Abstract
A juvenile female pig found dead was submitted for necropsy. A focal meningeal red mass measuring 3.5 cm in diameter was located over the left olfactory bulb. Histologically, the mass was composed of polygonal-to-spindle–shaped cells forming vascular clefts with focal invasion of the neuropil of the olfactory bulb. Immunohistochemistry revealed strong expression of vimentin, and a variable labeling of factor VIII–related antigen, smooth muscle actin, and neurotrophin receptor p75. The final diagnosis was a primary meningeal hemangiosarcoma.
Hemangiosarcomas represent a malignant transformation of endothelial cells forming blood-filled vascular clefts that may be accompanied by more solid areas resembling fibrosarcomas and other poorly differentiated sarcomas. 11 Local tissue invasion and distant metastases are common. Hemangiosarcomas have been reported most frequently in dogs but occur rarely in horses, cattle, cats, goats, and sheep. 3,5,6,12,16 In pigs, hemangiosarcoma has been observed in the skin and the testis, respectively, of only 2 individuals in a case study of 31 spontaneous neoplasms. 9 In another report, a hemangiosarcoma was suspected in the spleen of a 10-year-old potbellied pig; however, the diagnosis was not confirmed by histopathology. 14
Indogs, typical primary sites of origin of hemangiosarcoma include spleen, liver, and right atrium. These primary neoplasms are commonly associated with metastases to various tissues and organs 11 including the central nervous system (CNS) in dogs. 10 Only a single case of a primary hemangiosarcoma of the CNS in a juvenile dog has been published. 10 In other domestic species, hemangiosarcoma with metastases to the brain has been observed in a horse 12 and a bull (P. Wohlsein, personal observation, 1996). In human beings, primary intracranial vascular neoplasms are rare. 13
In pigs, neoplastic disorders are rarely encountered. 2,19 The paucity of reported tumors in pigs can in part be explained by the young age of these animals when they are slaughtered. 2,19 In contrast to other domestic species like dogs and horses, the majority of pigs are slaughtered as juveniles, an age that is statistically characterized by a lower tumor incidence. There are few surveys on tumors of domestic pigs, but a striking overrepresentation of hematopoietic tumors exists. 2,19 Only 2 cases of CNS neoplasms have been reported in 1 study, 9 including a glioblastoma and a meningeal hemangioma diagnosed by biopsy in 2 different feeder pigs. However, neither tumor was described in detail. The aim of the present study is to report the morphologic and immunohistologic findings of a primary meningeal hemangiosarcoma in a pig.
