Abstract
An outbreak of bovine tuberculosis (TB) in a Michigan dairy herd resulted in quarantine, depopulation, pathology, and epidemiologic investigations. This herd, compared to other TB-infected herds in Michigan, was unusual in the long-term feeding of waste milk to its replacement calves. The herd had 80 cattle with positive results on caudal fold test or gamma interferon testing, which were reclassified as suspects because the herd had never been known to be tuberculous previously. Autopsy revealed striking variation in the anatomic distribution of gross anatomic lesions, microscopic lesions, and culture-positive lymph nodes between the adult cattle, the calves, and the domestic cats present on the farm. Adult cattle had lesions and culture-positive lymph nodes predominantly within the thoracic lymph nodes, whereas cats had 50% of their lesions and culture-positive lymph nodes in their abdomens, and 50% of positive calves had culture-positive lymph nodes in their abdomens. This difference in anatomic distribution correlated with the likely routes of infection, which are believed to be by direct airborne transmission in adult cattle and indirect ingestion of contaminated milk in both calves and cats. Although TB literature over the past 100-plus years states that the route of infection may manifest itself in differences in lesion anatomic distribution, our team has been working with TB for over 20 years, and we have never encountered such striking variation between different groups of animals on the same farm.
Introduction
The state of Michigan has been dealing with an endemic bovine tuberculosis (TB) problem in its wild white-tailed deer (Odocoileus virginianus) since 1994. Over the past 21 years, the infection has spilled back into domestic cattle on at least 61 farms. In spite of the authors’ extensive experience with TB, new aspects of this old disease are still being encountered. In the case of the farm in the present report, we found an unusual distribution of tuberculosis lesions in adult cattle, calves, and domestic cats, in which the anatomic distribution of the lesions and culture-positive tissues varied considerably based on the likely route of exposure between the various animal groups affected. This was unique compared with our groups’ experience in both wild and domestic animals, and we believe it will be of interest and informative to others working in the tuberculosis area.
Materials and methods
A whole-herd tuberculosis test was performed because a cow at a local slaughter plant had tuberculosis-compatible lesions and was traced back to this particular farm. Tuberculosis testing of the entire herd (all adult cattle and all calves down to 1 day of age) was carried out using the caudal fold test method. There were 80 suspects out of 451 cattle tested (18% reactor prevalence); this included 56 suspect adult cattle and 24 suspect calves. Confirmatory gamma interferon testing was performed on all 80 suspect cattle. An initial submission of 9 adult cattle that were strong caudal fold test reactors (caudal fold swellings of several cm in diameter) was made to the Diagnostic Center for Population and Animal Health (DCPAH; Lansing, Michigan), where they were euthanized and subjected to full autopsies. Autopsy protocol involved gross examination of the head, thoracic, and abdominal lymph nodes and lungs, with collection of fresh and formalin-fixed sections of lymph nodes divided into these 3 anatomic locations following previously described methods. 4 Collection of lymph nodes included the following: head—both parotid, both submandibular, and both medial retropharyngeal lymph nodes; thoracic—a cranial mediastinal, a middle mediastinal, a caudal mediastinal, and a tracheobronchial lymph node; and abdominal—a mesenteric, a hepatic, an ileocecal, and a supramammary lymph node. Formalin-fixed samples were routinely paraffin embedded, sectioned at 5 µm, stained with hematoxylin and eosin as well as Ziehl–Neelsen acid-fast stains, and then examined microscopically by board-certified veterinary pathologists. The remaining 47 adult cattle that were classified as reactors were shipped directly to slaughter facilities, where they were examined by U.S. Department of Agriculture veterinarians similar to the autopsy protocol performed on the initial 9 cattle. Fresh and formalin-fixed samples of the same 3 lymph node pools from all 56 adult cattle were forwarded to the Bacteriology and Pathobiology Laboratories, National Veterinary Services Laboratories (NVSL; Ames, Iowa) where histological examinations, mycobacterial cultures, and polymerase chain reaction (PCR) tests on positive cultures were conducted. Detailed sample preparation, culture, and PCR methods were as previously described, with the exception that PCR was run on both fresh tissues and mycobacterial cultures.7,11 In addition, 19 cattle at the slaughter house had milk samples collected and forwarded to NVSL for mycobacterial culture.
Twenty-four strong caudal fold test reactor calves were subsequently submitted to and autopsied at DCPAH following protocols identical to those described for the adult cattle. Cultures were performed at NVSL; methods were similar to those used in adult dairy cattle.
