Abstract
An 11-year-old male neutered Pembroke Welsh Corgi dog displayed a mass measuring 7.5 cm × 6.6 cm × 1.6 cm in the skin. Neoplastic tissue was nonencapsulated, and the neoplastic cells showed infiltrative growth into the surrounding tissue on microscopic examination. The neoplastic tissue was mainly located from the dermis to the subcutis. Epidermotropism of neoplastic cells was not observed. The tissue was composed of irregular, solid nests of round to polygonal cells. Nests were separated by fine fibrovascular stroma. Mitotic index was high (7.90 ± 0.38 per high power field) and extensive necrosis was observed in the neoplastic tissue. Vascular invasion was often observed in the neoplastic tissue. Neoplastic cells were positive for vimentin, HLA-DR antigen, Iba1, CD18, and E-cadherin, but cells did not express cytokeratin, S100, CD20, CD79α, CD3, MUM-1, lambda light chain, kappa light chain, lysozyme, CD204, or CD11d by immunohistochemistry. Electron microscopic analysis revealed dendrites on these cells. From the above-mentioned findings, the tumor was diagnosed as a cutaneous histiocytic sarcoma with E-cadherin expression. It is possible that neoplastic cells in the present case were derived from cutaneous Langerhans cell. To our knowledge, cutaneous histiocytic sarcoma with E-cadherin expression in domestic animals has not been previously diagnosed in domestic animals.
Canine proliferative histiocytic diseases have been classified into 3 major categories: nonmalignant nonneoplastic, nonmalignant neoplastic, and malignant neoplastic disease. 5 These 3 categories involve 3 disorders: 1) reactive histiocytosis (nonmalignant nonneoplastic disease), 2) cutaneous histiocytoma (nonmalignant neoplastic disease), and 3) histiocytic sarcoma (malignant neoplastic disease).5,31 Reactive histiocytoses are histiocyte- and lymphocyte-rich inflammatory disorders related to immune dysregulation. 19 Reactive histiocytosis arises from activated interstitial dendritic cells.5,31 Canine cutaneous histiocytoma is a benign histiocytic tumor that shows Langerhans cell differentiation, and usually occurs as a solitary lesion in young dogs.19,31 Canine histiocytic sarcoma is a malignant histiocytic tumor arising from interstitial dendritic cells, which occurs in most tissues of the body.19,31 Histiocytic sarcomas may occur as multiple lesions in single organs and disseminate to many organs. 19
Although there are several reports of primary cutaneous malignant histiocytic proliferative diseases in dogs,17,30 canine primary cutaneous histiocytic sarcoma derived from Langerhans cell has not been reported, to our knowledge. Our report describes the macroscopic, histologic, immunohistochemical (IHC), and electron microscopic features of canine cutaneous histiocytic sarcoma with E-cadherin expression, which we suspect is a Langerhans cell sarcoma.
An 11-year-old male neutered Pembroke Welsh Corgi dog displayed a mass measuring 7.5 cm × 6.6 cm × 1.6 cm in the skin of ventral thorax (Fig. 1). The affected lesion was detected as a soybean-sized nodule 6 months before, and its volume increased rapidly from 2 months before examination. Surgical excision was done at Tottori University Veterinary Teaching Hospital (Tottori, Japan). The cut surface of the mass was solid and grayish-white. Fourteen days after surgical excision, recurrence of neoplastic lesion was observed in the affected part of skin. Orally administered lomustine (40 mg/m2 body surface area) every 3 weeks was elected post-recurrence.
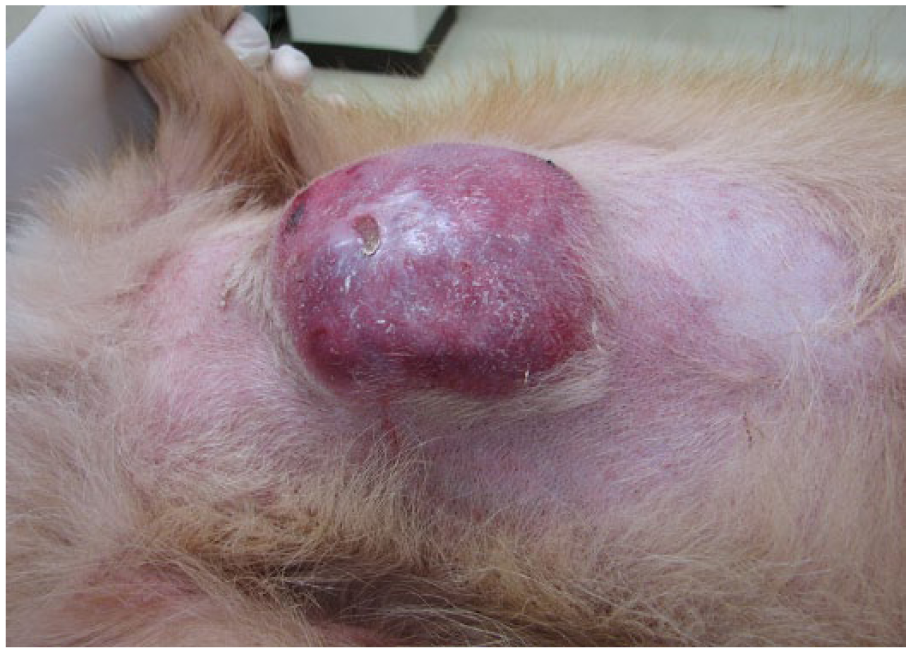
A single nodular mass occurred in the skin of the ventral thorax of an 11-year-old male neutered Pembroke Welsh Corgi dog.
