Abstract
The role of Bovine viral diarrhea virus (BVDV) in the development of Porcine circovirus-2 (PCV-2)-associated disease (PCVAD) was investigated in 2 experimental studies. In the first, separate groups of germ-free pigs were inoculated with filtered tissue homogenate (from diseased pigs) containing PCV-2b + BVDV-1–like virus (group 1), PCV-2a + BVDV-1–like virus (group 4), BVDV-1–like virus only (group 3), or PCV-2b + BVDV-1–like virus following a BVDV vaccination protocol (group 2). This last group was used to test if BVDV vaccination would prevent clinical PCVAD in this model. Many of the inoculated pigs had mild multisystemic inflammation consistent with classic PCVAD. One vaccinated, dually inoculated pig had acute respiratory distress followed by death at 21 days postinfection. Lesions in this pig resembled the severe form of PCVAD observed in the field since the fall of 2004, suggesting a role of ruminant pestiviruses and/or vaccination in the development of this disease. In the second study, cesarean-derived, colostrum-deprived pigs were inoculated with PCV-2b and a cytopathic strain of BVDV-1 (cpBVDV-NADL) either alone or in combination. Clinical signs of PCVAD were seen in a single animal inoculated only with PCV-2b. This pig had growth retardation followed by acute respiratory distress leading to death 30 days postinfection. Pulmonary lesions in this animal were similar to those seen in the pig that died in the first study. Infection with cpBVDV-NADL did not enhance PCV-2b replication or lesion formation.
Keywords
Introduction
Porcine circovirus-2 (PCV-2; family Circoviridae, genus Circovirus) is one of the most important viral pathogens in the U.S. pig population. 39 Systemic PCV-2–associated disease (PCVAD) was first recognized in Canada in 1991 as a chronic, insidious, and sometimes protracted disease characterized by progressive weight loss typically affecting pigs 2–3 weeks postweaning, or at about 5–6 weeks of age (Harding JCS, Clark EG: 1997, Diagnostic notes. Recognizing and diagnosing postweaning multisystemic wasting syndrome (PMWS). J Swine Health Prod 5:201–203). Morbidity is usually low, but case fatality rate is often high among affected animals. 32 In addition to wasting, clinical signs of PCVAD include pallor or icterus, respiratory distress, and diarrhea (Harding JCS, Clark EG: 1997, Recognizing and diagnosing PMWS). Gross lesions are observed primarily in the lungs, lymph nodes, and kidneys: lungs often fail to collapse and are mottled tan, lymph nodes are enlarged, and kidneys have scattered white foci (Harding JCS, Clark EG: 1997, Recognizing and diagnosing PMWS). Characteristic microscopic lesions consist of lymphoid depletion, lymphohistiocytic or granulomatous inflammation, and intracytoplasmic intrahistiocytic inclusions in 1 or multiple organs (Harding JCS, Clark EG: 1997, Recognizing and diagnosing PMWS). 45
The pathogenesis of PCVAD is still not fully understood. PCV-2 is considered essential for the development of the disease.10,22 One or more of a variety of co-factors are thought to be necessary for full expression of clinical disease.1,15,22,40 The co-factors include other infectious agents such as Porcine reproductive and respiratory syndrome virus (PRRSV), Porcine parvovirus (PPV), and Mycoplasma hyopneumoniae. Experimentally, immune stimulants such as keyhole limpet hemocyanin in incomplete Freund adjuvant and oil-in-water adjuvanted vaccines appear to similarly enhance PCV-2–associated lesions, suggesting that up-regulation of the immune system may contribute to the development of PCVAD.17,23,25 Additionally, factors inherent to the host and environment may play important roles in the development of PCVAD.
A significant increase in both the incidence and severity of PCVAD has been observed in North America since the fall of 2004, mainly in 10–15-week-old pigs rather than in recently weaned pigs.6,18 Animals that died had unique gross and histologic lesions, including cavitary effusions, marked pulmonary and mesenteric edema, typical PCV-2 intracytoplasmic inclusions in renal tubule and bronchial gland epithelial cells, and vasculitis.6,19,26 Links have been established between a new genotype of PCV-2 (PCV-2b), not reported earlier in North America, and the increased incidence and severity of PCVAD (DeLay J, McEwen B, Carman S, et al.: 2005, Porcine circovirus type 2-associated disease is increasing. AHL Newsletter 9:22).6,7,18,26 Reasons for this relatively sudden emergence of a more virulent viral strain include virus escape of existing herd immunity to previously circulating PCV-2a 18 or introduction of a formerly undetected cofactor, such as Bovine viral diarrhea virus (BVDV), that resulted in increased replication of a previously unrecognized type of PCV-2 (Segalés J, Torremorell M: 2006, What we know and do not know about PMWS. In: Proceedings of the Allen D. Leman Swine Conference, pp. 121–125. St. Paul, MN). 39
Pigs are susceptible to infection by all known pestiviruses. They are the only natural host of Classical swine fever virus and likely also of a novel pestivirus named Bungowannah virus that appears to be associated with the porcine myocarditis syndrome. 21 In addition, natural infection of swine with BVDV and Border disease virus (which have cattle and sheep as their primary host, respectively) have also been reported.29,42 Ruminant pestiviruses are pathogenic for pig fetuses, but are generally considered to be relatively nonpathogenic for pigs after birth. 29 Nonetheless, disease syndromes characterized by growth retardation and wasting have been reported in pigs infected with these viruses. 31 Concurrent infection with BVDV in pigs may intensify the effects of Transmissible gastroenteritis virus. 54 In addition to PCV-2, BVDV type 1 (BVDV-1) was isolated from an outbreak of acute, high mortality in pigs in southern Minnesota in 2004 (Torrison J: 2004, Clinical and diagnostic considerations for an acute, high mortality syndrome in grow-finish swine. In: Proceedings of the Swine Disease Conference for Swine Practitioners, pp. 92–100. Ames, IA). The Indiana Animal Disease Diagnostic Laboratory (West Lafayette, Indiana) has detected both BVDV-1–like virus and PCV-2b in tissues of diseased pigs from severe epidemic outbreaks of PCVAD since 2005. In these animals, the aforementioned unique gross lesions have been observed in conjunction with unique microscopic lesions, namely vasculitis and intracytoplasmic inclusions in renal tubule and bronchial gland epithelial cells (unpublished data), that mirror the tropism for endothelial and epithelial cells of various pestiviruses in their host species.8,50,53
The current study tested the hypothesis that BVDV enhances the pathogenicity of PCV-2, triggering the severe form of PCVAD, and that the cellular tropism of BVDV might expand the tropism of PCV-2 and explain the emergence of unique lesions in renal tubule and bronchial gland epithelium as well as in blood vessels. A vaccination protocol to BVDV was tested as a possible measure to prevent development of clinical disease. Fatal interstitial pneumonia and pulmonary edema in addition to other lesions were produced that were microscopically associated with vasculitis in 1 out of 4 vaccinated, germ-free pigs inoculated with PCV-2b and a previously unrecognized BVDV-1–like virus. This animal also had typical microscopic lesions of PCVAD in lymphoid tissues. The findings prompted a similarly designed study in cesarean-derived, colostrum-deprived (CDCD) pigs. In the second study, clinical signs and lesions typical of the more severe form of PCVAD occurred in 1 out of 8 pigs solely inoculated with PCV-2b. This suggests that this viral genotype is by itself capable of inducing severe disease in pigs.
