Abstract
Intraparenchymal spinal cord tumors in the cat are rarely reported and often as single case reports. In the current study, the clinical, magnetic resonance imaging (MRI), histologic, and immunohistochemical features of 7 cases of intraparenchymal spinal cord tumors in the cat are described. All cats were domestic breed, ranged from 4 to 12 years of age (median 8 years), and included spayed females (5/7) and neutered males (2/7). The duration of clinical signs ranged from 2 weeks to 3 months. MRI revealed lesions that were hyperintense on T2-weighted images with variable contrast enhancement. All 7 tumors had histologic features consistent with glial origin: 3 were astrocytic (gemistocytic or fibrous), and 2 were oligoastrocytic. Single cases of oligodendroglioma and gliomatosis cerebri were also present in the study. Glial fibrillary acidic protein immunoreactivity was robust in the tumors that were predominately astrocytic, and the gliomatosis cerebri case had extensive BLA.36 and Iba1 immunoreactivity. Ki-67 immunoreactivity was variable and most abundant in the case of malignant oligoastrocytoma. The majority of peritumoral lymphocytes were CD3 positive. The current study expands upon the known reports of spinal cord neoplasia in the cat, confirms a caudal cervical segment predilection, and includes a report of gliomatosis cerebri in the spinal cord of a cat.
Introduction
Neoplasia of the spinal cord is typically subdivided based on their location of origin: extradural, intradural–extraparenchymal, or intraparenchymal. Tumors in cats that arise within the intradural space are the most common and include lymphoma, meningioma, and histiocytic sarcoma.2,10,14,18 Intraparenchymal neoplasia is much less commonly recorded in cats and includes tumors that arise from glial cells (or their progenitors), including astrocytes, oligodendrocytes, ependymal cells, and microglial cells. Although the histologic appearance of many oligodendrogliomas and ependymomas are fairly uniform, tumors of astrocytic origin can have a markedly wide spectrum of histologic appearance and include fibrillary, protoplasmic, pilocytic, gemistocytic, astroblastic, and granular cell variants. Additionally, owing to the common cellular origin of some astrocytes and oligodendrocytes, tumors can exhibit differentiation toward both cell lineages.
In the cat, extraparenchymal lymphoma and osteosarcoma are reported to be the most common tumors affecting the spinal cord whereas intraparenchymal neoplasia is uncommon with only brief case reports of astrocytomas and oligodendrogliomas.5,11,19,20 A 2008 retrospective study detailing spinal neoplasia in cats reported low numbers (8/85) of cats affected by intraparenchymal spinal neoplasia. 11 Because of the relative lack of knowledge in regard to intraparenchymal spinal cord tumors in cats, the presentretrospective study was initiated to better describe the clinical presentations, magnetic resonance imaging (MRI) findings, pathologic, and immunohistochemical features of a cohort of cats with intraparenchymal spinal cord neoplasms.
Materials and methods
The pathology database at Cornell University College of Veterinary Medicine (Ithaca, New York) was searched for cases of spinal cord neoplasia in the cat from the period of 1980–2012. All cases were reviewed by 2 board-certified veterinary anatomic pathologists (Miller and Summers). Four of 7 cases were studied at necropsy (Cornell University) while the remainder was submitted by referring board-certified veterinary neurologists. All necropsies were performed within 6 hours of death. Tissues were collected, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 µm, and stained using hematoxylin and eosin.
In 6 out of the 7 cases, immunohistochemical staining (IHC) using an automated platform was performed. a One case did not have enough material to perform IHC (case 3). The antibodies used included: glial fibrillary acidic protein (GFAP b ; 1:5,000 dilution), Ki-67 b (1:50 dilution), BLA.36 c (1:600 dilution), MAC)387 b (1:300 dilution), Pax-5 a (ready to use), cluster of differentiation (CD)45 d (1:100), and CD3 a (ready to use). Detection was achieved through either the polymer refine DAB (3,3’-diaminobenzidine) kit a (CD3, BLA.36, Ki-67, and CD45) or the polymer refine red kit a (GFAP, MAC387, and PAX-5). Negative controls consisted of phosphate buffered saline substitution for the primary antibody. Positive control tissues consisted of lymph node (CD3, BLA.36, PAX-5, MAC387, Ki-67, and CD45) and brain (GFAP). Additional IHC for Iba1 e was performed under the following conditions: formalin-fixed, paraffin-embedded sections were deparaffinized, rehydrated, and subsequently blocked with hydrogen peroxide. Pretreatment involved microwaving for 20 min in 0.01 M citrate buffer followed by 20 min of cooling. All steps were followed by a Tris-buffered saline wash. Prior to application of primary antibodies, all slides were treated with protein block b for 10 min. Sections were incubated with anti-rabbit Iba1 (polyclonal, 1:1,000, 30 min at room temperature). Slides were then incubated with secondary antibody biotinylated goat anti-rabbit f (1:200, 30 min at room temperature) followed by 30-min incubation at room temperature with a commercial avidin–biotin–peroxidase complex kit. e The Iba1 slides were developed with DAB chromogen b and counterstained with Mayer hematoxylin. GFAP is an intermediate filament and is used to identify cells of astrocytic origin. Ki-67 is a proliferation marker and can be used to derive the labeling index within a tumor (the number of Ki-67–positive cells divided by total number of cells). BLA.36 is predominately expressed by B-lymphocytes and their progenitors; however, it is also known to recognize dendritic cells including microglia (Miller, personal observation, 2013). 13 MAC387 is expressed predominately on blood monocytes and tissue macrophages. PAX-5 is a transcription factor expressed through B-cell maturation. CD3 is a membrane antigen that stains T-lymphocytes. CD45 is a transmembrane glycoprotein involved in T-cell and B-cell receptor signaling. Iba1 is an ionized calcium-binding adapter molecule that is expressed on microglia.