Lomustine treatment was associated with regression of neoplastic tissue, but the lesion recurred. The dog died 193 days after the first lomustine treatment. Autopsy revealed 2 nodular lesions (15 mm × 13.5 mm × 12 mm, and 7 mm × 5 mm × 5 mm) in the skin of the ventral thorax, and no neoplastic lesions in intrathoracic, intra-abdominal organs or brain. The direct cause of death of the dog was unclear.
The aforementioned 3 nodular lesions were histologically analyzed. The excised tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned and stained with hematoxylin and eosin, periodic acid–Schiff (PAS) reaction, and Grimelius stain. Grimelius stain was used for differential diagnosis of neuroendocrine tumor. The mitotic index was calculated by determining the mean number of mitoses counted in 30 fields microscopically observed with a 400× magnification.
Immunohistochemical staining was performed by the peroxidase-conjugated immune-polymer method,a,b with primary antibodies against vimentin, c cytokeratin, d human leukocyte antigen (HLA)-DR antigen, d ionized calcium binding adaptor molecule 1 (Iba1), e lysozyme, d cluster of differentiation (CD)204, f CD18, g CD11d, g E-cadherin, h CD20, i CD79αcy, d CD3, d multiple myeloma oncogene-1 (MUM-1), j S100, d lambda light chain, d and kappa light chain d for differential diagnosis of histiocytic tumors, lymphoid tumors, plasmacytic tumors, and neuroendocrine tumors (Table 1). Antigen retrieval was performed for each antibody (Table 1). Endogenous peroxidase activity was quenched by immersing the sections in 3% hydrogen peroxide in methanol for 15 min. Then, the sections were incubated with the primary antibody and treated with the secondary antibody. The sections were exposed to a 3,3′-diaminobenzidine solution containing hydrogen peroxide to facilitate a peroxidase color reaction.
Details of primary antibodies and pretreatment procedures for antigen retrieval.*
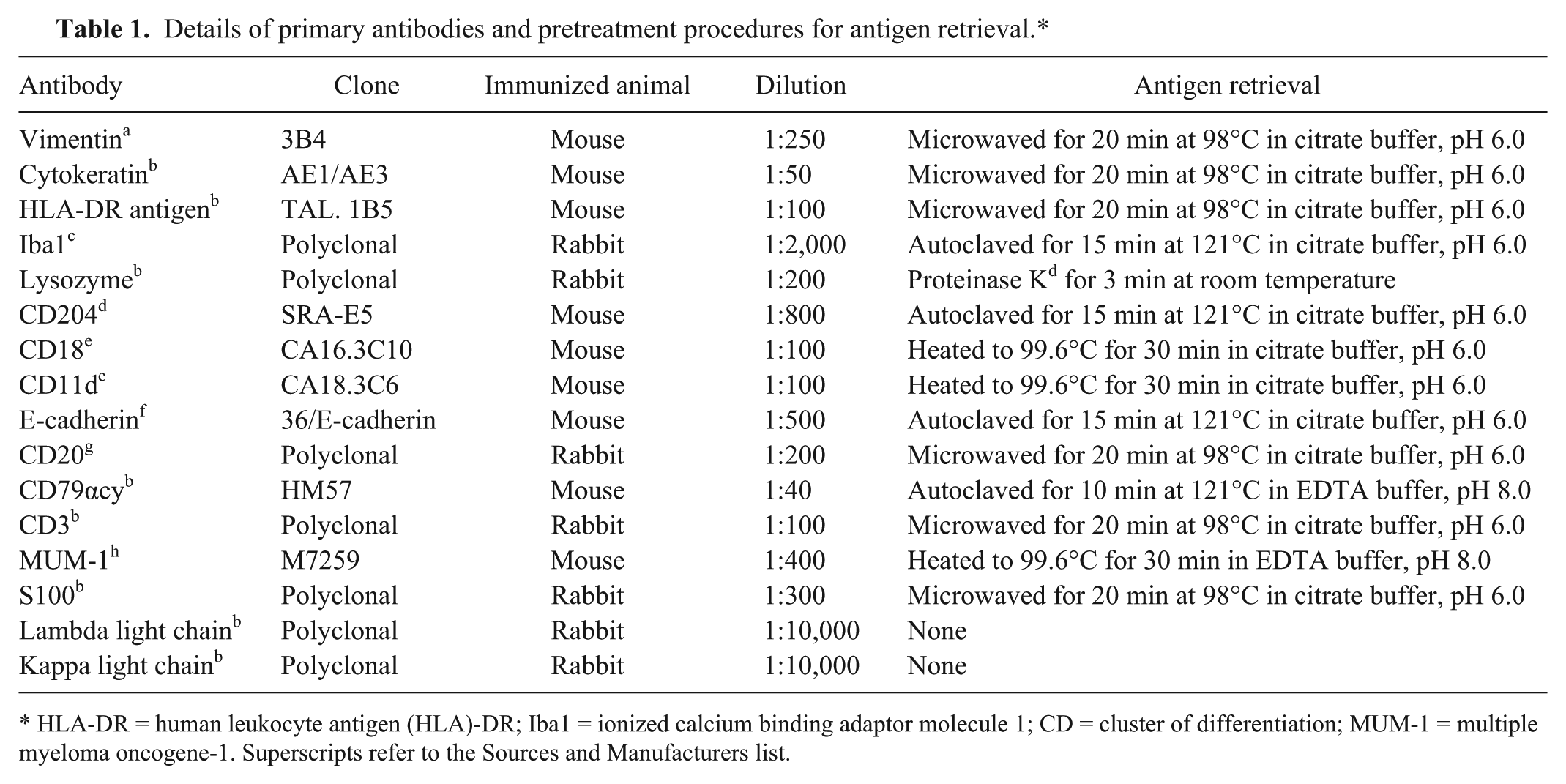
HLA-DR = human leukocyte antigen (HLA)-DR; Iba1 = ionized calcium binding adaptor molecule 1; CD = cluster of differentiation; MUM-1 = multiple myeloma oncogene-1. Superscripts refer to the Sources and Manufacturers list.
