Abstract
The ROTEM platelet device, a point-of-care whole blood platelet impedance aggregometer, is an add-on to the rotational thromboelastometry ROTEM delta device. The latter has been validated in dogs. We examined whether canine whole blood is suited for analysis with the ROTEM platelet device using adenosine-5′-diphosphate (ADP) and arachidonic acid (ARA) as agonists for platelet activation, and if there are significant differences between sample storage times and anticoagulants used. Subsequently, we determined canine reference intervals (RIs) for the ROTEM platelet device for ADP and ARA. In a pilot study, we examined whole blood from 7 dogs after 15-min and 60-min storage of lithium-heparinized samples and 40-min and 80-min storage of hirudinized samples. Statistical analysis showed no significant differences between ROTEM platelet device results for both ADP and ARA in lithium-heparin and hirudin anticoagulated canine whole blood. Lithium-heparinized blood samples analyzed after 15-min storage had the lowest coefficient of variation. RIs were determined for heparinized whole blood samples from 49 dogs after 15 min of storage.
Platelets play an important role in primary hemostasis. They are activated through exposed subendothelial surfaces or by the binding of specific agonists such as arachidonic acid (ARA), adenosine-5′-diphosphate (ADP), thrombin, or thromboxane A2 (TXA2). Platelet activation leads to platelet adhesion, aggregation, and release of granule contents that enable further hemostatic processes.8,20,48
Although thrombocytopenia is a common finding in various diseases in dogs, thrombocytopathy is identified less frequently. 20 Thrombocytopathy is classified as primary (congenital) and secondary (acquired). 13 Primary thrombocytopathy is seen with Glanzmann thrombasthenia or Bernard-Soulier syndrome, and is rare in dogs. 13 There are various conditions leading to secondary thrombocytopathy in dogs, such as acute hemorrhage, septic peritonitis, uremia, hepatopathy, monoclonal gammopathy, or treatment with non-steroidal anti-inflammatory drugs (NSAIDs).5,17,26,27,30,42,50–52 The opposite—an amplified activation of platelets, and thus an increased risk of thromboembolic disease—has been described in dogs with immune-mediated hemolytic anemia, sepsis, heartworm disease, lymphosarcoma, parvoviral enteritis, and in critically ill dogs.4,28,31,35,40,46
As the importance of thromboembolic disease is gaining more recognition in small animal medicine, anti-platelet drugs such as acetylsalicylic acid (ASA; inhibitor of ARA-dependent platelet activation) and clopidogrel (inhibitor of ADP-dependent platelet activation) are used increasingly.19,48 It is important to monitor the response to treatment because individual differences in response to treatment have been described in dogs for both ARA and ADP inhibitors.5,11,42 Assessment of platelet function is furthermore desirable before undergoing elective surgical procedures or after experiencing trauma to assess the risk of excessive bleeding in patients under anti-platelet drug therapy. 39
There are various methods to assess platelet function, such as flow cytometry, and adhesion or aggregation assays. 8 Light transmission platelet aggregometry is the gold standard and was first described in 1962. 3 This method has many disadvantages, such as the need to prepare platelet-rich plasma and the requirement for large sample volumes.7,20
Platelet impedance aggregometry using whole blood was first described in 1980. 6 In 2006, a point-of-care (POC) device using whole blood platelet impedance aggregometry (Multiplate analyzer; Roche) was introduced in human medicine and has since been evaluated in dogs, cats, sheep, pigs, and rats.1,10,16,18,19,29,49 The utilization of whole blood for platelet function testing enables the interactions of platelets with other blood cells in a physiologic environment, prevents the loss of platelet subpopulations during preparation of platelet-rich plasma, and requires less training for personnel.10,12,16,29 Whole blood platelet impedance aggregometry is based on a test cell containing 2 electrodes to which platelets adhere and aggregate after activation with specific agonists. Platelet aggregation leads to a change in impedance between the electrodes. The increase in impedance is measured over time and is used to quantify the degree of platelet activation. 6
Recently, a new POC whole blood platelet impedance aggregometer ROTEM platelet device (Tem International) was released as an add-on device to the ROTEM delta device (Tem International). It uses a method similar to the Multiplate analyzer; the Multiplate analyzer has 2 pairs of electrodes in a test cell as an internal control, whereas the ROTEM platelet device has only 1 pair of electrodes in each test cell. The ROTEM platelet device needs only a small sample of whole blood and is user friendly as a result of an automated pipetting system. The ROTEM platelet device allows ARA (ara-tem; Tem Innovation), ADP (adp-tem; Tem Innovation), and thrombin receptor–activating peptide (TRAP; trap-tem; Tem Innovation) dependent platelet activation. Based on the manufacturer’s instructions, fresh whole blood used for analysis is anticoagulated with citrate, hirudin, or heparin (https://www.ttuhsc.edu/medicine/odessa/internal/documents/ttim-manual/ROTEM_Analysis.pdf). Although the Multiplate analyzer using ADP, ARA, and collagen as agonists for platelet activation has been validated in dogs, to our knowledge, investigations have not reported the optimal sample storage time, anticoagulant, and agonist for the use of the ROTEM platelet device in dogs.10,16,29 Nor have corresponding RIs been determined.