Materials and methods
Animals
Experiment 1, germ-free pigs
Twenty near-term crossbreed pigs were derived by cesarean section from 2 sows seronegative for PRRSV and not viremic for PCV-2, PRRSV, or BVDV. Pigs were maintained in gnotobiotic isolator units in the Animal Resource Wing at South Dakota State University (Brookings, South Dakota). During the first 3 weeks of the study, animals were raised 4 to an isolator and thereafter were kept in groups of 2. Therefore, the initial number of 5 isolators was later increased to 10 units. Pigs were fed with commercial milk substitute a 3 times per day for the first 16 days, and 2 times per day for the rest of the study.
Experiment 2, CDCD pigs
Forty-six crossbreed pigs were derived by cesarean section from 4 high-parity sows from a commercial high-health (PRRSV- and PCVAD-negative) farm. Piglets were placed in individual Plexiglas partitions on raised wire decks equipped with 1 feeding pan each, and were initially fed a commercially available milk replacer a and provided fresh water ad libitum. The pigs were gradually weaned onto a standard commercial early weaning meal diet by 17 days of age when they were placed in groups in separate rooms on raised wire decks equipped with 1 nipple drinker and 1 self-feeder. The pigs were fed age-appropriate pelleted or meal diets thereafter.
Experimental design
Experiment 1, germ-free pigs
The experimental design is delineated in Table 1. The germ-free pigs were divided into 1 of 6 treatment groups. At 3 days of age (study day: 21), pigs in groups 2 and 5 were vaccinated through the intramuscular route using 2 ml of a commercial killed vaccine for BVDV-1 and -2. b The pigs received a booster of 2 ml of a Madin–Darby bovine kidney (MDBK) cell culture lysate containing the nonpathogenic, cytopathic NADL (National Animal Disease Laboratory) strain of BVDV-1 with a titer of 104 TCID50 at 10 days of age (day −14). When pigs were 24 days old (day 0), all animals in groups 1–4 were inoculated with 4 ml (1 ml each into both nostrils and right and left thigh musculature) of 1 out of 3 inocula: groups 1 and 2 received a filtered (through a 0.2-μm syringe filter) homogenate prepared from tissues of pigs with PCVAD known to contain PCV-2b (GenBank accession no. EU594437) and a previously unrecognized BVDV-1–like virus (with 88–91% homology with other BVDV-1 and BVDV-2 at the 5′-untranslated region), but determined to be free of PPV, PRRSV, Swine influenza virus (SIV), and M. hyopneumoniae by virus isolation, polymerase chain reaction (PCR), and/or serological testing (enzyme-linked immunosorbent assay [ELISA]); groups 3 and 4 received a filtered inoculum prepared from tissues of diseased pigs known to contain the same pestivirus, but free of PCV-2, PPV, PRRSV, SIV, and M. hyopneumoniae; and group 4 also received a porcine kidney (PK)-15 cell culture lysate containing PCV-2a (104.6TCID50/ml). Pigs in group 5 were vaccinated, but not challenged with any virus. Group 6 served as the negative control.
Experiment 1, germ-free pigs: design of experimental reproduction of Porcine circovirus-2–associated disease by coinfection of germ-free pigs with Bovine viral diarrhea virus 1 (bvdv-1)-like virus and Porcine circovirus-2 (PCV-2)
Animals were examined daily for alterations in clinical status. Specific clinical signs that were monitored included behavioral changes (such as lethargy), dyspnea, cough, icterus, and wasting. Two pigs from each group were humanely euthanized at days 10 and 21/22. At necropsy, samples of selected tissues were collected from each pig and preserved at −80ºC for virus detection and in 10% neutral buffered formalin for histopathologic analysis. The experimental protocol was approved by the Institutional Animal Care and Use Committees of Purdue University and South Dakota State University.
Experiment 2, CDCD pigs
The experimental design is delineated in Table 2. At 17 days of age, CDCD pigs were randomly assigned into 2 groups of 8 (groups 1 and 2) and 3 groups of 10 animals (groups 3–5). On the same day, the pigs in group 4 received a 2-ml intramuscular dose of a commercial killed vaccine for BVDV-1 and -2. b At 27 days of age, the animals in groups 1, 3, and 4 were challenged with 4 ml (1 ml each into both nostrils and thigh musculature) of a suspension containing a cytopathic strain of BVDV-1 (cpBVDV-NADL) propagated in MDBK cells (105.5 TCID50/ml). The same viral strain had been used in the previous study for booster vaccination. At 31 days of age (study day 0), pigs in groups 2–4 received a similar challenge with a PK-15 cell culture lysate containing a field isolate of PCV-2b (104.5 TCID50/ml) derived from the same tissue inoculum used in the germ-free study. Group 1, which served as a negative control, received a sham inoculum (PK-15 cell culture lysate free of PCV-2). The study was terminated 5 weeks after inoculation of PCV-2b.
Experiment 2, cesarean-derived, colostrum-deprived (CDCD) pigs: design of experimental reproduction of Porcine circovirus-2–associated disease by coinfection of cdcd pigs with Bovine viral diarrhea virus 1 (BVDV-1) and Porcine circovirus-2b (PCV-2b)
Pigs were kept under strict biosecurity conditions and were observed daily. Animals were scored for severity of respiratory disease, wasting, and icterus. At each of days 0, 4, and 14, 2 pigs from each group were euthanized; the remaining pigs of each group were euthanized at day 35. On the same days, serum samples were collected from all animals. At necropsy, samples of selected tissues were collected into 10% neutral buffered formalin for histopathologic examination and were frozen at −80°C for virus detection. Additionally, serum samples from each pig were tested for antibodies to PCV-2, BVDV, PRRSV, and PPV, and samples from pigs that remained until the end of the study were tested for antibodies to M. hyopneumoniae. The experimental protocol was approved by the Purdue University Animal Care and Use Committee.
In situ hybridization for PCV-2
In situ hybridization (ISH) to demonstrate PCV-2 nucleic acid was performed on sections of lung, lymph nodes, tonsil, spleen, ileum, liver, kidney, gonad, and brain and spinal cord of all pigs of the germ-free and CD/CD study. Two digoxigenin-labeled oligonucleotide probes (DIG-5′-AAGTTCAGCCA GCCCGCGG-3′ and DIG-5′-CATTCCAACGGGGTCTG ATTGCTGG-3′) were designed for ISH based on highly conserved regions of the PCV-2 genome (nucleotide positions 491–509 and 811–835, respectively). The technique was carried out using a manual capillary-action work station c as previously described 13 with some modifications. Briefly, tissue sections from inoculated and control animals were deparaffinized and pre-hybridized at 42°C for 1 hr. Hybridization was performed overnight at 42°C with specific digoxigenin-labeled 19- and 25-mer oligoprobes that hybridize with the genes coding for the replicase and capsid protein, respectively, of PCV-2. The detection system consisted of anti-digoxigenin antibody conjugated with alkaline phosphatase (dilution 1:500) and the substrates nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate. d Sections of lymph node and kidney of a pig infected with PCV-2 were used as positive control. The amount of PCV-2 nucleic acid was scored blind as follows: 0 = no positive cells present, 1 = average 1–9 positive cells present per high power field, 2 = average 10–19 positive cells present per high power field, and 3 = average more than 20 positive cells present per high power field.
