Abstract
Antigen enzyme-linked immunosorbent assay (ELISA) is used for the detection of Bovine viral diarrhea virus persistently infected (BVDV PI) cattle; however, colostrum-derived antibodies may interfere with antigen detection in serum from young PI calves. Our study aimed to assess serum pretreatment methods for reducing such interference. Dilution of PI serum with serum containing specific antibody showed that antibody levels equivalent to those observed in colostrum-fed calves were able to eliminate all antigen signals in a serum sample. Serum was treated with ethylenediamine tetra-acetic acid at pH 4.5, 5.5, 6.5, and 7.5, then boiled, centrifuged, and the supernatant-recovered. BVDV antibody was undetectable by ELISA in supernatants from treated samples, and the antigen ELISA signal was improved. Maximum antigen signal recovery of >90% was achieved at pH 5 ± 0.5. When this optimal treatment method was applied to field samples from 3 PI calves (which were negative in the antigen-capture ELISA without treatment), the antigen signal improved and gave a positive result in each case. Pretreatment may provide an improvement in the detection of young PI calves.
Accurate testing allows for the control and mitigation of losses associated with Bovine viral diarrhea virus (BVDV) through the identification and eradication of persistently infected (PI) cattle. Various methods are available for the detection of PI cattle.1,4 Of particular relevance to control and mitigation programs, where large numbers of animals may require testing, is the antigen-capture enzyme-linked immunosorbent assay (ELISA). The ELISA is a simple, rapid, inexpensive testing method that does not require specialized equipment and is well suited to large-scale testing. However, the antigen-capture ELISA detection methodology has one major problem: colostrum-derived, BVDV-specific antibodies may interfere with detection of antigen in the serum of very young PI calves.2,5 As a result, alternative testing strategies are often employed in this cohort.
One such strategy is to delay testing until calves are a minimum of 12 weeks of age. 6 However, given the highly infectious nature of PI calves, this delay may result in substantial viral spread and is therefore far from ideal. Another testing strategy is to use an alternative method, particularly reverse transcription transcription (RT)-PCR, which is not subject to the same interference. 2 RT-PCR, however, is more expensive, labor intensive, and time consuming than antigen ELISA. RT-PCR can be used effectively on pooled serum (or ear notch supernatants) to eliminate large numbers of negative samples and thereby minimize the number of samples that require individual testing.1,4 However, pools that return a positive RT-PCR result must be further broken down and individual animals tested—often by antigen ELISA—where similar difficulties are encountered. Alternatively, serum samples may be replaced by ear notch samples in young calves. Although ear notch samples show a lower level of interference by colostrum-derived antibodies,2,3 ear notches still suffer a dramatic drop in signal following colostrum ingestion,2,5 resulting in a very narrow testing window and leading to a debate regarding the sensitivity of detection of PI calves using ear notches (Hill FI. Bovine viral diarrhoea virus—what test can I use? VetScript 2013;July:26–28).
Therefore, serum remains a preferred sample for BVDV detection; all animals, regardless of age, can be sampled under a single protocol, and a multitude of tests can be performed on serum, 1 including parentage testing and antibody testing (e.g., screening for adequacy of colostrum intake). As such, the ability to reduce the interference of colostrum-derived antibodies with the detection of antigen in the serum of young PI calves (and to eliminate the “detection gap”) would be of substantial benefit to BVDV control and mitigation efforts. Therefore, the aim of our proof-of-concept study was to assess serum pretreatment methods to increase the signal in the antigen ELISA in young, colostrum-fed calves.
Serum collected from a cow previously confirmed as PI with BVDV was serially diluted in either sample diluent a or pooled serum from 17 antibody-positive cows, resulting in samples ranging from neat PI serum to neat diluent or pooled antibody-positive serum. All dilutions were tested for the presence of BVDV-specific antibody or antigen by ELISA, according to the manufacturer’s instructions, a with results expressed as sample-to-positive (S/P) ratios or corrected optical densities (ODs), respectively.
