Abstract
The objective of our study was to evaluate 2 pregnancy-associated glycoprotein (PAG)-based enzyme-linked immunosorbent assays (ELISAs) for use with either blood or milk. From 12 dairy farms, 116 Montbéliarde or Holstein cows were selected that had either undergone artificial insemination (AI;
Introduction
Accurate and timely detection of pregnancy in dairy cows is an essential component of today’s reproductive management programs. Veterinarians and farmers use early detection of nonpregnant (open) cows to enable faster rebreeding and shorten the calving interval, thereby maximizing milk production and farm revenue. Keeping cows open for additional days comes at a cost, estimated in U.S. dollars at $0.42 per day at 100 days open to $4.95 per day at 175 days open. 6 With a calving interval of 407 days instead of 1 year, the average economic loss is $0.62 (€0.57) per day of increase in calving interval, and can reach $0.76 (€0.70) per day with a calving interval of 507 days. 14 Thus, early detection of pregnancy is an essential component of reproductive management programs. Accurate and early diagnosis of open cows allows farmers and veterinarians to readapt their practices, such as better in-heat observation for these cows or implementing hormone treatment programs.
There are several methods available to determine if a cow is pregnant. The simplest is observation of estrus signs ~3 weeks after insemination. However, results obtained with this method depend on estrus detection ability. Mean detection rate is ~43.4% and thus far, too unreliable, 4 and ~5.7% of pregnant dairy cows show signs of estrus during pregnancy. 23 Concerning rectal palpation, premature examination can induce embryonic mortality, so in practice, this examination is generally performed at 50–55 days after artificial insemination (AI) 25 and thus, cannot qualify as “early” detection. However, with ultrasonography, transrectal examination becomes an accurate and simple method of diagnosis, with sensitivity reaching 100% 28 days post-AI (Filteau V, DesCôteaux L. Predictive values of early pregnancy diagnosis by ultrasonography in dairy cattle. Proc 31st AABP annual meeting; 1998 Sept 24–26; Spokane, Washington). Transrectal ultrasonography requires specific and expensive instruments, an experienced and skillful operator, and a good strain of animals. 1 In addition, there is also a set of analytical techniques to detect pregnancy. The oldest is a progesterone assay, ideally at 24 days post-AI, that can be performed with milk. 17 However, even though its sensitivity is acceptable (93.1%) its specificity is poor (39.3%), hence results are not usable in practice.
Finally, pregnancy can also be detected by detecting specific pregnancy-associated glycoproteins (PAGs). PAGs are powerful pregnancy markers in domestic cattle because they are part of a large family of glycoproteins expressed temporally across the gestational period. PAG expression occurs in the trophectoderm, predominantly by the binucleate trophoblast cells, and persists from the first day of gestation until calving. 9 At least 22 transcribed genes produce a pattern of these PAGs in cattle, but the expression and regulation of these genes vary according to the stage of pregnancy. 22 As PAGs are not expressed coordinately throughout pregnancy, early diagnosis by screening selected PAGs may be possible. 11 Publications report the use of PAG-1 (also known as pregnancy-specific protein B) in an immunoassay for the diagnosis of pregnancy using blood. 18 At 28 days post-AI, with a sensitivity of 93.9% and a specificity of 95.5%, this new method is promising. 18 Assays using bovine PAG-1 for the analysis of PAGs in milk have been demonstrated. 8 However, both the performance of the test, given the lower levels PAGs in milk relative to blood, and the described protocols suggest greater suitability in academic settings rather than test methods intended for use in higher throughput reference laboratories that generally prefer greater sensitivity and faster turnaround times. Some members of the PAG family, including PAG-4, PAG-5, and PAG-9, are expressed very early in pregnancy (by day 25) and have a short half-life 22 ; these specific PAGs are also undetectable by 6 weeks postpartum, making them ideal markers for detecting early pregnancy.
