Abstract
Production-limiting diseases in swine caused by Brachyspira are characterized by mucohemorrhagic diarrhea (B. hyodysenteriae and “B. hampsonii”) or mild colitis (B. pilosicoli), while B. murdochii is often isolated from healthy pigs. Emergence of novel pathogenic Brachyspira species and strains with reduced susceptibility to commonly used antimicrobials has reinforced the need for standardized susceptibility testing. Two methods are currently used for Brachyspira susceptibility testing: agar dilution (AD) and broth microdilution (BMD). However, these tests have primarily been used for B. hyodysenteriae and rarely for B. pilosicoli. Information on the use of commercial susceptibility testing products such as antibiotic gradient strips is lacking. Our main objective was to validate and compare the susceptibility results, measured as the minimum inhibitory concentration (MIC), of 6 antimicrobials for 4 Brachyspira species (B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii) by BMD and AD (tiamulin, valnemulin, lincomycin, tylosin, and carbadox) or antibiotic gradient strip (doxycycline) methods. In general, the results of a high percentage of all 4 Brachyspira species differed by ±1 log2 dilution or less by BMD and AD for tiamulin, valnemulin, lincomycin, and tylosin, and by BMD and antibiotic gradient strip for doxycycline. The carbadox MICs obtained by BMD were 1–5 doubling dilutions different than those obtained by AD. BMD for Brachyspira was quicker to perform with less ambiguous interpretation of results when compared with AD and antibiotic gradient strip methods, and the results confirm the utility of BMD in routine diagnostics.
Keywords
Introduction
Brachyspira-associated diarrheal conditions have a significant global impact on the health and productivity of commercial swine. The associated production losses in swine herds of decreased weight gain, high morbidity, and cost of treatment and/or culling are economically significant. 29 Swine dysentery, typically caused by Brachyspira hyodysenteriae, is characterized by mucohemorrhagic diarrhea in grower-finisher pigs. 9 In the late 2000s, however, a novel pathogen, designated as “Brachyspira hampsonii”, was isolated from swine dysentery-like cases in the United States. 2 A less severe disease characterized by mild colitis in grower-finisher pigs is caused by Brachyspira pilosicoli, 26 and, although Brachyspira murdochii is mostly considered a commensal, it has occasionally been isolated from some cases of mild diarrhea. 17 Because of the absence of any commercially available vaccines for brachyspiral diseases, control strategies focus on biosecurity and antimicrobial use. These efforts have been complicated by increasing reports of reduced susceptibility of Brachyspira toward commonly used antimicrobials worldwide.11,18,20,25 Yet, despite the central role of antimicrobials in the treatment and control of these diseases, there are no Clinical and Laboratory Standards Institute (CLSI) guidelines for susceptibility testing of Brachyspira.
Members of the genus Brachyspira are gram-negative, anaerobic spirochetes that show beta-hemolysis on tryptic soy agar containing 5% sheep blood (5% SB-TSA). 12 Although Brachyspira species, particularly the strongly beta-hemolytic species such as B. hyodysenteriae and “B. hampsonii”, show prominent zones of hemolysis, they show limited surface growth on an agar medium. 28 Their growth characteristics, hemolytic patterns, and fastidious nature has resulted in difficulty standardizing a method for in vitro antimicrobial susceptibility testing of Brachyspira species.
Agar dilution (AD) was often used until the late 1990s in both research and diagnostic settings to determine the susceptibilities of Brachyspira species toward commonly used antimicrobials.7,18,27 This method involved manual production of agar plates containing a defined concentration of antimicrobial and was labor-intensive and difficult to compare between laboratories. In the early 2000s, however, broth microdilution (BMD) was developed for evaluating the antimicrobial susceptibility of Brachyspira species. 13 This method utilized a readymade panel that is reported to yield reproducible results comparable between laboratories, thus showing better potential for use in routine diagnostic settings. None of the antimicrobials commonly used for controlling Brachyspira infections in swine herds are available in the form of commercially available products such as antibiotic gradient strips (hereafter, strip test). Furthermore, disc diffusion is not recommended for anaerobic bacteria by CLSI due to poor correlation with other methods for multiple antimicrobials, 5 and has also shown poor performance with Brachyspira species. 24
A majority of research studies evaluating the antimicrobial susceptibilities of Brachyspira over the past decade have used BMD rather than AD for Brachyspira susceptibility testing.10,11,15,16,22,25,30 Almost all of these studies have used BMD for evaluating the susceptibility of the pathogenic B. hyodysenteriae,10,11,15,16,25,30 and occasionally for B. pilosicoli. 22 Commensal species such as B. murdochii and novel pathogenic species such as “B. hampsonii” have only occasionally been evaluated by BMD.13,19 Additionally, studies comparing the susceptibility results of the 2 methods have only been conducted with B. hyodysenteriae, and most of them have been limited to pleuromutilin drugs.13,16,25 Information on the comparison of susceptibility results against a range of antimicrobials for diverse Brachyspira species by these 2 methods is currently lacking. Given the emergence and increased isolation of diverse Brachyspira species and the reports of reduced susceptibilities to a variety of commonly used antimicrobials globally, a comparative analysis of in vitro antimicrobial susceptibility testing methods is required.
Our study aims to meet the following objectives: 1) to evaluate the effect of incubation periods and their influence on antimicrobial activity when using BMD for diverse Brachyspira species; 2) to validate 3 in vitro antimicrobial susceptibility testing methods (BMD, AD, and strip test) for B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii; and 3) to study how the susceptibility results of other methods compare with BMD for 4 Brachyspira species (B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii), including 5 ATCC (American Type Culture Collection) reference strains.
Materials and methods
Brachyspira isolates
A set of 40 Brachyspira field isolates (10 each of B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii) were used in the comparison of susceptibility testing methods. These isolates were obtained from the University of Minnesota’s Veterinary Diagnostic Laboratory and were selected to represent diverse epidemiological sources (different swine production systems and geographical regions and/or states) in the United States. The following reference and type strains were also included in the comparative analysis: B. hyodysenteriae B-78T (ATCC 27164), B. hyodysenteriae B204 (ATCC 31212), B. pilosicoli P43/6/78 (ATCC 51139), “B. hampsonii” NSH-16 (ATCC BAA-2463), and “B. hampsonii” NSH-24 (ATCC BAA-2464). All cultures were revived from frozen ampules that were thawed. The cultures were passaged at least twice on 5% SB-TSA (incubated anaerobically at 37°C for 3 days) and checked for purity by phase-contrast microscopy before being evaluated by the susceptibility testing methods.