For electron microscopy, formalin-fixed samples were further fixed in 2.5% glutaraldehyde, postfixed in 2% osmium tetroxide, and embedded in epoxy resin. Ultrathin sections were cut and stained with uranyl acetate and lead citrate, then examined under an electron microscope.
Neoplastic tissue was mainly located within the dermis and the subcutis (Fig. 2). The lesion was nonencapsulated, highly necrotic, and infiltrative (Figs. 2, 3), extending into the cutaneous muscles (Fig. 3). Epidermotropism or the extension of the tumor tissue to the epidermis was not observed (Fig. 2). Neoplastic tissue was composed of irregular and solid nests of round to polygonal cells with well-defined cell borders (Fig. 4), separated by fine fibrovascular stroma (Fig. 4). Neoplastic cells had pale, fine granular cytoplasm and oval nuclei with a coarse chromatin pattern (Fig. 4). The nuclei were located in the central regions of these neoplastic cells. Moderate anisokaryosis and anisocytosis were observed. There were many mitoses in neoplastic tissue (7.90 ± 0.38 per field microscopically observed with a 400× magnification; Fig. 4). Neoplastic cells were negative for PAS and Grimelius stains. These cells did not show metachromasia with toluidine blue stain. The electron microscopic analysis demonstrated that the neoplastic cells had a distinct cell membrane, dendrites, and developed rough-surfaced endoplasmic reticulum and a few mitochondria (Fig. 5). There were no Birbeck granules in the cytoplasm of the neoplastic cells (Fig. 5).
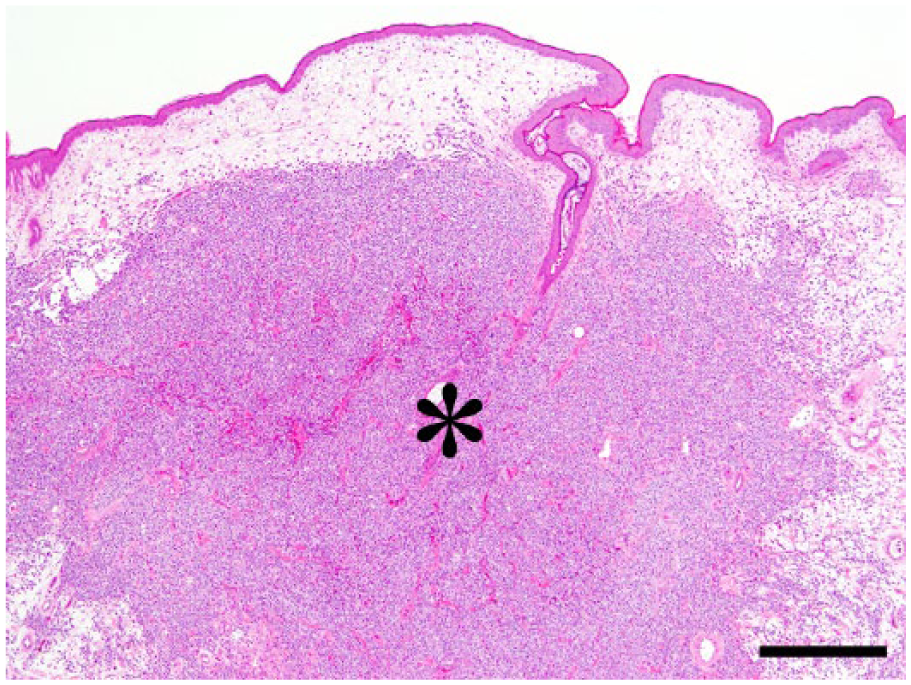
Microscopically, neoplastic tissue (*) was mainly located within the dermis and the subcutis. The tissue was nonencapsulated and infiltrative. The continuity of tumor tissue with the epidermis was not observed. Bar = 500 µm.