Results
Clinicopathologic features
Seven cats were included in the present study. Five of the 7 cats were Domestic Shorthair, and 2 were Domestic Longhair. Five of the 7 were spayed females and 2 were neutered males. Ages ranged from 4 to 12 years of age with a median age of 8 years. The duration of clinical signs ranged from 2 weeks to 3 months. Details of signalment, clinical signs, and neuroimaging results are noted in Table 1. Ancillary testing (complete blood cell count, chemistry, urinalysis, cerebrospinal fluid analysis, and serology) was normal except for
Clinicopathologic features of spinal gliomas in 7 cats.*
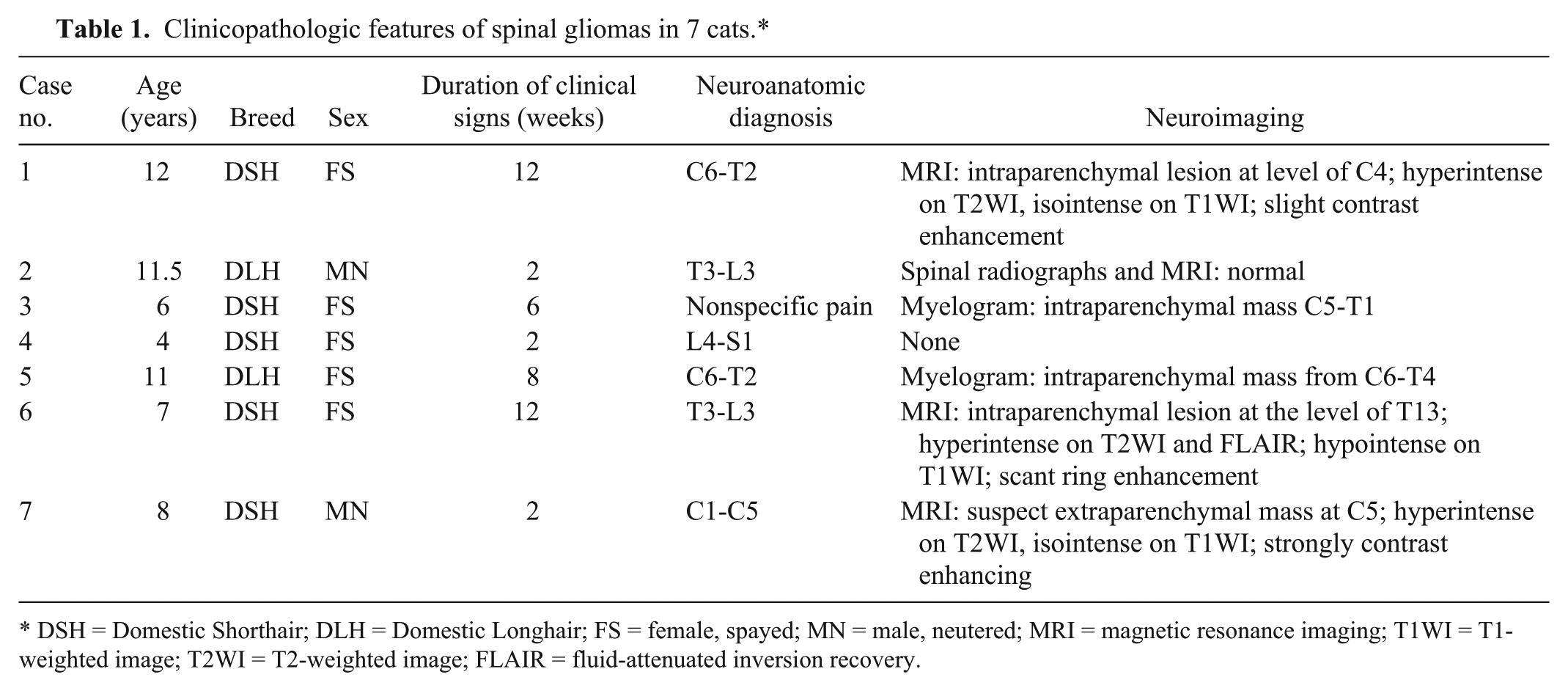
DSH = Domestic Shorthair; DLH = Domestic Longhair; FS = female, spayed; MN = male, neutered; MRI = magnetic resonance imaging; T1WI = T1-weighted image; T2WI = T2-weighted image; FLAIR = fluid-attenuated inversion recovery.