Considering these facts, a test method based on short-lived early-expressed PAGs for early and reliable pregnancy detection would be of interest. Two commercial PAG-based enzym-linked immunosorbent assays (ELISAs) can be used on either bovine blood samples a or bovine milk samples. b
The objectives of our study were 1) to test the blood pregnancy test (BPT) and the milk pregnancy test (MPT); 2) to compare the test results among sample matrices (serum, plasma, milk) and stage of pregnancy; 3) to determine the optimal cutoff for the MPT (the cutoff for the BPT was known prior to this study); and, 4) to verify the absence of interference of PAGs from previous pregnancy with PAGs produced since the last insemination. For this purpose, 116 dairy cows were monitored after calving and after insemination. Results obtained by BPT and MPT were compared with results obtained by the reference method, transrectal ultrasonography (TU).
Materials and methods
Animals, dairy farms, and sampling
Two veterinarians from the same practice selected animals from 12 French dairy farms located in the Bourgogne area. Cows from each breed, Montbéliarde and Holstein, were chosen for a total of 116 animals that were between 2.5 and 9 years old. Both veterinarians performed on-site sampling and examinations between February and May of 2011. Of the 116 cows in this study timeframe, 102 had been artificially inseminated. The remaining 14 cows had last calved 2–3 months prior but had not undergone AI since, but were being prepared for AI.
Blood samples were collected from the median caudal vein or artery into a 5-mL red-top serum tube containing a clot activator, and a 5-mL lavender-top tube containing 100 µL of a 15% solution of ethylenediamine tetra-acetic acid (EDTA) for plasma. Milk samples were collected from only 1 healthy quarter into a 25-mL screw-cap tube after elimination of foremilk. The 102 AI cows were sampled for serum, plasma, and milk at ~16, 30, and 41 days post-AI. Approximately 53 days post-AI, farmers obtained only milk samples from these cows. Milk and blood samples were taken from the 14 non-AI cows to verify the absence of PAGs that may persist after calving.
Sample storage
Serum and plasma were processed 1–3 h after sampling. After centrifugation at 3,000 ×
Pregnancy diagnosis by ultrasound
To confirm pregnancy, TU was performed on each cow that had been artificially inseminated. The procedure was conducted by a veterinarian who has 10 years of experience in performing this procedure on large ruminants. Pregnancy was detected if fluid and an embryo were observable in the uterine lumen. All confirmations of pregnancy by this method were completed on the same day as the third consecutive drawing of blood and milk, ~41 days post-AI. TU examinations were performed with a portable instrument c with a 5 megahertz linear array transducer. The use of TU at least 31 days post-AI is considered the reference test method, claiming 100% accuracy. 19 This method of confirming pregnancy was chosen for accuracy as well as safety because the use of TU at this stage of development has been demonstrated not to increase the rate of fetal loss. 2
Laboratory analysis
Samples were assigned unique identifiers, and technicians performing the ELISA were blinded to the pregnancy status of the cows. All samples were maintained frozen until the completion of the study. BPT and MPT are ELISAs designed for early detection of PAGs as a marker for pregnancy in bovine serum and plasma (BPT) or milk (MPT). Briefly, microtiter plates were coated with anti-PAG antibodies. After dilution (for serum and plasma only) and incubation at 37°C (1 h for serum and plasma, 2 h with shaking for milk), captured PAGs were detected with a PAG-specific antibody in the detector solution and a horseradish peroxidase conjugate. Unbound conjugate was washed away, and 3,3′,5,5′-tetramethylbenzidine substrate was added for color development, which is proportional to the amount of PAGs in the sample. The total assay times were ~2.5 h for BPT and 3.5 h for MPT. No defatting of milk samples is required to perform the MPT.
Interpretation of results
Two optical density (OD) values are measured for each individual sample and control at 450 nm and a reference wavelength of 620–650 nm. Corrected OD values for all samples and controls are the OD value at 450 nm minus the OD value at the reference wavelength. A “signal minus noise” (S-N) value is then determined for each corrected sample OD value by subtracting the mean of the corrected negative control OD values. Concerning BPT, if the S-N value is <0.30, the animal is considered not pregnant (open), and if the S-N value is ≥0.30, the cow is considered pregnant. Although evaluating an S-N cutoff value for MPT was an objective of our study, OD value measurements, OD value correction, and S-N calculations were handled similarly to those for serum and plasma.
