Abstract
The aim of the present study was to establish a relationship between the results obtained using the enzyme-linked immunosorbent assay (ELISA) technique for antibodies in blood serum and milk at herd level. For this purpose, 325 samples of bulk tank milk were analyzed with 4 antibody ELISAs from dairy herds with a prevalence of seropositive animals; seroprevalence was also evaluated. Data were arranged to analyze the sensitivity of the bulk tank milk test to detect herds with high risk of active infection (>65% seroprevalence) and the specificity to detect those with very few (<5%) or no (0%) seropositive animals, respectively. The sensitivity values ranged from 0.92 to 0.70 and the specificity from 0.83 to 0.54 to detect free herds (0% seroprevalence) and from 0.88 to 0.77 to detect herds with <5% of seropositive animals. In a quantitative approach, Pearson correlation coefficients, reported as a measure of linear association between herd seroprevalence and transformed optical density values recorded in bulk tank milk, ranged from 0.71 to 0.86. According to these results, the 4 antibody ELISAs would be valid tests for carrying out a herd classification program using milk samples.
Bovine viral diarrhea virus 1 and 2 (BVDV-1, BVDV-2; family Flaviviridae, genus Pestivirus) cause great economic losses because of reduced milk production, increased mortality of young animals, and the reproductive, respiratory, and intestinal problems that they produce. Infected animals are also more susceptible to other diseases.1,7 In adult animals, bovine viral diarrhea is typically subclinical, and neutralizing antibodies can be detected at 2–3 weeks postinfection. 10
To estimate the prevalence of BVDV antibodies, enzyme-linked immunosorbent assay (ELISA) is the most frequently used technique for individual samples of serum or milk. 3 Using ELISA to detect anti-p80/NS3 antibodies allows for distinction between antibodies resulting from natural infections and those originating from inactivated vaccines. Animals vaccinated with live vaccines also develop antibodies against p80/NS3 protein. 12
The adaptation of the ELISA technique for detecting antibodies from bulk tank milk (BTM) samples constitutes an inexpensive and reliable alternative for monitoring the evolution of disease control programs within dairy herds, as the test can provide information about the status of a large group of animals (lactating cows) from a single sample. 13 Along this line, several European countries have monitored dairy herds for several years using BTM antibody ELISAs.2,5,9,14 Previous studies have shown that the sensitivity of tests using antibody levels in bulk milk to predict the presence of persistently infected animals is close to 1, whereas the specificity is lower.2,9,14 The objective of the current study was to examine the relationship between within-herd seroprevalence and BTM antibody levels against BVDV using 4 commercial ELISAs.
For this purpose, a study was performed in 2004 in Galicia in northwestern Spain, which is the Spanish region with the greatest milk production (35% of total production). To evaluate the relationship between the levels of antibodies in a sample of BTM and the seroprevalence of the herd, 325 samples of BTM were used from 325 dairy herds with a prevalence of seropositive animals. Serum from every animal that was lactating at the time of serum collection was gathered less than 1 week prior to BTM collection. From the farms sampled, 24% (78/325) used polyvalent inactivated vaccines, whereas the rest of the herds were not vaccinated. The serum samples were obtained by puncture of the caudal vein. The samples were left to clot, and then centrifuged at 700 rcf for 5 min at room temperature. The serum was separated, and the samples were frozen at −20°C until used for antibody determination. The BTM samples were collected after homogenization and were also stored at −20°C after elimination of the fat fraction. After serum sampling, the farms were classified into the following categories according to the prevalence of seropositive animals: free herds (0% seropositive), low prevalence herds (<5%), herds with seroprevalence of 5–65%, and high prevalence herds (>65%), where BVDV is more likely to be present. 13 When tested blood samples indicated a possible active infection, this was confirmed in double serum or ear-notch sampling 2–3 weeks apart to detect viremic animals and determine the true status of the herd. Double sampling allows the differentiation of transient and persistent infections.
The farms were selected by means of stratified random sampling from samples submitted to the Laboratory of Animal Health and Production in Galicia (Laboratorio de Sanidade e Produción Animal de Galicia) in 2004. Farms were divided into strata based on herd size (<24, 24–32, 32–44, and >44 animals), which were the quartiles of the herd size variable in the studied population. Then, within each stratum, a simple random sampling was used. The mean number of lactating cows in these herds was 33.2. For analysis of the serum samples, a competitive ELISA was used, a and the cutoff point (≤50% inhibition) for the interpretation of the results was recommended by the manufacturer.