Gross, histologic, and immunohistochemical features
Gross and histologic features are detailed in Table 2. In 3 out of 7 cats, the lesion resulted in a visible, segmental enlargement of the spinal cord. On transverse section, lesions consisted of an intraparenchymal, yellow-tan, expansile mass with no discernible borders with normal parenchyma.
Gross, histologic, and select immunohistochemical features of spinal cord gliomas in the cat.*
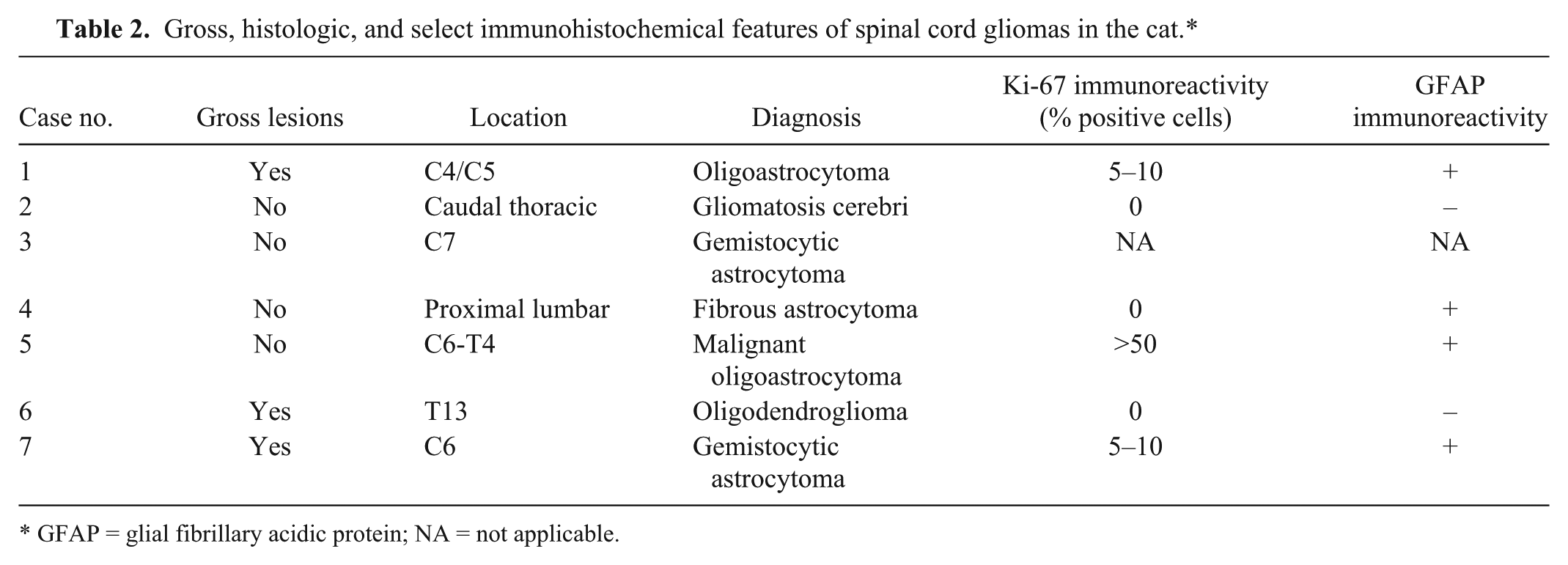
GFAP = glial fibrillary acidic protein; NA = not applicable.