Fluorescent antibody test for BVDV
Frozen samples (preserved at −80°C) of lymph node, lung, and ileum collected from germ-free pigs inoculated with the BVDV-1–like pestivirus and from vaccinated and unvaccinated negative control germ-free pigs were cut using a cryostat. Sections were mounted on glass slides, air-dried, and fixed with acetone. After dipping into 3% Tween 80, sections were incubated in a humid chamber for 30 min at room temperature with anti-BVDV polyclonal antiserum conjugated to fluorescein-isothiocyanate e at a dilution of 1:50. Sections were washed with phosphate buffered saline, air dried, mounted in glycerol, and examined under a fluorescent microscope.
Virus isolation
Samples of lung, spleen, ileum, and lymph node and tonsil pool collected from germ-free and CDCD pigs at necropsy and preserved at −80ºC were homogenized by grinding to a 10% suspension in phosphate buffered salt solution (PBSS). For PCV-2, tissue homogenate and a suspension of PCV-free PK-15 cells in minimum essential medium, f supplemented with 10% fetal bovine serum, L-glutamine, N′-(2-hydroxyethyl)piperazine-N′-(2-ethanesulfonic acid), sodium pyruvate, f and an antibiotic–antimycotic solution (gentamicin sulfate, streptomycin sulfate, and penicillin G potassium), f were inoculated into plates. These plates were placed into a 37°C humidified CO2 incubator. After 48 hr of incubation, 300 mM of D-glucosamine f was added to the resulting cell monolayer. After an additional 24 hr of incubation, the cells were fixed in cold 80% acetone. Virus growth was confirmed by indirect fluorescent antibody test (IFAT) using specific antibodies to PCV-2 as previously described.43,49 For isolation of BVDV, the tissue homogenate was inoculated into a suspension of MDBK cells, which was maintained in Dulbecco modified Eagle medium supplemented with 5% horse serum, antibiotic–antimycotic solution, and 800 mM of L-glutamine. Plates were placed into a 37°C humidified CO2 incubator, and cells were evaluated for cytopathic effect 96 hr later, followed by IFAT using specific antibodies to pestiviruses from the National Veterinary Services Laboratories (Ames, IA) and commercial anti-BVDV polyclonal antiserum. e
Serology
Serum samples were collected from sows 4 weeks prior to cesarean section, from germ-free pigs at days −21, −14, 0, 10, and 21/22, and from CDCD pigs at days −31, −24, −14, −4, 0, 4, 14, and 35. A PCV-2 ELISA, based on the recombinant open reading frame 2 capsid protein of PCV-2, was performed on all serum samples. 36 Positive results were confirmed by IFAT using a goat anti-swine immunoglobulin G (H+L) conjugated with fluorescein isothiocyanate.g,43 Serum samples of the germ-free pigs were submitted to a BVDV ELISA using protein A conjugated to horseradish peroxidase as the secondary antibody conducted according to the manufacturer’s instructions. h Additionally, a virus neutralization assay for BVDV was performed on all serum samples in 96-well microtitration plates as previously described 55 with some modification, using MDBK cells as the indicator and a 1:4 starting dilution.
Serum of sows and pigs was also tested by ELISA for the presence of specific antibodies to PPV, i PRRSV, j and M. hyopneumoniae. k Positive results for PPV by ELISA were confirmed by IFAT. This assay was performed in a similar manner as for PCV-2, except for the use of PPV and the exclusion of the addition of glucosamine.
Polymerase chain reaction
Polymerase chain reaction for PCV-2 and pestiviruses was performed on serum, lung, lymph node and tonsil pool, spleen, ileum, liver, kidney, gonad, and brain and spinal cord pool of germ-free and CDCD pigs. Sections of tissue were homogenized in PBSS at a final concentration of 10% (w/v) using a tissue homogenizer. DNA extraction was performed manually on samples of germ-free pigs using a commercial blood and cell culture DNA kit l and automatically m on samples of CDCD pigs using a commercial kit. l Quantitative real-time PCR for PCV-2 and reverse transcription (RT)-PCR for pestiviruses were performed using sequence-specific oligonucleotide primers and a 6-carboxyfluorescein (FAM)-labeled probe as previously described.16,41 Serum collected from germ-free pigs at days 0, 10, and 21/22 of the first study and from CDCD pigs at days −31, −24, and 35 of the second study was tested by quantitative real-time RT-PCR for PRRSV using a commercially available one-step multiplex RT-PCR test n that can detect both North American and European strains of the virus. 51 Finally, serum of germ-free pigs collected at days 0, 10, and 21/22 of the first study, and serum of CDCD pigs collected at days −31, −24, and −14 of the second study was tested for PPV viremia by gel-based PCR as previously described. 3
Filtered inocula and serum collected from each pig at the beginning and end of each study was tested for the presence of both Torque teno virus 1 and 2 (TTV-1 and -2, respectively). Real-time PCR for TTV-1 was performed using sequence-specific primers and a probe bearing a 5′ FAM and 3′ MGB (minor groove binder) quencher. 12 Nested PCR for TTV-2 were performed as previously described. 20
Light microscopy
Samples of trachea, lung, heart, tonsil, thymus, spleen, lymph nodes (sublumbar, inguinal, mesenteric, tracheobronchial, and submandibular), liver, kidney, jejunum, ileum, spiral colon, gonad, brain, spinal cord, skin (pinna and gluteal region), and femoral bone marrow were collected for histopathology. Tissues were trimmed after fixation in 10% neutral buffered formalin, processed routinely for paraffin embedding, sectioned at 5 µm, and stained with hematoxylin and eosin for microscopic examination. The microscopic examination and scoring were performed in a blinded fashion. The scoring system ranged from 0 for normal to 3 for severe lesions.
Statistical analysis
Differences in serologic responses, levels of viremia, lesion severity, and amount of intralesional PCV-2 nucleic acid based on ISH were evaluated using Kruskal–Wallis one-way analysis of variance by ranks.
Results
Clinical signs
Experiment 1, germ-free pigs
Clinical signs are summarized in Table 3. Ataxia and hind limb paresis were observed in 2 dual-infected (PCV-2b/BVDV-1–like virus) germ-free pigs. Signs were transient in 1 animal (days 10–16). The other pig also had partial tongue paralysis; signs in this animal were present at days 10–22 (end of study). At day 21, one group 2 pig (vaccination + PCV-2b/BVDV-1–like virus) died following sudden onset of anorexia, lethargy, and marked respiratory distress. No clinical signs were observed in other groups.
Experiment 1, germ-free pigs: clinical signs and postmortem lesions of germ-free pigs experimentally inoculated with Bovine viral diarrhea virus 1 (BVDV-1)-like virus and Porcine circovirus-2 (PCV-2).*
Reported as (Incidence) clinical sign/lesion.
Experiment 2, CD/CD pigs
Clinical signs are summarized in Table 4. One animal inoculated with PCV-2b only developed clinical signs of PCVAD (failure to gain weight, rough hair coat, cutaneous pallor, and mild lethargy) over 12 days followed by acute onset of marked respiratory distress and death at day 30. Control pigs and all other inoculated pigs did not exhibit any clinical signs attributable to viral inoculation. At 6–7 weeks of age, several animals in each group had signs of arthritis characterized by weight shifting and mild joint swelling. Affected animals were treated intramuscularly with 10 mg/kg of gentamicin o twice daily for 3 days. Animals recovered uneventfully, supporting the clinical impression of bacterial arthritis.
Experiment 2, cesarean-derived/colostrum-deprived (CDCD) pigs: clinical signs and postmortem lesions of CDCD pigs experimentally inoculated with Bovine viral diarrhea virus 1 (BVDV-1) and Porcine circovirus-2b (PCV-2b).*

Reported as (Incidence) clinical sign/lesion.