Our aims were 2-fold. We conducted a pilot study to determine the optimal anticoagulant and sample storage time for the ROTEM platelet device and to determine the functionality of assays with canine blood. Our second aim was to establish canine RIs for ROTEM platelet device parameters in samples handled based on the results of the pilot study.
Materials and methods
Study population
Our prospective observational study, which took place between February and September 2017 at the small animal clinic of the Vetsuisse Faculty (University of Zurich, Switzerland), was approved by the local ethics committee on animal research (ZH 042/15). We recruited 53 healthy staff- and client-owned dogs; informed owner-consent was obtained for each dog.
Dogs enrolled in the study were adult (> 12 mo old) and weighed > 5 kg for the pilot study or > 3 kg for the determination of RIs, respectively. Dogs were not enrolled if, in the 4 wk before blood collection, they had donated blood, were vaccinated, had been treated with NSAIDs, paracetamol, dipyrone, steroids, anti-platelet drugs (e.g., clopidogrel), tranexamic acid, or colloids, or had a history of coagulation problems (e.g., excessive bleeding during surgery). To avoid breed-specific results, a maximum of 4 dogs of the same breed were included in the study, and no Cavalier King Charles Spaniels were enrolled given their known inherited giant platelet disorder. 9
The health status of each dog was verified by history, physical examination, CBC (XT-2000iV; Sysmex), microscopic evaluation of a blood smear, serum biochemistry panel (Cobas 600; Roche), and venous blood gas analysis (RAPIDPoint 500 blood gas system; Siemens). Activated partial thromboplastin time (aPTT), prothrombin time (PT), thrombin time (TT), Clauss fibrinogen (STart 4 hemostasis analyzer; Stago), and ROTEM delta device analyses were conducted for a concurrent study. 23
Feathered edge and monolayer areas of blood smears were reviewed by experienced laboratory technicians for the presence of platelet aggregates; presence or absence of aggregates was semiquantitatively graded as absent, mild, moderate, or marked. Dogs were excluded from statistical analysis in cases of moderate or marked platelet aggregation or if any other significant laboratory abnormalities were detected. Dogs with marginal thrombocytopenia were not excluded.
Sampling
Dogs were fasted for at least 4 h before blood collection. Applying minimal stasis of the vein, blood was collected by jugular venipuncture with gentle aspiration using a 20-G needle and a 10- or 20-mL syringe. Dogs were either sitting or in lateral recumbency during blood withdrawal. If venipuncture was problematic, the attempt was stopped, and the other jugular vein was punctured after a brief pause for the dog to recover. If a satisfactory venipuncture was not possible, the dog was excluded from the study. Blood collection tubes were filled immediately after sampling in the following order: 3 mL of serum (4.5-mL serum tube; Sarstedt) for a serum biochemistry profile; 2 × 1.3-mL sodium-citrate (Micro tube 9NC; Sarstedt) for traditional coagulation times (aPTT, PT, TT), and Clauss fibrinogen and ROTEM delta device analysis; 2 × 1.3-mL lithium-heparin (Micro tube LH; Sarstedt) for ROTEM platelet device analysis and 0.5 mL of lithium-heparin for venous blood gas analysis; 2.7 mL of hirudin (S-Monovette hirudin; Sarstedt; only if the dog was part of the pilot study) for ROTEM platelet device analysis; and 0.5 mL of K3-EDTA (Microvette K3E; Sarstedt) for CBC and blood smears. Each tube was carefully inverted 10 times immediately after filling. All venipunctures and platelet impedance aggregometry analyses were performed by the same person.