Histologically, all tumors displayed behavior consistent with cells of glial origin. All tumors effaced spinal cord structures; however, no invasion into the subarachnoid space, through the dura, or into nerve roots was noted. Tumors were predominately located in the gray matter in cases 4 and 5, white matter in case 1, mixed gray and white matter in cases 3 and 7, and was diffuse in case 2. Microscopically, the tumors were well demarcated in cases 3, 6, and 7. In the remaining cases, regions of extensive invasion into the adjacent, less-affected white matter resulted in myelin loss with the formation of digestion chambers (ellipsoids) containing invading macrophages with disintegration and loss of axons. Large gemistocytic astrocytes (as part of pure astrocytomas or mixed gliomas) were the predominant astrocyte phenotype in 4 of the 7 animals (cases 1, 3, 5, and 7; Fig. 1A). In these cases, the gemistocytes, with abundant eosinophilic cytoplasm and eccentrically located nuclei, were often arranged in loose aggregates and cords and were interspersed with fine, often fibrillary eosinophilic matrix. There was little cellular and nuclear atypia in this population of cells. Two cases (nos. 1 and 5) had regions of more atypical growth where astrocytes formed ill-defined sheets with occasional pseudorosettes and showed marked cellular and nuclear atypia (Fig. 1B). The mitotic rate in these areas was greater than 3–5 per 400× field. Case 4 was a small mass composed of small fibrous astrocytes that had mild to moderate amounts of eosinophilic cytoplasm and blended seamlessly with the adjacent parenchyma.
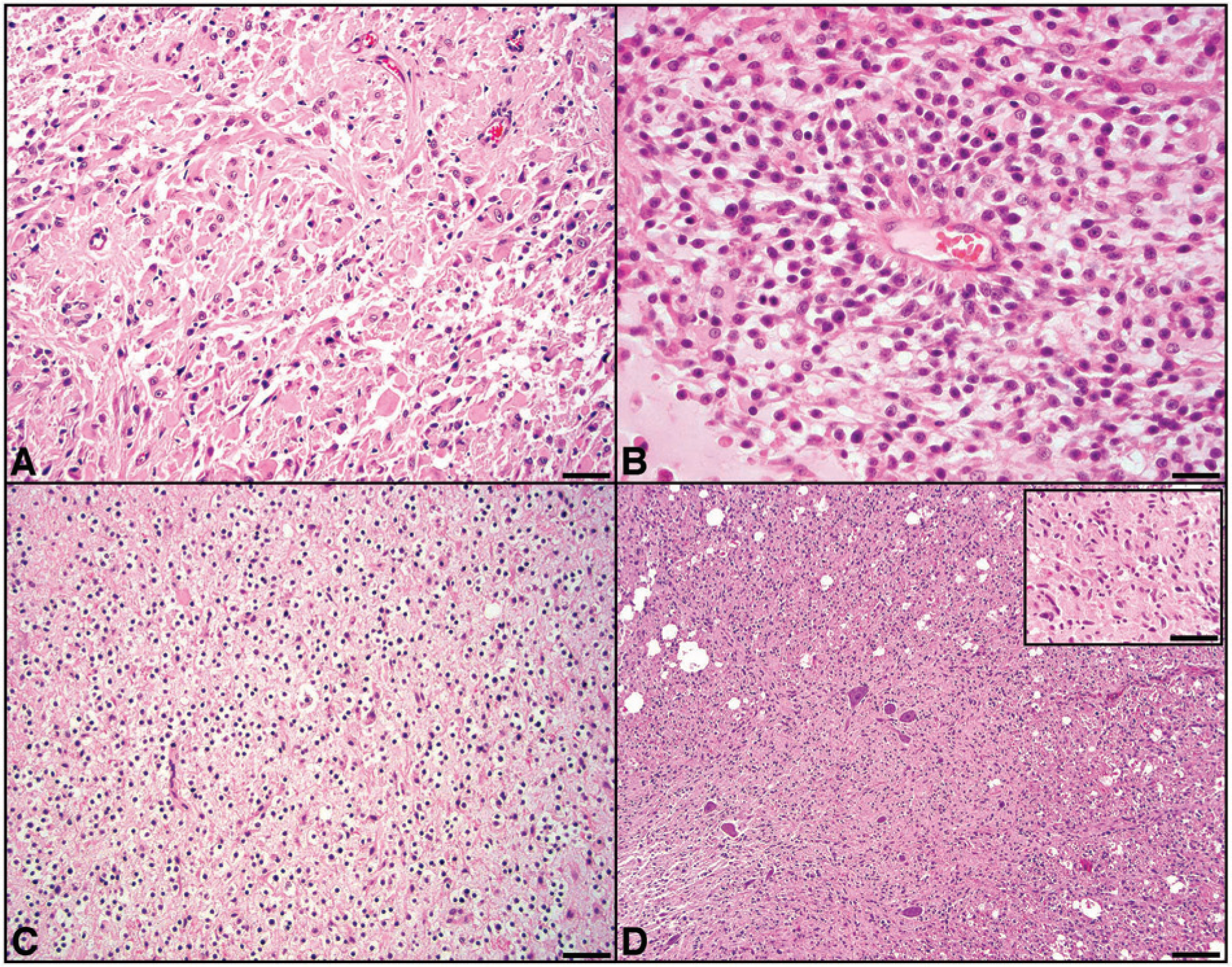
Feline spinal glioma.