Macroscopic lesions
Experiment 1, germ-free pigs
Lesions are summarized in Table 3. At both necropsy dates, gross lesions were restricted to enlarged lymph nodes draining the inoculation sites (inguinal and mandibular nodes). The group 2 pig (vaccination + PCV-2b/BVDV-1–like virus) that died at day 21 had generalized lymph node enlargement, pulmonary consolidation (approximately 10% of cranial lung lobes), and marked interlobular septal edema (Fig. 1). The small intestine was thickened, with a corrugated appearance, the mesenteric lymphatics were enlarged and multifocally thickened (beaded appearance), and the mesentery was markedly edematous (Fig. 2). Additional lesions included mild chyloabdomen, mild splenomegaly, and mild edema in the renal pelvis.
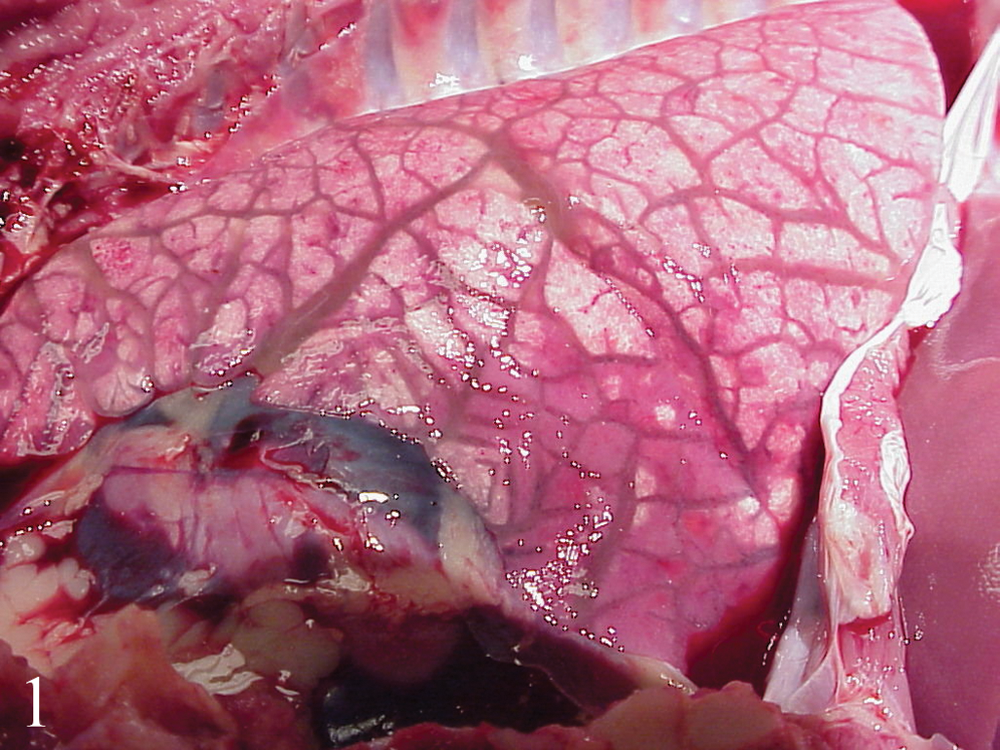
Opened thorax of a germ-free pig vaccinated for Bovine viral diarrhea virus (BVDV) and inoculated with tissue homogenate containing Porcine circovirus-2b (PCV-2b) and BVDV-1–like virus (group 2). The lung failed to collapse, and interlobular septa are prominent due to marked fluid accumulation (edema).
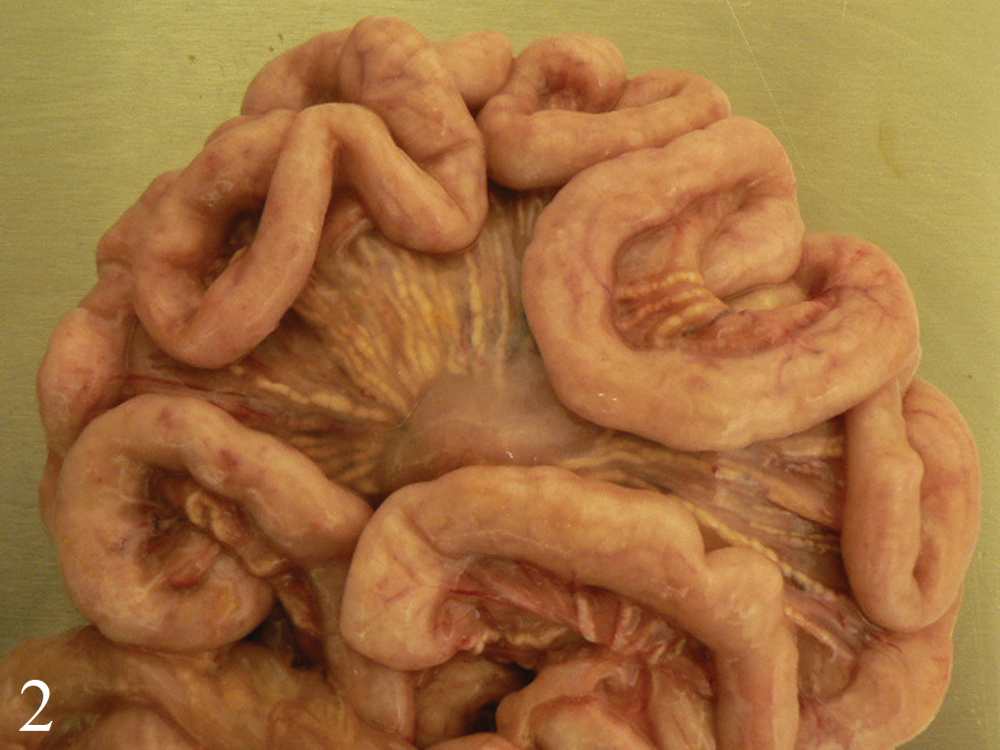
Small intestine of a germ-free pig vaccinated for Bovine viral diarrhea virus (BVDV) and inoculated with tissue homogenate containing Porcine circovirus-2b (PCV-2b) and BVDV-1–like virus (group 2). The jejunal wall is thickened and corrugated. Prominent lymphatics with a beaded appearance are present in the edematous mesentery.
Experiment 2, CDCD pigs
Lesions are summarized in Table 4. At day 14, 3 out of 6 pigs infected with PCV-2b had generalized lymphadenomegally. At day 35, 7 out of 11 PCV-2b–inoculated pigs had generalized lymphadenomegally and pinpoint white foci scattered throughout the kidneys (nephritis). The animal that died at day 30 had abundant hydrothorax, moderate interstitial edema, and cranioventral pneumonia in both lungs (approximately 25% of the total parenchyma). These lesions were similar to those seen in the pig that died in the first study. In the animals clinically diagnosed with arthritis, synovial thickening, excessive joint fluid, and fibrous tags on the serosal surface of multiple viscera indicated previous septicemia.
Microscopic lesions
Experiment 1, germ-free pigs
Lesions are summarized in Table 3.
Day 10 euthanasia
—All group 1 pigs (PCV-2b/BVDV-1–like virus) and 1 of the two group 2 pigs (vaccination + PCV-2b/BVDV-1–like virus) had mild, focal to multifocal lymphohistiocytic inflammation in lung (typically peribronchiolar), liver, and choroid plexus of the lateral and fourth ventricles of the brain.