Platelet impedance aggregometry with the ROTEM platelet device
Blood samples were held at room temperature in the designated space on the ROTEM platelet device. Diluent (0.9% sodium chloride with preservatives; Tem Innovation) was warmed to 37°C in the designated heating area prior to analysis. Analysis was performed as described by the manufacturer. Briefly, 0.15 mL of diluent and 0.15 mL of whole blood are pipetted into a single-use cuvette containing a stirring bar and 2 electrodes (ROTEM platelet cuvette; Tem Innovation). The sample is incubated for 3 min while the device determines an impedance baseline value of the system. During this time, 0.02 mL of diluent are added to the reagent to reconstitute the lyophilized agonist for platelet activation. After determination of a baseline, platelets are activated by addition of 0.012 mL of reconstituted reagent into the cuvette containing whole blood and diluent. Aggregation of the activated platelets leads to increasing electrical impedance, which is recorded over 6 min. The increase of impedance is directly proportional to the number of platelets involved in coating the electrodes by aggregation. The recorded impedance slope is processed with specific software and includes the following parameters: 1) the area under the aggregation curve (AUC; Ω × min) for overall platelet aggregation, 2) the amplitude of measured impedance at 6 min (A6; Ω) for the extent of platelet aggregation, and 3) the maximum slope (MS; Ω/min) for the rate of aggregation (https://www.ttuhsc.edu/medicine/odessa/internal/documents/ttim-manual/ROTEM_Analysis.pdf).
ARA and ADP were evaluated as agonists for platelet activation using ara-tem and adp-tem reagents for the ROTEM platelet device. Each ROTEM platelet device features 2 channels (A and B). Because samples were run in duplicate for both agonists, we used 2 ROTEM platelet devices for 4 parallel assays.
Pilot study: comparison of 2 anticoagulants and storage times
We enrolled the first 8 dogs recruited in the pilot study. Hirudin and lithium-heparin were chosen as anticoagulants, and analyses were performed after storage of 15 min and 30 min for lithium-heparin and 40 min and 80 min for hirudin. Sample storage times were chosen based on the available literature as well as clinical and logistic reasons.24,29,34,36 Exact times varied ± 3 min—depending on order of analyses—given the time needed to perform each test step. At each sample storage time, analyses with ara-tem and adp-tem were performed in duplicate.
Reagents and order of analysis were assigned randomly (www.randomizer.org) to 1 of the 2 devices, but the same reagent was run in duplicate on the same device to assess differences between the 2 channels.
Determination of RIs
Based on the results of the pilot study, RIs were established with lithium-heparin anticoagulated blood after 15 min of storage. If an error occurred and the test had to be repeated, a maximum of a 10-min delay from the predetermined time was tolerated (a maximum of 25 min after sampling). Similar to the pilot study, samples were analyzed in duplicate simultaneously on 2 ROTEM platelet devices in randomly assigned order (www.randomizer.org).
Statistical analysis
Statistical analysis was performed using the Analyse-it (Analyse-it Software) add-in for Excel 2016 (Microsoft) and Reference Value Advisor v.2.1 add-in for Excel macroinstruction set. 14 Statistical significance was set at p ≤ 0.05 for all tests. Figures were generated with Prism v.9 (GraphPad).
Pilot study: comparison of 2 anticoagulants and storage times
Overall platelet aggregation was quantified as AUC for each channel as well as for means of channel A and B for the same reagent and sample storage time. Data were tested for normality using the Shapiro–Wilk test. For normally distributed data, mean and SD as well as median and range were calculated, and, for non-normally distributed data, median and range were calculated. Comparison of storage time and anticoagulant was performed using the Levene test for homogeneity of variances and the Kruskal–Wallis test for location.
Overall CV of AUC was calculated for every group (designated by storage time, anticoagulant, and reagent). The percentage difference between simultaneous measurements on channel A and B of every sample was calculated.
Determination of RIs
Serving as a quality control, the percentage difference between simultaneous measurements of AUC on channel A and B of every sample was calculated. If results differed by >20% from the mean of the 2 channels, the samples were excluded.
Normality was tested using the Anderson–Darling test, and outliers were identified by Dixon–Reed and Tukey tests. The mean results of the 2 channels were determined for AUC, A6, and MS, and used for determination of RIs by the reference value advisor program. As determined by the program, nonparametric RIs were reported, and a bootstrap method was used for determination of CIs. RIs are reported as mean, median, SD, range, and 2.5th–97.5th percentile with 90% CI.
Results
Study population characteristics
Of the 53 dogs recruited for blood withdrawal, 3 dogs were excluded because of moderate platelet aggregates and 1 dog because of anemia, resulting in 7 dogs included in the pilot study and 49 dogs for RI determination. Results of the pilot study for the chosen storage time and anticoagulant were included in the determination of RIs. Mean platelet count of all included samples was 263 × 109/L (range: 122–513 × 109/L); 17 of 49 (35%) samples had minor platelet aggregates.