A female grower pig weighing 33 kg was found dead and submitted for necropsy. Anamnestically, the animal did not exhibit any clinical signs, and neurologic symptoms were not observed. At necropsy, the pig was in a moderate nutritional condition and showed bilateral ear tip necrosis. The lung had severe generalized pulmonary edema with multifocal interstitial hemorrhages and a moderate suppurative pneumonia. Virologic examination by means of conventional polymerase chain reaction (PCR) demonstrated
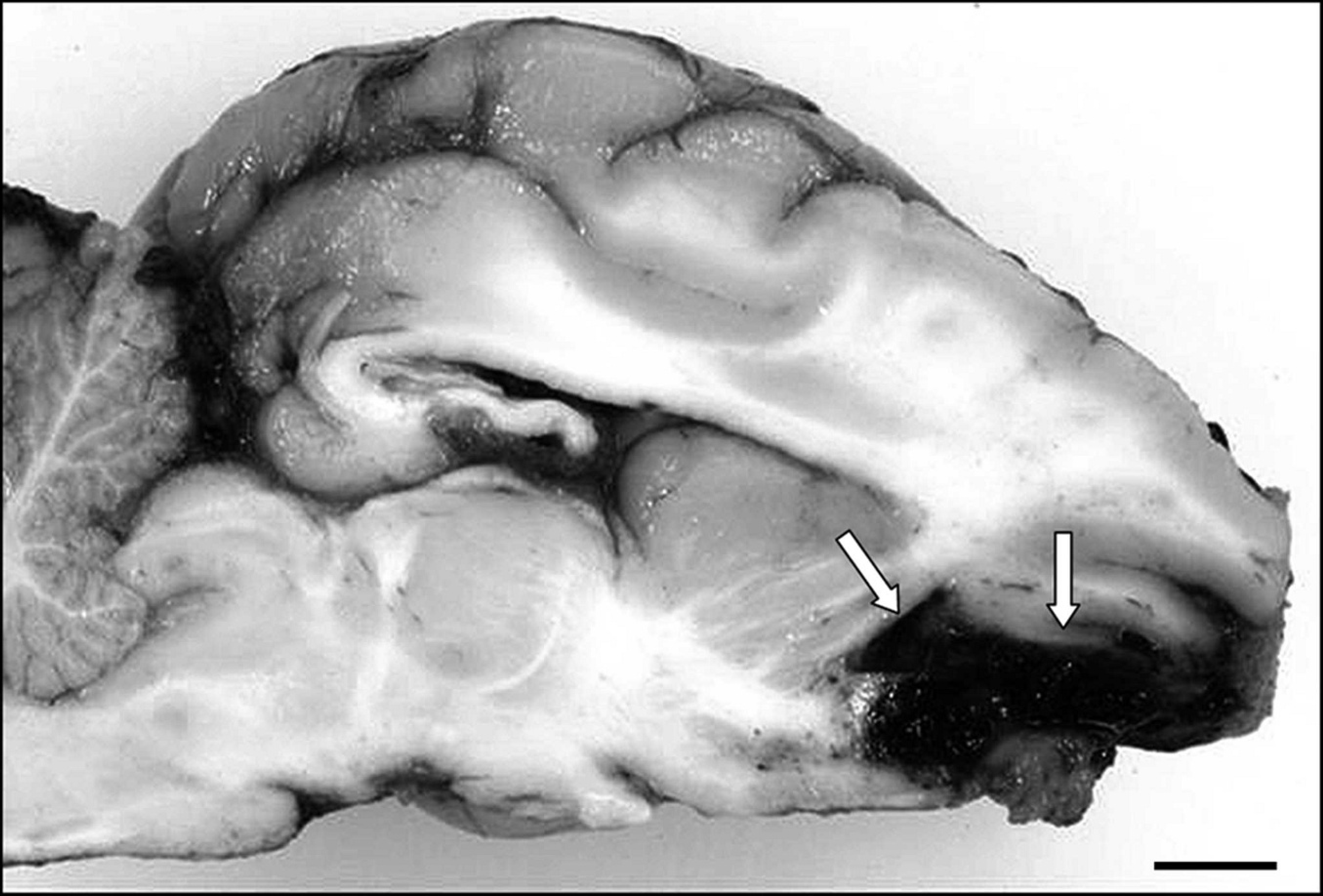
Brain of a juvenile pig with a dark-red friable mass at the meningeal surface of the left olfactory bulb (arrows). Bar = 1 cm.
Samples were collected from various organs and tissues including the brain, lymph nodes, and lung; fixed in 10% neutral buffered formalin; and processed by routine methods (i.e., embedded in paraffin wax, sectioned at 5 μm, and stained with hematoxylin and eosin). Reticulin fibers were demonstrated by Gomori silver staining. 15 Immunohistochemistry was performed using the avidin–biotin–peroxidase method, a according to the manufacturer's instructions, and various monoclonal and polyclonal antibodies (Table 1) as described previously. 7 Briefly, after dewaxing, tissue sections were immersed in H2O2 0.5% in methanol for 20 min. Nonspecific primary antibody binding was blocked by incubation with inactivated goat serum diluted 1:5 in phosphate buffered saline (PBS; pH 7.1). Then, the primary antibody diluted in PBS was applied, and the tissue sections were incubated in a moist chamber at 4°C overnight. After washing, tissue sections were incubated with a biotin-labeled goat–anti-mouse or goat–anti-rabbit immunoglobulin G. a The chromogen solution consisted of 0.05% 3,3′-diaminobenzidine tetrahydrochloride b with 0.03% H2O2 as substrate in 0.1 M Tris buffered saline c (pH 7.6). Tissue sections were counter-stained with Mayer hematoxylin, coverslipped, and examined microscopically. For negative control purposes, the primary antibody was replaced by ascitic fluid from nonimmunized BALB/cJ mice d or rabbit serum. b As positive controls, various tissues were used as specified in Table 1.