Antimicrobial agents
The antimicrobials evaluated in our study were selected because they are commonly used to control Brachyspira infections in swine and/or were available in the commercial BMD panel that is used as the reference test in this study. A total of 6 antimicrobials were evaluated by at least 2 susceptibility testing methods each and included pleuromutilins (tiamulin fumarate and valnemulin hydrochloride), a lincosamide (lincomycin hydrochloride), a macrolide (tylosin tartrate), a quinoxaline (carbadox), and a tetracycline (doxycycline hyclate). Doubling dilutions of all 6 antimicrobials were evaluated by BMD, of which 5 antimicrobials were included in a commercially available antimicrobial panel, a and carbadox was included in a specially requested custom-made BMD panel. For comparative purposes, doxycycline was evaluated by strip test b (as this was the only one of these antimicrobials available from the vendor), and doubling dilutions of the other 5 listed antimicrobials were evaluated by AD. The range of antimicrobial concentrations tested by BMD that were also used for comparative purposes with other test methods were as follows: tiamulin (0.063–8 µg/mL), valnemulin (0.031–4 µg/mL), lincomycin (0.5–64 µg/mL), tylosin (2–128 µg/mL), carbadox (0.002−0.125 µg/mL), and doxycycline (0.125–16 µg/mL).
In vitro antimicrobial susceptibility testing protocol for all 3 methods
In addition to purity checks before testing, every isolate was also checked for purity by phase-contrast microscopy and subculturing of material from the control well (BMD), control plate (AD), and growth zone (strip test method), at the time of recording the results. All isolates were tested in duplicate to evaluate the reproducibility of each method, and isolates with results differing more than 2 dilutions were repeated a third time to obtain a consensus value. Each batch of isolates evaluated by all methods included B. hyodysenteriae B-78T as a control strain. Bacteroides fragilis (ATCC 25285) was also included with each batch of isolates evaluated by the strip test method for additional quality control.
Broth microdilution method
Broth microdilution was performed as described previously 13 by using commercially available antimicrobial panels containing doubling dilutions of antimicrobials. a Briefly, 3-day old Brachyspira cultures were harvested from 5% SB-TSA plates and suspended in brain heart infusion broth c containing 10% fetal bovine serum d (10% FBS-BHI) to obtain a concentration of 1 × 108 organisms/mL. A 30-mL volume of final inoculum containing ~1 × 106 organisms of Brachyspira per mL of 10% FBS-BHI was produced by a 1:100 dilution. Previously prepared Brachyspira standards (confirmed by hemocytometer counts) were used for turbidity comparison to estimate the concentration of Brachyspira inocula by BMD. Each well was filled with 0.5 mL of this final inoculum, and the plates were incubated anaerobically at 37°C for 4 days on a shaker. The in vitro antimicrobial susceptibility was reported as the minimum inhibitory concentration (MIC), which is the lowest concentration of antimicrobial that inhibits visible growth (seen as turbidity in the well). Each panel contained 1 well with no antimicrobial that served as a growth control for each isolate evaluated.
In addition to B. hyodysenteriae strain B-78T used for quality-control purposes of BMD, 1 isolate each was selected to represent B. hyodysenteriae, “B. hampsonii” genetic group (formerly, clade) I, “B. hampsonii” genetic group (formerly, clade) II, B. pilosicoli, and B. murdochii for a comparison of the effect of incubation time on MIC results. The BMD protocol was followed as above; however, triplicate panels were inoculated with each isolate, and the results were recorded after each of 3 incubations periods (48, 72, and 96 hr).
Quality-control strains Enterococcus faecalis (ATCC 29212) and Staphylococcus aureus subsp. aureus (ATCC 29213) with available CLSI-accepted MIC ranges for veterinary-purpose antimicrobials (tiamulin and tylosin) were evaluated to determine the activities of these antimicrobials in the panels over time. The recommended incubation period for these quality-control strains is 24 hr, and the recommended incubation period for Brachyspira susceptibility testing by BMD is 96 hr. Therefore, to determine whether the antimicrobials degraded over the longer incubation period used by the Brachyspira BMD method, a similar protocol as detailed above was followed with minor modifications. These modifications included the use of 1-day-old cultures of these strains to create the inocula, and the use of 2 incubation periods (24 and 96 hr) each to evaluate the stabilities and activities of antimicrobials over the longer incubation (96 hr) required by BMD. Specifically, the longer (96 hr) incubation period involved an initial incubation of the panel (without bacteria) for 72 hr, followed by a second incubation of the panel (with bacterial inoculum) for 24 hr.
Agar dilution method
Antimicrobial plates containing concentrations of antimicrobials within the defined ranges were produced in the laboratory by using a single stock of each antimicrobial. The stock solutions of tiamulin, e valnemulin, f lincomycin, g and tylosin h were prepared by dissolving the powders in sterile distilled water. Carbadox i was dissolved in 1 N NaOH followed by sterile distilled water. These stock solutions were divided into aliquots needed for working concentrations and frozen at −20°C until further use. Test plates with defined antimicrobial concentrations were prepared by pouring the molten agar mixture (containing molten tryptic soy agar, sheep blood, and antimicrobial) into petri dishes. Control plates were prepared in a similar manner; however, the antimicrobial was substituted with sterile distilled water. All cooled plates were used for susceptibility testing on the same day as they were produced. A similar procedure of susceptibility was followed as those of previous studies,6,7,15,18,20 with some modifications. Briefly, 3-day-old Brachyspira cultures were harvested from 5% SB-TSA plates and suspended in 10% FBS-BHI to obtain an approximate concentration of 2 × 107 organisms/mL for each isolate evaluated. A 5-µL inoculum of each isolate (1 × 105 organisms/spot) was spotted onto the agar surface of each test and control plate, and a cut was created to allow for better visualization of hemolysis. The results were recorded after 2 days and 4 days of anaerobic incubation at 37°C. The in vitro antimicrobial susceptibility was reported as the MIC, seen as hemolysis around the inoculum cut.
Strip test method
Because members of genus Brachyspira are not listed within the CLSI or manufacturer’s recommendations, initial comparisons were made between commercially available Brucella blood agar j (strip test manufacturer’s recommendation for fastidious anaerobes) and Mueller–Hinton agar with 5% sheep blood k (5% SB-MHA; strip test manufacturer’s recommendation for doxycycline testing of Campylobacter spp.—an enteric fastidious microaerophilic organism). These initial comparisons used Bacteroides fragilis (recommended for strip test quality control), and B. hyodysenteriae B-78T (a control strain for in vitro antimicrobial susceptibility testing of Brachyspira). 21 As no differences in results were found during the initial comparisons, subsequent testing utilized 5% SB-MHA only for all isolates. The strip test was performed per manufacturer’s instructions using doxycycline strips (range: 0.016–256 µg/mL) available for research use. Briefly, 3-day-old Brachyspira cultures were harvested from 5% SB-TSA and suspended in BHI broth and then compared with a commercial turbidity standard l to obtain an approximate concentration of 3 × 108 organisms/mL. A cotton swab immersed in the suspension was used to evenly spread the inoculum onto the 5% SB-MHA plates and, after drying, a strip was applied onto the surface. After incubation anaerobically at 37°C for 4 days, the results were recorded as the MIC, which is the point of intersection between the edge of the growth zone and the strip.