The 49 dogs included 30 females (19 intact, 39%; 11 spayed, 22%) and 19 males (7 intact, 14%; 12 castrated, 25%). Median age was 48 mo (range: 12–116 mo), and median weight was 24 kg (range: 4.7–63 kg). Breeds represented were 15 of 49 (31%) mixed breed, 4 of 49 (8%) Flat-Coated Retriever, 3 of 49 (6%) Border Collie, 2 of 49 (4%) of the following: Belgian Shepherd, Berger Blanc Suisse, Dutch Shepherd, German Pointer, Golden Retriever, Leonberger, Samoyed, and 1 of 49 (2%) of the following: Alaskan Malamute, Beagle, Cocker Spaniel, Dalmatian, Great Dane, Greater Swiss Mountain Dog, Labrador Retriever, Lagotto Romagnolo, Little Lion Dog (Löwchen), Pug, Rottweiler, Siberian Husky, and Pembroke Welsh Corgi.
Pilot study: comparison of 2 anticoagulants and storage times
Mean platelet count in the dogs of the pilot study was 270 × 109/L (range: 160–372 × 109/L). One dog had mild platelet aggregation. CV of all measurements in a group was lowest for assays using lithium-heparin at 15-min storage with adp-tem (7.3%), and highest for hirudin at 40-min storage using