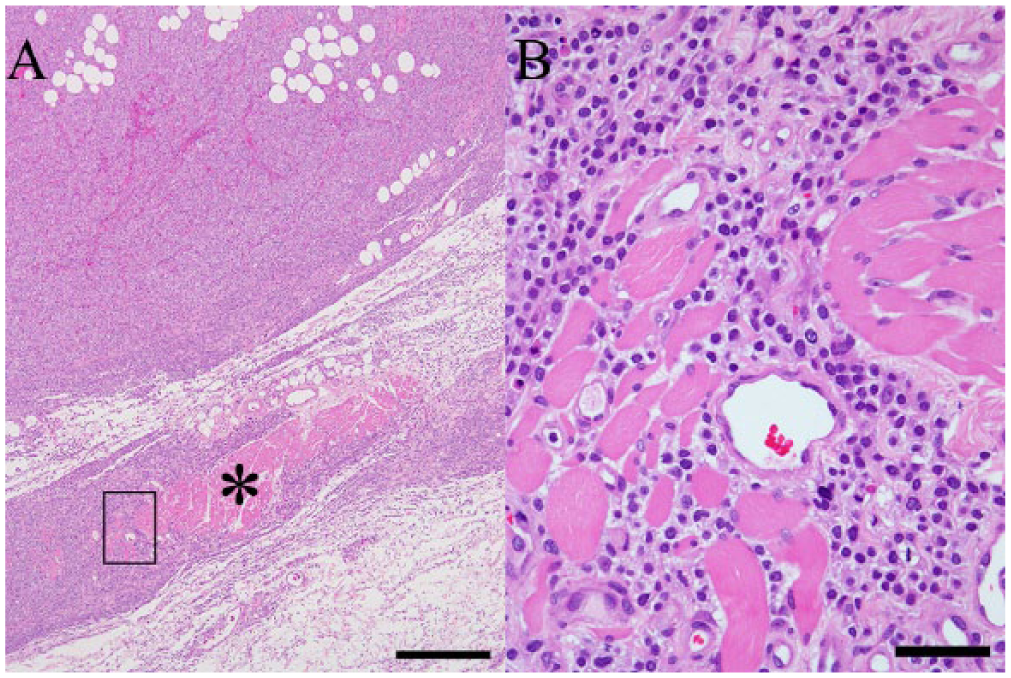
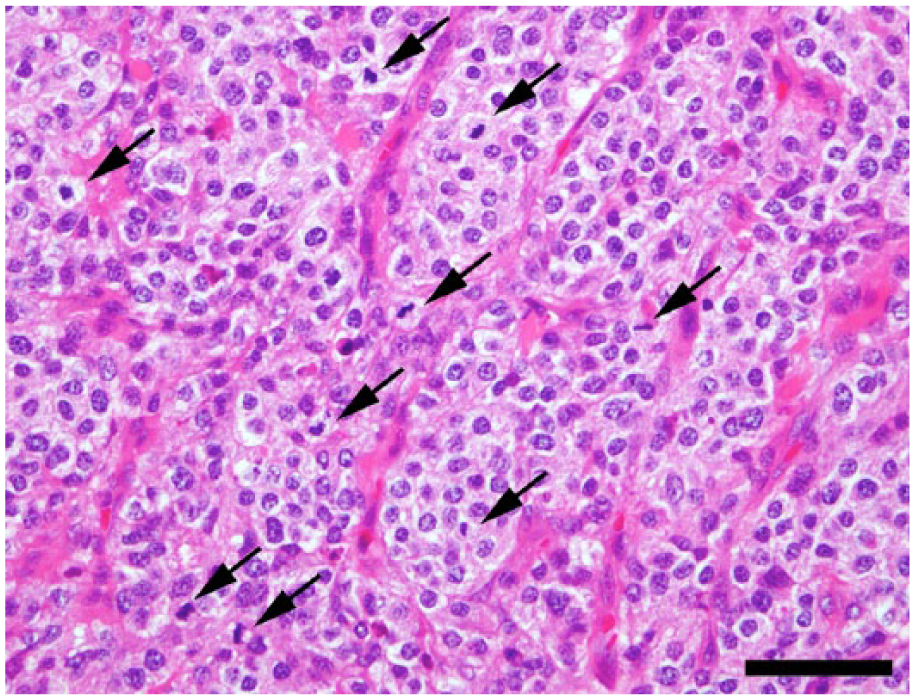
Neoplastic tissue was composed of irregular and solid nests of neoplastic cells. The nests were separated by fine fibrovascular stroma. Cells had pale and fine granular cytoplasm and oval nuclei. There were many mitoses in neoplastic tissue. Arrows show mitotic cells. Bar = 50 µm.
