Abstract
Antibody responses are useful indicators of Mycobacterium bovis infection in cattle. Many studies have evaluated the ability of immunoglobulin G (IgG) to serodiagnose bovine tuberculosis (TB). In the current study, immunoglobulin A (IgA) and IgG responses against the MPB70 and MPB83 antigens of M. bovis, the 38 kDa phosphate-binding lipoprotein (PstS1) that is a well-known serodiagnostic M. tuberculosis antigen, and a newly identified protein, termed Rv1483c, were compared in M. bovis–infected and noninfected cattle as well as in field samples. The diagnostic utility of the IgA antibody to MPB70 and MPB83 for bovine TB was superior or comparable to that of the IgG antibody, and the sensitivity of serodiagnosis increased when the results of antigen binding by IgA and IgG were combined. The sensitivities of the IgG and IgA antibodies to the Rv1483c and PstS1 proteins were significantly lower than those to MPB70 and MPB83, and no diagnostic utility for Rv1483c was observed in field samples. Importantly, the IgA antibody reacted strongly to the MPB70 and MPB83 antigens and differentiated cattle with TB from healthy cattle in a multiantigen printed immunoassay. The results of this study support the feasibility of using IgA antibody against the MPB70 and MPB83 antigens to detect bovine TB. In addition, approaches using assays for both IgA and IgG antibodies may increase detection accuracy.
Introduction
Mycobacterium bovis causes bovine tuberculosis (TB), a major economic problem in many countries. 25 Mycobacterium bovis also infects and causes TB in a wide range of wild animals, livestock, and humans. 28 Traditional test-and-slaughter policies based on skin testing with a purified protein derivative (PPD) have not been fully successful, because wildlife species and other livestock animals continuously act as infection reservoirs.27,28 Therefore, new diagnostic tools that can be applied to a variety of wild animals and cattle are urgently required.
The diagnostic tests most widely used for bovine TB are based on detection of the cell-mediated immune response (i.e., intradermal tests) and interferon-gamma (IFNγ) release assays. 19 Although skin tests are a comparatively inexpensive and simple procedure, these tests are not suitable for large-scale epidemiological surveys. In addition, intradermal tests require 2 visits to a farm, a requirement that is not practical for testing wildlife species, and may produce a false-negative result in advanced disease stages with a high bacterial load.18,28 Interferon-gamma release assays are more specific than intradermal tests, but are comparatively expensive and require trained staff. 26
Antibody-based assays for detecting bovine TB are attractive due to their simplicity, flexibility, and economic benefits. However, the lower sensitivity of antibody-detection assays remains a major concern. 26 Several attempts have been made to develop a serological assay for diagnosing bovine TB, and serological tests with promising accuracy have been reported.19,24,27,28 In addition, previous studies have demonstrated that serological assays can detect infected animals that were missed with intradermal tests and IFNγ release assays.1,28 Together, these results suggest that serological testing can be used as a supplement for diagnosing bovine TB, or as a surveillance tool in a variety of animals.
The sensitivity of diagnostic assays can be improved using multiple antigens. Several proteins and lipids with strong seroreactivity have been identified and tested as assay antigens for diagnosing bovine TB.26,27 The typical antigens used in many studies are MPB70 and MPB83, which are very promising candidates for the serodiagnosis of bovine TB.26,29 Other antigens such as ESAT-6, CFP-10, and Ag85 proteins have also been used as antigens, but yielded an insufficient sensitivity compared to MPB70 and MPB83 proteins.6,24,26 Some studies have demonstrated that a diagnostic system using a fusion protein or cocktail antigens can increase sensitivity. 15 Therefore, studies are warranted to identify additional M. bovis proteins with seroreactivity and to evaluate their diagnostic usefulness.
Studies on human TB suggest that the serum immunoglobulin A (IgA) antibody has a promising role in the diagnosis of active human TB.3,14,23 It is well known that mucosal IgA plays a major role in defense against invading pathogens, such as bacteria and viruses, via mucosal surfaces. 13 However, most studies on serodiagnosis in livestock species such as cattle have focused on assessing the immunoglobulin G (IgG) antibody.6,27,28 A previous report suggested that the immunoglobulin M (IgM) antibody is not valuable for diagnosing human TB. 20 It may be possible to improve test sensitivity by detecting both serum IgG and IgA antibodies against a specific antigen rather than only detecting IgG. Therefore, the objective of the present study was to assess the usefulness of IgA antibodies for diagnosing bovine TB.