ara-tem and lithium-heparin at 60-min storage using adp-tem, respectively (20.3%). All sample groups had results with a difference between measured AUC of channel A and B > 20%, except for analyses at 15-min storage using lithium-heparin for both reagents (Table 1; Fig. 1).
Pilot study. Platelet impedance aggregation in 7 healthy dogs measured with the ROTEM platelet device using adp-tem (ADP) and ara-tem (ARA) as agonists in heparinized or hirudinized whole blood at different sample storage times.
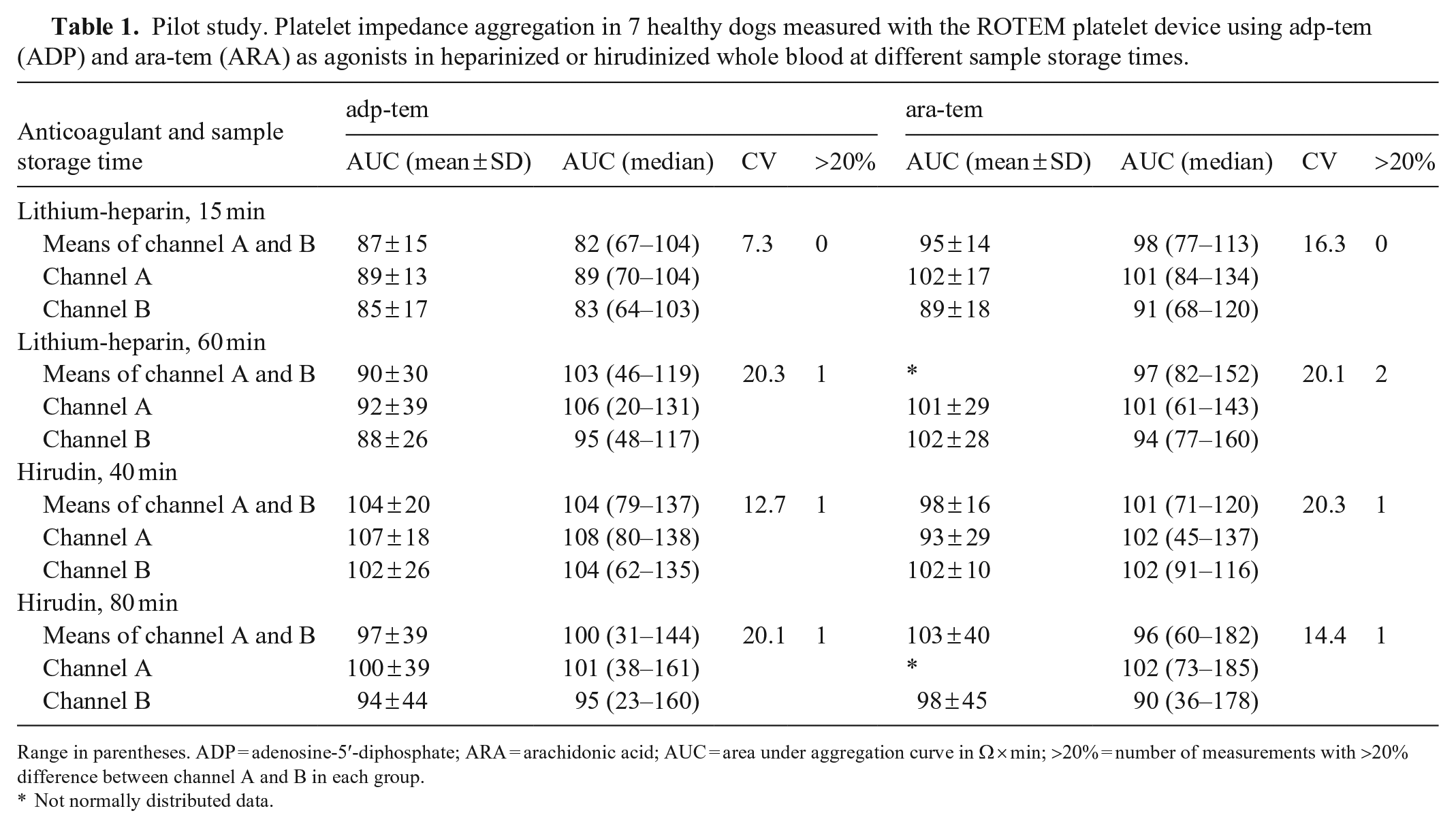
Range in parentheses.
ADP = adenosine-5′-diphosphate; ARA = arachidonic acid; AUC = area under aggregation curve in Ω × min; >20% = number of measurements with >20% difference between channel A and B in each group.
Not normally distributed data.
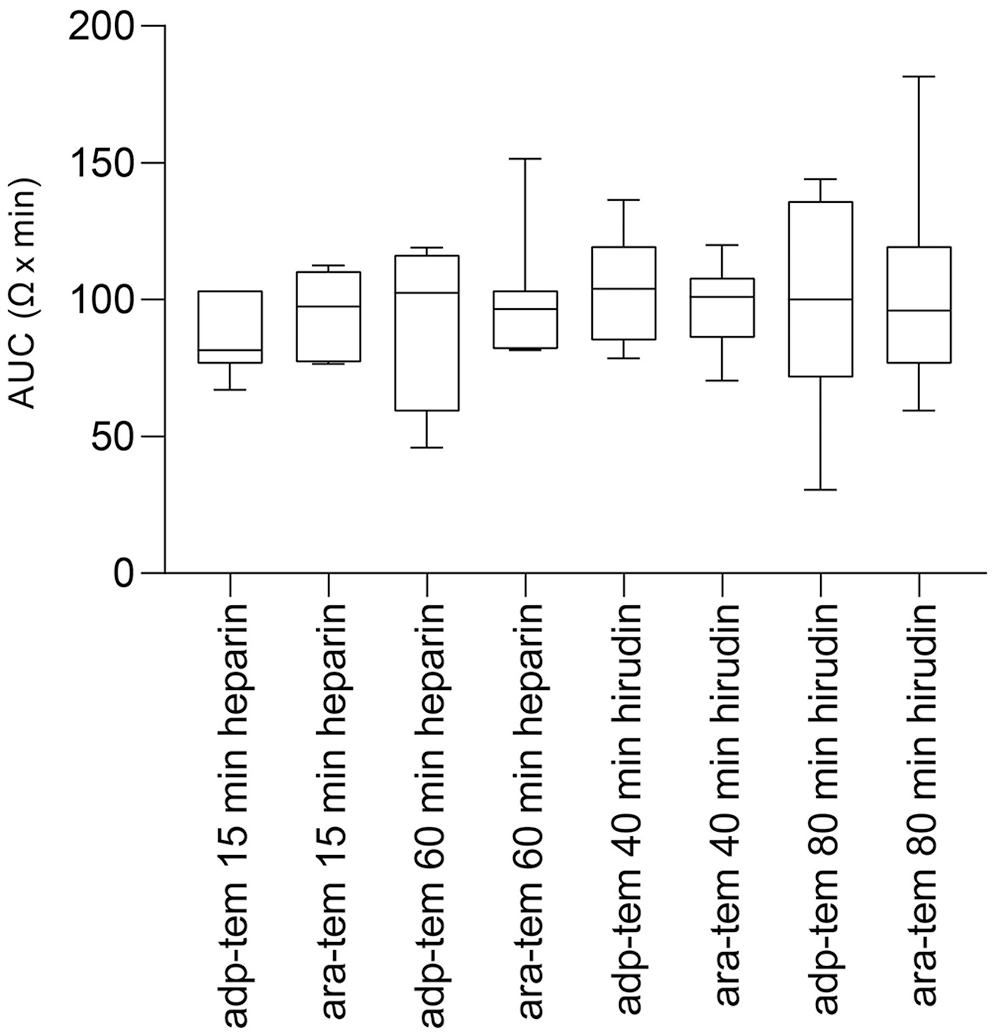
Boxplots of the mean AUC of parallel analyses on 2 channels measured with the ROTEM platelet device using adp-tem (ADP) and ara-tem (ARA) as agonists in heparinized or hirudinized whole blood samples from 7 dogs at different sample storage times.