Statistical analyses
Transrectal ultrasonography results taken at 41 ± 2 days post-AI were the reference test method results against which the ELISA results from serum, plasma, and milk samples were compared. Paired sample
Sensitivity indicates the probability that the ELISA correctly indicates pregnancy (Table 1; sensitivity = A/(A + C) × 100). Specificity is the probability that the ELISA correctly indicates a cow as open (specificity = D/(D + B) × 100). The positive predictive value (PPV) is the proportion of cows truly pregnant among cows identified as pregnant by ELISA (PPV = A/(A + B) × 100). The negative predictive value (NPV) is the proportion of cows truly open among cows identified as not pregnant by ELISA (NPV = D/(D + C) × 100). Accuracy is the proportion of true results (both true positives and true negatives) as determined by the reference test method relative to the total number of ELISA (accuracy = (A + D)/(A + B + C + D)).
16
For all parameters of sensitivity, specificity, PPV, NPV, and accuracy, 95% confidence intervals (CIs) for proportions were calculated according to the efficient-score method.
24
The characteristic test proportions for each sampling period were cross-compared using a McNemar chi-square test. A difference was considered significant at
Table used for calculations of sensitivity, specificity, positive predictive value, negative predictive value, and accuracy of the enzyme-linked immunosorbent assay (ELISA).

To determine an optimal cutoff value to interpret the pregnancy test on milk samples, a receiver operating characteristic (ROC) curve method was used.
26
A ROC curve analysis of the optimal cutoff S-N value was performed by plotting the true-positive rate (sensitivity, on the
Results
For the 4 sessions of sampling, the number of days post-AI was 14–19 days for the first session, 27–32 days for the second, 37–44 days for the third, and 52–56 days for the last session. Of 102 inseminated cows, 63 (61.8%) were identified as pregnant by TU, performed ~41 days post-AI, and 39 (38.2%) were identified as open at this stage. No false-positive results were observed when the serum, plasma, and milk of the 14 noninseminated cows were tested using samples obtained ~80 days after calving.