When diluted in sample diluent, the PI serum was diluted 1:127 (<1% PI serum) before producing a negative antigen result (Fig. 1), whereas dilution in pooled antibody-positive serum resulted in a negative corrected OD from a dilution of 3:1 (75% PI serum) onward. This dilution series clearly demonstrated that the presence of BVDV-specific antibodies in a serum sample can eliminate the antigen detection signal when the same sample is tested by antigen ELISA. Antibody titers up to 104.8 have been observed in young PI calves following ingestion of colostrum. 2 Antibody ELISA results as high as a S/P ratio of 2 have been observed in PI calves in the first week of life. 5 Equivalent antibody levels (producing a S/P ratio of 2 or higher) are shown here to be sufficient to eliminate an antigen ELISA result of corrected OD > 3. Very low antibody levels (S/P ratio 0.3–0.5) produced a 3-fold decrease in antigen signal. A sample consisting of 50% pooled antibody-positive cow serum and 50% PI cow serum was identified as producing ELISA results that mimic those observed in young PI calves soon after colostrum ingestion when maximal interference is observed (antibody ELISA S/P ratio >2, negative antigen ELISA result) and, therefore, this 50/50 mixture was subsequently used as the experimental sample (Table 1).
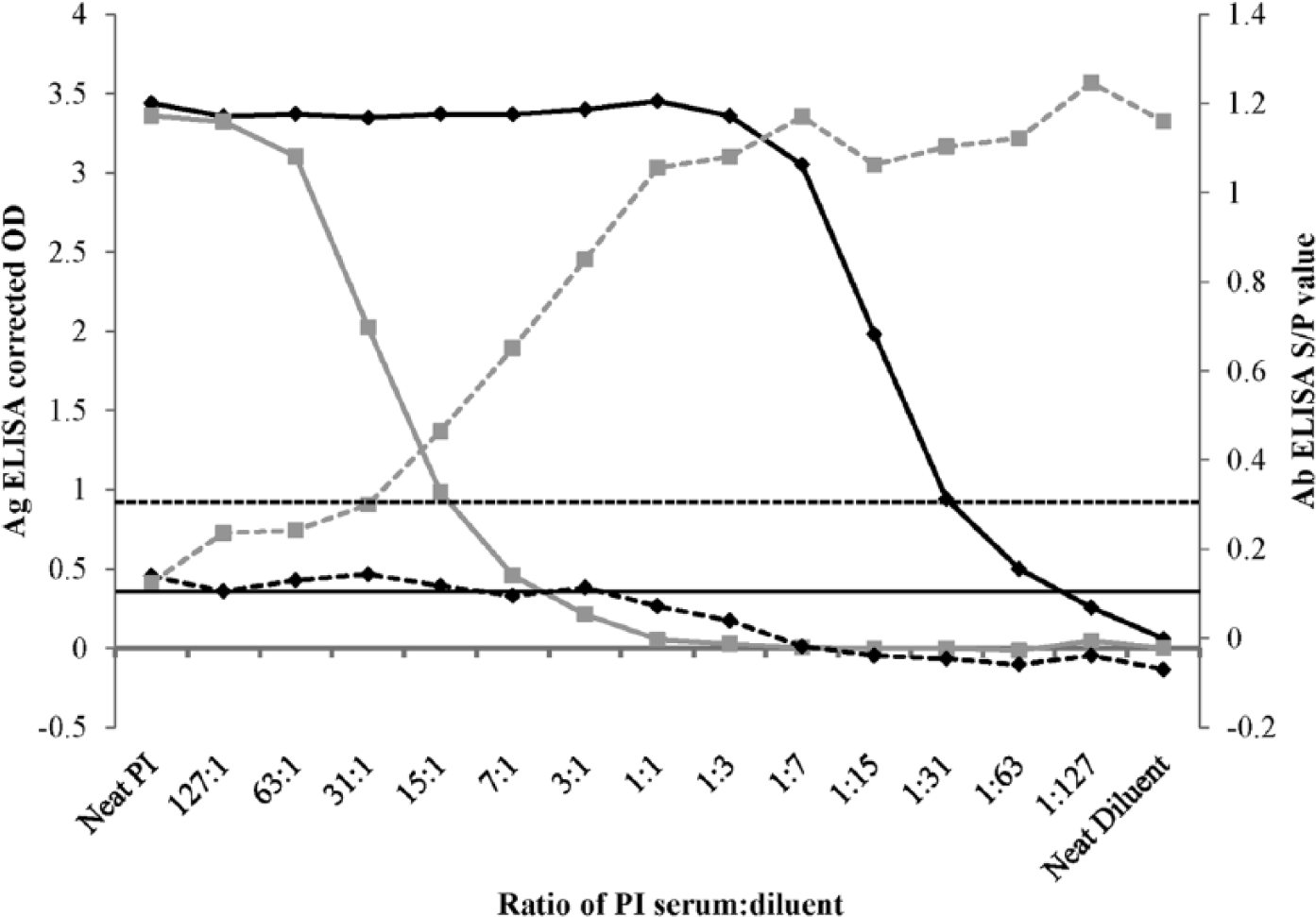