The Levene test for dispersion showed variances of populations to be equal for means (p = 0.11) and single channels alone (p = 0.20). The Kruskal–Wallis test for location detected no significant differences between medians of populations (p = 0.91 for means; p = 0.89 for single channels). Therefore, no significant differences were detected between groups. Based on the low CV and the lack of AUC measurements with > 20% channel difference, the use of lithium-heparinized blood at 15-min storage was chosen for the establishment of RIs.
Reference intervals
Results of AUC differing > 20% between channel A and B resulted in the additional exclusion of values from 4 dogs each for the calculation of RIs with adp-tem and ara-tem. The affected measurements were from different dogs for adp-tem and ara-tem. RI for adp-tem and ara-tem were determined (Table 2).
RIs for the ROTEM platelet device for adp-tem (ADP) and ara-tem (ARA), measured after 15-min storage of lithium-heparinized whole blood samples from 45 healthy dogs.
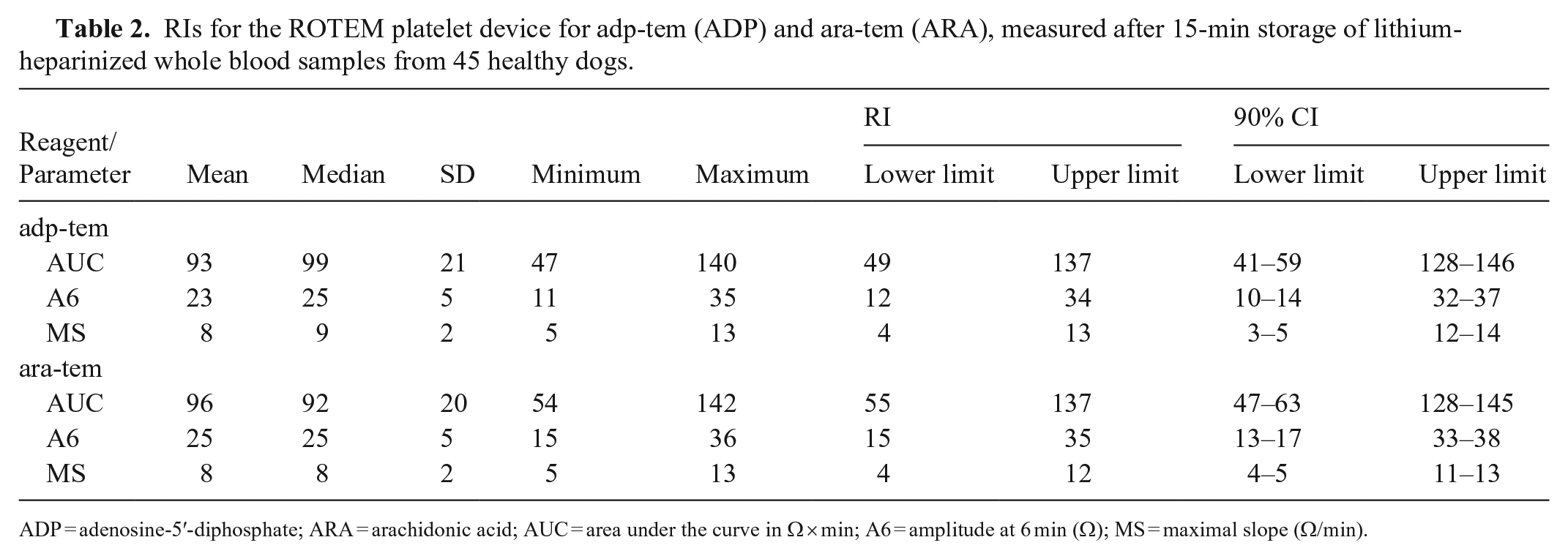
ADP = adenosine-5′-diphosphate; ARA = arachidonic acid; AUC = area under the curve in Ω × min; A6 = amplitude at 6 min (Ω); MS = maximal slope (Ω/min).