Analysis
For a standardized comparison between the methods, doxycycline strip test results beyond the BMD test range were converted to “>maximum value” or “<minimum value” of the latter method. Similarly, all strip test MIC values were converted to the evaluated doubling dilution values of the BMD method. For AD, MIC results recorded at 4 days were utilized in all comparisons. For all 3 antimicrobial susceptibility testing methods, consensus values were used when available and, in the case of occasional isolates with varying results, the median value was used for evaluation. For comparing the differences between the in vitro antimicrobial susceptibility testing methods, all results were converted to logarithmic values (log2), and the differences in log2-transformed dilutions were determined. The agreement between the BMD and AD or strip test methods was determined by the ±1 log2 agreement (the percentage of MIC pairs that differed by ±1 log2 dilution or less) known as the “essential agreement” and ±2 log2 agreement (the percentage of MIC pairs that differed by ±2 log2 dilution or less) values. Bias between the BMD and AD or strip test methods was determined by paired t-tests between the log2-transformed MIC data for each pair of antimicrobial and Brachyspira species evaluated. 1 Positive or a negative bias value is reported as an overestimation or underestimation, respectively, of the test method (AD or strip test) as compared to the reference test method (BMD). All statistics were performed using a commercial software package, m and all analyses were supported by 95% confidence interval values to interpret their statistical significance.
Results
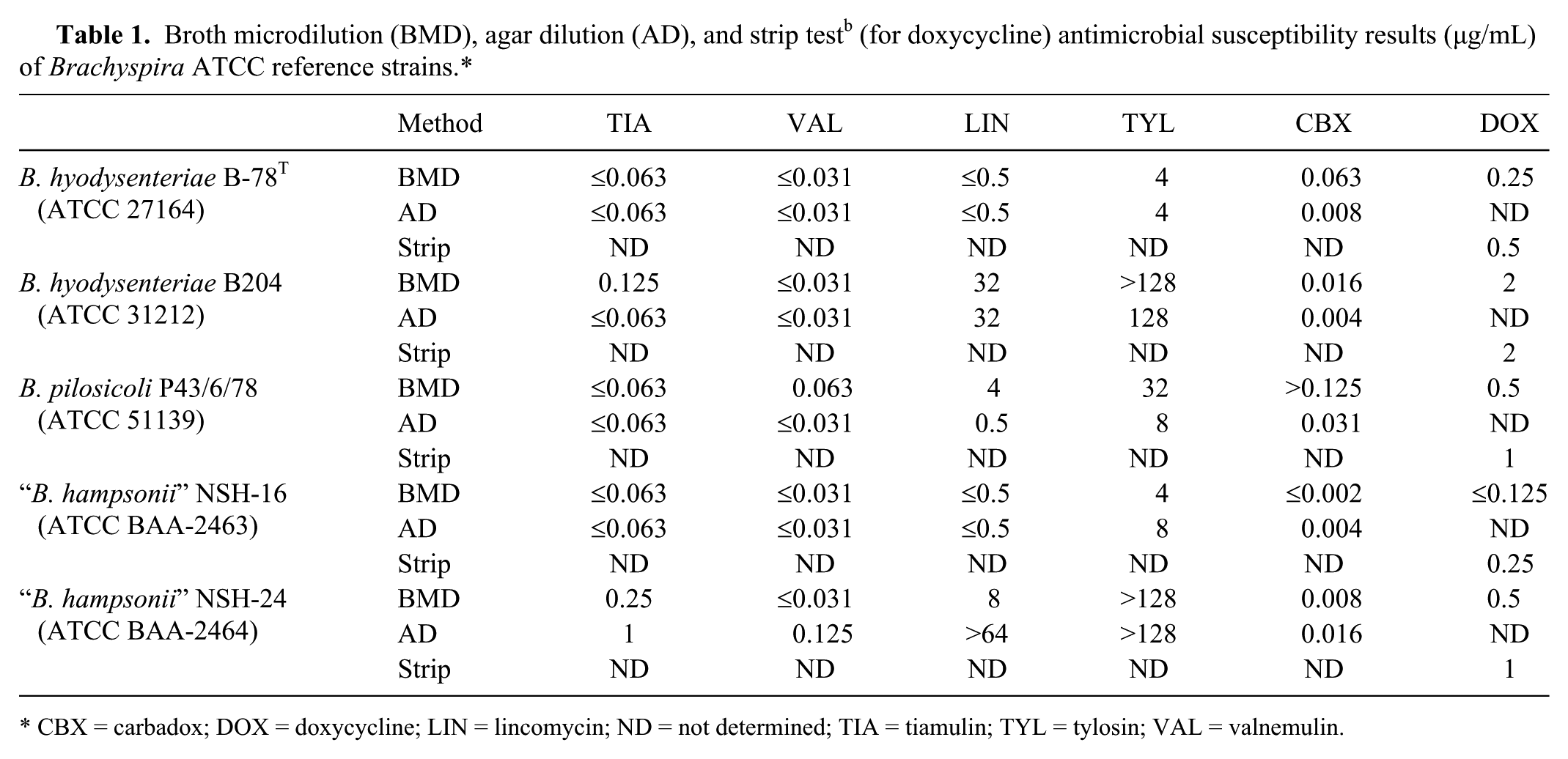
The MIC results of all Brachyspira ATCC type and/or reference strains are listed in Table 1. The results of strain B. hyodysenteriae B-78T for BMD were within the proposed quality-control ranges for evaluated antimicrobials. 21 In general, the tiamulin, valnemulin, lincomycin and tylosin MIC results for ATCC strains using the AD method (and strip test in the case of doxycycline) were either equal to or within 1 doubling dilution of those obtained by the BMD method. In contrast, carbadox MIC results by AD differed (usually lower) by 1–3 dilutions as compared with those obtained by BMD. However, “B. hampsonii” NSH-24 instead showed a different trend of lower MICs for tiamulin, valnemulin, and lincomycin by BMD than by AD, and showed closer agreement for carbadox MIC values between the 2 methods. Interestingly, when using BMD, B. pilosicoli P43/6/78 showed growth in wells with higher antimicrobial (lincomycin or tylosin) concentration despite the fact that inhibition of growth already occurred at a lower concentration, a phenomenon previously described as “skipped wells” 14 This resulted in 2 possible MIC values for lincomycin and tylosin for this strain and, to exclude the effect of resistance, the lower MIC values were reported in Table 1.
CBX = carbadox; DOX = doxycycline; LIN = lincomycin; ND = not determined; TIA = tiamulin; TYL = tylosin; VAL = valnemulin.