Histologic examination revealed a neoplastic growth in the leptomeninges of the olfactory bulb with focal invasion of the brain, predominantly associated with the leptomeningeal surroundings of preexisting blood vessels (Fig. 2). The neoplasm was composed of numerous, irregularly shaped, mostly ectatic, thin-walled vascular channels lined by pleomorphic polygonal-to-spindle–shaped tumor cells. Individual neoplastic cells had a small amount of homogenous eosinophilic cytoplasm and a round-to-oval, predominantly eccentrically located nucleus (Fig. 3). Mitotic figures were rare. Some tumor cells showed clumping and condensation of chromatin indicative of apoptosis. Approximately 15% of the tumor showed multifocal-to-coalescing areas of necrosis. In addition, confluent hemorrhages were present with infiltration of hemosiderophages. Gomori silver stain demonstrated a predominance of smooth, immature, reticulin fibers emanating from neo-plastic vascular clefts.
Primary antibodies, clonality, dilution, and immunohistochemical staining results of the meningeal tumor in the pig in the current study.
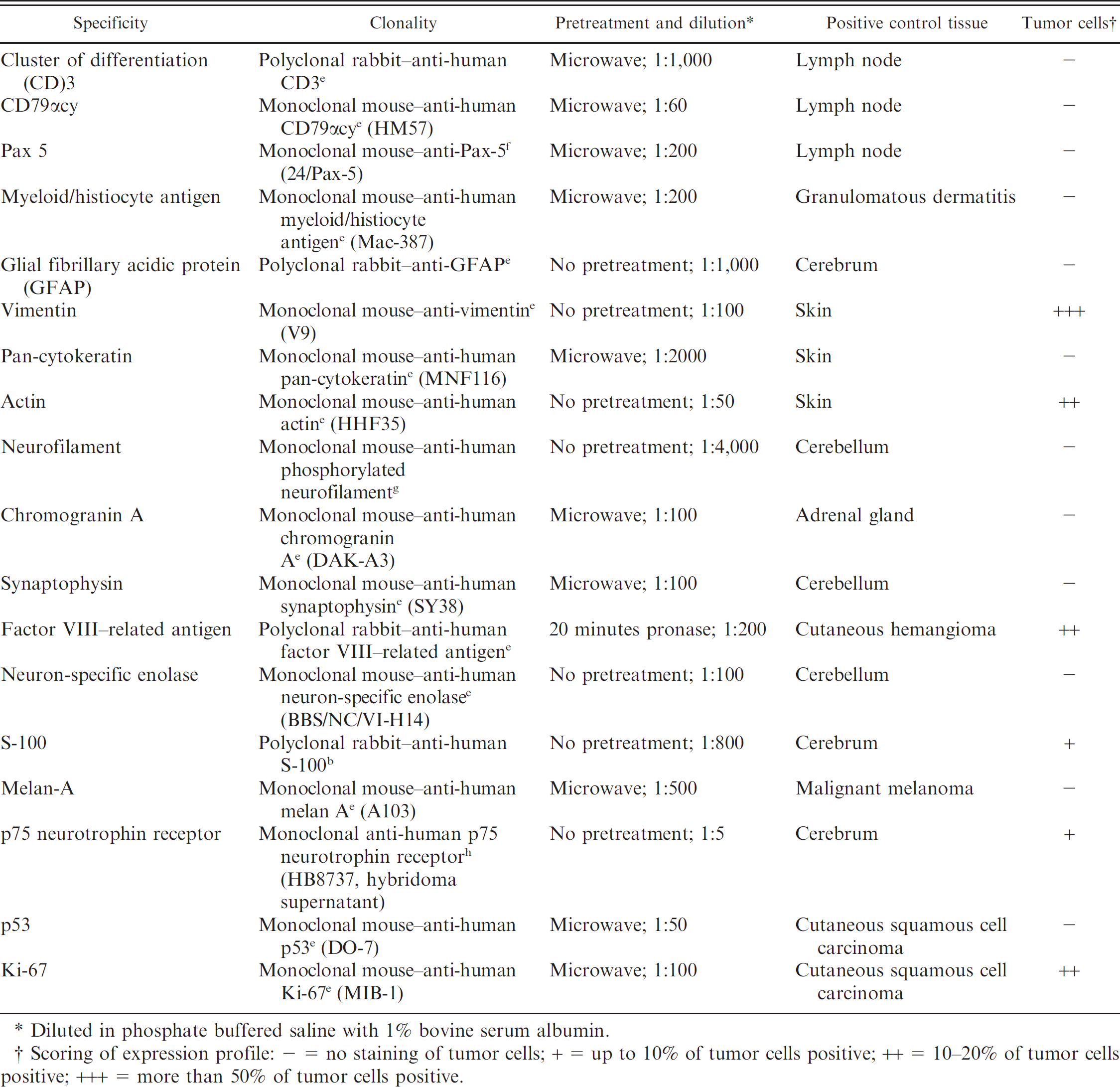
Diluted in phosphate buffered saline with 1% bovine serum albumin.
Scoring of expression profile: − = no staining of tumor cells; + = up to 10% of tumor cells positive; ++ = 10–20% of tumor cells positive; +++ = more than 50% of tumor cells positive.
The results of immunohistochemical staining are summarized in Table 1. The tumor cells had extensive expression of vimentin. Factor VIII–related antigen (FVIIIrA) was expressed variably, and smooth muscle actin was found in single tumor cells. Labeling of the neurotrophin receptor p75 was observed in approximately 30% of the tumor cells. Using Ki-67 antibody, the proliferation index of the tumor cells was approximately 20%. The remaining markers for neuroepithelial, epithelial, and hematopoietic cells were negative. The immunohistologic profile of the tumor cells was most compatible with a mesenchymal neoplasm of vascular origin, leading to a final diagnosis of primary meningeal hemangiosarcoma.
To the authors' knowledge, the present case represents the first report of a primary intracranial hemangiosarcoma in a pig, and moreover, the first detailed report of a tumor affecting the CNS of a pig in general. The pig was found dead without exhibiting observable neurologic symptoms. Pathologic investigation revealed pneumonia with bacterial infection that most likely was responsible for the death of this animal and suggested that the meningeal tumor represented an incidental finding.