The BTM samples were analyzed with 4 commercial antibody ELISA tests. The ELISAs were designated as follows: ELISA A (BVD/MD/BD p80, used for the analysis of serum samples), a ELISA B (Civtest bovis BVD/BD p80), b ELISA C (HerdChek BVDV), c and ELISA D (Svanovir BVDV-Ab). d The A and B ELISAs are based on the detection of antibodies against the p80 structural protein. The other 2 tests indirectly detect total antibodies. All analyses were performed following the recommendations of the manufacturer, including the procedures for the determination of cutoff points (determined from the optical density [OD] of samples and controls). In ELISA A, the values were expressed as percentage inhibition [(sample/positive control) × 100], in ELISA B as [100 – percentage inhibition], in ELISA C as sample/positive control index, and in ELISA D as OD. Presence of viremic cattle was evaluated by antigen-capture ELISA based on the detection of the Erns viral protein (antigen serum plus BVDV test kit e ).
For analysis, to estimate the accuracy of BTM samples to detect free herds (0% prevalence), herds with low prevalence (<5%), and high positive herds (>65%), threshold values of transformed OD were calculated for each of the mentioned groups for the 4 ELISAs. For this purpose, initially, the mean values of transformed OD resulting from analysis of BTM samples were estimated for each of the prevalence groups. From these estimated mean values and their corresponding confidence interval, threshold values that allow the prediction of herd seroprevalence status from BTM were established. This was carried out by simple (linear) interpolation, as the straight line between the upper limit of the confidence interval of one prevalence group and the lower limit of the next one.
Subsequently, and according to these cutoff points, the accuracy of the 4 BTM tests was evaluated in terms of sensitivity (Se), as the proportion of herds where BVDV were more likely to be present (>65% prevalence), and specificity (Sp), as the proportion of free (0%) or low positive (<5%). A t-test was conducted to evaluate the effect of vaccination on the OD values. Scatter plots were created by evaluating the correlation between within-herd prevalence and antibody levels in BTM using a quantitative approach along with the corresponding Pearson correlation coefficients (ρ).