Twenty-one domestic cats were collected on the farm, euthanized, and submitted to DCPAH for autopsy. In a similar fashion to the cattle, head, thoracic, and abdominal lymph nodes were collected from the cats and submitted for histologic examination, mycobacterial culture, and PCR testing at DCPAH. The head and thoracic samples were pooled for 19 of the cats; abdominal samples were kept separate for all 21 cats. Cultures and PCR were performed by methods similar to those used in adult cattle and calves.
Statistics
Differences in the distribution of lesions and positive mycobacterial culture results within the body between the different hosts (adult cows, calves, cats) were assessed using Fisher exact 2-tailed test (the low number of adult cows with detailed autopsy findings invalidated the use of the Mantel–Haenszel χ2 statistic). Overall differences in the distribution of positive findings between hosts were assessed using the Cochran–Mantel–Haenszel χ2 statistic for general association.
Results
The results for the 9 cattle that had autopsies performed at DCPAH are summarized below. All 9 cattle had gross and histologic caseogranulomas compatible with TB. These lesions were present in the thoracic lymph nodes of all 9 cattle. No cattle had lesions in their head lymph nodes, and only 1 cow had lesions in its abdominal lymph nodes (Fig. 1). Culture-positive results for Mycobacterium bovis were identical to lesion distribution: all 9 cattle were positive by culture from their thoracic lymph nodes; no cattle were culture positive in their head lymph nodes; and only 1 cow had culture-positive results from abdominal lymph nodes (Fig. 2).
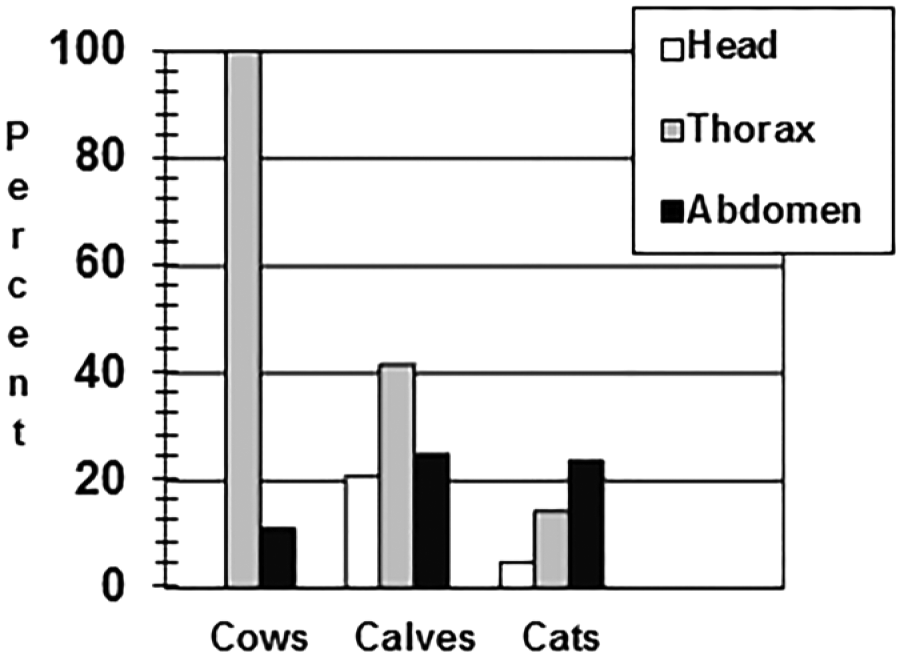
Percent gross anatomic and microscopic lesions by anatomic location in suspect bovine tuberculosis adult dairy cows (n = 9), dairy calves (n = 24), and domestic cats (n = 21).
