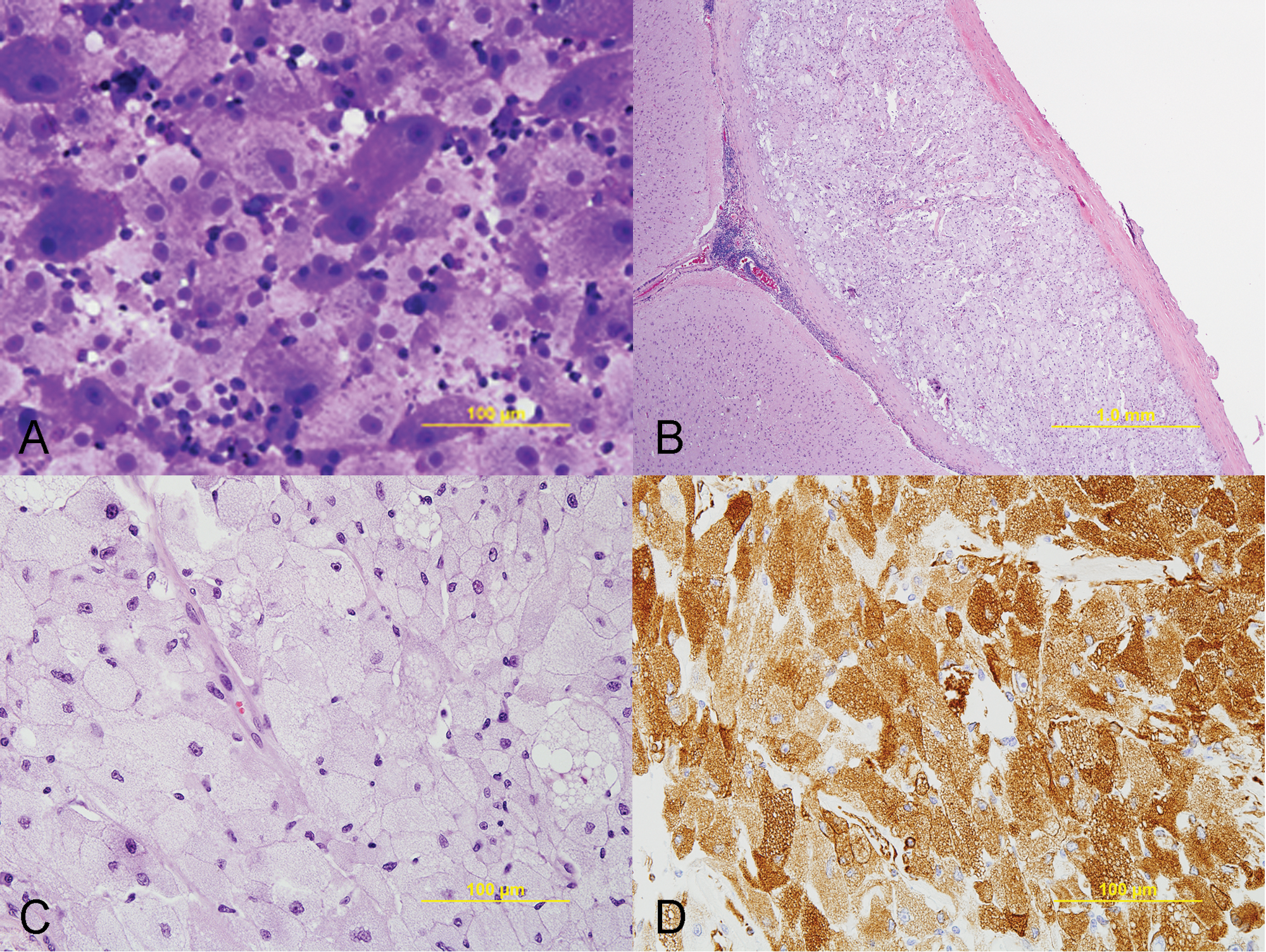
Meninges, dog.
Histological features of the neoplastic cells were similar to cytologic findings. The leptomeninges were expanded by a densely cellular, poorly circumscribed, unencapsulated, and infiltrative neoplasm composed of spindloid to polygonal cells arranged in sheets (Fig. 3B). The neoplastic infiltrate covered the gyri and extended down into the sulci resulting in compression of the underlying cerebral cortex. In addition to the cerebral hemisphere, neoplastic cells were also observed in the meninges overlying the C1 to C2 spinal cord segments.