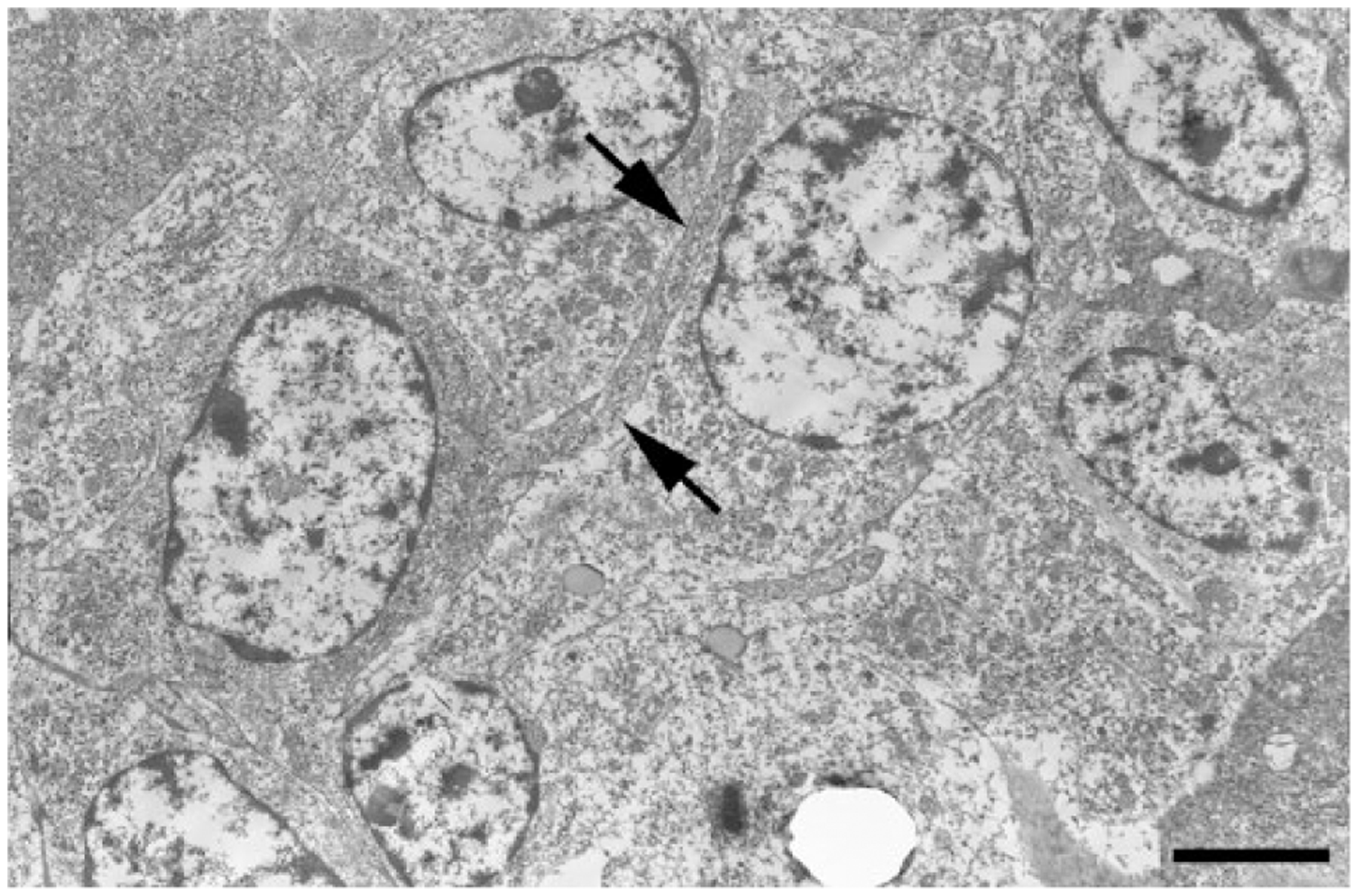
Electron microscopic analysis demonstrated that neoplastic cells had several dendrites (arrows). There were no Birbeck granules in the cytoplasm of the neoplastic cells. Bar = 3 μm.
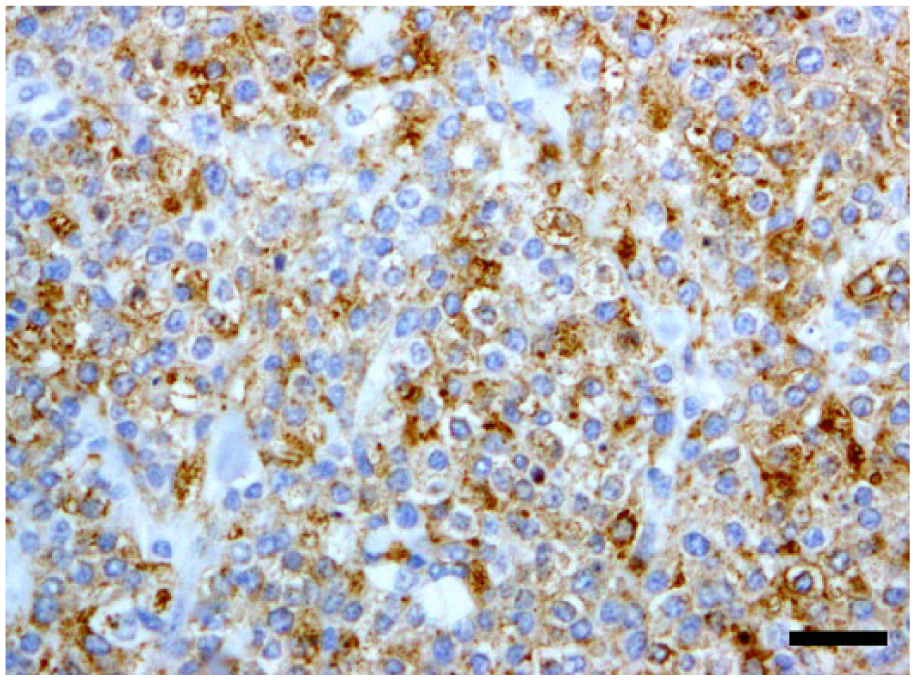
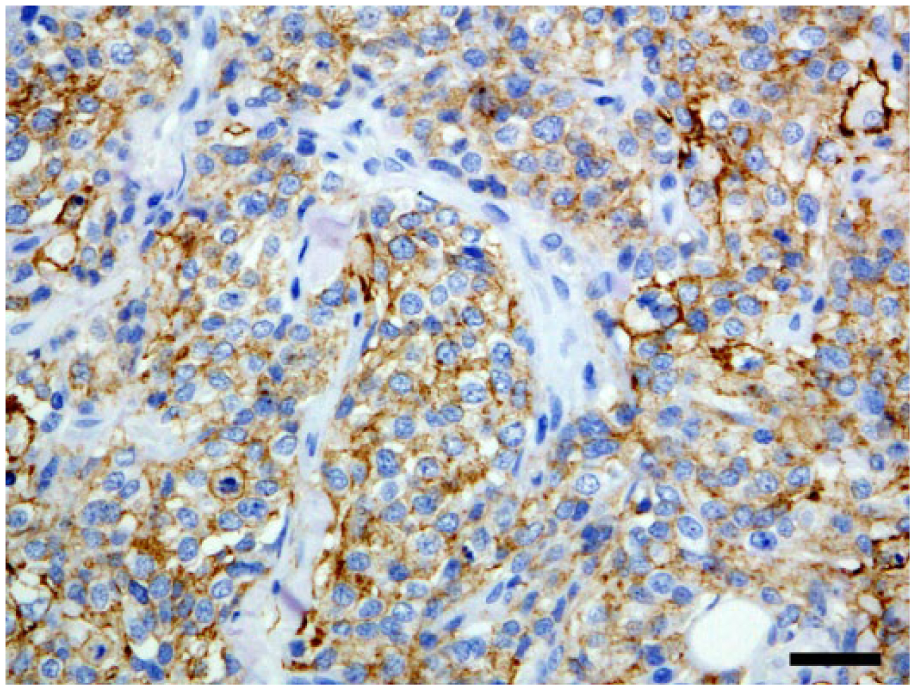
Immunohistochemical analysis demonstrated that almost all neoplastic cells expressed vimentin, HLA-DR antigen, Iba1, and CD18 (Figs. 6–8). The degree of E-cadherin immunoexpression varied, but the neoplastic cells were convincingly positive for E-cadherin (Fig. 9). However, the neoplastic cells were negative for cytokeratin, S100, CD20, CD79α, CD3, MUM-1, lambda light chain, kappa light chain, lysozyme, and CD204 or CD11d.
Neoplastic cells were immunohistochemically positive for human leukocyte antigen (HLA)-DR and alpha-chain. Bar = 30 µm.
Neoplastic cells were immunohistochemically positive for ionized calcium binding adaptor molecule 1 (Iba-1). Bar = 30 µm.
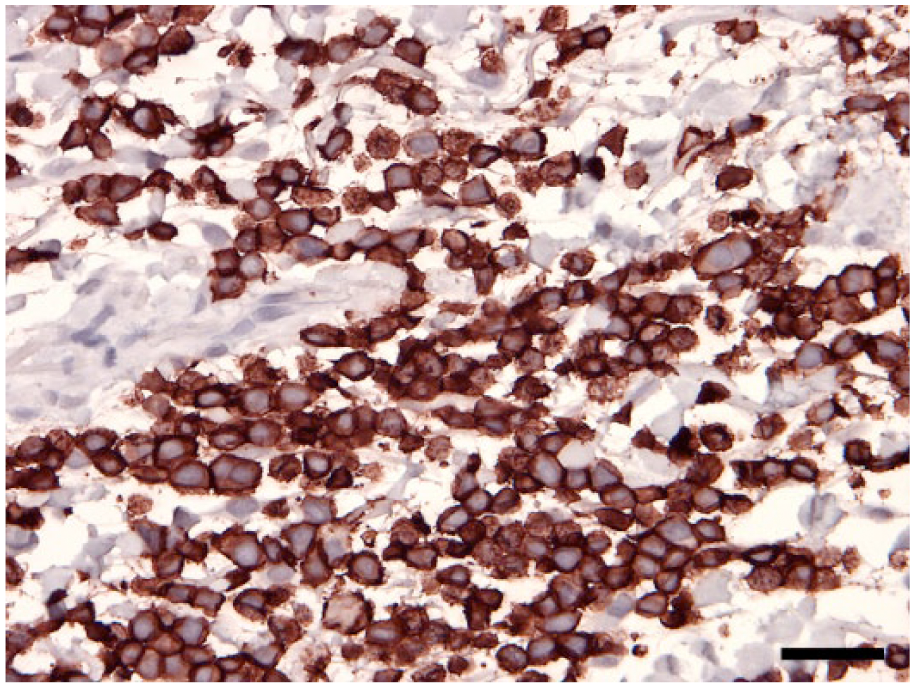
Neoplastic cells were immunohistochemically positive for cluster of differentiation (CD)18. Bar = 50 µm.