All ELISA results from serum, plasma, and milk samples obtained ~16 days post-AI were negative. This included negative results for all cows subsequently sampled and identified as pregnant as well as open via TU at ~41 days post-AI, with 1 exception. There was 1 milk sample that, when tested, had an unexpectedly high S-N value of 0.901 (Fig. 1). However, both the serum and plasma samples matched to that milk sample were negative.
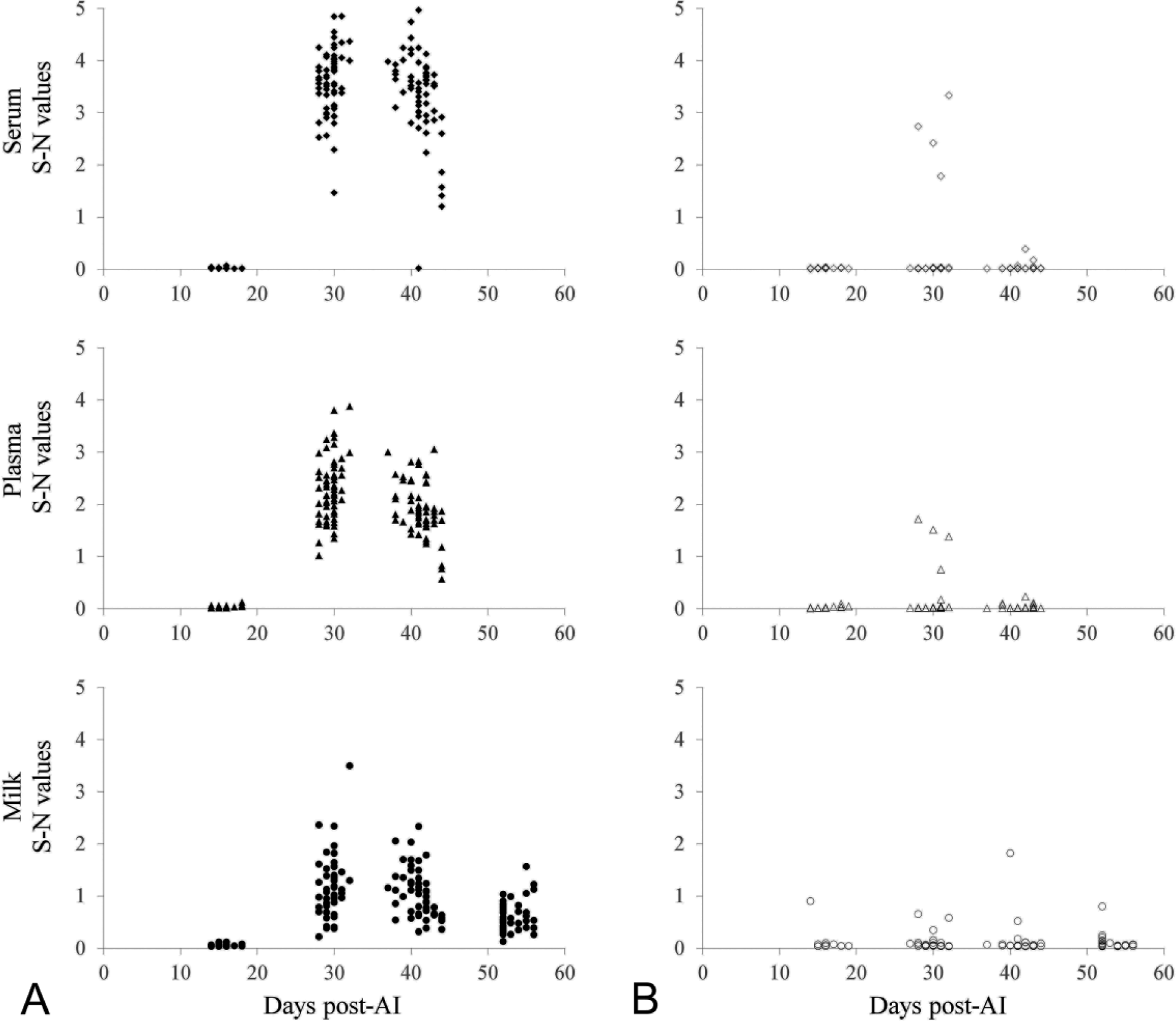
“Signal minus noise” (S-N) values from the serum, plasma, or milk enzyme-linked immunosorbent assays at 16, 30, 41, and 53 days post–artificial insemination (AI) for cows identified as either pregnant (
Of those cows identified as open by TU, 4 animals were indicated as pregnant by the serum and plasma ELISAs at 30 days post-AI. At 41 days post-AI, only 1 of those 4 animals initially identified as as pregnant remained positive in the serum ELISA, whereas all 4 were negative in the plasma ELISA (Fig. 1).
Of those cows identified as pregnant by TU, the serum ELISA result from only 1 animal was wrong. Mean S-N values (± standard error of the mean [SEM]) observed 30 days post-AI for serum and plasma were 3.613 (±0.080) and 2.290 (±0.076), respectively (
For serum and plasma obtained at ~30 days post-AI, sensitivity was 100% for both; specificity was 88.6% and 88.9%, respectively; PPV was 93.8% for both; NPV was 100% for both; and accuracy was 95.8% and 95.9%, respectively (Table 2). For serum and plasma sampled at ~41 days post-AI, test performance criteria were all between 96.8% and 100%. There were no significant differences in results at 30 days post-AI versus 41 days post-AI (
Sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and accuracy of 2 pregnancy-associated glycoproteins (PAG)-based enzyme-linked immunosorbent assays (ELISAs; a blood pregnancy test [BPT] on blood samples and a milk pregnancy test [MPT] on milk samples) using samples obtained on different days post–artificial insemination (AI).* The status of each animal was determined by transrectal ultrasound at 41 ± 2 days post-AI.
