Abstract
The transmission of bovine viral diarrhea virus (BVDV) from persistently infected (PI) heifers to adult seronegative goats was examined in this study. Ten seronegative adult goats were exposed to 4 PI heifers. None of the goats developed any clinical signs but all goats seroconverted by 42 days after exposure to the PI cattle. Results indicate that goats are susceptible to BVDV infection when housed with PI cattle.
Bovine viral diarrhea virus (BVDV) is a significant cause of morbidity and mortality among cattle in the United States, causing respiratory and reproductive disease, both of which lead to decreased productivity. The most damaging effect of BVDV infection is the establishment of a persistently infected (PI) animal; these cattle may appear to be asymptomatic while shedding the virus and infecting other animals. 5,10 The goat population in North America has increased dramatically in the past few years, in part as a result of increased demand for goat meat from ethnic populations. 25 In addition, livestock producers are encouraged to commingle goats with cows to efficiently use forage that cattle eat less preferentially and to control internal parasites by reducing the parasite load in the environment. 20 Pasturing goats with cattle, however, may increase the risk of BVDV infection in goats. The potential for BVDV transmission from cattle to goats and the generation of PI goats would have practical implications, such as BVDV abortions in goats and propagating PI goats that can serve as sources of BVDV infection for cattle and goats.
Pestiviruses and their natural hosts include BVDV of cattle, border disease virus (BDV) of sheep, and classical swine fever virus (CSFV) of pigs. These viruses are typically isolated from primary host species, but are capable of infecting other species. 17 The 4 recognized pestivirus species, BVDV 1, BVDV 2, BDV, and CSFV are genomically and antigenically very similar to each other. 1 Reverse-transcription polymerase chain reaction (RT-PCR) amplification and sequencing of the viral genomes has facilitated the identification of individual pestiviruses obtained from cases of naturally occurring disease. 13 For example, both BVDV and BDV may be isolated from cases of border disease (BD) in sheep or goats. 18
BVDV also infects pigs, 17 alpacas, 9 sheep, 2,11,23 goats, 4 and wild deer. 6,26 Of these species, BVDV infection of sheep and goats could be of the most importance in transmission to cattle because of the frequent pasturing of these species in common. Several outbreaks of BVDV infection in sheep have been reported, 2 where the most common manifestation of disease is reproductive losses. Inoculation of pregnant ewes at different points of gestation results in abortions and PI lambs. 23 Bovine viral diarrhea virus infections in nonpregnant goats are also subclinical. Disease in goats due to BVDV infection differs in sheep in that abortions are more commonly reported than PI offspring. 4,18
Goats have been reported to be infected with BVDV by natural transmission. 7,14,15,27 Seropositivity among goat populations varies around the world: 1.8% 7 to 16% in North America (1982–1983) 15 ; 31.4% in Africa (1998) 27 ; and 11.5% in Europe, 2006. 14 Review of the current literature indicates that goats can be experimentally infected with BVDV by direct inoculation. 4,16,19 In a previous study, 12 pregnant goats were injected intramuscularly with a Norwegian cytopathic BVDV of unknown type, and all were found to be seropositive by 3 weeks after inoculation. 16 A subsequent study by the same investigators found that 50 goats inoculated intramuscularly with a noncytopathic ovine pestivirus or a cytopathic bovine pestivirus all developed pestivirus-neutralizing antibodies within 5 weeks of inoculation. 19 In a third study, 25 goats were inoculated with either a cytopathic or a noncytopathic virus via the intranasal or subcutaneous route of administration. All 25 of these goats produced neutralizing antibodies within 3 weeks of inoculation. 4
Production of a PI goat kid as a result of exposure of the dam to a PI calf at an unspecified time during gestation has been reported (C. Bachofen, H. Vogt, H. Stalder, M. Hilbe, P. Tshcudi, and E. Peterhans, 6th European Society for Veterinary Virology, abstract 42, 2005). The PI was verified by demonstrating viral antigen in the skin and repeated isolation of a noncytopathic BVDV isolate identical to the BVDV isolate from the PI calf. The PI kid remained clinically healthy until it became anemic and died at 18 months of age.
These studies and case reports indicate that goats are susceptible to BVDV infection and that BVDV transmission from cattle to goats occurs. The present study attempts to evaluate the transmission of BVDV of known genotypes from PI cattle to serologically negative adult goats.
Ten healthy, nonpregnant female goats between 4 and 6 years old were obtained for the study. Of the goats, 6 were dairy type (predominantly Alpine breeding) and 4 were meat type. All goats were tested by serum neutralization (SN), virus isolation (VI), and immunohistochemistry (IHC) of formalin-fixed ear-notch skin biopsies to ensure that they were negative for BVDV as well as for antibody to the virus. Before the study began, all goats were vaccinated against Clostridium perfringens types C and D and Clostridium tetani and dewormed with an oral moxidectin drench. a Experimental protocols were approved by the Oklahoma State University Institutional Animal Care and Use Committee.