Superficially, the neoplastic cells were surrounded by thick bundles of connective tissue consistent with dura mater. The cells were also occasionally separated by moderately thick bands of connective tissue and supported by a fine fibrovascular stroma. The neoplastic cells had distinct cytoplasmic borders, with densely packed, vacuolated, amphophilic cytoplasm, and 1 distinct nucleus, often peripherally located, with finely stippled chromatin and 1 prominent magenta nucleolus (Fig. 3C). The more spindloid neoplastic population was more closely associated with the dura mater and gradually transitioned to the more polygonal, vacuolated population. There was marked anisocytosis and anisokaryosis, with neoplastic cells ranging from 10 to 60 µm in diameter and a nucleus to cytoplasmic ratio of 1:1 to 1:50. Mitotic figures were not observed. Within the neoplasm, there also were scattered aggregates of lymphocytes and plasma cells.
In the cerebrum adjacent to the neoplasm, there were multifocal areas of perivascular edema in the cortex. In the white matter, the myelin sheaths were often dilated. In both the gray and white matter, there were moderately increased numbers of reactive astrocytes (gliosis). In the olfactory lobe, the white matter was multifocally rarefied, with moderate accumulations of foamy macrophages (gitter cells). The lateral and the third ventricles were moderately dilated and lined by attenuated ependymal cells.
Histochemically, the neoplastic cells did not stain using oil red O and PAS (with and without diastase). Immunohisto-chemically, the neoplastic cells were diffusely positive for vimentin (Fig. 3D), but did not express S100, pancytokeratin, cytokeratin AE1/AE3, E-cadherin, lysozyme, or GFAP. Some of the neoplastic cells were mildly positive for NSE. Ultrastructurally, the cytoplasm of the neoplastic cells contained numerous membrane-bound vesicles filled with variable amounts of variably electron-dense, irregular granules, including residual bodies (Fig. 4). Moreover, numerous empty vesicles, multivesicular bodies, and membranous myelin-like whorls were observed. Mitochondria were dilated with marked loss of cristae. The peripherally located nucleus was indented as a result of compression by autophagic vacuoles. Based on the cytological, histomorphological, and ultrastructural observations, a diagnosis of an atypical meningeal GCT was made.
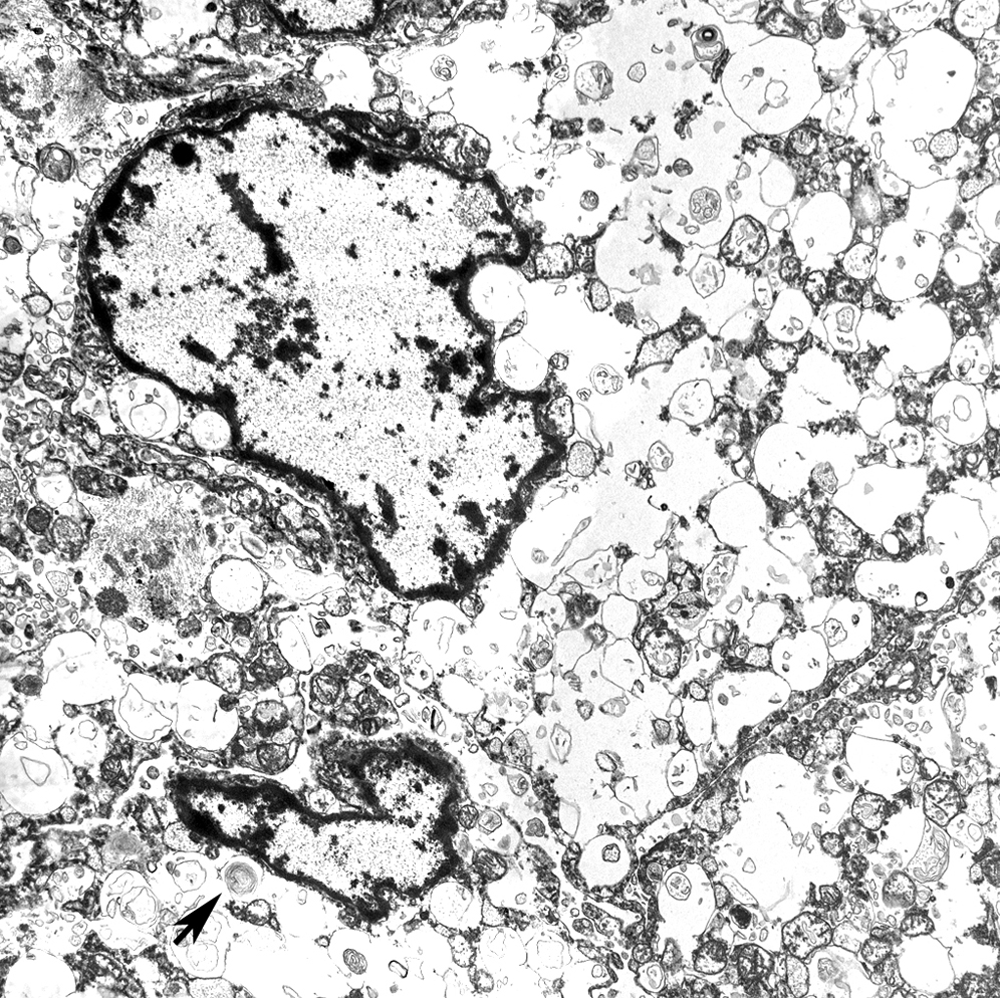
Electron microscopy, leptomeninges, dog. The cytoplasm of the neoplastic cells contains numerous, distended, membrane-bound vacuoles filled with variably electron-dense, organelle debris, including membranous whorls (arrow).