Of the 325 farms used in the study, 77 had 0% seroprevalence, 18 had greater than 0–5% of seropositive animals, and 61 had more than 65%. The remaining 169 were in the interval of 5–65%. The cutoff points, calculated to establish the intervals of transformed OD values that enabled classification of a herd within a corresponding seroprevalence group from a BTM sample, are summarized in Table 1. Using these cutoff points, with respect to the reference test (results in blood serum), the highest Se of a BTM ELISA to detect herds suspected of harboring an active infection (>65% seroprevalence) was for ELISA A (0.92), followed by ELISA C (0.89), ELISA B (0.75), and ELISA D (0.70).
Threshold values of transformed optical density from different enzyme-linked immunosorbent assays (ELISAs) in bulk tank milk samples for the established herd seroprevalence levels.*
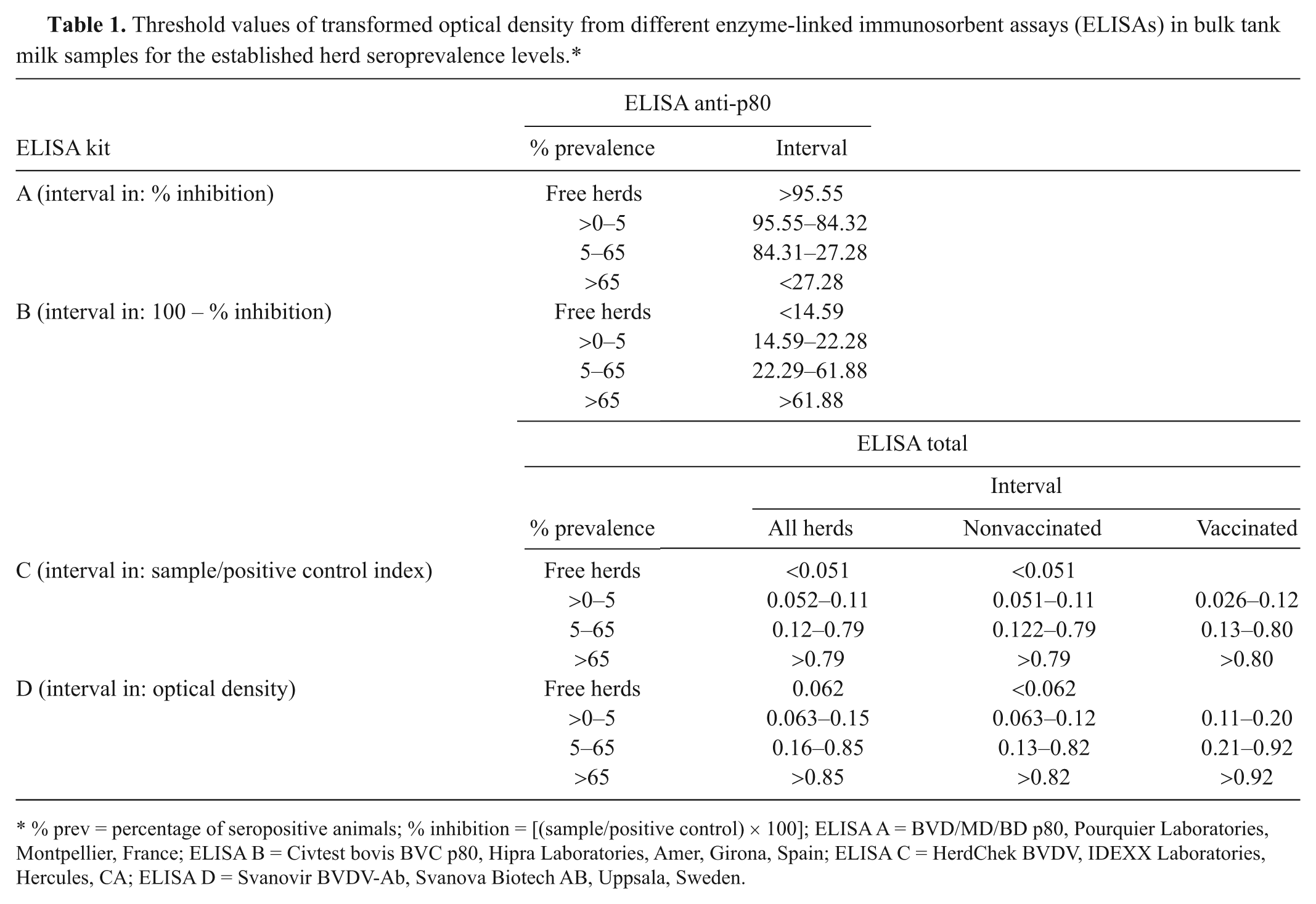
% prev = percentage of seropositive animals; % inhibition = [(sample/positive control) × 100]; ELISA A = BVD/MD/BD p80, Pourquier Laboratories, Montpellier, France; ELISA B = Civtest bovis BVC p80, Hipra Laboratories, Amer, Girona, Spain; ELISA C = HerdChek BVDV, IDEXX Laboratories, Hercules, CA; ELISA D = Svanovir BVDV-Ab, Svanova Biotech AB, Uppsala, Sweden.
For the group of herds with >65% of seropositive animals, 33.4% were harboring an active infection. The BTM antibody levels of infected herds were indistinguishable from those with high prevalence levels but cleared of BVDV. In the seroprevalence group of 5–65%, only 2.3% of the herds were infected (the lower within-herd prevalence found in an infected herd was 26.1%).
The highest Sp to merit classification of free herds (0% seroprevalence) was for ELISA D (0.83), followed by ELISA C (0.78), ELISA B (0.54), and ELISA A (0.61). Considering a herd as free with <5% seropositive animals, the Sp values increased to 0.77, 0.83, 0.86, and 0.88 for ELISAs A–D, respectively. Herds in this group, whose BTM samples predicted a prevalence higher than 5%, were mainly in smaller herds (<32 animals). With respect to results in serum, the test for BTM correctly classified inside the 5–65% group 49.1%, 60.3%, 63.9%, and 51.5% of the herds with ELISAs A–D, respectively.