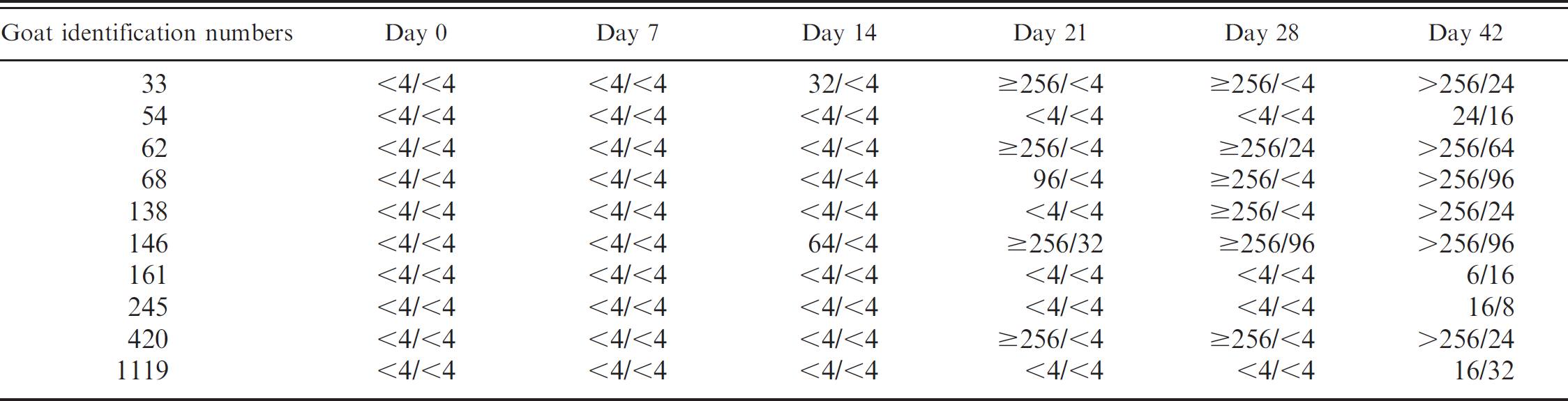
BVDV 2/BVDV 1 serum neutralization (SN) ratios.
Four PI heifer calves aged approximately 10 to 12 months were used for this study. Diagnosis of BVDV PI status was made on the basis of IHC performed on an ear-notch biopsy. The diagnosis was confirmed and the virus typed by RT-PCR. 13 The results indicated that 1 calf was persistently infected with BVDV lb and the other 3 with BVDV 2a.
Both does and heifers were housed together for the duration of the study in a 10,000-square-foot pen with a shed for shelter. All does and heifers had free access to prairie grass hay, approximately 20 lb of 14% protein alfalfa-based range cubes per day, and water from an automatic waterer. The goats were observed using the same waterer and feeding equipment as the heifers.
Serum was collected from each goat immediately before (day 0), and 7, 14, 21, 28, and 42 days after exposure to the heifers. Full-thickness ear-notch biopsies were also collected from all does upon completion of the study (day 42), and VI and SN against BVDV 1 and BVDV 2 were performed on serum samples from all goats at the initiation of the study.
The authors performed RT-PCR on RNA extracted from serum (total RNA was extracted from serum samples using QIAamp RNA blood purification kit e ) obtained 42 days after exposure using the Invitrogen One Step RT-PCR b with Platinum Taq. c Two universal BVDV primers were used: UTR DL1 (5‘gcc atg ccc tta gta gga cta gc-3’) and UTR DL4 (5‘-caa ctc cat gtg cca tgt aca gc-3’). The probe used was the TaqMan Probe c (5‘-FAM-tgg gca tgc cct cgt cca cgt ggc-BHQl-3’). Each reaction mixture contained 1− buffer, 1.8 mM magnesium sulfate, 0.3 μM UTR DL1, 0.3 μM UTR DL4, 0.1 μM probe, 0.5 μl reverse transcriptase and Platinum Taq DNA polymerase, and 4.85 μl RNase free water. The reaction conditions were as follows: 1 cycle of cDNA synthesis at 48°C for 30 minutes; 1 cycle of denaturation at 94°C for 2 minutes; and 45 cycles of denaturing at 94°C for 15 seconds and annealing at 60°C for 1 minute. 13
Immunohistochemical staining to detect the presence of BVDV antigen was performed on ear-notch skin biopsies obtained from goats 42 days after exposure. Paraffin-embedded skin was serially sectioned and mounted on poly-l-lysine-coated slides. The samples were stained for BVDV using monoclonal antibody BVD MAb 3.12Fl. d Positive controls consisted of fixed-skin specimens from calves that were confirmed to be PI by multiple viral isolations and positive BVD IHC staining. For the negative control, a nonimmune IgG fraction of mouse serum was substituted for the primary antibody. The slides were evaluated for the presence of positive staining characterized by red, distinct, granular intracytoplasmic staining in the epithelium of the stratum spinosum and stratum basale of the epidermis and follicular infundibulum in more than one location.