One case (no. 6) had diffuse infiltration of the parenchyma by small round cells with hyperchromatic nuclei and inapparent cytoplasm, resembling a “fried egg” appearance, consistent with an oligodendroglioma (Fig. 1C). These neoplastic cells lacked apparent atypia, and no mitoses were noted. Cases 1 and 5 also had small foci of oligodendroglial differentiation comprising roughly 30–40% of the tumor population. The oligodendroglial component of these 2 cases had prominent atypia with 2–4 mitotic figures per 400× field.
Finally, case 2 had extensive and diffuse invasion of the white and gray matter by neoplastic glial cells that had inapparent cytoplasm and elongate to angular nuclei histomorphologically resembling microglial cells (Fig. 1D). These neoplastic cells displayed moderate atypia. There was moderate anisocytosis and anisokaryosis, and the mitotic rate was 1–3 per 400× field. Within some areas of the white matter, neoplastic cells extended to the subpial region. In the gray matter, there were foci of neuronal satellitosis by the neoplastic cells.
Inflammation was present in 6 out of 7 cases (nos. 1–3, 5–7) and consisted of perivascular aggregates of small lymphocytes. The inflammatory foci were typically concentrated around the periphery of the neoplasms; however, scattered foci of intratumoral inflammation were noted. Reactive blood vessels were noted in all cases; however, glomeruloid blood vessels suggestive of malignancy were only noted in case 1.
Immunohistochemically, 4 of 6 cases had immunoreactivity for GFAP. Cases 4 and 7 had diffuse cytoplasmic immunoreactivity consistent with their histologic features of pure astrocytomas (Fig. 2A). As expected, cases 1 and 5 stained uniformly positive for GFAP in astrocytic areas whereas areas of oligodendroglial development displayed either scant or absent immunoreactivity (Fig. 2B). No GFAP immunoreactivity was noted in cases 2 and 6. Ki-67 immunoreactivity was varied (see Table 2) and was most abundant in case 5 (malignant oligoastrocytoma; Fig. 2C). Ki-67 immunoreactivity was scattered in cases 1 and 7 and was not noted in cases 2, 4, and 6. Case 2, which was negative for GFAP and Ki-67, had strong cytoplasmic immunoreactivity for BLA.36 (but negative for PAX-5 and CD45; Fig. 2D). Roughly 75% of the neoplastic cells in case 2 were also strongly positive for Iba1 (Fig. 2D, inset). Tumor-associated inflammation was assessed with CD3, MAC387, and PAX-5. Variable, scattered infiltrates of CD3-positive cells (average 2–5 per 400× field) were noted in and around the tumor in 5 out of 6 cases. Three of 6 cases had rare MAC387-positive macrophages. Only 1 case (no. 7) had a single perivascular cuff of PAX-5–positive lymphocytes.