On comparison of incubation times for the BMD method, visible growth was seen for B. hyodysenteriae and B. pilosicoli within 48 hr of incubation and, for all 4 Brachyspira species, after 72 and 96 hr of incubation. Comparison between the results of 3- and 4-day incubation periods for all 5 Brachyspira isolates showed no change in MIC, although occasionally the growth was easier to visualize after the 96-hr incubation period. Susceptibility testing of E. faecalis and S. aureus subsp. aureus strains by BMD produced MIC results within the CLSI-recommended ranges for tiamulin and tylosin after the standard (24 hr) and longer (96 hr) incubation periods (Table 2).
Evaluation of antimicrobial activity (in µg/mL) in the broth microdilution panel after 24-hr and 96-hr incubation periods.*
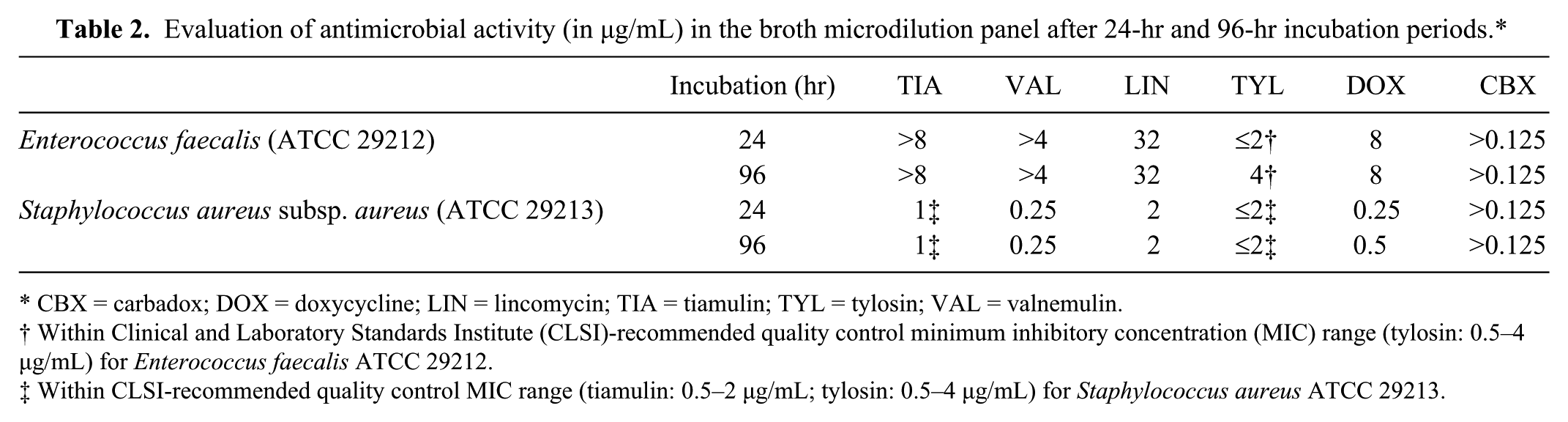
CBX = carbadox; DOX = doxycycline; LIN = lincomycin; TIA = tiamulin; TYL = tylosin; VAL = valnemulin.
Within Clinical and Laboratory Standards Institute (CLSI)-recommended quality control minimum inhibitory concentration (MIC) range (tylosin: 0.5–4 µg/mL) for Enterococcus faecalis ATCC 29212.
Within CLSI-recommended quality control MIC range (tiamulin: 0.5–2 µg/mL; tylosin: 0.5–4 µg/mL) for Staphylococcus aureus ATCC 29213.
Comparisons in the doubling dilution differences between the susceptibility results of all field isolates were analyzed by Brachyspira species and antimicrobial agent (Figs. 1, 2). While null dilution difference indicates the same result, positive dilution differences or negative dilution differences indicate that the BMD method results are higher or lower, respectively, than the AD or strip test method. When evaluated by Brachyspira species (Fig. 1) or specific antimicrobial (Fig. 2), a high percentage of isolates showed AD or strip test MIC results within 1 dilution of those obtained by BMD; a small percentage differed by 2 dilutions, and only rarely were differences more than 2 dilutions. However, a high percentage of all Brachyspira species tested against carbadox differed by more than 2 dilutions between BMD and AD, and this percentage was even higher for the weakly hemolytic species B. pilosicoli and B. murdochii. The susceptibility results of field isolates were stratified by Brachyspira species and summarized to compare the differences obtained by the 2 testing methods. The summary values include MIC50 (MIC value to which 50% tested isolates are susceptible), MIC90 (MIC value to which 90% tested isolates are susceptible), and the mode MIC values (Fig. 3). In general, MIC summary values were within 1 doubling dilution for BMD and AD or strip test methods, although carbadox BMD method values were higher than those obtained by AD.
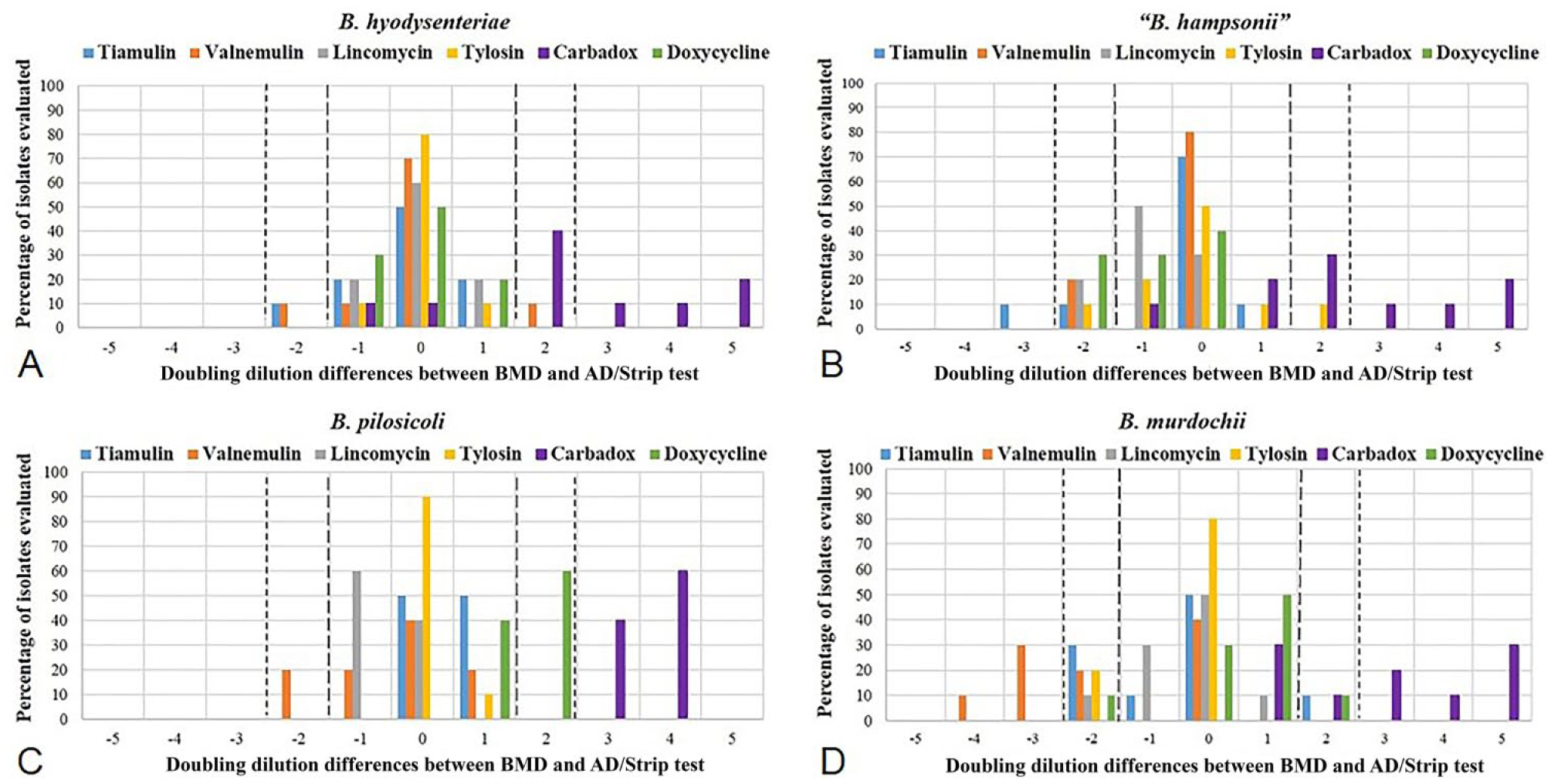
Distribution of differences in log2-transformed minimal inhibitory concentration data of 6 antimicrobials (tiamulin, valnemulin, lincomycin, tylosin, carbadox, and doxycycline) stratified by Brachyspira species.