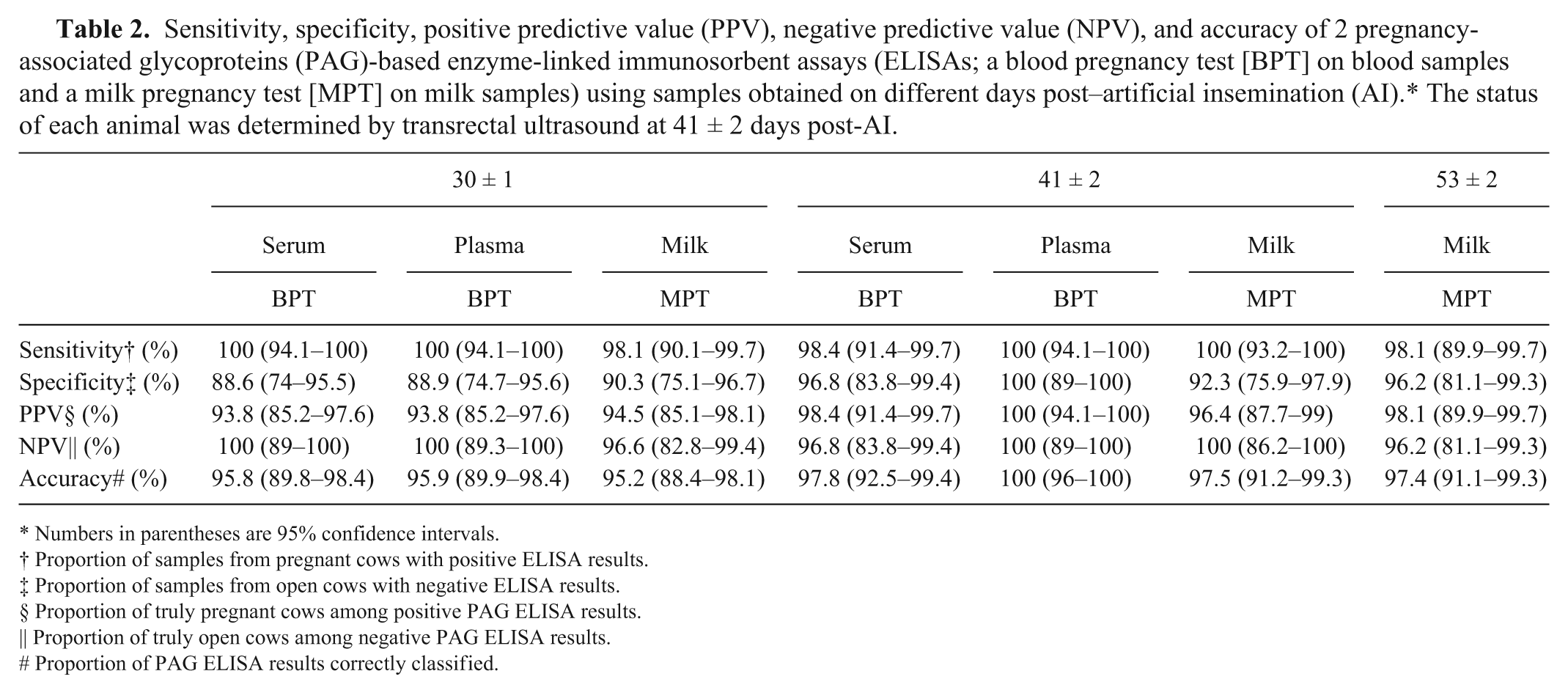
Numbers in parentheses are 95% confidence intervals.
Proportion of samples from pregnant cows with positive ELISA results.
Proportion of samples from open cows with negative ELISA results.
Proportion of truly pregnant cows among positive PAG ELISA results.
Proportion of truly open cows among negative PAG ELISA results.
Proportion of PAG ELISA results correctly classified.
Of those cows identified as open by TU, mean S-N values (±SEM) observed at 30, 41, and 53 days post-AI for milk were 0.105 (±0.027), 0.147 (±0.070), and 0.105 (±0.031), respectively (Fig. 1). Of those cows identified as pregnant by TU, mean S-N values (±SEM) observed at 30, 41, and 53 days post-AI for milk were 1.171 (±0.079), 1.028 (±0.064), and 0.581 (±0.040), respectively.
A key objective of our study was to determine the S-N cutoff value for MPT, using an ROC curve analysis. This approach for milk samples resulted in an optimal cutoff S-N value of 0.25 for MPT, with an AUC of 0.976 (Fig. 2). A milk sample with an S-N value of <0.25 would identify the cow as being open, whereas a milk sample with an S-N value that was >0.25 would identify the cow as being pregnant. At an S-N cutoff value of 0.25 for MPT, the following results were obtained: sensitivity was 98.1% for milk sampled at ~30 days post-AI; specificity was 90.3% (Table 2). For milk obtained ~41 days post-AI, sensitivity was 100%; specificity was 92.3%. For milk obtained ~53 days post-AI, sensitivity was 98.1%; specificity was 96.2%.