Materials and methods
Cattle samples
Sixty sera samples were obtained from cattle at the Animal and Plant Quarantine Agency bank (Anyang, South Korea). Thirty samples originated from M. bovis–infected cattle, as determined by mycobacterial culture and/or the presence of macroscopic lesions compatible with TB infection. For bacterial culture, the tissues obtained from the TB lesions of organs were dissected and homogenized with sterile phosphate buffered saline (PBS), and decontaminated by 10% oxalic acid solution for 10 min at room temperature. After centrifugation (1,000 × g for 10 min), the pellets were inoculated onto Lowenstein–Jensen medium with pyruvate. The remaining 30 samples were obtained from a bovine TB-free herd that has had skin test–negative results for 7 years.
In addition, sera (n = 155) from cattle on 10 farms in Gyeongnam Province, Korea during 2011–2013 were examined. On these farms, intradermal testing was performed by injecting M. bovis PPD under the skin of the caudal fold of cattle aged >12 months. Dermal swelling >5 mm after 72 hr was considered positive and swelling <3 mm was considered negative. Sera were collected from cattle that were PPD skin test positive (PPD(+); n = 59) and skin test negative (PPD(–); n = 96).
Antigen preparation
Native MPB70 protein was purified from M. bovis strain BCG (Bacillus Calmette–Guérin; Tokyo strain) culture filtrate proteins (CFPs) using a 2-step biochemical method as previously described.9,17 Briefly, M. bovis BCG was grown at 37°C as a surface pellicle on Sauton medium for 6 weeks, after which the CFPs were filter sterilized with a 0.2-μm membrane. The 50–80% ammonium sulfate precipitate (ASP) of the CFPs was dialyzed against 1 mM potassium phosphate buffer (PPB, pH 7.0), loaded onto a hydroxyapatite column equilibrated with 1 mM PPB, and eluted with 1 mM PPB because MPB70 was not retained on the column. Further purification was performed by anion-exchange chromatography a and a linear salt gradient elution. The MPB70 protein was eluted in 20 mM Tris–HCl (pH 8.0) containing 10–50 mM NaCl. The eluted protein was pooled, concentrated, and stored at −70°C. Protein concentration was estimated using the bicinchoninic acid protein assay kit b with bovine serum albumin (BSA) as the standard. The native 38-kDa phosphate-binding lipoprotein (PstS1) was purified from M. tuberculosis CFPs as previously described. 10
The gene for MPB83 was amplified by a polymerase chain reaction (PCR) assay using M. bovis strain AN5 genomic DNA as the template and the following primers: forward, 5′-CATATGTTC TTAGCGGGTTGTTCGAGC-3′; reverse, 5′-AAGCTTCTGTGCCGGGGGCATCAGCAC-3′. The PCR product was cut with NdeI and HindIII and inserted into the pET22b(+) vector. c The recombinant protein was overexpressed in Escherichia coli BL21 cells carrying the DE3 bacteriophage, and were purified by nickel–nitrilotri-acetic acid (Ni-NTA) affinity chromatography in accordance with the manufacturer’s instructions. d The purified protein was pooled, concentrated, dialyzed against PBS (pH 7.4), and stored at −70°C.
Identification and production of the seroreactive Rv1483c protein
Mycobacterium tuberculosis CFPs were used to screen for seroreactive proteins in cattle sera to identify a novel antigen. Simple fractionation was performed to separate the complex M. tuberculosis antigens, as previously described. 21 Briefly, the CFPs were precipitated with ammonium sulfate (50% and 80% saturation). The resulting ASP was dissolved in 50 mM PPB containing 1 M ammonium sulfate, separated by hydrophobic interaction chromatography (HIC), a and then eluted with 50 mM PPB and 1 mM PPB. The pass fractions, the 50 mM PPB eluate, and the 1 mM PPB eluate were further fractionated by ion-exchange chromatography and a linear salt gradient of 0–300 mM NaCl in 20 mM Tris–HCl (pH 8.0). Individual fractions were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and reactivity with cattle sera was analyzed by Western blotting. Bands that reacted strongly with sera from cattle infected with M. bovis were selected and subjected to liquid chromatography–electrospray ionization–mass spectrometry (LC-ESI-MS) at Yonsei Proteomic Research Centre (Seoul, South Korea), as previously described. 22
The recombinant Rv1483c protein was produced in E. coli as described above. The corresponding gene was amplified by PCR using M. tuberculosis genomic DNA as a template with the following primers: forward, 5′-CATATGACCGCTCAGAACCCCAACCTG-3′, reverse, 5′-AAGCTTTTGGGCAGCGGTGTCGAGATG-3′. The PCR products were digested with NdeI and HindIII and inserted into the pET22b(+) vector. The recombinant proteins were overexpressed in E. coli BL21 and purified by Ni-NTA affinity chromatography.