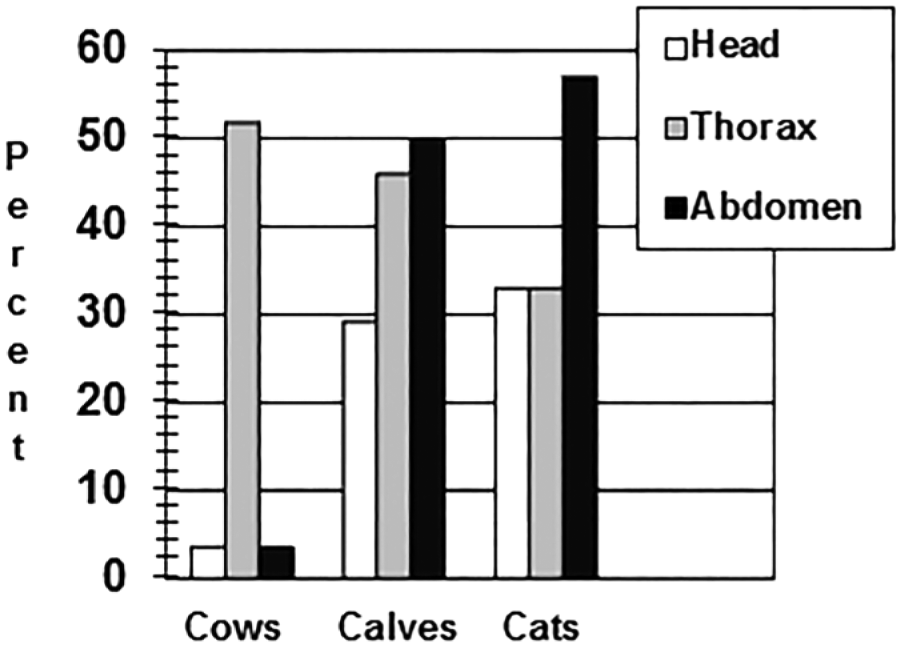
Percent positive Mycobacterium bovis culture results by anatomic location in suspect bovine tuberculosis adult dairy cows (n = 56), dairy calves (n = 24), and domestic cats (n = 21).
Detailed records of the gross anatomic and histologic lesions found in the 47 cattle that went to slaughter and were inspected and collected at that site are not available. However, culture results indicated 20 M. bovis–positive cattle out of the 47. Of these, 18 cattle were positive in thoracic lymph nodes; 2 cattle were positive in head lymph nodes; and only 1 was positive in abdominal lymph nodes (Fig. 2). Additionally, M. bovis was cultured from 2 of the 19 milk samples collected from reactor cattle sent to the slaughter house; neither of these 2 animals had gross anatomic or histologic lesions compatible with TB within their supramammary lymph nodes or mammary glands.
Results for the 24 calves submitted to DCPAH for detailed autopsies are summarized below. Thirteen calves had gross anatomic and histologic lesions compatible with TB; 5 had lesions in their head lymph nodes, 10 calves had lesions in their thoracic lymph nodes, and 6 calves in their abdominal lymph nodes (Fig. 1). Fifteen total calves were positive by culture (including all 13 calves with gross anatomic and histologic lesions) and PCR for M. bovis (Fig. 2). The distribution of M. bovis–positive cultures was 6 calves with positive head lymph nodes, 11 calves with positive thoracic lymph nodes, and 12 calves with positive abdominal lymph nodes.
Results for the 21 cats examined are summarized below. For the 21 cats, only 1 cat had gross lesions in the head, 3 cats had thoracic lesions, and 5 cats had abdominal lesions (a total of 5 cats had gross anatomic lesions in any site; Fig. 1). Mycobacterial culture and PCR testing identified 7 additional TB-infected cats. Seven cats had culture-positive head and thoracic lymph node pools, and 12 cats had culture-positive abdominal lymph nodes (Fig. 2).
Statistics
By testing modalities, there were significant differences in the distribution of positive lesions at autopsy (χ2 = 9.3, 2 df, p = 0.0096): adult cows had higher levels and cats had lower levels of thoracic lesions. There were significant differences in the culture results (χ2 = 19.81, 2 df, p < 0.0001): adult cows had significantly lower levels of positive head and abdominal cultures than calves and cats ( p = 0.0003 and p < 0.0001, respectively).