Days 21/22 euthanasia
—Vaccinated and unvaccinated pigs inoculated with PCV-2b/BVDV-1–like virus (groups 1 and 2) and 1 pig in group 4 (PCV-2a/BVDV-1–like virus) had more extensive and severe inflammatory lesions than those necropsied at day 10. Focal to multifocal lymphohistiocytic inflammation was present to variable extent in tracheal submucosa, pulmonary parenchyma, myocardium, glossal skeletal muscle, esophageal, gastric and intestinal submucosa, renal interstitium, urinary bladder wall, meninges, neuroparenchyma of the brain and spinal cord, and choroid plexus of these animals. Small granulomas were noted in the inguinal lymph nodes of 3 pigs inoculated with PCV-2b/BVDV-1–like virus (groups 1 and 2). Lesion distribution and severity in the 2 pigs with ataxia and hind limb paralysis did not differ from those in the others. The group 2 pig (vaccination + PCV-2b/BVDV-1–like virus) that died at day 21 with severe clinical signs had lymphohistiocytic bronchointerstitial pneumonia, lymphohistiocytic enteritis, marked pulmonary and mesenteric edema, necrotizing (fibrinoid) arteritis, and mesenteric and small intestinal lymphangitis and lymphangiectasia (Fig. 3). 27 Lymphoid tissues of this pig were markedly depleted of lymphocytes, accompanied by multifocal to coalescing macrophage infiltrates. Pigs inoculated only with BVDV-1–like virus (group 3) and vaccine-only pigs (group 5) had, at days 21/22, minimal lymphocytic inflammation in lung, heart, stomach, and brain (choroid plexus). No microscopic lesions were identified in the tissues of the other negative control pigs in groups 5 and 6.
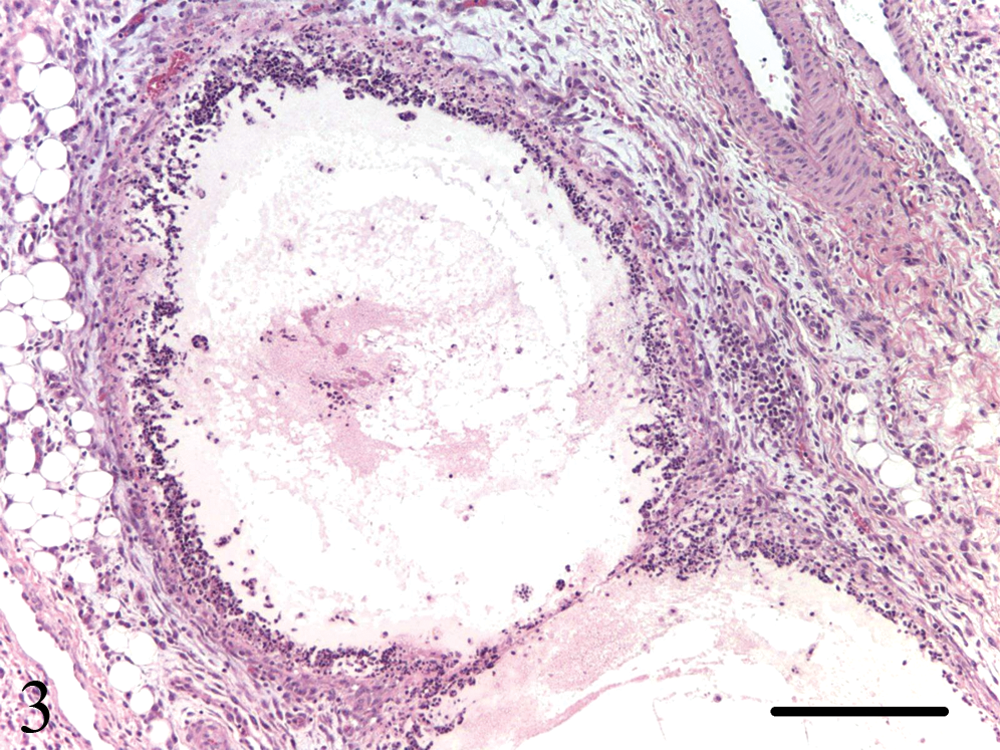
Mesentery of a germ-free pig vaccinated for Bovine viral diarrhea virus (BVDV) and inoculated with tissue homogenate containing Porcine circovirus-2b (PCV-2b) and BVDV-1–like virus (group 2). The wall of this markedly ectatic lymphatic is diffusely necrotic and inflamed (lymphangitis). Hematoxylin and eosin stain. Bar = 100 µm.
Experiment 2, CDCD pigs
Lesions are summarized in Table 4.
Day 14 euthanasia
—The PCV-2b–inoculated pigs (5/6) had lymphohistiocytic inflammation in lymph nodes, spleen, tonsil, lung, kidney, liver, heart, esophagus, stomach, and adrenal gland.
Day 35 euthanasia
—The PCV-2b–inoculated pigs (11/11, groups 2–4) had lymphohistiocytic inflammation in lymph nodes, spleen, tonsil, lung, kidney, liver, heart, esophagus, stomach, adrenal gland, and brain. Inflammatory lesions in kidney, lung, and brain at 35 days post–PCV-2 infection were significantly (p < 0.05) more severe in pigs inoculated with PCV-2b/BVDV-1 (group 3) than in the other pigs inoculated with PCV-2b (groups 2 and 4). Lymphoid lesions were most prominent in animals inoculated only with PCV-2b (group 2) and were minimal or absent in the pigs of the negative control group (group 5) and the pigs inoculated only with BVDV-1 (group 1). Chronic perivasculitis and vasculitis were identified in 1 out of three group 2 pigs (PCV-2b only), 1 out of four group 4 pigs (vaccination + PCV-2b/BVDV-1) and 3 out of four group 3 pigs (PCV-2b/BVDV-1). These lesions were characterized by endothelial cells hypertrophy, neointimal proliferation, segmental fibrinoid medial degeneration, and medial and adventitial lymphocytic inflammation, and affected medium- and small-sized arteries in the renal medulla, sublumbar lymph nodes, spleen, liver, and esophagus. The animal singly inoculated with PCV-2b that died had marked pulmonary edema associated with acute vasculitis characterized by hemorrhage and fibrinoid degeneration of small-sized arteries and arterioles in the lung and tubaric branches of the ovarian artery. The vascular lesions have been described in more detail elsewhere. 27 Negative control pigs and pigs singly inoculated with BVDV-1 had minimal lymphohistiocytic inflammation of the serosa, synovium, choroid plexus, liver, and lung.
In situ hybridization for PCV-2
In both studies, PCV-2 nucleic acid was demonstrated in tissues from all pigs inoculated with this virus independently of the viral subgroup they had received (Table 5). As previously described, 45 nucleic acid was most frequent in the cytoplasm and was most abundant in mononuclear and multinucleate cells in lymphoid tissues. Small amounts of nucleic acid were also demonstrated in nonlymphoid tissues, primarily in infiltrating macrophages and rarely in epithelial and endothelial cells. In the 2 animals that died, large amounts of intralesional PCV-2 nucleic acid were detected in both lymphoid and nonlymphoid tissues. In the germ-free pig, labeling was most intense in mononuclear cells infiltrating the lung, small intestine, and mesocolonic lymph vessels. In the CDCD pig, which had only minimal to mild inflammation of nonlymphoid tissues, large amounts of PCV-2 nucleic acid were detected in parenchymatous cells and resident mononuclear cells. In both animals, abundant intracytoplasmic PCV-2 nucleic acid was located in endothelial cells, smooth muscle–like cells, and inflammatory cells within and around affected arteries. 27 PCV-2 nucleic acid was also detected in rare endothelial cells in nonaffected vessels in multiple tissues of the other PCV-2–infected pigs with lesions. No PCV-2 nucleic acid was detected in any tissue of animals not inoculated with this virus.