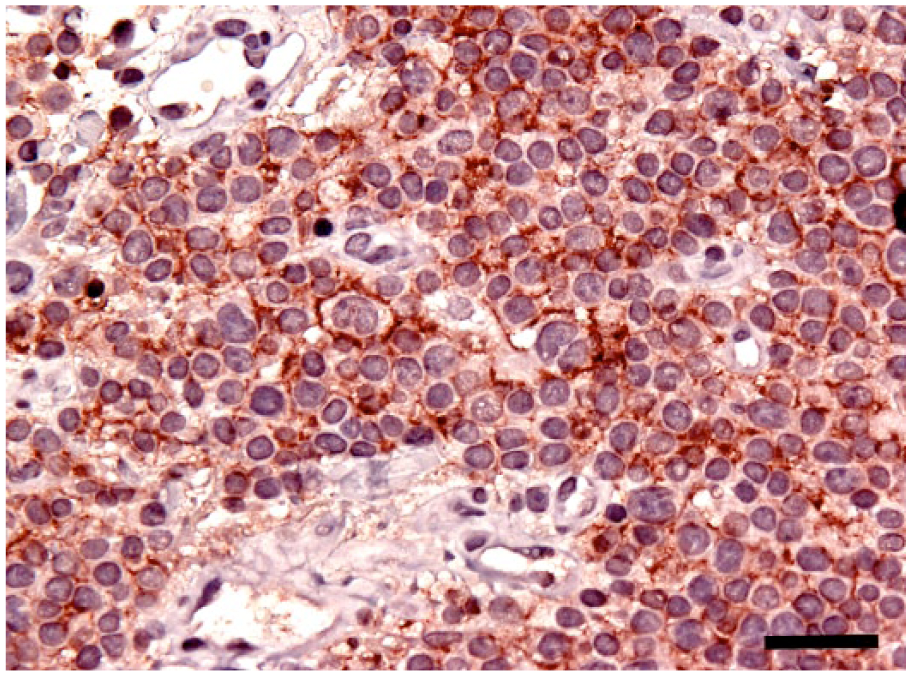
The neoplastic cells were immunohistochemically positive for E-cadherin. Bar = 50 µm.
Human leukocyte antigen–DR is expressed in antigen-presenting cells such as dendritic cells, macrophages, and B lymphocytes. 13 Iba1 is a 147–amino acid, calcium-binding protein involved in the cytoplasmic actin cross-linking activity, and is associated with phagocytosis, membrane ruffling, and motility of cells. 23 Iba1 is expressed by almost all subpopulations of cells of monocyte and macrophage lineage. 24 CD18 is a panleukocyte marker, 35 and CD18 was expressed in canine histiocytic tumors such as canine cutaneous histiocytomas, histiocytic sarcomas, and malignant fibrous histiocytomas. 8 The aforementioned IHC features suggested that the neoplastic cells in the present case were histiocyte derived. Based on the histological, IHC, and electron microscopic findings, the animal was diagnosed with a cutaneous histiocytic sarcoma with E-cadherin expression.
Langerhans cells immunoexpress HLA-DR, Iba1, CD18, and E-cadherin. 8 CD204 is the class A macrophage scavenger receptor and is expressed in macrophages. 29 CD11d encodes the latest alpha-subunit of the leukocyte integrin family, and it is expressed predominantly in myelomonocytic cells. 22 Previous studies showed that CD11d expressed in splenic red pulp macrophages and resident bone marrow macrophages in dogs.20,26 Langerhans cells were negative for CD204 or CD11d.8,29 The neoplastic cells in the present case immunoexpressed HLA-DR, Iba1, CD18, and E-cadherin, but not CD204 or CD11d. The expression of these IHC markers in the neoplastic cells of the present case coincided with those of Langerhans cells, and are consistent with a Langerhans cell origin. To our knowledge, there are no reports of cutaneous Langerhans cell–derived malignant tumor in animals, and its histological features have not been elucidated.
In humans, tumors derived from Langerhans cells are classified by the World Health Organization into Langerhans cell sarcoma and Langerhans cell histiocytosis according to the degree of cytologic atypia and clinical aggressiveness. 11 The most prominent feature of Langerhans cell sarcoma in humans is the malignant cytology of a pleomorphic tumor. 11 Almost all cases of Langerhans cell sarcoma in humans reported previously were in adults. 11 Langerhans cell sarcoma occurs commonly in the skin and underlying soft tissue, with multiorgan involvement that includes lymph node, lung, liver, spleen, and bone. 11 Langerhans cell sarcoma behaves aggressively with an overall survival of <50%. 11 On the other hand, human Langerhans cell histiocytosis covers a spectrum of disease ranging from localized to rapidly progressive systemic disease. 6 Although human Langerhans cell histiocytosis can occur in individuals of any age, it is more common in children aged 1–3 years, but rare in adults. 34 Nuclear atypia is minimal in human Langerhans cell histiocytosis. 11 Various kinds of inflammatory cells such as lymphocytes, eosinophils, macrophages, and plasma cells infiltrate the Langerhans cell histiocytosis lesions, which produce cytokines to facilitate development of Langerhans cell histiocytosis. 3 The present case had an onset of this neoplastic disease in adulthood, showing malignant histological and cytologic features such as high mitotic rate and extensive necrosis. There were no inflammatory cells in tumor tissue of the present case. It is suggested that the present case is more similar to Langerhans cell sarcoma than Langerhans cell histiocytosis. However, nuclear and cytologic atypia or pleomorphism of neoplastic cells was not observed in the present case.
The epidermotropism of neoplastic cells or the continuity of tumor tissue with the epidermis was not observed in the present case. A previous study demonstrated that epidermotropism was not evident in human Langerhans cell sarcoma. 16 Canine cutaneous histiocytoma, a Langerhans cell–derived tumor, arises in the dermis and may invade the epidermis in a subset of lesions. 19 Canine cutaneous histiocytomas do not arise directly from intraepidermal Langerhans cells. 19 It is quite possible that they arise from dermal precursors of Langerhans cell similar to the cells described previously.14,19 Similar to canine cutaneous histiocytoma, the neoplastic cells in the present case may have arisen in the dermis. This may be why the epidermotropism of neoplastic cells and the extension of the tumor tissue to the epidermis were not observed in the present case.