Mean ROTEM platelet device parameters were not significantly different between female and spayed-female dogs or between female and male/castrated-male/spayed-female dogs (data not shown).
Discussion
Evaluation of platelet function has become increasingly important in both clinical cases and research studies. Interpretation of test results require species-specific RIs and optimal sample handling. Our study results show that canine blood is suited for analysis with the ROTEM platelet device. No significant differences were detected between anticoagulants and sample storage times in a pilot study including 7 dogs; however, heparinized samples analyzed with adp-tem after 15 min of storage had the lowest CV.
The storage of samples and the effect of anticoagulants on platelet function testing have been studied in animals and in people on various devices, but to our knowledge, there have been no reports of the use of the ROTEM platelet device in dogs and only a few reports of studies in people.34,37,38 The stability of human blood samples anticoagulated with hirudin and citrate analyzed with the ROTEM platelet device were compared in people. 34 Although citrated blood is recommended by the manufacturer for ROTEM platelet device determination of platelet function in people, citrated blood yielded significantly lower results. 34 The same finding was detected in dogs using the Multiplate analyzer.10,25,29 Furthermore, given that spontaneous aggregation in canine blood anticoagulated with citrate may occur, either lithium-heparin or hirudin tubes are recommended for use with the Multiplate analyzer. 10 Citrate was therefore not considered as an anticoagulant in our study. Based on the above-mentioned studies, lithium-heparin and hirudin were chosen as anticoagulants in the pilot study for determination of canine platelet function using ROTEM platelet device analysis. Although sodium-heparin showed accurate results for a longer sample storage in the Multiplate analyzer than lithium-heparin in people, 24 we preferred lithium-heparin because it is routinely used and available at the clinic where our study took place.
In people, aggregation in ara-tem and adp-tem ROTEM platelet device assays increased from baseline to 15-min storage and stabilized from 15-min to 60-min storage in hirudin tubes. 34 We therefore chose a resting time of at least 15 min for our pilot study. Our results show that the use of hirudin offers no advantages over lithium-heparin in canine whole blood samples analyzed with the ROTEM platelet device after storage of 15–80 min. However, if longer storage is a prerequisite, use of hirudin as an anticoagulant may be considered. Canine hirudinized, but not heparinized samples, had spontaneous aggregation in a Multiplate analyzer. 29 We did not evaluate spontaneous aggregation by means of ROTEM platelet device aggregation without an activator, but none of the samples in our study had moderate or marked platelet aggregation as evaluated on a blood smear prior to analysis.
Three agonists for the activation of platelets are available as reagents for the ROTEM platelet device: TRAP (trap-tem), ADP (adp-tem), and ARA (ara-tem; https://www.ttuhsc.edu/medicine/odessa/internal/documents/ttim-manual/ROTEM_Analysis.pdf). Although thrombin is a strong agonist for platelet activation in vivo, a study with the Multiplate analyzer has shown that the TRAP-6 reagent used in tests designed for people does not induce significant platelet activation in dogs, presumably caused by species differences in the thrombin receptor, and our own experience has shown the same for the ROTEM platelet device.10,25 Therefore, we did not evaluate trap-tem in our study.
ADP binds to specific transmembrane receptors of platelets, activates G-proteins and other secondary pathways, eventually leading to the release of ARA (which is then converted to TXA2), the secretion of platelet granules, and a change in platelet shape and size. 48 A study using light transmission platelet aggregometry with platelet-rich plasma showed that a fraction of dogs does not respond to ADP as an agonist. 15 In contrast to this, all samples were aggregated by ADP in our study, which is in accord with studies using platelet impedance aggregometry to measure platelet function in dogs.10,16,29,45
The agonist ARA is a precursor of TXA2, one of the most important physiologic agonists of platelet activation, and serves as an agonist in the activation of platelets. 48 Tests based on ARA activation have resulted in higher imprecision and interindividual variation than ADP in dogs as assessed with the Multiplate analyzer, which was however not confirmed by our pilot study.10,25 Similar to ADP, ARA was also described to inconsistently activate canine platelets in a study using platelet-rich plasma in an optical aggregometer,21,22 a finding not supported by our study.