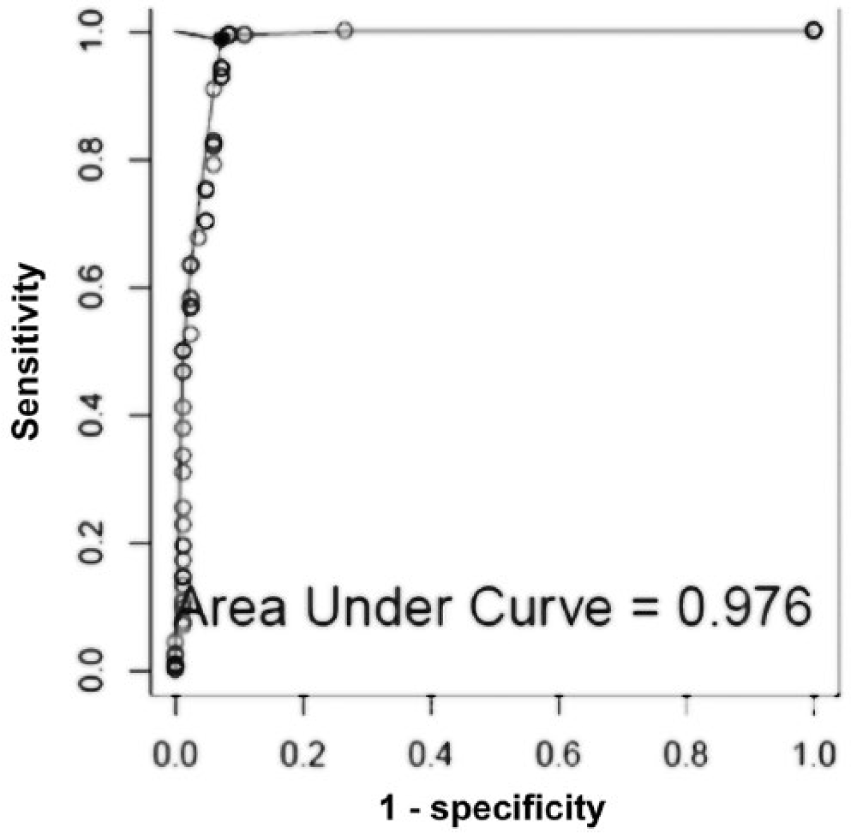
Receiver operating characteristic curve for “signal minus noise” values obtained from milk analysis.
Discussion
Blood tests based on detection of PAG-1 can only be performed on blood >90 days postpartum given the half-life of PAG-1. 12 In contrast, the reagents for both BPT and MPT used in our study were designed to specifically react with PAGs that are expressed early in pregnancy, allowing detection as soon as 25 days post-AI. 13 This subset of PAGs is also cleared more rapidly than PAG-1 from the blood, which allows sampling of cows as early as 60 days postpartum.
In this study, the performance of 2 ELISAs for the detection of PAGs was evaluated using paired blood (serum, plasma) and milk samples from AI (
Examination by TU was performed at 41 days post-AI, to avoid false-negative results as well as possible induction of embryonic loss as a result of the invasiveness of the procedure. If TU were performed 30 days post-AI, sensitivity and specificity of the method drops from 100% to 95% (95% CI: 89.7–100% and 92.7–99.5%, respectively). 19 Additionally, pregnancy detection made by ultrasound at 27 days post-AI have required reexamination at 39 days post-AI to increase diagnostic confidence.11,20 However, one disadvantage to having performed TU relatively late at 41 days post-AI is that potential pregnancy losses between 30 and 41 days post-AI would not be discovered.