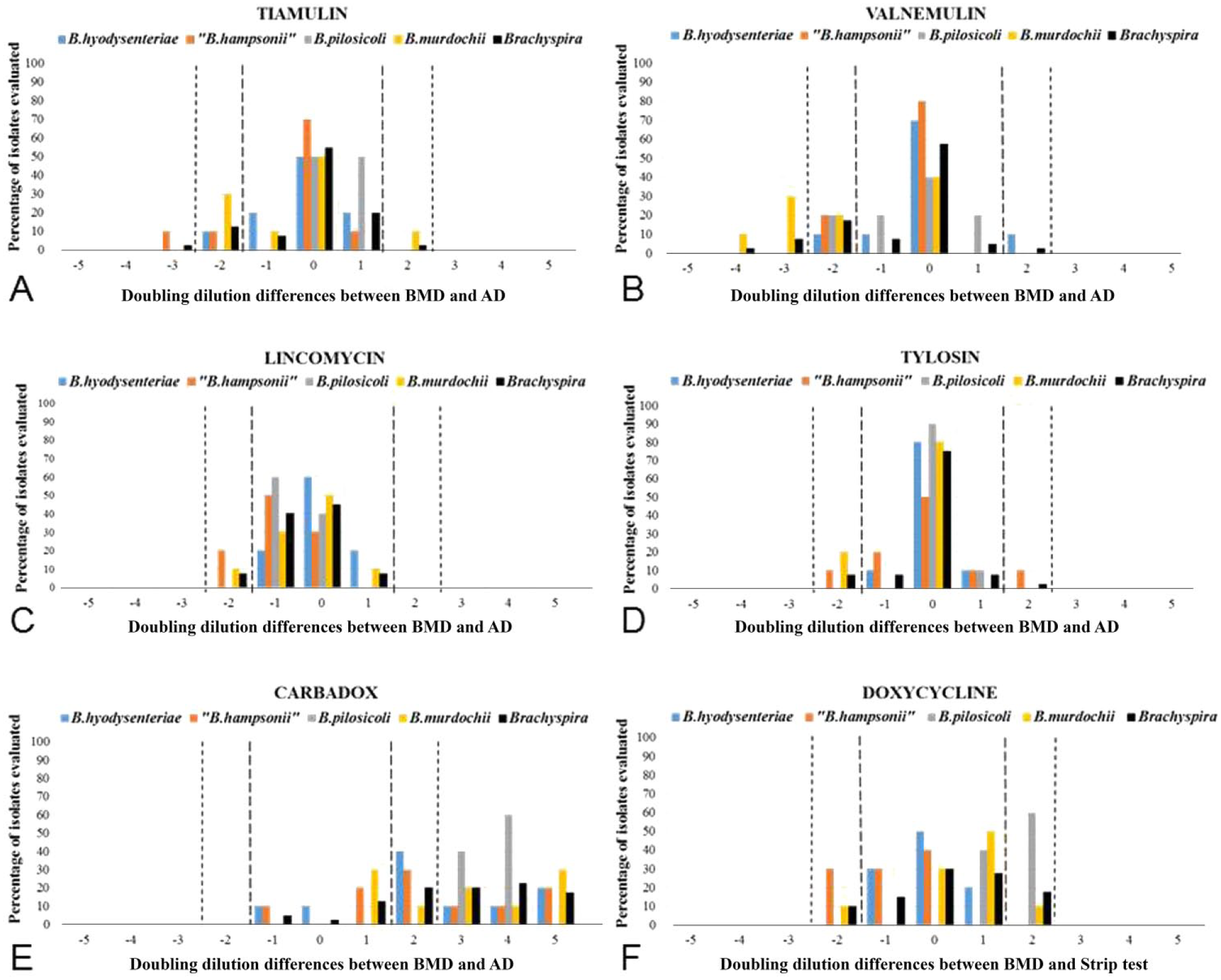
Distribution of differences in log2-transformed minimal inhibitory concentration (MIC) data of 4 Brachyspira species (B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii) stratified by 6 antimicrobials.