Experiment 1, germ-free pigs: laboratory test results following experimental reproduction of Porcine circovirus-2–associated disease by coinfection of germ-free pigs with Bovine viral diarrhea virus 1 (BVDV-1)-like virus and Porcine circovirus-2 (PCV-2).*
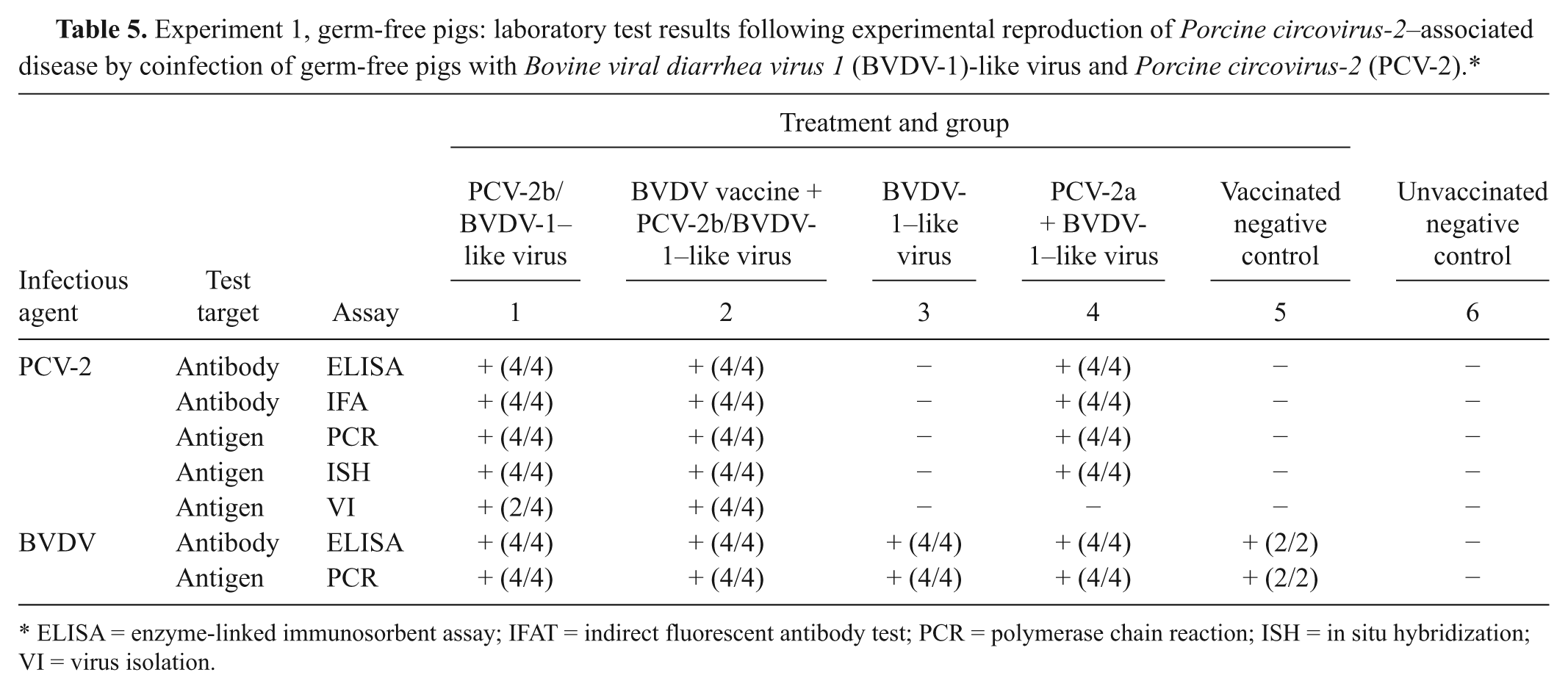
ELISA = enzyme-linked immunosorbent assay; IFAT = indirect fluorescent antibody test; PCR = polymerase chain reaction; ISH = in situ hybridization; VI = virus isolation.
Fluorescent antibody test for BVDV
Frozen sections of lung, ileum, and lymph node of BVDV-1–like pestivirus-inoculated germ-free pigs contained scattered positive cells with cytoplasmic immunofluorescence. In the lung, immunofluorescence was seen primarily in cells forming acinar-like structures near large airways (interpreted to be bronchial glandular epithelial cells). In the ileum and lymph node, immunofluorescent cells were scattered singly throughout the intestinal wall and the center of the node. Sections of neither unvaccinated nor vaccinated negative control pigs showed any staining.
Virus isolation
Experiment 1, germ-free pigs
PCV-2 was recovered at day 10 from several tissues of 4 out of 4 pigs inoculated with PCV-2b/BVDV-1–like virus (groups 1 and 2), as well as at days 21/22 from several tissues of 2 out of 2 pigs that received this inoculum following vaccination (group 2; Table 5). Virus was most consistently isolated from lymphoid tissues (lymph node pool, spleen, and ileum).
Experiment 2, CDCD pigs
PCV-2 was recovered from pools of lymph nodes and tonsils from 18 out of 24 PCV-2b–inoculated pigs (groups 2–4) that were euthanized at days 4, 14, and 35. BVDV was recovered from lymph node pool and lung of only the BVDV-1–inoculated pigs (groups 1, 3, and 4) that were euthanized at days 0 and 4.
Serology
Experiment 1, germ-free pigs
The results for PCV-2 and BVDV are summarized in Table 5. All pigs were seronegative for PCV-2 on days 0 and 10 by ELISA and IFAT. Serum antibodies to PCV-2 were detected at days 21/22 in all pigs that received this virus (groups 1, 2, and 4). The pig of group 2 that died had an IFAT titer against PCV-2 of 1:80. The other pigs that were inoculated with PCV-2b/BVDV-1–like virus (groups 1 and 2) had an IFAT titer against the same virus of 1:1,280. The pigs inoculated with PCV-2a/BVDV-1–like virus (group 4) had an IFAT titer against PCV-2 of either 1:80 or 1:320. All pigs tested were negative for PPV, PRRSV, and M. hyopneumoniae–specific antibodies.
All pigs that had been vaccinated against BVDV (groups 2 and 5) were seropositive for this virus at day 0. In the animals inoculated with PCV-2b/BVDV-1–like virus (group 2), the antibody titer against BVDV increased steadily until the end of the study. In the vaccinated negative control pigs (group 5), the titer peaked at day 10. Of the unvaccinated pigs inoculated with BVDV-1–like virus, at the time of euthanasia at days 21/22, 1 out of 2 animals in each group (1, 3, and 4) were regarded as having a suspect antibody titer, and 1 out of 2 pigs in group 1 was seropositive. All pigs were negative for PPV, PRRSV, and M. hyopneumoniae–specific antibodies throughout the experiment.