Four cows identified as open via TU at 41 days post-AI were indicated as pregnant by ELISA using all 3 sample types (serum, plasma, milk) at 30 days post-AI. By 41 days post-AI, the serum result from only 1 of the 4 cows remained positive, having an S-N value just slightly above the cutoff, but well below the average of those cows in that group that were both positive in the ELISA and identified as pregnant via TU. A study of 19 cows that had aborted found that, within 9 days following fetal loss, PAG concentrations return to levels observed in open cows. 11 It is likely that 4 cows (6%) experienced fetal loss in our study. Pregnancy loss generally ranges from 0.4% to 10.6%, 5 and a 2006 study described a pregnancy loss rate of 3.7% in the 33–40-day post-AI stage, 21 which is consistent with the value obtained herein. If the data from these 4 cows were excluded, and the performance of the ELISAs reevaluated at 30 days post-AI, sensitivity, specificity, and accuracy for the BPT would be 100%, and accuracy for MPT would be 98.8%. Pregnancy losses could well occur during routine testing. With a pregnancy ELISA based on the detection of PAGs, false-positive results may be obtained when sampling occurs relatively early at 30–40 days post-AI (Cosson JL, et al. Application of a new pregnancy test for the economic management of breeding livestock. Proc National Meeting Rural Vets [GTV]; 2011 May 11–13; Nantes, France). Decrease in PPV because of embryonic mortality has a potential adverse impact on the value of such testing by falsely identifying cows as pregnant. Fetal loss will then only be detected if these cows are discovered to be in estrus or open at the next pregnancy check. However, resampling to identify embryonic loss was found to be economically advantageous taking into account multiple factors, including the cost of additional testing. 10
Negative ELISA results from cows that had calved 2–3 months earlier demonstrate the specificity of serum, plasma, and milk tests. To establish a baseline more representative of a nonpregnant population, the mean S-N values (±SEM) were reevaluated with the results excluded from the 4 cows falsely identified as pregnant. The mean S-N values (±SEM) for the open cows at 30 days post-AI were 0.018 (±0.001), 0.024 (±0.005), and 0.059 (±0.005) for the serum, plasma, and milk ELISAs, respectively. The mean S-N values (±SEM) for the open cows at 41 days post-AI were 0.020 (±0.002), 0.025 (±0.005), and 0.149 (±0.072) for the serum, plasma, and milk ELISAs, respectively.
Furthermore, S-N values of the pregnant cows observed with the MPT are lower than values observed with BPT. This is likely because of the lower concentrations of PAGs found in milk relative to PAG concentrations found in blood. 8 Lower S-N values were observed with milk samples obtained at 53 days post-AI relative to those samples obtained earlier, suggesting that PAG concentrations for individual animals may decrease by this time. In testing performed 60 days post-AI in a population of 683 lactating cows, with a pregnancy prevalence of 97% determined by veterinary diagnosis, the milk ELISA sensitivity and specificity (with 95% CI) were 99.2% (98.2–99.7%) and 95.5% (78.2–99.2%), respectively. 15 This indicates that, despite a possible decrease in PAG concentrations at 53 days post-AI, testing at 60 days post-AI using the milk ELISA is reliable and accurate. However, the MPT used in our study has been optimized to reliably detect PAGs in milk and allow a pregnancy diagnosis within the same timeframe and with similar levels of accuracy as that which can be obtained by the BPT. Before the development of this milk ELISA, results obtained with milk samples using a PAG-1–based ELISA would only be capable of detecting pregnancy >150 days. 7
The price of an MPT is ~$5.50 (€5). 3 However, maintaining an open cow costs from $0.62 (€0.57) to $0.76 (€0.70) per day, 14 and correctly identifying cows as pregnant helps to decrease the calving interval. So, using BPT or MPT to optimize the calving interval could help to increase the efficiency and profitability of the herd.
Footnotes
Authors’ contributions
L Commun contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. K Velek contributed to analysis of data; drafted the manuscript; and gave final approval. JB Barbry contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and gave final approval. S Pun contributed to acquisition of data; critically revised the manuscript; and gave final approval. A Rice contributed to analysis of data; critically revised the manuscript; and gave final approval. A Mestek contributed to interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. C Egli contributed to conception of the study and critically revised the manuscript. S Leterme contributed to conception and design of the study; contributed to interpretation of data; critically revised the manuscript; and gave final approval. L Commun, K Velek, JB Barbry, S Pun, A Rice, and S Leterme agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Bovine pregnancy test, IDEXX Laboratories Inc., Westbrook, ME.
b.
Milk pregnancy test, IDEXX Laboratories Inc., Westbrook, ME.
c.
KX5200VET, Echomedic, Brussels, Belgium.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: K Velek, S Pun, A Rice, A Mestek, C Egli, and S Leterme are employees of IDEXX Laboratories Inc., Westbrook, Maine. L Commun and JB Barbry declared no potential conflicts of interest.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