Canine AUC, A6, and MS measured from heparinized samples yielded results comparable to a preliminary study including 20 people (https://www.ttuhsc.edu/medicine/odessa/internal/documents/ttim-manual/ROTEM_Analysis.pdf). To our knowledge, no studies have established canine ROTEM platelet device RIs for AUC, A6, and MS. ROTEM platelet device AUC is not comparable to the AUC of the Multiplate analyzer, and the Multiplate analyzer does not yield results for A6 and MS. 38 Although our study and other studies in people report RIs for A6 and MS, these parameters do not seem to be used commonly in the interpretation of platelet function analysis in people, and their use has yet to be determined in veterinary clinical and laboratory studies. 34
A common finding in all RIs established for platelet impedance aggregometry for people and animals is the relatively large range of RIs, which makes the clinical interpretation of results challenging. However, dogs treated with ADP or ARA antagonists, such as clopidogrel or ASA, are expected to have a very low AUC in the respective test. AUC decreased significantly in dogs treated with clopidogrel and tested in a Multiplate analyzer using ADP as agonist, and therefore, distinguishing impaired from normal platelet function is still possible.43,47 Poor correlation of methods of platelet function testing in dogs was identified comparing thromboelastographic platelet mapping to whole blood platelet aggregometry with the Multiplate analyzer and platelet function analysis using citrated whole blood. It is suggested that population-based RIs are unsuitable for most platelet function tests, and that subject-based RIs are preferable because biological variation in platelet aggregation should be considered. 2 Hence, subject-based RIs for ADP- and ARA-induced platelet aggregometry in the Multiplate analyzer are more sensitive in detecting abnormalities in platelet function in dogs. 44
In people, sex influences whole blood platelet aggregation RIs for the ROTEM platelet device, and day-to-day variation may occur and may be more pronounced in citrate-anticoagulated samples. 34 Age, however, does not seem to influence platelet aggregation in the Multiplate analyzer in people. 41 In our study, mean values of all variables were not significantly different between female and spayed-female dogs as well as female and male/castrated-male/spayed-female dogs. This result must be interpreted in view of the small group size, and further studies are needed to determine if population-based RIs are useful clinically. RIs in ROTEM delta device parameters were impacted by female sex. 23
Results of platelet impedance aggregometry have been described to vary among breeds of dogs, which is why we included a maximum of 4 dogs of a breed in our study. 33
There are several limitations of our study. Blood was withdrawn and analyzed at the described storage times consecutively on 2 devices. This resulted in a difference between ara-tem and adp-tem measurements of up to 3 min. Given that the results of ara-tem and adp-tem were not compared directly, this is judged to be of little importance. Sample storage time was different for both anticoagulants in our study because of lack of additional analyzers. RIs were established from the means of parallel measurements on 2 channels; results differing > 20% between channels were excluded. This approach was chosen because the ROTEM platelet device features only 1 pair of electrodes per test cell, whereas the Multiplate analyzer contains 2 pairs of electrodes in each test cell serving as internal control by excluding results differing > 20% from each other. 38 We decided that duplicate measurements were more important than uniform sample storage times between anticoagulants, given that duplicate measurements can improve analytical performance. 2
Sample collection may have an influence on results. We minimized this effect by using gentle suction, a large needle, and jugular venipuncture, as well as exclusion of patients in which venipuncture was difficult. Additionally, samples were analyzed for aggregation on blood smears, and samples with moderate or severe platelet clumping were excluded. We did not exclude samples with minor platelet clumping because we observe mild platelet aggregation frequently in our laboratory, hence the samples included in our study are consistent with our reference population. A study examining the effect of storage conditions and anticoagulant on platelet clumping in healthy dogs observed that 12.9–31.4% of EDTA-anticoagulated samples had platelet aggregation; the authors concluded that platelet clumping is common in blood samples of healthy dogs and is difficult to avoid. 32
The following limitation is given by the device and could not be addressed by a different study design. The analysis time in the ROTEM platelet device is limited to 6 min compared to 12 min with the Multiplate analyzer. We deemed this of little importance, given that short analysis times of 6 min are preferable over 12 min on the Multiplate analyzer when using canine whole blood. 29 Further studies are needed to examine if ROTEM platelet device analyses can reliably monitor anti-platelet drug therapy and recognize abnormal values in clinical patients.
Footnotes
Acknowledgements
We thank all dog owners and the Small Animal Foundation (Stiftung für Kleintiere) of the Vetsuisse Faculty, University of Zurich.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was funded by the Small Animal Foundation (Stiftung für Kleintiere) of the Vetsuisse Faculty, University of Zurich, Switzerland (ROTEM material), and by Axonlab, Baden, Switzerland (discount on ROTEM material).