Discussion
The differences in anatomic distribution between the adult cattle versus the calves and cats from this facility were striking. In the adult cattle, gross anatomic, histologic, and culture-positive results were predominantly within their thoracic lymph nodes, whereas both calves and cats had statistically significant higher prevalence of culture positives in their abdominal lymph nodes compared to adult cattle. This difference in anatomic distribution appears to correlate well with the likely routes of transmission of TB in this case. The cows housed together in the same barn most likely spread tuberculosis to one another by airborne transmission, whereas the calves and cats that were fed waste milk (colostrum and antibiotic-contaminated milk) most likely contracted the disease by oral ingestion. The additional 46% prevalence of thoracic culture positives in the calves is most likely related to lateral transmission of tuberculosis by aerosol route, as these calves started out in individual hutches, but after 3–6 weeks they were pen-raised in similar age groups allowing for aerosol transmission between calves. The aerosol route of infection is believed to be responsible for 80–90% of all tuberculosis infections in cattle. 8
The authors do believe that variation in route of infection can result in differences in the anatomic distribution of TB lesions; they have been working on an ongoing TB outbreak for the past 20 years. In their experience, with over 60 infected domestic cattle herds to date in Michigan and over 750 infected white-tailed deer, such a difference in anatomic distribution of lesions is rarely encountered under natural conditions. Part of this may relate to the routes of transmission and subsequent spread through the animal. TB is most commonly transmitted by either airborne inhalation or oral ingestion, and, in either route, the tonsils are one of the earliest sites to develop infection based on experimental models.5,6 Tonsils have been shown to be a good site with which to experimentally inoculate both cattle and deer with TB; once the tonsil is infected, the efferent lymphatics drain to the medial retropharyngeal lymph nodes.5,6 Therefore, the head lymph nodes are commonly affected by both routes of transmission and are not helpful in discriminating the initial route of transmission. Another confounding factor to determining the route of transmission is that M. bovis tends to spread over long periods of time to many different lymphoid tissues throughout the body, thus masking early differential anatomic distribution of lesions. In the cases of primary pulmonary infection, the tuberculosis organisms may spread through lymphatics, hematogenously, or by granulomas rupturing into airways, which can allow the organisms to be coughed up, and subsequently swallowed and enter the gastrointestinal tract.
In the current case, the principal underlying factors that allowed variable differential tissue distribution of lesions were how the animals were housed, and how the waste milk from infected cattle was handled. The adult cattle were kept in pens and barns in close proximity and separated from their calves except during the first day post-calving. This “sharing” of air contributed to the lateral transmission of M. bovis among adult cattle. Both the calves and cats were fed waste milk on a daily basis for many months (up to 6 months of age) allowing for oral ingestion of milk contaminated by M. bovis. It has been experimentally shown that infection with TB by oral inoculation in either cattle or sheep requires 16–18 million times the infective dose compared to respiratory inoculation. 10 In this herd, at least 2 adult milking cows were shedding M. bovis into their milk, and because only 19 of >300 adult milking cattle had their milk tested, there were likely additional cows shedding M. bovis. Because waste milk was routinely fed to calves for up to 6 months and to cats continuously, this provided ample opportunity to expose these animals by ingestion. The feeding and management of TB-contaminated milk has been previously shown to be a significant risk factor in infection of dairy calves. 9 Of course, contaminated milk also poses a potential zoonotic threat to humans, which is why pasteurization of commercial milk has been widely accepted and utilized in the United States since 1924. 3
All calves selected for autopsy were between 6 and 9 months of age, based on these calves exhibiting the largest reaction (up to several centimeters) to the caudal fold test. Therefore, the calves had less time to develop widespread disseminated lesions than did their adult cohorts on this farm. The fact that milk transmission was likely to have occurred on this farm is given further credence by the fact that 2 other local dairy farms developed M. bovis in their calves that had been fed waste milk from this index herd (CS Bruning-Fann, personal communication, 2015). No adult cattle tested positive for M. bovis on either of the secondary herds, and no movement of cattle occurred between the farms.
Another interesting feature of this outbreak was the predominantly abdominal distribution of lesions in cats, which is in contrast to reports from Great Britain, where most cats have been limited to skin lesions associated with wounds in regions where tuberculosis is endemic in cattle and badgers.1,2 In Great Britain, it is believed that the initial spillover of TB into the cat population was by way of ingestion of contaminated milk on infected dairy farms, and in years prior to 2004, M. bovis infections in cats were more commonly associated with the alimentary tract and with the ingestion of unpasteurized milk, similar to our report from Michigan. 1
This TB outbreak in Michigan is a striking example of an old axiom that the route of TB transmission may correlate with differences in the anatomic distribution of tuberculosis lesions. In this case, airborne transmission between adult cattle resulted in predominantly thoracic lesions, whereas cats that ingested milk contaminated by M. bovis developed predominantly abdominal lesions, and calves developed similar numbers of both thoracic and abdominal lesions due to initial oral exposure to contaminated milk and subsequent pen rearing allowing for additional aerosol transmission. This is also an important reminder of the continued zoonotic threat that unpasteurized milk poses to the health of humans and why continued surveillance and control of M. bovis is still important even in industrialized countries like the United States. Finally, our study points out how the epidemiologic study of cattle herds infected with TB can provide important information on the routes of disease transmission and for improved control of TB.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for the research, authorship, and/or publication of this article.