Clinical signs attributable to BVDV exposure, including general signs of malaise, lethargy, or inappetance, were not observed in the goats during the 42-day exposure period. The calves and goats did not often closely associate with each other in the pen, but were frequently within 10 feet of each other as they were eating, drinking, or resting. The 2 species ate together, and there was no evidence of bullying by the larger calves. All goats were seronegative (anti-BVDV antibody titer <4) for both types 1 and 2 BVDV-neutralizing antibodies 3 weeks before initiation of the study and on day 0. Ear notches from the goats were negative for BVDV by IHC staining, and serum was negative by virus isolation. The RT-PCR amplification and BVDV genotyping of serum samples from the PI calves confirmed prior test results.
Serologic responses against both BVDV 1 and BVDV 2 were detected in all goats by 42 days after exposure. Seroconversion to type 1 was detected in 1 of 10 goats 21 days after exposure and 2 of 10 by 28 days after exposure; all 10 were seropositive 42 days after exposure. Serum-neutralizing antibody titers to BVDV 1 at day 42 ranged from 8 to 96.
Serum-neutralizing antibody titers to BVDV type 2 were detected in 2 of 10 goats by day 14 after exposure and 5 of 10 by day 21 after exposure. At day 42, all 10 goats were seropositive; 6 of 10 samples had BVDV 2 SN titers >256, while the remaining 4 weakly positive samples had BVDV 2 SN titers from 6 to 24.
The serologic responses to BVDV of the goats in this study are similar to those found in cattle. While the SN antibody titers to BVDV in response to natural or experimental infection of cattle appear to be variable, 22 vaccination studies have shown that antibody titers in cattle may range from 8 to 32 at day 21 and from 16 to 128 on day 30. 12
Differences in time of infection and dose of virus for individual does may have occurred in this experimental exposure to PI cattle. Timid goats may not have been infected as quickly or with as large a dose as the more aggressive does that may have had some nose-to-nose contact with the PI heifers; this would explain the variation in time to seroconversion and the differences in SN titers at each time point. Transmission of the viruses from transiently infected goats to other goats could also explain these differences but was not examined in this study.
Although all goats seroconverted to BVDV, neither viral RNA nor viral antigen was found in serum and ear skin collected at 42 days after exposure. The lack of viral RNA in serum reflects the timing of sample collection, which was past the period of viremia characteristic of most acute BVDV infections. In acutely infected animals, viremia is transient and present up to 15 days after infection. 24 In addition, serum is less likely to contain virus as BVDV infects monocytes and lymphocytes found in the buffy coat fraction of whole blood. 8 Generally, only PI animals will have viral antigen detectable in skin, whereas transiently infected animals may not. 21
Serum-neutralizing antibodies from animals infected with one type of BVDV will cross-react with the alternate type in SN assays. 3 Therefore, it is not possible to determine which type of BVDV each individual goat was exposed to from the SN titer data. Future experiments exposing goats to PI cattle with a single BVDV type would provide clearer serology test results.
The results of this study demonstrate that goats exposed to PI cattle become infected, as evidenced by seroconversion. The results indicate that BVDV can be transmitted under natural conditions from PI cattle to goats. Previously published 1.8% and 16% prevalence rates of seropositive goats in Louisiana 7 and Quebec 15 indicate that BVDV infection in goats, although uncommon, does occur in North America. These data have important implications for the management of these two species with respect to shared facilities and pasture. Further research to assess the effect of PI BVD cattle on pregnant goats and their offspring is planned.
Acknowledgements. This work was supported by a grant from the Department of Clinical Sciences, Center for Veterinary Health Sciences, Oklahoma State University. The authors wish to thank Mr. Erick Loetz and the American Institute for Goat Research, Langston University, Langston, Oklahoma for providing the goats used in this study. Mr. A. C. Box of the Oklahoma State University Center for Veterinary Health Sciences Ranch was instrumental in providing daily care of the animals.
Footnotes
a.
Cydectin, Fort Dodge Animal Health, Overland Park, KS.
b.
Invitrogen, Carlsbad, CA.
c.
Life Technologies, Gaithersburg, MD.
d.
Oklahoma Animal Disease Diagnostic Laboratory, Stillwater, OK.
e.
Qiagen, Valencia, CA.