Enzyme-linked immunosorbent assay
An enzyme-linked immunosorbent assay (ELISA) was performed as previously described. 22 Briefly, the protein antigens (1 µg/mL) were diluted in coating buffer, e and 100 µLwas added to each well of 96-well microtiter plates. f After an overnight incubation at 4°C, the plates were washed 3 times in PBS with 0.05% Tween 20 (PBST), and the wells were blocked in 3% BSA in PBS at 37°C for 2 hr. After 3 more washes, 100 µL/well of cattle serum (1:50 dilution) was added to each plate, and the plates were incubated at 37°C for 1 hr. After washing, 100 µL of alkaline phosphatase (AP)-conjugated mouse anti-bovine IgG g or IgA h was added to each well at a 1:5,000 dilution. The plates were incubated for an additional 1 hr at 37°C. After 7 more washes, the reactions were developed with a p-nitrophenol phosphate tablet and a Tris buffer tablet g in 5 mL of water. The reaction was stopped with 2 M sodium hydroxide after a 30-min incubation in the dark. Optical density (OD) was measured at 405 nm with an ELISA microplate reader. i
Multiantigen print immunoassay
The native MPB70 and recombinant MPB83 proteins were immobilized on nitrocellulose membranes as narrow bands using a commercial printer. j The proteins were printed at concentrations of 0.8–1 mg/mL in PBS. A 30-µL aliquot of protein solution was sufficient to print a 30-cm long band. After antigen printing, the membrane was cut perpendicular to the antigen band into 5-mm wide strips. The nitrocellulose strips were blocked for 1 hr in 5% BSA in Tris buffered saline (TBS), and incubated at 4°C overnight with serum samples diluted 1:100 in blocking buffer. After washing with TBS with 0.05% Tween 20 (TBST), the strips were incubated at room temperature for 2 hr with AP-conjugated mouse anti-bovine IgG g or IgA h diluted 1:2,000 with 5% BSA in TBS, and washed again with TBST. Enzyme activity was visualized by incubating the strips for 3–5 min with nitro-blue tetrazolium chloride and 5-bromo-4-chloro-3′-indolyphosphate p-toluidine salt substrate. The strips were extensively rinsed in 0.01% acetic acid to stop the reaction.
Evaluation of tests and statistical analysis
The mean plus standard deviation (SD) OD of each group was used, and values are expressed as mean OD ± SD. The mean plus 2 SDs from the negative control cattle was used as the cutoff value to distinguish between positive and negative results. Differences between the 2 groups and mean differences were evaluated by a receiver operator characteristic curve analysis and the nonparametric Mann–Whitney test. Additionally, the differences between sensitivities of IgG and IgA assays for the antigens, analyzed as the number of positive samples, were examined by the chi-square test. Statistical analysis were performed using a commercial software package. k
Results
Antigen production
The native MPB70 protein was precipitated with 50–80% ammonium sulfate and did not bind to hydroxyapatite resin. Using these characteristics, MPB70 from M. bovis CFP was purified in a 2-step hydroxyapatite chromatography process followed by anion-exchange chromatography (Fig. 1A). The recombinant MPB83 was expressed as a 6× His-tagged protein in E. coli, and purified by Ni-NTA affinity chromatography (Fig. 1B). The native PstS1 protein was purified as a major single band from M. tuberculosis CFPs (Fig. 1C).