Experiment 2, CDCD pigs
All pigs were seronegative for PCV-2 and BVDV at the beginning of the study (day 0). Serum antibodies for PCV-2 were initially detected at day 14; all PCV-2–inoculated animals (groups 2–4) had seroconverted to this virus by day 35. The animal that showed clinical disease and died at day 30 was seronegative for PCV-2 at days 4 and 14. No serum sample was available from this animal thereafter. Samples of thoracic and lung fluid were tested instead, but antibodies to PCV-2 were not detected. The pigs in the negative control group and in the group inoculated only with BVDV-1 remained seronegative to PCV-2 throughout the experiment.
Neutralizing antibodies to BVDV were first detected at day 14. Whereas all animals singly inoculated with this virus (group 2) were seropositive at this day, only 2 out of 6 of the dual-inoculated animals (group 3) and 4 out of 6 of the vaccinated, dual-inoculated animals (group 4) were seropositive. By day 35, all BVDV-1–inoculated animals had seroconverted to this virus except for 1 pig in group 1.
All serum samples from all pigs at necropsy were seronegative to PRRSV and M. hyopneumoniae. One dually inoculated pig (group 3) seroconverted to PPV at day 14. At the termination of the study, the other 3 animals of the same group had also seroconverted to this virus.
Polymerase chain reaction
Experiment 1, germ-free pigs
PCV-2 nucleic acid was detected by quantitative real-time PCR in all serum samples and tissues collected at days 10 and 21/22 of pigs inoculated with this virus (groups 1, 2, and 4; Table 5). Most of the inoculated animals presented the peak of viremia at day 10, except for the pig that died at day 21. In tissues, circoviral DNA was generally most abundant in the lymph node and tonsil pool (mean average of 1.21 × 107 PCV-2 genome copy numbers per ml of 10% [w/v] tissue homogenate in all PCV-2–inoculated pigs except for the one that died, which had 3.98 × 107 PCV-2 genome copies), followed by lung, spleen, liver, and ileum, which had relatively equal amount of nucleic acid (mean average in most pigs varying between 9.96 × 105 and 2.53 × 105 PCV-2 genome copies per ml of 10% [w/v] tissue homogenate except for the one that died, which had between 3.09 × 106 PCV-2 genome copies in the liver and 1.53 × 108 PCV-2 genome copies in the spleen). PCV-2 DNA was not detected at any time during the experiment in samples from control pigs or animals not inoculated with this virus (groups 3, 5, and 6). Pestiviral infection was confirmed by quantitative real-time reverse transcription (RT)-PCR in all animals except for the strict negative control group. Pestiviral RNA was most abundant in kidney, liver, spleen, ileum, and gonads. All pigs were negative for PRRSV and PPV by PCR.
The PCR for TTV was negative on the filtered inoculum known to contain only BVDV-1–like virus, and was positive for genogroups 1 and 2 on the filtered inoculum containing PCV-2b and BDVD-1–like virus. TTV-1 was detected in the serum of the 2 pigs from group 2 that were vaccinated with BVDV, received the PCV-2b/BDVD-1–like virus inoculum, and euthanized at day 10. One of these pigs was already positive for TTV-1 at day 0. The other 2 pigs of this group, as well as those that received the same inoculum but had not been vaccinated (group 1), were positive for TTV-2 by the end of the study. Additionally, 1 pig from group 3 inoculated with the filtered inoculum known to contain only BVDV-1–like virus was also positive for TTV-1 at day 21.
Experiment 2, CDCD pigs
PCV-2 DNA was detected in serum and tissues of all pigs inoculated with this virus and necropsied on days 4, 14, and 35. Circoviral nucleic acid was also detected in aseptically collected joint fluid samples of pigs necropsied at day 14, following the arthritis outbreak. Levels of viremia for PCV-2, which peaked at day 14, were similar among the animals of the PCV-2b only (group 2; average of 1.66 × 105 PCV-2 genome copy numbers per ml) and PCV-2b/BVDV-1–inoculated (group 3; average of 8.24 × 104 PCV-2 genome copy numbers per ml) groups, and were slightly lower in the vaccinated, dually inoculated animals (group 4; average of 4.42 × 103 PCV-2 genome copy numbers per ml). Of the 3 pigs that remained until the end of the study in the group solely inoculated with PCV-2b (group 2), 1 had steadily increasing levels of viremia, reaching 4.28 × 104 PCV-2 genome copy numbers per ml at the time of euthanasia. The amounts of PCV-2 DNA increased most in the CDCD pig that showed clinical signs and died, with a mean average of 3.72 × 106 PCV-2 genome copy numbers per ml of 10% (w/v) tissue homogenate in all tissues examined. In the other clinically healthy animals inoculated with PCV-2, the average amount of PCV-2 DNA was highest at day 35 in PCV-2b/BVDV-1–infected pigs (average of 2.12 × 106 PCV-2 genome copy numbers per ml of 10% [w/v] lymph node pool, kidney, and brain homogenate, compared to an average of 8.90 × 105 PCV-2 genome copy numbers per ml of 10% [w/v] same tissue homogenates in the other animals).
Presence of pestiviral RNA was confirmed by RT-PCR in serum and multiple tissues of the animals infected with BVDV-1. Viremia was only transient in all groups. The viral load in the serum started day 0 (i.e., 4 days post-BVDV infection), peaked at day 4, and had disappeared by day 35. Based on RT-PCR, the levels of pestiviral nucleic acid were lower in serum and tissues of vaccinated, dually inoculated pigs than in samples of animals from the other 2 groups that received this virus.
Serum collected at days −31, −24, and 35 of the study was negative for PRRSV nucleic acid, and serum collected at days −31, −24, and −14 was negative for PPV nucleic acid. Although most of the pigs were negative for TTV-1 and -2 at the beginning of the study, all but 1 pig in each group (or 2 in the negative control group) were positive by PCR for TTV-1 at the time of euthanasia. Among the pigs that remained negative for TTV until the end of the study was the pig that died at day 30.
Discussion
Clinical signs and lesions typical of those observed in North America since 2004 in a more severe form of PCVAD were experimentally reproduced in 1 pig of each of the experimental studies. The germ-free pig that died was vaccinated for BVDV followed by inoculation with filtered homogenate containing PCV-2b and a previously unrecognized BVDV-1–like virus. A two-step vaccination protocol was used in order to combine the safety of an inactivated vaccine (administered at an age when the immune system was still developing in the piglets) with the good immune response induced by an attenuated live cytopathic vaccine strain (administered when the pigs were old enough to mount an adequate immune response). It was anticipated that clinical PCVAD in the vaccinated pigs would be prevented; instead fatal disease in 1 of 2 pigs that remained until the end of the study was produced. Therefore, vaccination with a commercial inactivated BVDV vaccine followed by infection with a viable cytopathic vaccine virus did not prevent PCVAD in this model. This was thought to be due to 1) the effect of immune stimulation (a single dose of a vaccine containing the adjuvant Quil A, a saponin derivative, was used), or 2) the much higher dose and effective replication of pestivirus used as modified live vaccine as compared to the low dose of the BVDV-1–like virus in nonvaccinated pigs, or 3) a combination of these 2.