Levels of Bovine viral diarrhea virus (BVDV)-specific antibodies (Ab; dashed lines) and antigen (Ag; solid lines) as measured by enzyme-linked immunosorbent assay (ELISA) a in serum from a persistently BVDV infected (PI) cow, when serially diluted in either sample diluent a (black) or pooled serum from 17 antibody-positive cows (gray). Ag ELISA results are expressed as corrected optical density (OD) values. Ab ELISA results are expressed as sample-to-positive (S/P) value. Manufacturer’s cutoff thresholds are shown by horizontal lines for both Ab (dotted line) and Ag (solid line) ELISAs.
Composition of 3 samples artificially created to mimic Bovine viral diarrhea virus–specific antigen and antibody content of serum collected under various biological situations.*

PI = persistently infected.
Sample treatment methods were adapted from a published method. 7 An aliquot of 100 µL of each sample was treated by: 1) boiling for 7 min; or 2) addition of an equal volume (100 µµ) of 0.1 M Na2-EDTA (ethylenediamine tetra-acetic acid disodium salt), b at pH 4.5, 5.5, 6.5, or 7.5 (± 0.1) followed by boiling for 7 min. All treated samples were then centrifuged at 16,000 × g for 7 min and supernatant recovered. All supernatants, along with the untreated samples, were tested for BVDV-specific antigen by ELISA. a The treated and untreated negative antigen controls (Table 1) were also tested for BVDV-specific antibodies by ELISA. a Signal recovery was calculated as:

All sample treatments produced an increase in antigen signal in the experimental sample and a decrease in signal in the positive antigen control (Table 1; Fig. 2). The negative antigen control sample tested negative for antigen (corrected OD <0.1) regardless of treatment, and tested positive for antibodies when untreated (S/P ratio 1.4), but negative for antibodies (S/P ratio <0.1) after any treatment, suggesting that the specific antibodies were successfully removed from the samples. Treatment with 0.1 M Na2-EDTA pH 4.5 or 5.5 resulted in the highest signal recovery of 95% and 93%, respectively, while resulting in only small decreases in the signal of the positive antigen control. As such, treatment with EDTA pH 5 ± 0.5 was applied to field serum samples collected every 2 days, from the day of birth until 14 days of age and weekly until 5 weeks of age, from 3 PI and 7 non-PI calves born following natural in utero BVDV infection under experimental conditions as part of a previous research trial (University of Adelaide Animal Ethics Committee project S-2012-087). 5
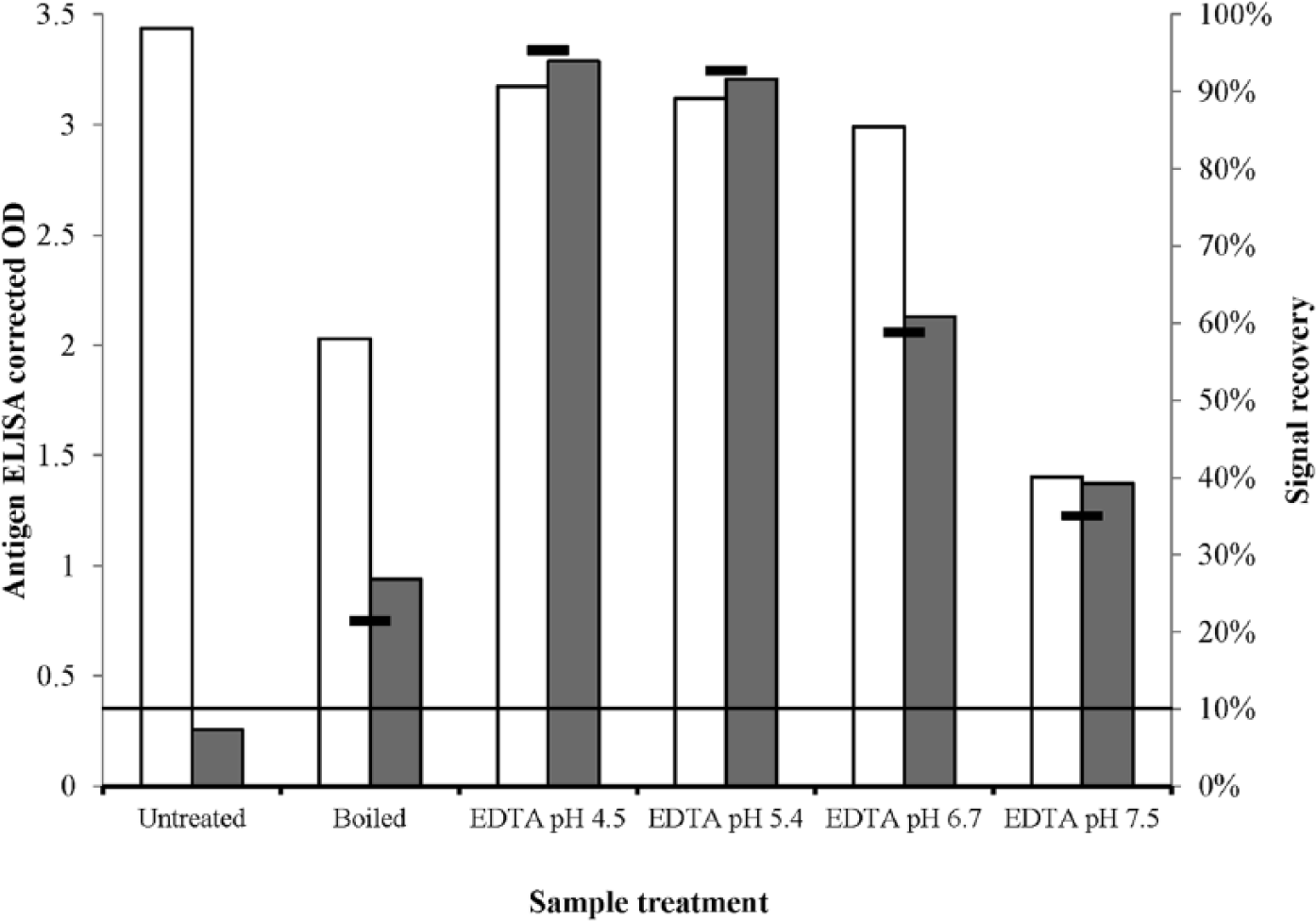
Detectable levels of Bovine viral diarrhea virus–specific antigen (Ag) as measured by enzyme-linked immunosorbent assay (ELISA) a and expressed as a corrected optical density (OD) in positive control (white column) and experimental sample (gray column), and the signal recovery (black dash) when treated: 1) by boiling for 7 min; or 2) by addition of an equal volume of 0.1 M Na2-EDTA b at pH 4.5, 5.5, 6.5, or 7.5 prior to boiling for 7 min. All samples were centrifuged and supernatant recovered and tested. Manufacturer’s cutoff threshold for Ag ELISA is shown by horizontal line.