Typically, the diagnosis of GCT is based on histomorphological, cytochemical, immunohistochemical, and ultrastructural features, with the proposed cell of origin being from the corresponding anatomic site of the tumor. Yet, the histogenesis of GCTs remains controversial due to different proposed cellular origins, even though GCTs from different anatomic sites share similar histomorphologic features.1,2,7 In human beings, most intracranial GCTs occur in the neurohypophysis and are thought to be derived from pituicytes.11,14 Associated with cranial nerves, intracranial GCTs of Schwann cell origin have been reported and are characterized by the presence of basal lamina, frequent proximity of the lesion to unmyelinated nerve fibers, and positive staining with S100 antibodies.3,17 Although uncommon, GCTs located within the cerebrum are thought to have an astrocytic origin based upon immunohistochemistry.1,3,7,17 In addition, oligodendrogliomas may also display histomorphologic features of GCTs.4,27 Ultrastructural oligodendrogliomas with granular cell changes consist of neuritic structures composed of stubby cytoplasmic processes containing many microtubules of 20 nm in diameter and neurosecretory granules. 27 Rarely, GCTs may arise in the meninges in human beings. 26
Like human beings, GCTs in animals arise in both non-neural and neural tissues. 19 In rats, GCTs have been reported in the meninges surrounding the brain and, based upon immunohistochemical and ultrastructural studies, the progenitor of meningeal GCTs in rodents is considered to be meningothelial arachnoid cells. 29 Canine cerebral GCTs have been reported to be derived from meningeal, mesenchymal, or neuronal cells.10,12,22,26 Despite these reports, the cell of origin of meningeal GCT in dogs is still controversial, with conclusions based primarily on exclusionary rather than confirmatory data. Based on immunohistochemistry, the identity of the origin of the neoplastic cells in the present case could not be determined; however, strong binding of vimentin antibody supports a mesenchymal origin.
Given the anatomic location of the tumor, the diagnosis of meningioma was considered. In human beings, the meningioma variant, vacuolated meningioma, is histologically similar to GCT. 8 However, given the lack of positive staining for E-cadherin and S100, combined with a lack of intercellular bridging, which is a hallmark of meningiomas,20,29 a diagnosis of meningioma was unlikely. Similarly, negative results for GFAP staining and the anatomic location excluded an astroglial origin. Likewise, uniformly negative staining with NSE and S100 antibody was unsupportive of a neuronal origin. Finally, the lack of lysosome staining excluded a histiocytic origin. In the case presented herein, the single disparate finding from those typical of GCT was a lack of histochemical positive staining with PAS. In the end, the neoplasm in the current case did not show glial, histiocytic, or neuronal cell characteristics. Ultimately, based upon the gross anatomic location along with the cytologic and histomorphologic observations, combined with exclusion of other differential diagnoses, a diagnosis of an atypical meningeal GCT is warranted in the present case despite the absence of PAS, NSE, or S100 positivity.
Footnotes
Acknowledgements
The authors thank Dr. Elizabeth Howerth for editing this manuscript, Drs. Summers and deLahunta for their assistance in evaluating the histopathologic specimens, and Abbie Butler for performing the immunohistochemistry.
Notes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