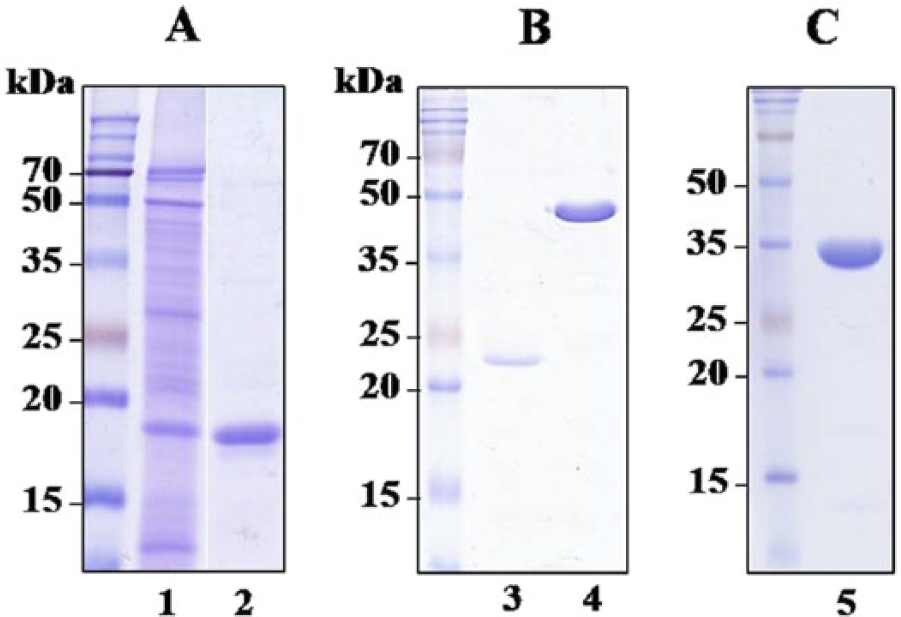
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis of the purified antigens.
Identification and production of Rv1483c
The complexity of M. tuberculosis CFPs is a barrier against searching for antigens related to new vaccines or diagnostic targets. Therefore, broad-scale fractionation of the CFPs was performed, and the seroreactivities of the fractions were determined by immunoblotting. The 0–50% ASP and 50–80% ASP of the CFPs were fractionated using HIC. The pass fraction, the 50 mM PPB eluate, and the 1 mM PPB eluate were analyzed by SDS-PAGE, and an immunoblot was performed with 2 sera (1 pool of 5 M. bovis–infected cattle and 1 pool of 5 noninfected cattle). Some protein bands reacted with sera from healthy cattle as well as cattle with bovine TB (data not shown). One band contained in the HIC 80% ASP pass fraction (Fig. 2A, arrow) reacted strongly with sera from M. bovis–infected cattle but not with control cattle (latter not shown). However, this gel band stained with Coomassie blue was very faint (Fig. 2A, arrow), and was not clearly separated by 2-dimensional electrophoresis (data not shown). Next, the HIC 80% ASP pass fraction was further separated by ion-exchange chromatography to concentrate the minor bands. As shown in Figure 2B, the seroreactive band, previously identified in Figure 2A, was detected in fractions 56–63 as a major band (marked by arrow in Fig. 2B). This band was then subjected to LC-ESI-MS analysis. The protein with the highest scoring match to the query and the highest amino acid sequence homology was Rv1483c. The DNA sequences of the Rv1483c gene are identical in both M. bovis AN5 and M. bovis BCG strains. The Rv1483c protein was produced as a 6× His-tagged recombinant protein in E. coli, and the purified protein appeared as a major band of ~47 kDa (Fig. 1B).