On untreated serum samples from PI calves, the antigen ELISA returned a negative result until between 6 days and 4 weeks of age (Fig. 3). Following treatment, all 3 PI calves returned a strong positive antigen ELISA result (corrected OD >1.8) at all time points, providing evidence that pretreatment of serum samples can eliminate interference by colostrum-derived antibodies and improve the sensitivity of detection of PI calves. By comparison, the ear notch results of these same calves suffered a larger decline. 5 In particular, calf 03 returned antigen ELISA corrected ODs on ear notches below 1.5 (at times below 0.5), up to 2 weeks of age. 5 It is possible that use of this treatment method could also improve antigen detection in ear notch supernatants; however, sufficient supernatant volume was not available for inclusion in this study.
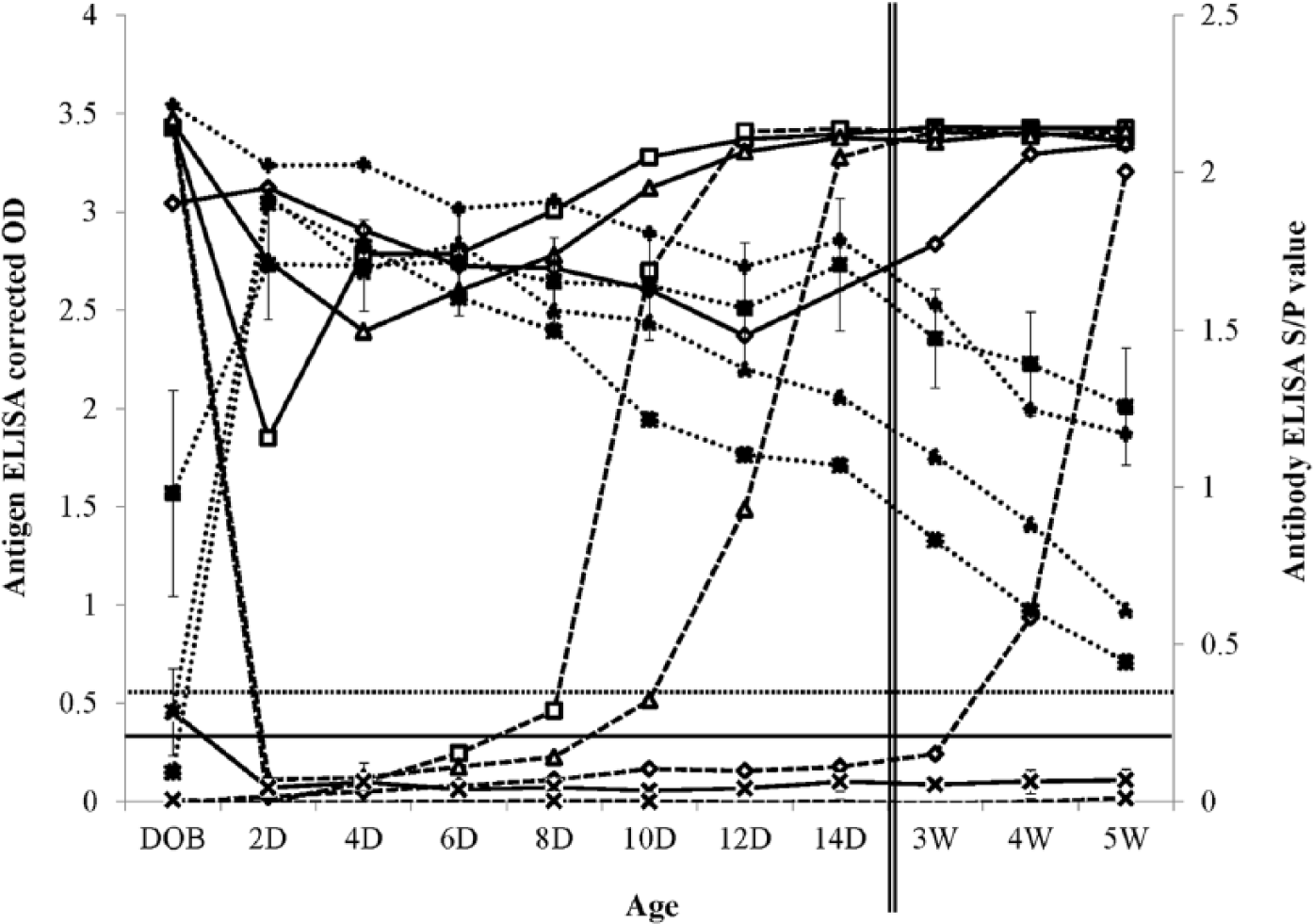
Detectable levels of Bovine viral diarrhea virus (BVDV)-specific antigen (Ag) and antibodies (Ab) as measured by enzyme-linked immunosorbent assay (ELISA) a in serum from 3 colostrum-fed persistently BVDV infected (PI) calves (03 ◊, 07 □, and 11 ∆) and the mean levels in 7 colostrum-fed non-PI calves (×) from day of birth (DOB) to 5 weeks of age (5W) when untreated (Ag, dashed lines; Ab, dotted lines) or treated by addition of an equal volume of 0.1 M Na2-EDTA b at pH 5 ± 0.5 prior to boiling for 7 min, centrifugation at 16,000 × g for 7 min, and recovery of the supernatant for testing (Ag only, solid lines). Error bars indicate 95% confidence intervals. Manufacturer’s cutoff thresholds are shown by horizontal lines for both Ab (dotted line) and Ag (black) ELISAs. D = days; W = weeks; corrected OD = corrected optical density; S/P value = sample-to-positive value.
When untreated, all (n = 7) non-PI calves returned negative antigen ELISA results at all time points (Fig. 3). When treated, 69 of 70 (98.6%) samples from non-PI calves returned negative antigen ELISA results at ≥2 days of age. On the day of birth, 4 of 7 non-PI calves returned positive antigen results (corrected OD >0.3; range: 0.37–0.91). This may be associated with the in utero BVDV infection that these particular calves underwent. 5 Further analysis of specificity of this method on non-PI calves and naive calves that did not undergo any in utero infection is necessary. An adjustment in the positivity threshold for use on treated samples, or further development of the method to reduce nonspecific reactions (e.g., blocking with bovine albumin or milk), may be appropriate.