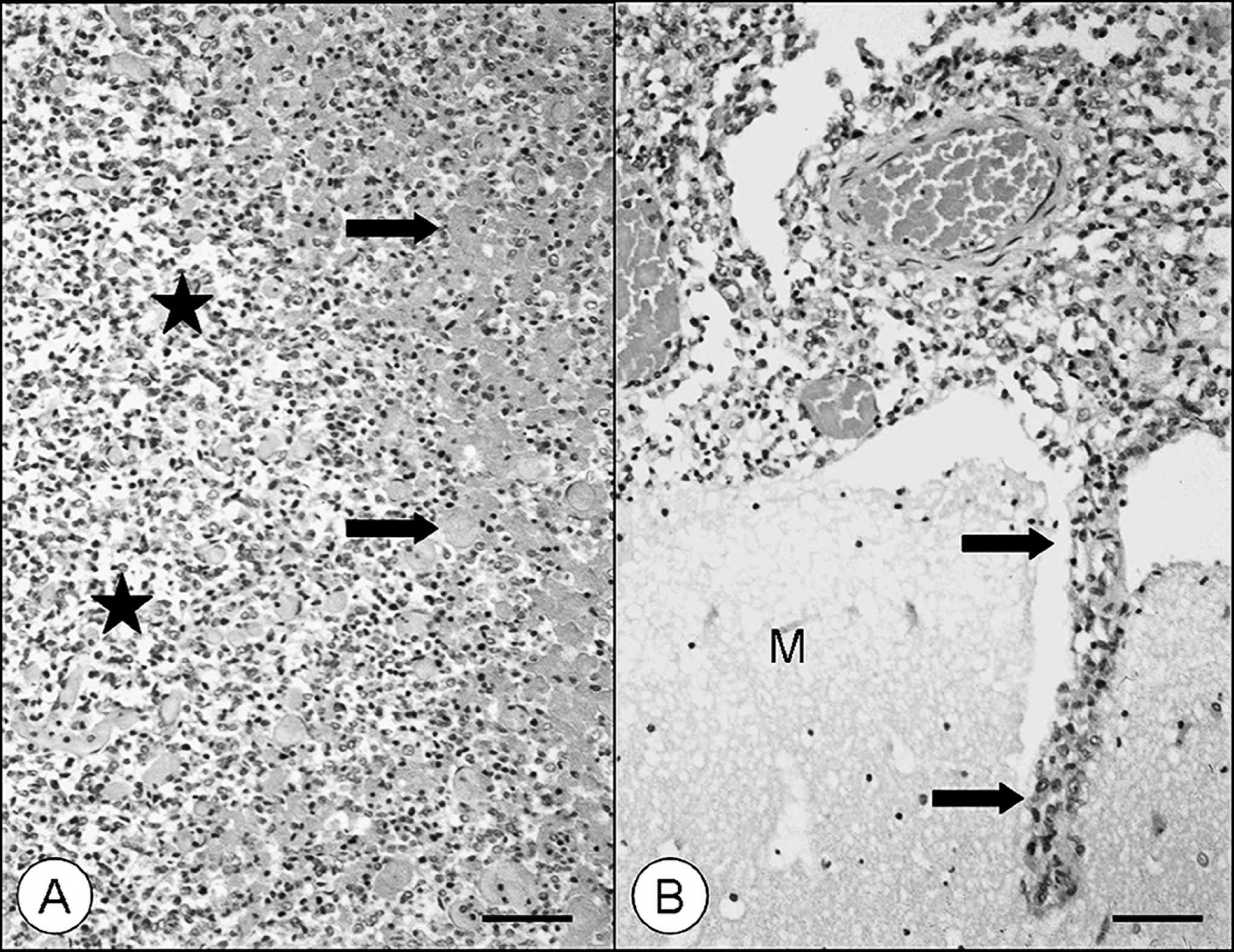
Meningeal hemangiosarcoma in a pig.
The histomorphologic appearance of the tumor was characterized by vascular channels with massive hemorrhages. With regard to differential diagnoses of primary vascular neoplasms in the CNS, several tumors have to be considered. In human beings, hemangioblastoma represents a rare primary vascular neoplasm of the brain affecting predominantly cerebellum, brain stem, and spinal cord of children and adolescents. 1 However, this differential diagnosis appears unlikely because characteristic lipid-laden stromal cells admixed with capillary-forming neoplastic cells were not detected in the present neoplasm. The tumor also showed distinct morphologic characteristics of malignancy, including areas of necrosis and infiltrative growth into the brain that distinguished it from potential vascular malformations and benign vascular neoplasms such as hamartomas and hemangiomas, respectively. A meningeal cavernous hemangioma was recorded in a case study of porcine neoplasms; however, gross and histologic descriptions of this neoplasm were lacking. 9 Moreover, the relatively high proliferative index of the present neoplasm demonstrated by immunohistochemistry is interpreted as additional evidence of malignancy. Although tumors are rare findings in pigs, malignant blastomas are much more common than benign ones. 19 Hemangiosarcomas represent common malignant neoplasms of endothelial origin that can metastasize to the brain with an incidence of 14% in dogs. 18 However, primary hemangiosarcoma of the brain represents a rare tumor in veterinary medicine and has only been reported in 1 juvenile dog with origin in the right cerebral hemisphere. 10 In the present case, no evidence of neoplastic growth was detected in other organs and tissues, excluding a diagnosis of metastasis.