Results of the germ-free study prompted a similarly designed study in CDCD pigs using the same PCV-2b virus and modified live vaccine BVDV as used in the first study. This culturable BVDV strain was chosen over the field virus because it allowed for a higher inoculum titer and therefore a better evaluation of the effect of BVDV infection in pigs co-inoculated with PCV-2b. In the second study, clinical signs and lesions typical of the more severe form of systemic PCVAD occurred in 1 out of 4 pigs solely inoculated with PCV-2b. The results contradict the hypothesis that ruminant pestivirus is a novel triggering agent in systemic PCVAD that accounts for the difference in clinicopathologic manifestation of the disease. The results are also in contrast to recent observations that PCV-2b is by itself not sufficient to induce severe disease. 14
The occurrence of high viral load and lack or reduced amounts of anti–PCV-2 antibodies in the pigs that died supports a strong relationship between immune response, viral load, and outcome of viral infection. The efficacy of the immune response early in the viral infection seems to determine whether clinical PCVAD develops.11,35,37 It has been hypothesized that most animals become infected during the preweaning period.28,46 Passively acquired antibodies through ingestion of colostrum and cellular immunity have previously been discussed as important in reducing viremia levels and protecting pigs from severe PCV-2–associated lesions.2,34
Vasculitis accompanied by pulmonary and mesenteric edema and cavitary effusions in the pigs that died was likely induced by PCV-2, given the intense labeling for circoviral nucleic acid within and around the affected vessels. In the germ-free study, the vascular lesions were restricted to the animal that died naturally. In the CDCD study, however, 6 out of the 12 animals inoculated with PCV-2b and necropsied at days 30 or 35 had vascular lesions, 1 with acute and the other 5 with more chronic vascular and perivascular lesions. These vascular lesions were recently described elsewhere. 27 Interestingly, all 6 CDCD pigs were the offspring of 2 related Landrace sows. This suggests a hereditary predisposition to the development of vascular lesions. This is consistent with previously reported experimental evidence that host genetics may increase susceptibility of animals to PCV-2–associated lesions and disease. 38
The pigs inoculated solely with BVDV-1 lacked gross lesions. Microscopic alterations were subtle and restricted to mild lymphocytic inflammation in multiple tissues. This confirms previous observations that BVDV infection in pigs usually develops without any clinical signs or significant lesions.29,31 Subclinical infection and mild disease are also the most common consequence of post-natal infection with BVDV in cattle. 44 Infected animals of this species may be viremic from 4 to 15 days after initial infection. Similarly, pigs in the current study were only transiently viremic, with lower levels of virus being present in the serum of vaccinated animals.
Multiple pigs in both studies were positive for TTV-1 and/or TTV-2 by the end of the experiments. TTV has been shown to be highly prevalent and widely distributed in pigs. 33 Infection with this group of viruses has been found in association with co-infections with several other viruses, primarily PCV-2 and porcine Boca-like virus. 4 Both TTV-1 and -2 have been implicated by some authors as another virus that facilitates PCV-2–induced systemic disease.9,20 Lesions of porcine dermatitis and nephropathy syndrome were detected in pigs with combined experimental infection with TTV and PRRSV, 24 and there may be a relationship between TTV-2 infection and reproductive failure in sows. 47 Other studies demonstrated that pigs with PCVAD were similarly more likely infected with TTV-2 than nonaffected pigs.4,20 In a different study, however, TTV viral load did not differ significantly between pigs that were PCV-2–negative and those affected with clinical disease by PCV-2. 30 Additionally, it has been shown that also pigs without PCVAD were positive simultaneously for PCV-2, TTV-1, and TTV-2.4,20,30 In the animal experiments reported herein, TTV-1 and -2 were detected in the filtered inoculum containing PCV-2b and BVDV-1–like virus. As a result of this, vaccinated germ-free pigs that received this inoculum were positive for TTV-1 at day 10, and all (vaccinated and nonvaccinated) germ-free pigs that received this inoculum were positive for TTV-2 at days 21/22, including the germ-free pig that died. In the second study, all but 1 CDCD pig of each group (or 2 in the negative control group) were positive for TTV-2 at the time of euthanasia. Most importantly, the CDCD pig that died in the second study was negative for TTV. Thus, the role of TTV as an aggravating factor of PCV-2 infection remains to be determined.
In the CDCD pig that died, no infectious agent that would serve as an immunostimulatory co-factor was detected. This is in contrast to the multiple other pigs in the same study that became infected with TTV or PPV before the termination of the study. PPV is a well-known co-factor of PCVAD. 1 Although co-infection with PPV did not lead to development of clinical disease, it may have influenced the severity of the lesions and the amount of circoviral nucleic acid, which at 35 days post–PCV-2 infection were higher in multiple tissues of the animals co-infected with PPV than in the other pigs inoculated with PCV-2b.
Multiple CDCD pigs of various treatment groups developed polyarthritis at 6–7 weeks of age that resolved in 1–3 days in association with antibiotic therapy. Polyarthritis is a common problem in young pigs that is usually a sequel to bacteremia or septicemia. 52 Early treatment reduces the duration of illness and mortality. Bacterial cultures were negative on joint fluid collected from affected animals that were necropsied at the time of the outbreak (day 0). This was likely due to interference from therapeutic antibiotics. PCV-2 and BVDV were detected in synovial fluid of affected animals in the groups inoculated with each respective virus; however, negative control pigs had similar clinical and pathologic findings despite testing negative for both viruses. The animals with polyarthritis had fibrous tags on multiple serosal surfaces, in addition to the typical lymphocytic synovitis that may persist after acute fibrinous synovitis. 48 These findings are indicative of infectious arthritis associated with bacteremia. Lack of colostral immunoglobulins likely predisposed the animals to such disease, 48 which was probably further complicated by PCV-2 and BVDV infection. The potential for interference by such a systemic bacterial infection in the proliferation of PCV-2 and/or in the immune response to this virus cannot be disregarded in the CDCD pig that died.
Evidence was found that variability in (subclinical) disease expression may be influenced by host genetics, which is different from the so-called “sow effect” on litter mortality as it relates to sow PCV-2 viremia and antibody titer against this virus. 5 Additionally, the findings suggest that the vascular system is a replication site of PCV-2, which could explain the multisystemic distribution of lesions in PCVAD. Further experimental studies are necessary to confirm these findings.
Footnotes
Acknowledgements
The authors would like to thank the staff of the Virology section of the Animal Disease Diagnostic Laboratory, Purdue University, West Lafayette, IN and the faculty and staff at the Animal Disease Research & Diagnostic Laboratory, South Dakota State University, Brookings, SD for their help in conducting these studies. Brian Denton is acknowledged for his help in the statistical analysis, and Kristy A. Mietelka for the revision of this manuscript.
a.
Esbilac®, PetAg Inc., Hampshire, IL.
b.
Triangle 1, Fort Dodge Animal Health, Fort Dodge, IA.
c.
MicroProbe, Fisher Scientific, Pittsburgh, PA.
d.
Roche Diagnostics Corp., Indianapolis, IN.
e.
VMRD Inc., Pullman, WA.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Kirkegaard and Perry Laboratories Inc., Gaithersburg, MD.
h.
HerdChek BVDV Ab, IDEXX Laboratories Inc., Westbrook, ME.
i.
PPV-Ab Svanovir, Svanova, Uppsala, Sweden.
j.
HerdChek PRRS, IDEXX Laboratories Inc., Westbrook, ME.
k.
HerdChek Mhyo, IDEXX Laboratories Inc., Westbrook, ME.
l.
MagAttract® Virus Mini M48 kit, Qiagen Inc., Valencia, CA.
m.
KingFisher, Thermo Fisher Scientific Inc., Waltham, MA.
n.
Tetracore Inc., Rockville, MD.
o.
Gentocin®, Schering Plough Animal Health Corp., Union, NJ.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The experiments were funded by the National Pork Board.