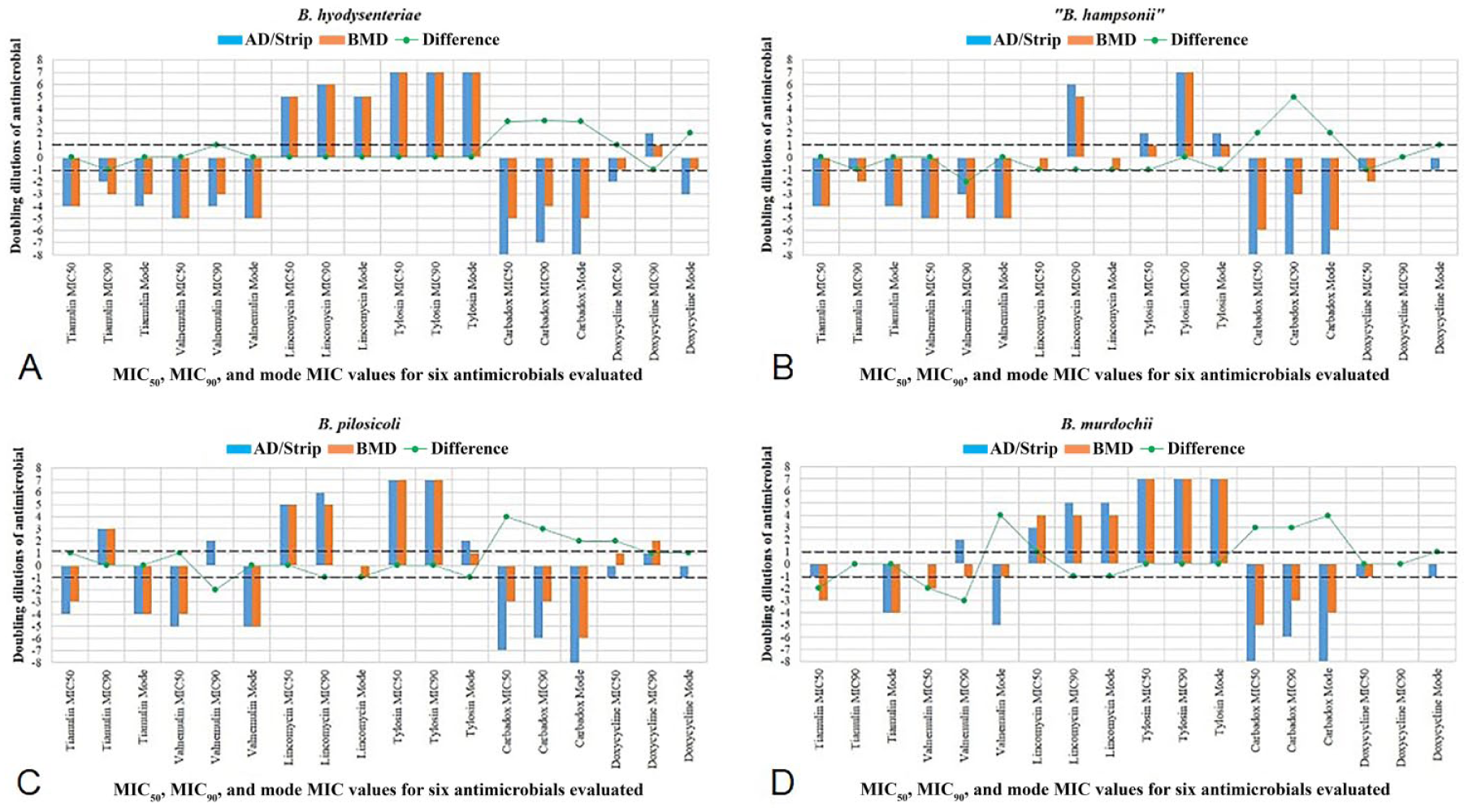
Distribution of differences in the log2-transformed minimal inhibitory concentration (MIC)50, MIC90, and mode MIC values of 6 antimicrobials against 4 Brachyspira species by the broth microdilution (BMD) and agar dilution (AD; tiamulin, valnemulin, lincomycin, tylosin, and carbadox) or strip test (doxycycline) methods.
A comparison was made between the BMD and the AD or strip test methods by the agreement and bias measures for each antimicrobial and Brachyspira species (Table 3). The average ±1 log2 agreement between BMD and AD or strip test methods for tiamulin, valnemulin, lincomycin, and tylosin were as follows: B. hyodysenteriae (mean: 92.5%, range: 80–100%), “B. hampsonii” (mean: 80%), B. pilosicoli (mean: 95%, range: 80–100%), and B. murdochii (mean: 67.5%, range: 40–90%). Similarly, the average ±2 log2 agreement between BMD and AD or strip test methods for these 4 antimicrobials were as follows: B. hyodysenteriae (mean: 100%), “B. hampsonii” (mean: 97.5%, range: 90–100%), B. pilosicoli (mean: 100%), and B. murdochii (mean: 90%, range: 60–100%). The ±2 log2 agreement values for carbadox were as follows: B. hyodysenteriae (60%), “B. hampsonii” (60%), B. pilosicoli (0%), and B. murdochii (40%). The agreement (±1 log2, ±2 log2) values between the BMD and strip test methods for doxycycline were as follows: B. hyodysenteriae (100%, 100%), “B. hampsonii” (70%, 100%), B. pilosicoli (40%, 100%), and B. murdochii (80%, 100%). A positive bias or a negative bias value (Table 3) indicates that, in general, AD or strip test tends to overestimate or underestimate the MIC values, respectively, when using BMD as the reference method for comparison. A statistically significant bias >1 doubling dilution between BMD and AD was not detected for any of the 4 Brachyspira species tested against tiamulin, valnemulin, lincomycin, and tylosin, with a single exception—a positive bias for valnemulin for B. murdochii isolates (AD results were ~1–2 dilutions higher than BMD results). In contrast, carbadox showed a statistically significant negative bias that was >1 doubling dilution between BMD and AD for all 4 Brachyspira species (AD results were ~3 dilutions lower than the BMD results). When compared with BMD, the strip test did not show a statistically significant bias for Brachyspira species tested against doxycycline, with the exception of B. pilosicoli, which showed a statistically significant negative bias (strip test results were ~1–2 dilutions lower than BMD results).