With regard to the expression of mesenchymal (vimentin) and vascular markers (FVIIIrA) in association with smooth muscle actin expression, the histologic diagnosis is most consistent with a primary meningeal hemangiosarcoma. The reported immunohistochemical phenotype of hemangiosarcomas comprises a variable expression of FVIIIrA, which is consistent with the findings of the present case. 5,17 The expression of smooth muscle actin, representing the contractile ability of neoplastic endothelial cells, is similar to the findings of others. 17 To the author's knowledge, this is the first report of neurotrophin receptor p75 expression in a hemangiosarcoma affecting an animal. Neurotrophin receptor p75 belongs to the tumor necrosis factor-receptor superfamily and is expressed by neural and non-neural cells. An induction of neurotrophin receptor p75 expression in vascular cells under pathological conditions has been reported (for review of neurotrophins and their receptors, see Caporali and Emanueli) 4 . Moreover, neurotrophin receptor p75 expression has been documented in a variety of non-neural mesenchymal tumors including hemangioendothelioma and angiosarcoma in human beings. 8 The lack of p53 staining is consistent with reports of hemangiosarcomas in dogs and cattle in which the majority of the investigated tumors did not express p53. 5,20 In a recent study on p53 expression in dogs, an accumulation of p53 was found in 17.9% of the investigated tumors. 20 However, the study failed to prove a mutation of the p53 gene, leading to the conclusion that inactivation of the p53 pathway might be involved in the pathogenesis of canine hemangiosarcoma to some extent. 