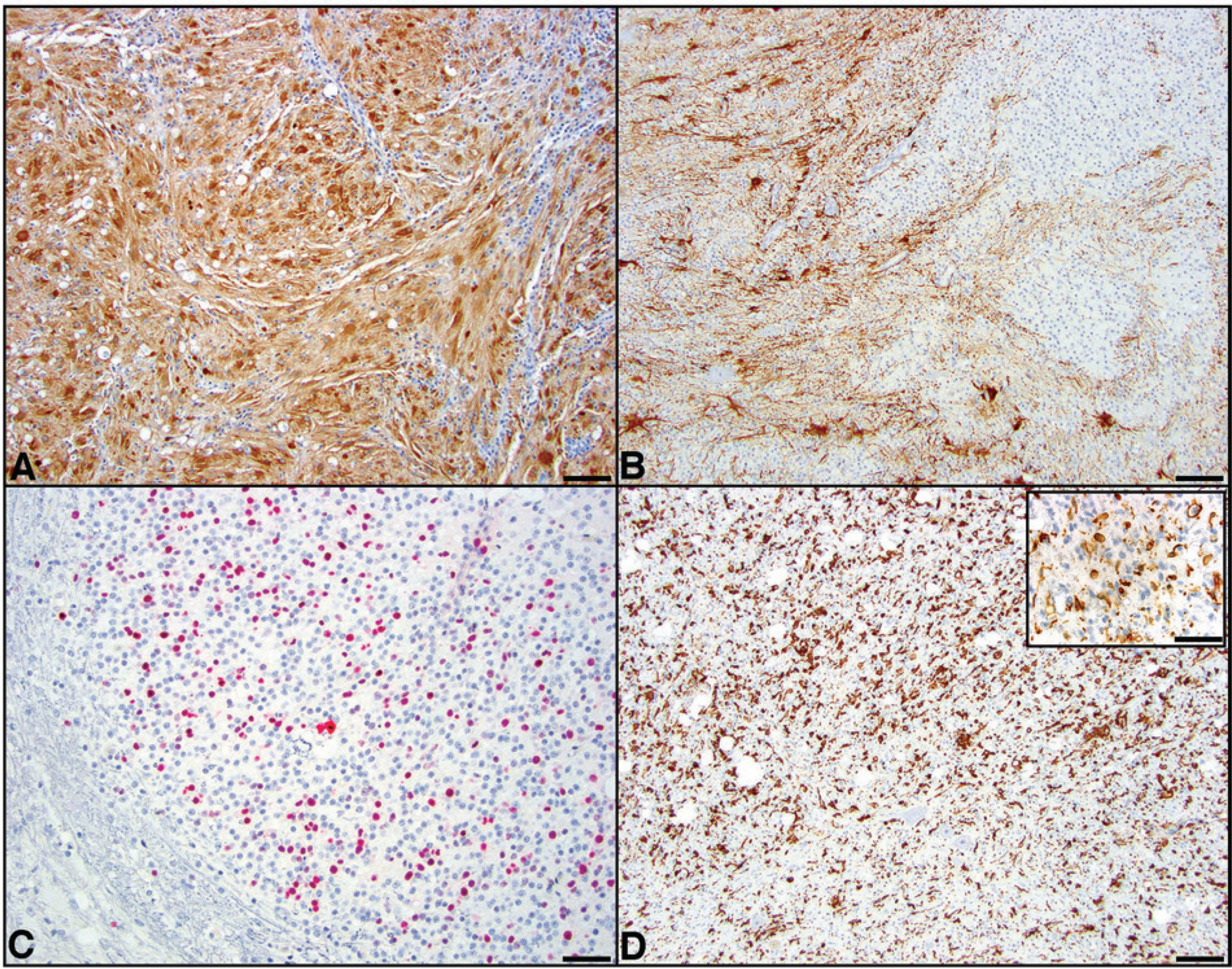
Feline spinal cord glioma.
Discussion
Reports of neoplasms of the feline central nervous system largely describe tumors that arise adjacent to the brain and spinal cord with few reports concentrated on intraparenchymal neoplasia. In a 2008 retrospective report of spinal neoplasia in the cat, lymphoma accounted for almost 39% of neoplasia affecting the spinal cord whereas only approximately 9% of reported tumors were glial in origin. 11 Of the glial tumors in that report, 3 out of 8 were astrocytomas, 2 were ependymomas, and 6 cases were located within the cervical portion of the spinal cord. 11 In the current report, 4 out of the 7 cases involved the caudal cervical spinal cord with the other 3 located in either the thoracic or lumbar regions. Although reported cases are few, there may be an increased likelihood for intraparenchymal spinal cord tumors in the cat to arise in the cervical spinal cord.
In the current report, astrocytic tumors were the most frequently reported. This is consistent with the previous literature. Both fibrous and gemistocytic variants of astrocytoma were noted. Gemistocytes are a common astrocyte phenotype that can predominate in both reparative and neoplastic lesions. Although not relevant for clinical staging, a differential diagnosis for a gemistocytic astrocytoma is the giant cell astrocytoma. 4 Unlike the tumors described herein (and even less common), giant cell astrocytomas arise in a subventricular location and have diffuse distribution of the gemistocytes. In addition, the giant cell variant tends to be more circumscribed rather than blending seamlessly with the adjacent parenchyma as did the gemistocytic variants reported in the current case series.