Comparison of susceptibility results obtained by broth microdilution and agar dilution or strip test methods (for doxycycline) for 6 antimicrobial agents against 4 Brachyspira species.*
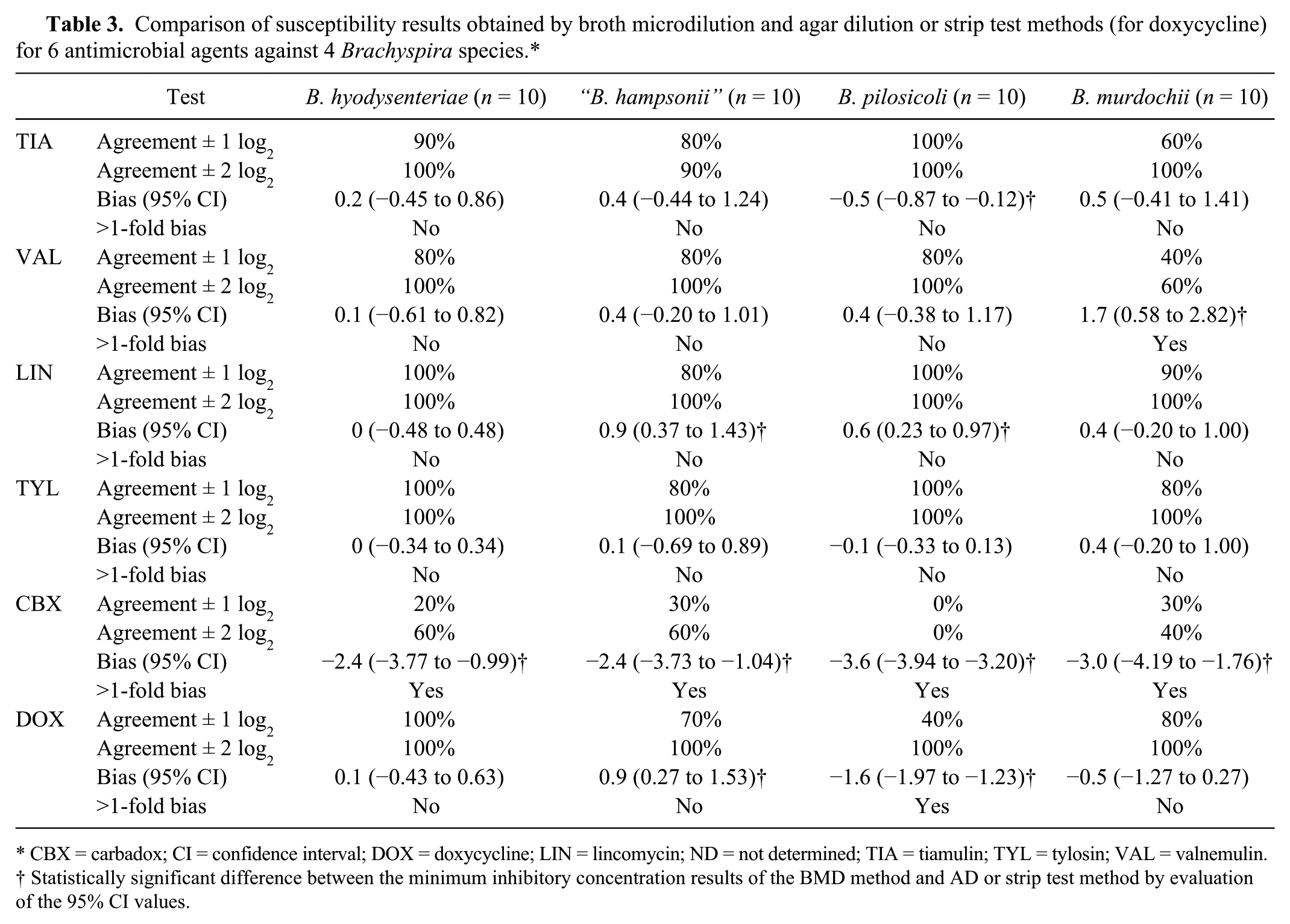
CBX = carbadox; CI = confidence interval; DOX = doxycycline; LIN = lincomycin; ND = not determined; TIA = tiamulin; TYL = tylosin; VAL = valnemulin.
Statistically significant difference between the minimum inhibitory concentration results of the BMD method and AD or strip test method by evaluation of the 95% CI values.
Discussion
Our study performed a direct comparison between susceptibility techniques (BMD vs. AD or strip test) for pathogenic and commensal Brachyspira species, including B. hyodysenteriae, “B. hampsonii”, B. pilosicoli, and B. murdochii against 5 groups of commonly used antimicrobials. The results help understand the role that bacterial species, phenotypic traits, antimicrobials, and testing conditions can play in the results of in vitro susceptibility testing for these diverse species. Because of the absence of CLSI-approved interpretive criteria for the susceptibility testing of Brachyspira species in swine, categorical agreements between the 3 susceptibility testing methods could not be calculated. Other measures including essential agreement, bias, and dilution differences were calculated to determine the method most appropriate for in vitro susceptibility testing of swine Brachyspira species.
Agar dilution showed reproducibility between replicates; however, the technique was laborious with a higher possibility for error. The method required production of test plates (antimicrobial in 5% SB-TSA) where evaluation of very low concentration of antimicrobials (e.g., 0.002 µg/mL carbadox) could potentially introduce measurement errors and thus lower comparability between different laboratories. The modification of this method to include a cut in the agar did result in better visualization of hemolysis (data not shown). Regardless, the recording of results of the AD method can be subjective particularly due to the presence of smaller spots or zones of hemolysis. The other drawback of this technique is its reliance on an indirect indicator of growth (hemolysis), rather than directly observing growth. Surface growth is limited for Brachyspira species, thus requiring such a reliance on hemolysis as an indicator of growth; however, it is possible that concentrations of an antimicrobial that inhibit hemolysis are not sufficient to completely inhibit growth of the bacteria itself.
The strip test method showed lower reproducibility (for doxycycline) than AD or BMD. Although it is recommended to record the edge of the zone of growth (instead of hemolysis) to record the MIC cutoff, the limited surface growth of Brachyspira made it difficult to record the results for doxycycline by strip test. Additionally, several isolates produced “double zones” of hemolysis, further complicating the observation of the MIC.
In comparison, the BMD results were easier to record and less subjective. The reliance on the presence or absence of growth (visualized as turbidity in the wells of the plate) overcomes the subjectivity in recording results by AD. BMD showed a high reproducibility for most antimicrobials and Brachyspira species. Our comparison between species, however, did result in the finding that “B. hampsonii” genetic group II isolates sometimes require the entire 96-hr duration to ensure growth in the broth medium. Furthermore, the minimal effect of the incubation period on the stability of the antimicrobials in broth, and the fastidious nature of “B. hampsonii” genetic group II supported the use of a 96-hr incubation period, as recommended 14 for BMD. The availability of a standardized commercial panel makes BMD suitable for routine diagnostic testing and inter-laboratory comparisons. Overall, by comparing the skill and time required for performing susceptibility tests on Brachyspira species by these 3 methods, as well as the subjectivity, ease of reading results, and reproducibility of the methods, BMD was found to be most suitable for evaluating in vitro susceptibility of Brachyspira.
The susceptibility results of the B. hyodysenteriae B-78T strain were found to be consistently within the range proposed for quality control. 21 The reproducibility of the results of this strain and the agreement between the results by BMD and AD or strip test methods confirmed its utility as a control strain for the latter 2 techniques, as previously recommended. 21 The agreement between the MIC results of the various Brachyspira ATCC strains evaluated by BMD and AD or strip test techniques for all antimicrobials, except carbadox, mirrored the general trend discussed in the comparison of the different methods. For instance, multiple testing of the “B. hampsonii” genetic group II strain NSH-24 yielded lower MICs for tiamulin, valnemulin, and lincomycin by BMD than by AD. It is possible that this is caused by its lowered growth in broth when compared to agar. The “skipped well” phenomenon observed with B. pilosicoli strain P43/6/78 against lincomycin and tylosin has been reported for B. pilosicoli field isolates previously, 14 wherein it was attributed to mutations associated with in vitro resistance that develops on exposure to an antimicrobial in the incubated broth medium. As suggested previously, 14 although this complicates the recording of results for such B. pilosicoli isolates, it is easier to detect development of in vitro resistance in isolates tested by BMD that may go unnoticed if tested by AD.