20 In cattle, p53 expression in hemangiosarcomas of the urinary bladder is restricted to neoplasms with muscular invasion and is positively correlated to invasiveness. 5 With regard to the present case, the significance of the p53 pathway remains unclear as a single case does not provide enough evidence to draw any conclusions from the lack of p53 immunoreactivity.
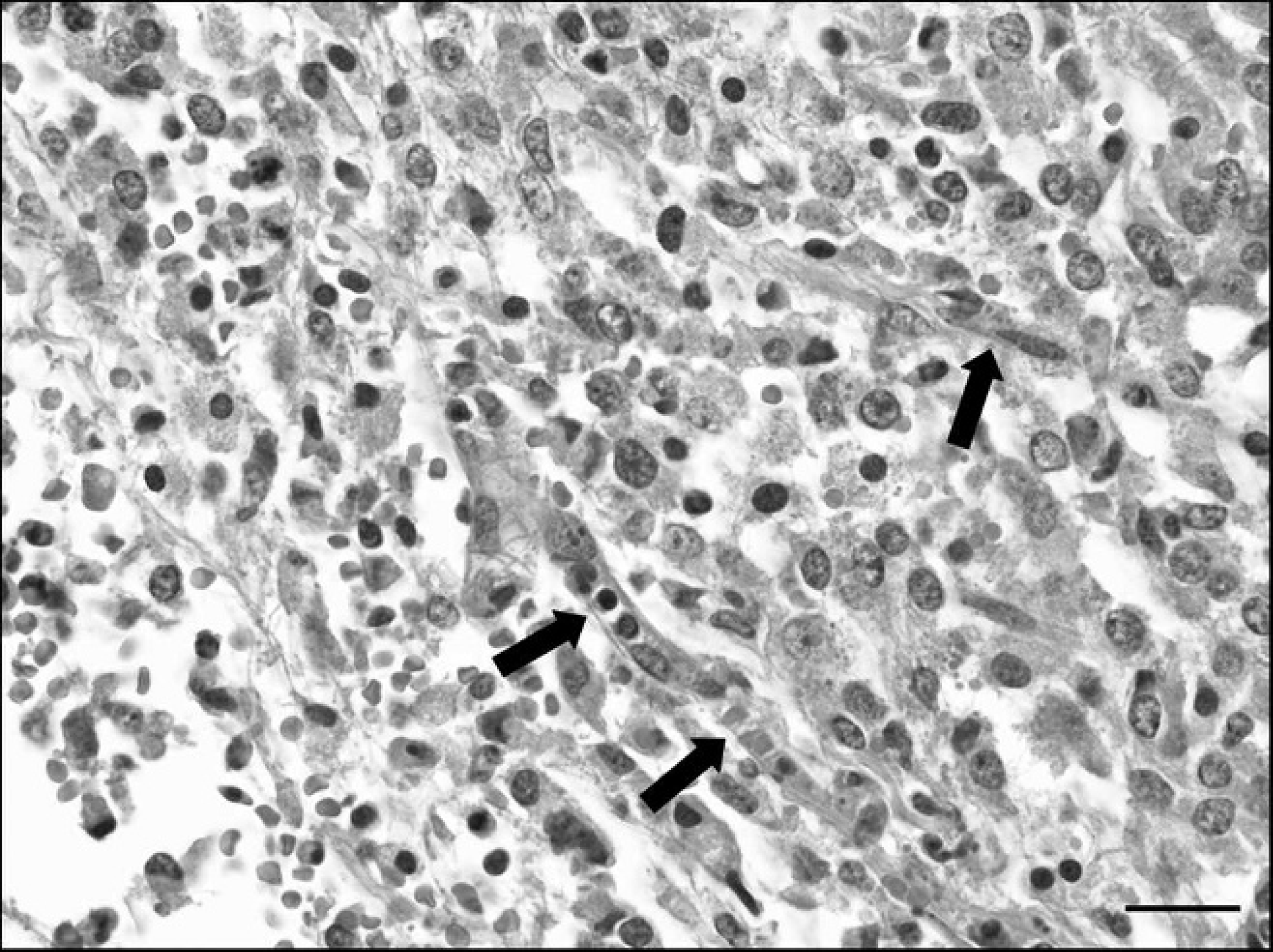
Meningeal hemangiosarcoma in a pig. Pleomorphic tumor cells with oval nuclei and small cytoplasm that are forming vascular clefts (arrows). Hematoxylin and eosin. Bar = 40 μm.
The strikingly rare occurrence of tumors in common domestic pigs may be mainly related to the limited age of most pigs due to slaughtering. 2,9,19 For example, a previous study documented 15.88 tumors per 100,000 slaughtered animals with a relatively higher incidence in animals over 6 months of age. 19 However, similar to human beings, some neoplasms are exclusively seen in younger patients, and, in particular, specific brain tumors have a higher incidence in children and young adults. 13 A previously reported primary intracranial hemangiosarcoma was found in a juvenile dog 10 suggesting that this neoplasm might occur preferentially in young and adolescent animals. The current report shows that a primary hemangiosarcoma should be considered as a rare differential diagnosis of diseases affecting the CNS of young and adolescent domestic pigs.
Footnotes
a.
Vector Laboratories Inc., Burlingame, CA.
b.
Sigma-Aldrich Chemie Gmbh, Munich, Germany.
c.
Merck KGaA, Darmstadt, Germany.
d.
Biologo, Kronshagen, Germany.
e.
Dako Denmark A/S, Glostrup, Denmark.
f.
Becton Dickinson GmbH, Heidelberg, Germany.
g.
Sternberger Monoclonals Inc., Baltimore, MD.
h.
American Type Culture Collection, Manassas, VA.