In the p80 ELISAs (A and B), no differences in the OD transformed values were observed between vaccinated and nonvaccinated herds for the same seroprevalence group. Otherwise, in the ELISAs for total antibody detection, significant differences were observed between the mean values of the group of herds with <5% prevalence in ELISAs C and D.
The scatter plots showed good correlation between within-herd prevalences and transformed OD values from BTM samples for the 4 ELISAs in a quantitative approach (although the strongest correlation was with ELISA C; Fig. 1), which was also reflected in the ρ values: 0.76 for ELISA A, 0.71 for ELISA B, 0.86 for ELISA C, and 0.74 for ELISA D (P < 0.001 for all coefficients). Observed Se and Sp values, along with the scatter plots, reflected good accuracy and correlation of the results from the 4 antibody ELISAs that were tested in BTM samples.
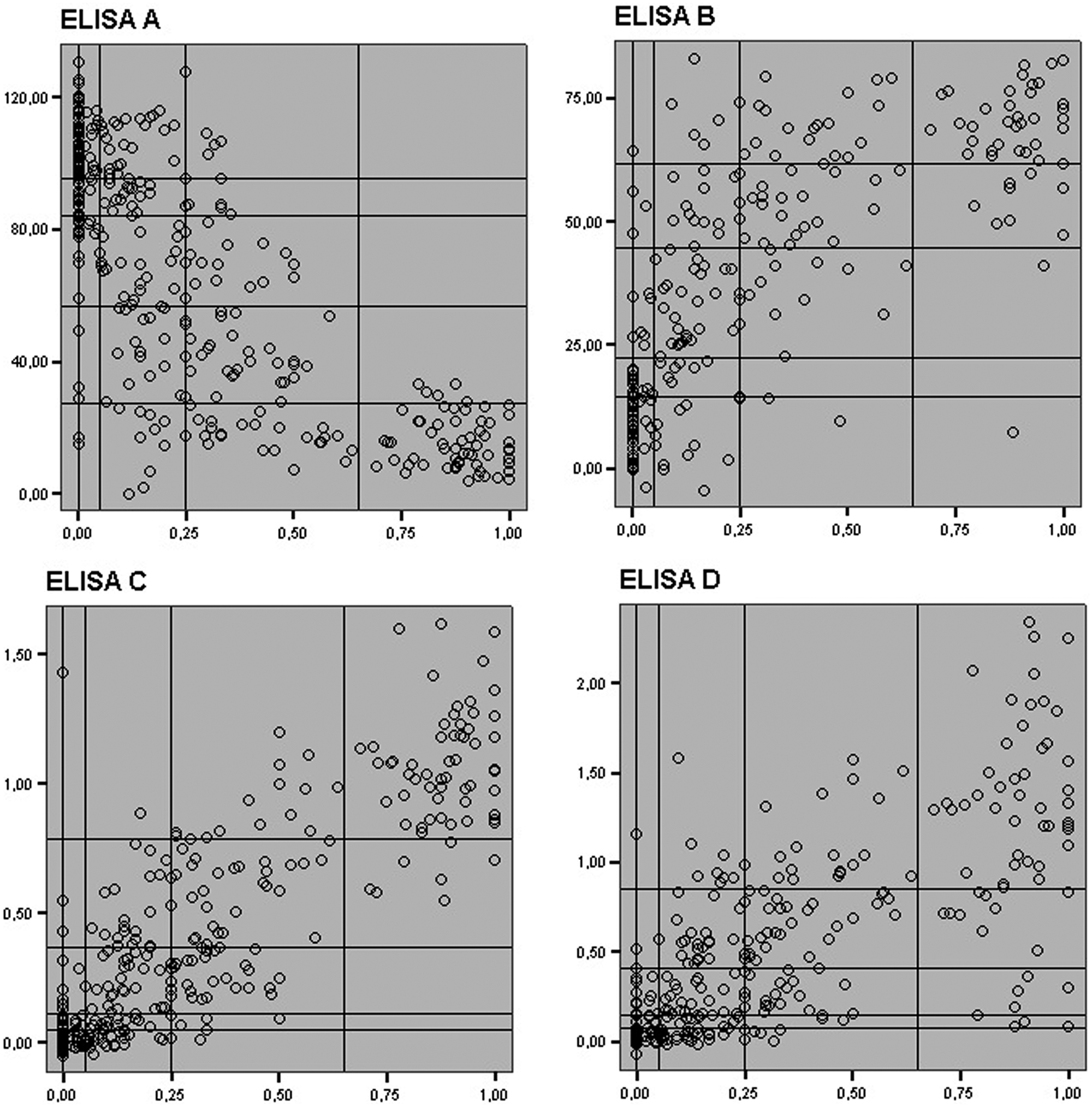
Scatter plots of herd seroprevalence (x-axis) and the level of antibodies in bulk tank milk (BTM; y-axis) expressed as % of inhibition (ELISA A), 100 – % inhibition (ELISA B), sample/positive control index (ELISA C), and optical density (ELISA D). ELISA = enzyme-linked immunosorbent assay.