Although some previous studies have compared the susceptibility results of B. hyodysenteriae isolates by BMD and AD,13,16,25 to our knowledge, there is no information on the comparability of these methods for other Brachyspira species such as “B. hampsonii”, B. pilosicoli, and B. murdochii. For anaerobes, MICs determined by BMD can be 1 doubling dilution different from those determined by AD. 3 Additionally, 1 doubling dilution variation is acceptable for susceptibility test procedures, 4 and is thus commonly accepted as a criteria for identifying the essential agreement of 2 susceptibility tests. In our study, the susceptibility results were most comparable (within 1 doubling dilution) between the BMD and the AD methods for tylosin, lincomycin, tiamulin, and valnemulin. These findings are similar to those obtained in previous studies evaluating the susceptibility of B. hyodysenteriae against these antimicrobials.13,16,25 The essential agreement (±1 log2) between BMD and AD for tylosin, lincomycin, tiamulin, and valnemulin was high for all 4 Brachyspira species with the exception of B. murdochii susceptibility against pleuromutilins, which showed relatively lower essential agreement values. The ±2 log2 agreements between the BMD and strip test methods for these 4 antimicrobials were high for all Brachyspira species. With the exception of B. murdochii susceptibility toward valnemulin, no statistically significant bias >1-fold dilution between BMD (reference test method) and AD was detected for Brachyspira species tested against these 4 antimicrobials. Because the differences in agreement or bias values are unique to a specific pair of evaluated antimicrobial and bacterial species, it is possible that these differences can be attributed to the differences in microbial growth characteristics in different media and growth conditions.
In contrast, the comparability and essential agreement between the BMD and AD methods for carbadox were much lower than with other antimicrobials, with the broth MICs being ~3 dilutions (ranging from 1 to 5 dilutions) higher than those in agar for all 4 Brachyspira species evaluated. These observations were in agreement with a previous study that compared the 2 methods for susceptibility testing of B. hyodysenteriae isolates against carbadox. 13 Because these differences were consistently detected for all Brachyspira species in our study, it is unlikely that the difference in results can be attributed to the species of Brachyspira, and instead is more likely associated with the effect of carbadox on the growth of the bacterium in the 2 different media. It is possible that subinhibitory concentrations of carbadox may be capable of inhibiting hemolysis (used as an indirect indicator of growth for the AD method), thus contributing to the difference in MIC results by the 2 methods. A previous study 28 developed a novel microtiter broth dilution method that measured growth by correlating direct counts of B. hyodysenteriae with the decrease in absorbance associated with the hemolysis of bovine erythrocytes in a broth medium caused by the growth of the bacteria. Interestingly, that previous study showed similar carbadox MIC results between BMD and AD, and was attributed to the fact that hemolysis was used as an indirect indicator of growth in both methods. Additionally, the carbadox MIC results of our study for the B. hyodysenteriae ATCC reference strains B-78T and B204 were within the ranges for each as reported by previous studies using the same BMD method,8,13 thus supporting the carbadox susceptibility results by BMD in our study. Therefore, the detection of a statistically significant bias suggests that AD might underestimate the carbadox MIC results by 1–5-fold dilutions when compared with the reference BMD method.
The susceptibility results of the BMD and strip test methods for doxycycline were less comparable, and the strip test results were often very difficult to interpret. In general, the essential agreement (±1 log2) values were moderate, and no statistically significant bias >1 doubling dilution was detected between BMD and strip test methods while evaluating Brachyspira species against doxycycline. For B. pilosicoli, however, a lower essential agreement (±1 log2) and a statistically significant bias >1 doubling dilution between BMD and the strip test were identified, thus suggesting that the strip test tends to underestimate the MIC results of B. pilosicoli by 1–2 dilutions as compared to BMD.
The agreement values help determine which bacterial species and/or antimicrobial is most affected by the choice of a particular test method and also helps compare MIC results obtained by different methods that may be used by different laboratories. Similarly, the detection of a statistically significant bias between in vitro antimicrobial susceptibility methods helps inform diagnosticians and clients about the likelihood of one test method to underestimate or overestimate the MIC results as compared with another test method. The detection of a statistically significant bias, however, does not imply that the bias is also clinically significant, 1 as such interpretations require comparisons of categorical agreements. Currently, proposed wild-type cutoff values for several antimicrobials against B. hyodysenteriae and possibly B. pilosicoli are used for monitoring in vitro susceptibilities 23 ; however, CLSI interpretive criteria for pathogenic Brachyspira are urgently needed for classifying isolates as clinically susceptible or resistant and subsequently determining the categorical agreements of different test methods.
The importance of reliable in vitro susceptibility testing methods for Brachyspira cannot be overstated given the increased reports of reduced susceptibility toward antimicrobials11,18,20,25 and the emergence of novel pathogenic Brachyspira species. In this study, we have evaluated the advantages and limitations of BMD for susceptibility testing of various pathogenic and commensal swine Brachyspira against 5 groups of antimicrobials and compared it with other susceptibility methods including AD and strip test methods. Our results confirm that BMD represents a standardized technique that can be used for routine diagnostic testing and for monitoring any changes in the in vitro susceptibility of Brachyspira in and between farms, regions, or countries.
Footnotes
Acknowledgements
We thank the University of Minnesota’s Veterinary Diagnostic Laboratory for providing us with Brachyspira field isolates for antimicrobial susceptibility testing. We also thank Aaron Rendahl for help with statistical analyses and interpretations.
Authors’ contributions
NS Mirajkar contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. CJ Gebhart contributed to conception and design of the study, and contributed to interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
VetMIC Brachy, National Veterinary Institute (SVA), Uppsala, Sweden.
b.
Etest doxycycline DC 256, bioMérieux, Lombard, IL.
c.
Bacto brain heart infusion, BD, Franklin Lakes, NJ.
d.
Fetal bovine serum, Sigma-Aldrich, St. Louis, MO.
e.
Tiamulin fumarate, VETRANAL; Sigma-Aldrich, St. Louis, MO.
f.
Valnemulin hydrochloride, Novartis Animal Health US Inc., Greensboro, NC.
g.
Lincomycin hydrochloride, Sigma-Aldrich, St. Louis, MO.
h.
Tylosin tartrate, VETRANAL; Sigma-Aldrich, St. Louis, MO.
i.
Carbadox, Sigma-Aldrich, St. Louis, MO.
j.
BBL Brucella agar with 5% sheep blood, Hemin, and Vitamin K1 prepared media, BD, Franklin Lakes, NJ.
k.
BBL Mueller Hinton agar with 5% sheep blood, BD, Franklin Lakes, NJ.
l.
Remel McFarland equivalence turbidity standard, Thermo Scientific, Pittsburgh, PA.
m.
Statistica, StatSoft Inc., Tulsa, OK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with research and/or publication of this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Novartis Pharmaceuticals Corp. [11651-GEBHART-NOVARTIS PHARM-CON000000033714-3000].