Three of the cases had features of oligodendroglial differentiation with 2 diagnosed as mixed oligoastrocytoma. Oligoastrocytomas, well recognized in human beings, are thought to arise from a multipotential progenitor cell that undergoes malignant transformation and can develop dually into astrocytic and oligodendrocytic cell lines. 17 Although some degree of dual astrocytic and oligodendrocytic differentiation in glial tumors occurs commonly, the diagnosis of oligoastrocytoma requires at least 30% of the tumor be comprised by one of the neoplastic cell populations. It is thought that these multipotential progenitor cells reside in the subependymal rests and can give rise to neoplasms that have varied histologic appearance based on the section examined. Judging the malignancy of gliomas based on histologic examination can be difficult; however, the presence of glomeruloid blood vessels, increased atypia, and robust Ki-67 immunoreactivity are all suggestive of increased malignancy (and was variably present in cases 1 and 5).15,20
While histologic features remain the most reliable way to accurately diagnose neoplasia of the central nervous system, ancillary testing like IHC can aid greatly in diagnosis. GFAP, an intermediate filament largely of astrocytes and a reliable immunohistochemical marker, was positive in 4 out of 6 cases consistent with the histologic features. Immunohistochemical diagnosis of oligodendrogliomas remains less consistent as markers such as Olig2 can stain both oligodendrocytes and astrocytes and cannot be relied on as the sole criterion for making a diagnosis. 6 Interestingly, in the current report, case 2 had no immunoreactivity for GFAP, but did have strong immunoreactivity for BLA.36. 16 BLA.36 is a leukocyte marker that can be used to stain B-lymphocytes and also is known to stain epidermal dendritic cells. 13 To the authors’ knowledge, BLA.36 has not previously been used to diagnose central nervous system tumors; however, it has strong immunoreactivity with normal feline microglia (data not shown). In addition, the majority of neoplastic cells in case 2 were strongly positive for Iba1 further suggesting microglial origin. This case also failed to display immunoreactivity with either PAX-5 or CD45 indicating that this tumor was likely not of lymphocyte origin. Although the cell of origin in gliomatosis cerebri remains controversial, in most human cases it is believed to be derived from astrocytes; however, based on the microscopic and immunohistochemical features of the case presented herein, this represents an extremely uncommon variant of gliomatosis cerebri derived from microglia.
In the current study, the median age at diagnosis was 8 years (6/7 cases were aged 6 years or older) with the average duration of clinical signs between 2 weeks to 3 months. In 5 of 7 cases, the owners elected euthanasia at time of diagnosis based on the presumed poor long-term prognosis. The 2 patients treated with steroids were euthanized 2 weeks later because of a poor response to therapy. Treatment of intraparenchymal tumors is limited. No known reports exist in the veterinary literature of surgical therapy for spinal glial tumors in the cat. In human medicine, therapy is controversial and includes dural decompression, radiation therapy, and radical excision of the tumors.3,8 Aggressive surgical therapy is rarely considered in veterinary medicine because of the concern of neurologic deterioration postoperatively. However, radical surgical excision has not been shown to cause significant clinical worsening in human beings and thus may be a consideration for veterinary patients with well-defined intraparenchymal tumors found to be well defined with MRI. Outcome after radical excision in human beings depends on tumor grade and neurologic status prior to surgery. In addition, intraoperative monitoring, such as evoked potentials, intraoperative ultrasound, ultrasonic aspirator, and surgical microscopes have been shown to be beneficial in defining the safe extent of resection during surgery in people. Owing to the invasive nature of these neoplasms (observed grossly and microscopically in the cases reported herein), the benefit of surgical resection is likely low. Little is known about the response rate with radiation therapy, chemotherapy, or multimodal therapy, and it is likely that treatment options will vary based on a variety of tumor-related characteristics (i.e., proliferative fraction, degree of invasiveness, etc.).
Magnetic resonance imaging enables localization of spinal cord tumors in relationship to the dura as extradural, intradural-extraparenchymal, or intraparenchymal tumors. An accurate anatomic diagnosis not only provides the clinician the ability to form a list of differentials based on location of the lesion, but also allows for more specific diagnostics and treatment options. MRI has been reported to accurately diagnose the location of spinal cord tumors in human beings. However, limited reports are available on the MRI of spinal cord tumors in dogs and cats.1,9,12,21 Some characteristics of intraparenchymal tumors include enlargement of the spinal cord on multiple planes, thinning of the subarachnoid space at the level of the lesion, and contrast enhancement within the spinal cord. 7 In the current case series, 4 out of 7 patients had MRI in order to characterize the cause of the neurological dysfunction. In these cases, MRI accurately determined the intraparenchymal location of the tumor in 3 cats. In 1 cat, the lesion was thought to be extradural. Lesions were hyperintense on T2-weighted image (T2WI) in 3 out of 4 cases. Additionally, 3 out of 4 of the lesions had minimal contrast enhancement or ring enhancement as is classically seen with intracranial tumors of glial origin (Fig. 3). Table 3 presents a comparison of imaging findings based on tumor location. Despite these criteria, definitive anatomic localization can be difficult, especially between the parenchymal and extraparenchymal compartments. Reasons for difficulty in determining the correct anatomic space include poor spatial resolution due to size of spinal cord, prevention of cerebrospinal fluid pooling between the mass and spinal cord, and invasiveness of the tumor.