In our case, a high mitotic rate was observed (7.9 ± 0.38 per high power field). A similar histological finding was observed in cutaneous Langerhans cell sarcomas of humans. 11 Previous studies showed a mitotic rate of >30–60 per 10 high power fields in cutaneous Langerhans cell sarcomas in humans.2,7,15,32 In the present case, the high mitotic rate may be associated with the repeated recurrence after lomustine treatment.
Neoplastic tissue in our case was composed of irregular, solid nests of round to polygonal epithelioid neoplastic cells with well-defined cell borders. A previous study showed sheet-like epithelioid tumor cell growth with a vaguely nested pattern in human cutaneous Langerhans cell sarcoma. 25
Langerhans cells in humans contain Birbeck granules. 1 The formation of Birbeck granules is mediated by internalization of langerin (CD207) expressed on the surface of Langerhans cells. 19 Birbeck granules are cytoplasmic structures that define Langerhans cells and distinguish them from interstitial dendritic cells. 19 A previous study served to identify Birbeck granules ultrastructurally in the cytoplasm of 60% of tumor cells in human cutaneous Langerhans cell sarcoma. 2 However, Birbeck granules have not been observed in canine Langerhans cells. 21 In the present case, electron microscopic examination demonstrated the absence of Birbeck granules in the cytoplasm of the tumor cells. On the basis of this finding, it is not possible to directly link the neoplastic cells in the present case to a Langerhans cells origin based on Birbeck granule formation, because canine Langerhans cells do not contain Birbeck granules. 21
Langerhans cells use E-cadherin to localize in the epidermis via homotypic interaction with E-cadherin expressed by keratinocytes. 27 E-cadherin is a lineage-associated marker but not a lineage-specific marker for Langerhans cells. 19 Migration of Langerhans cells from skin to the draining lymph node is associated with a downregulation of E-cadherin expression. 9 A previous study 9 showed that lack of E-cadherin expression on neoplastic cells was associated with dissemination of neoplastic cells and poor outcome in human Langerhans cell histiocytosis. In the neoplastic cells of the present study, the degree of E-cadherin immunoexpression varied. Thus, the present findings may be related to tumor cell aggressiveness in the present tumor.
Lysozyme immunoexpression was not observed in the neoplastic cells in our study. Lysozymes occurred within macrophages and monocytes; however, Langerhans cells did not contain appreciable lysozyme. 18 Lysozyme is a useful marker of histiocytic differentiation in malignant or systemic histiocytosis, but it was a less reliable histiocytic marker in canine cutaneous histiocytoma; 40% of the tumors in our study were negative for lysozyme expression. 18 Lysozyme content is reportedly low in human Langerhans cell histiocytosis. 11 Another earlier study showed a lysozyme-positive rate of 40% in human Langerhans cell histiocytosis. 33
The overall results of our study suggested that the neoplastic cells in this case may have originated from Langerhans cells. Canine cutaneous histiocytoma is known as a cutaneous histiocytic proliferative disease deriving from Langerhans cells in dogs. The pathological condition of the present case differs from that of canine cutaneous histiocytoma in some respects: 1) while a canine cutaneous histiocytoma usually occurs as a solitary lesion and spontaneously regresses,19,28 the lesion volume in the present case gradually increased daily for 6 months; 2) although the age-specific incidence rate for canine cutaneous histiocytoma drops precipitously after 3 years, 28 the cutaneous neoplastic lesion in the present case occurred in an 11-year-old dog; and 3) canine cutaneous histiocytomas are progressively infiltrated by lymphocytes after a variable time course, 4 but there was no lymphocyte infiltration of tumor tissue in our case.
A previous study showed 7 (47%) Pembroke Welsh Corgis among the 15 dogs diagnosed with subdural histiocytic sarcomas. 10 Another study showed 11 cases (22%) of Pembroke Welsh Corgis among 50 dogs with canine histiocytic sarcomas. 12 Histiocytic sarcoma may occur more commonly in Pembroke Welsh Corgis than other canine breeds. To our knowledge, canine cutaneous histiocytic sarcoma with E-cadherin expression has not been reported in animals.
Footnotes
a.
EnVision, Dako Japan Inc., Kyoto, Japan.
b.
Histofine simple stain MAX-PO (M or R), Nichirei Biosciences Inc., Tokyo, Japan.
c.
YLEM, Avezzano-Aquila, Italy.
d.
Dako Japan Inc., Kyoto, Japan.
e.
Wako Pure Chemical Industries Ltd., Osaka, Japan.
f.
Trans Genic Inc., Kobe, Japan.
g.
Leukocyte Antigen Biology Laboratory, Davis, CA.
h.
BD Transduction Laboratories, Lexington, KY.
i.
Thermo Fisher Scientific Inc., Rockford, IL.
j.
Dako Denmark A/S, Glostrup, Denmark.
Authors’ contributions
A Hirako contributed to conception of the study, and analysis and interpretation of data. A Sugiyama contributed to conception and design of the study, and to acquisition, analysis, and interpretation of data. M Sakurai, K Ozaki, H Sakai, T Takeuchi, T Morita, and PF Moore contributed to analysis of data. All authors drafted the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