Interference with the antigen ELISA by antibody in the serum of colostrum-fed PI calves was substantially reduced by pretreating serum samples using the method described. This method requires further validation beyond our limited proof-of-concept study, with inclusion of a greater number of PI calves and the use of truly BVDV naïve calves as controls. However, our study provides evidence that this method may provide an additional and important tool in the early detection of PI calves using serum samples and represents a significant advance in the field of BVDV detection. The method is simple, rapid, and inexpensive. It requires no more specialized equipment than a centrifuge, and further adaptation of this method to the 96-well format (which the authors believe will prove a simple task) could make this method suitable for large-scale testing, and open the possibility of incorporation into automated systems in the future.
Footnotes
Authors’ note
Preliminary results were presented as an abstract at the XXVIII World Buiatrics Congress, 27 July–1 August 2014; Cairns, Australia.
Authors’ contributions
SR Lanyon contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. MP Reichel contributed to analysis and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
BVDV Total Ab ELISA, BVDV Serum/Ag Plus ELISA; IDEXX Laboratories, Rydalmere, New South Wales, Australia.
b.
Sigma Aldrich Pty. Ltd., Sydney, New South Wales, Australia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The larger trial, from which the calf serum samples utilized in this project were sourced, was funded by Meat and Livestock Australia (MLA; project no. B.AHE.2014). All ELISA kits were supplied by IDEXX Laboratories Inc. Neither Meat and Livestock Australia nor IDEXX had any role in the study design, the collection, analysis, and interpretation of data, the writing of the manuscript, or the decision to submit the manuscript for publication.