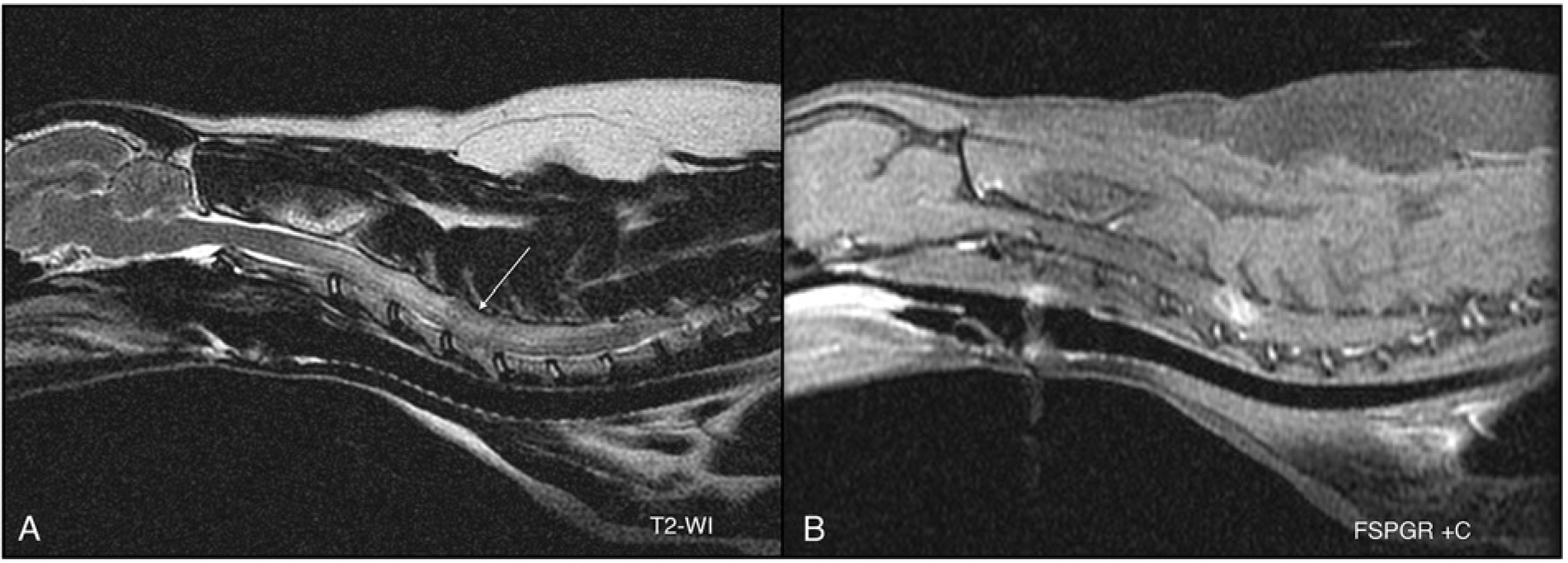
Feline spinal cord glioma.
General patterns for localization of spinal neoplasia based on magnetic resonance imaging.*

CSF = cerebrospinal fluid; SAS = subarachnoid space.
The current retrospective study further advances the literature regarding intraparenchymal spinal cord tumors in cats. Although this cohort of cases was small, a clear breed or sex predilection (5/7 female) was not observed. Likewise, despite the paucity of reported cases, these tumors may have a predilection for the cervical spinal cord with the majority having an astrocytic morphology. Oligodendrocytic and mixed astrocytic–oligodendrocytic neoplasms may also be observed. MRI can greatly aid in the diagnosis and better direct the clinician to the cause of the neurologic signs in the affected animal.
Footnotes
Acknowledgements
The authors thank the histopathology technicians at Cornell University College of Veterinary Medicine for assistance with immunohistochemistry and Jen Patterson for assistance with images. The authors also thank Don Levesque for one of the submitted cases.
a.
Bond-Max IHC/ISH platform, Bond polymer refine DAB kit, Bond polymer refine red kit; Leica Biosystems, Newcastle Upon Tyne, United Kingdom.
b.
Dako North America Inc., Carpinteria, CA.
c.
BioGenex Laboratories Inc., San Ramon, CA.
d.
Pharmingen, BD Biosciences, San Jose, CA.
e.
VECTASTAIN Elite ABC, Vector Laboratories Inc., Burlingame, CA.
f.
Wako Chemicals USA Inc., Richmond, VA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