A previous study, using ELISA D of the present study, considered that BTM samples with OD ≤20 had few (<26.5%) or no antibody-positive cows and could be considered not infected. 11 The present study includes separate cutoffs for herds with low prevalence (<5%) and those without positive animals, reaching good Sp values, especially in the first case. A BTM test with good accuracy to detect herds with less than 5% can be considered a good tool because these herds can be considered naive. 13 In the present study, none of the infected herds had a prevalence level lower than 26.1%.
Therefore, it is necessary to take into account that exceptions could lead to an incorrect classification of herds. Some discrepancies could be explained by varying contributions of individual cows to the BTM, as well as different milk yields of antibody-negative and -positive cows, or could even reflect the incorporation of new seropositive animals. 14
One BTM test does not distinguish actively infected herds from herds early infected but cleared of the virus, as it is known that after virus elimination, infected cows remain seropositive for years. However, a BMT sample that indicates a within-herd prevalence >65% seems to be sufficient to establish further investigations, as 33.4% of these herds were genuinely infected. It is also possible that the existence of a small group of animals with high antibody titers gave rise to a high level of antibodies in the BTM samples, indicating a higher seroprevalence than the actual prevalence,6,11 such that was observed in smaller herd sizes.
Conversely, if the infection is recent, levels of antibodies in BTM could still be low. Some authors also support the hypothesis that the presence of persistently infected animals in the lactation group may result in the neutralization of antibodies present in BTM samples from the virus excreted by these animals. 11 These factors should therefore be considered when interpreting BTM ELISA results. A repeated bulk milk test a few weeks later would solve such problems. 9
Once Se and Sp values have been estimated, predictive values can be estimated for a considered population based on its prevalence. When the results of vaccinated and nonvaccinated herds within each prevalence group were compared, there were only significant differences in values from the groups with low prevalence in the total antibody ELISAs. This difference could be because animals with natural infections have a level of antibodies that is much higher than levels found in vaccinated animals. 8 Many farms vaccinated with polyvalent inactivated vaccines showed low antibody levels, which could indicate that total antibody ELISAs were not sensitive enough or that vaccines did not induce a strong humoral response. 4 The vaccinated farms with low total antibody levels were consistent with those that had begun to vaccinate less than 2 years ago.
Therefore, the 4 ELISAs used in the current study showed acceptable correlation between the level of antibodies in BTM samples and herd seroprevalence that constitute another tool for controlling BVD in herds. Consequently, an initial herd classification could be determined and then monitored with additional samples to help control and observe bovine viral diarrhea infection over time. 5
Footnotes
Acknowledgements
The authors thank the visiting veterinarians and those working at the Laboratorio de Sanidade e Produción Animal de Galicia for their help in collecting the serum samples, as well as their colleagues from Seroloxía, ADSG, and Campañas de Saneamento Gandeiro for their help in processing and analyzing the samples.
a.
BVD/MD/BD p80, Pourquier Laboratories, Montpellier, France.
b.
Civtest bovis BVC p80, Hipra Laboratories, Amer, Girona, Spain.
c.
HerdChek BVDV, IDEXX Laboratories, Hercules, CA.
d.
Svanovir BVDV-Ab, Svanova Biotech AB, Uppsala, Sweden.
e.
Antigen serum plus BVD test kit, IDEXX Laboratories, Hercules, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by project PGIDIT02RAG50701PR of the Xunta de Galicia.