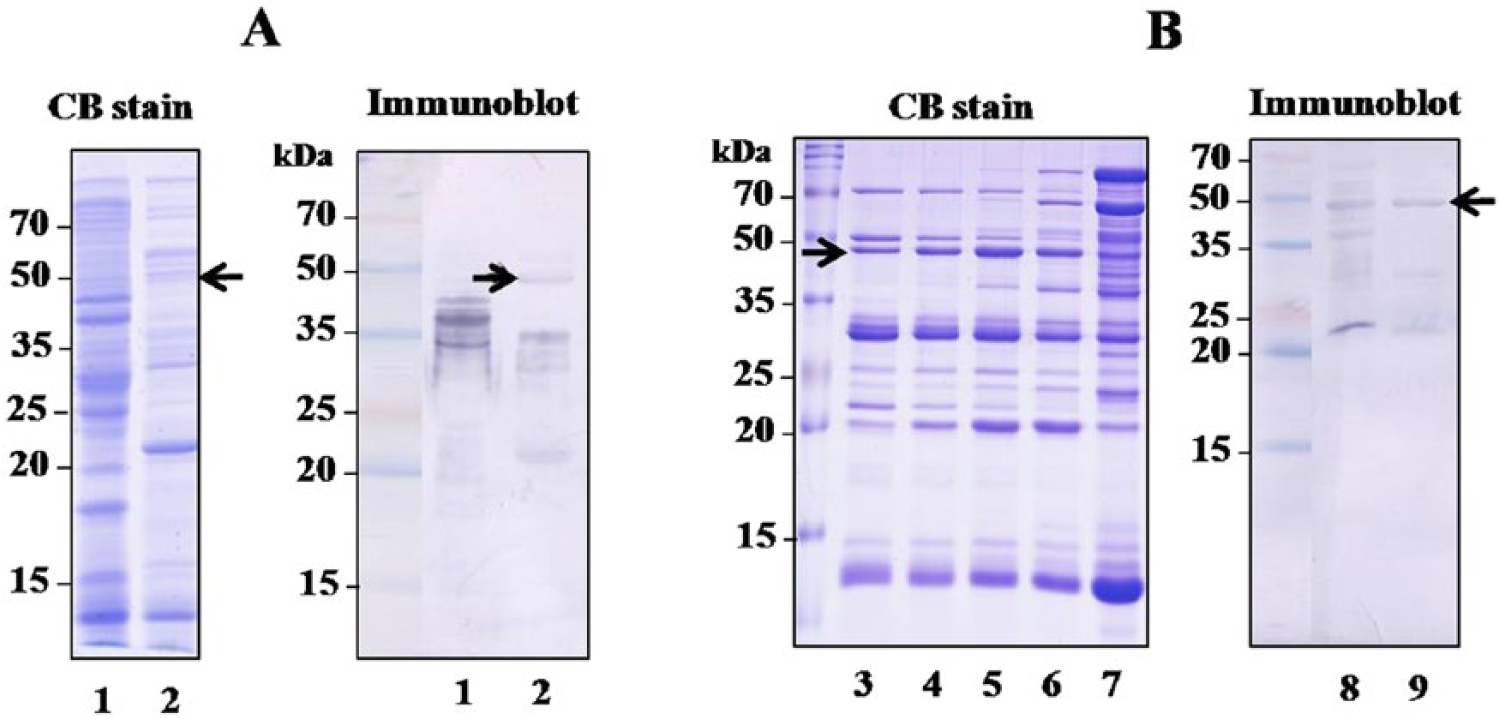
Identification of the Rv1483c protein by sodium dodecyl sulfate–polyacrylamide gel electrophoresis analyzed by either Coomassie blue (CB) stain or immunoblot with a pooled serum from 5 Mycobacterium bovis–infected cattle.
Detection of IgG and IgA antibody responses in Mycobacterium bovis–infected cattle
To evaluate the serodiagnostic potential of the IgA antibody against various antigens, serum IgG and IgA levels against the MPB70, MPB83, PstS1, and Rv1483c proteins were measured in cattle with culture-confirmed bovine TB and in noninfected cattle. Both IgG and IgA antibody responses to all 4 antigens were significantly higher in M. bovis–infected cattle than in healthy noninfected cattle (Table 1). Mean serum IgG OD values to MPB70 were significantly higher than those of IgA to this antigen in cattle with bovine TB (P < 0.0001). The IgA antibody response to Rv1483c was significantly higher than that of IgG to this antigen (P < 0.0001). No other antigen showed a significant difference between the mean values for the IgA and IgG assays. The sensitivities of anti-MPB70 and MPB83 IgA antibodies in culture-confirmed bovine TB were 100% and 96.7%, respectively. There was no significant difference between the sensitivities of IgG and IgA to the antigens except between sensitivities of anti-MPB70 IgG and anti-MPB70 IgA antibodies (P = 0.04). The sensitivities of the IgG and IgA antibodies to Rv1483c and PstS1 were significantly lower than those to MPB70 and MPB83 (P < 0.001). The specificities of the IgG and IgA antibodies to the MPB83 antigen were both 100%. These results suggest that M. bovis–infected cattle show a significant IgA antibody response that may be useful for serodiagnosis.
Immunoglobulin G (IgG) and immunoglobulin A (IgA) antibody responses to each of the 4 antigens in Mycobacterium bovis–infected and noninfected cattle.*
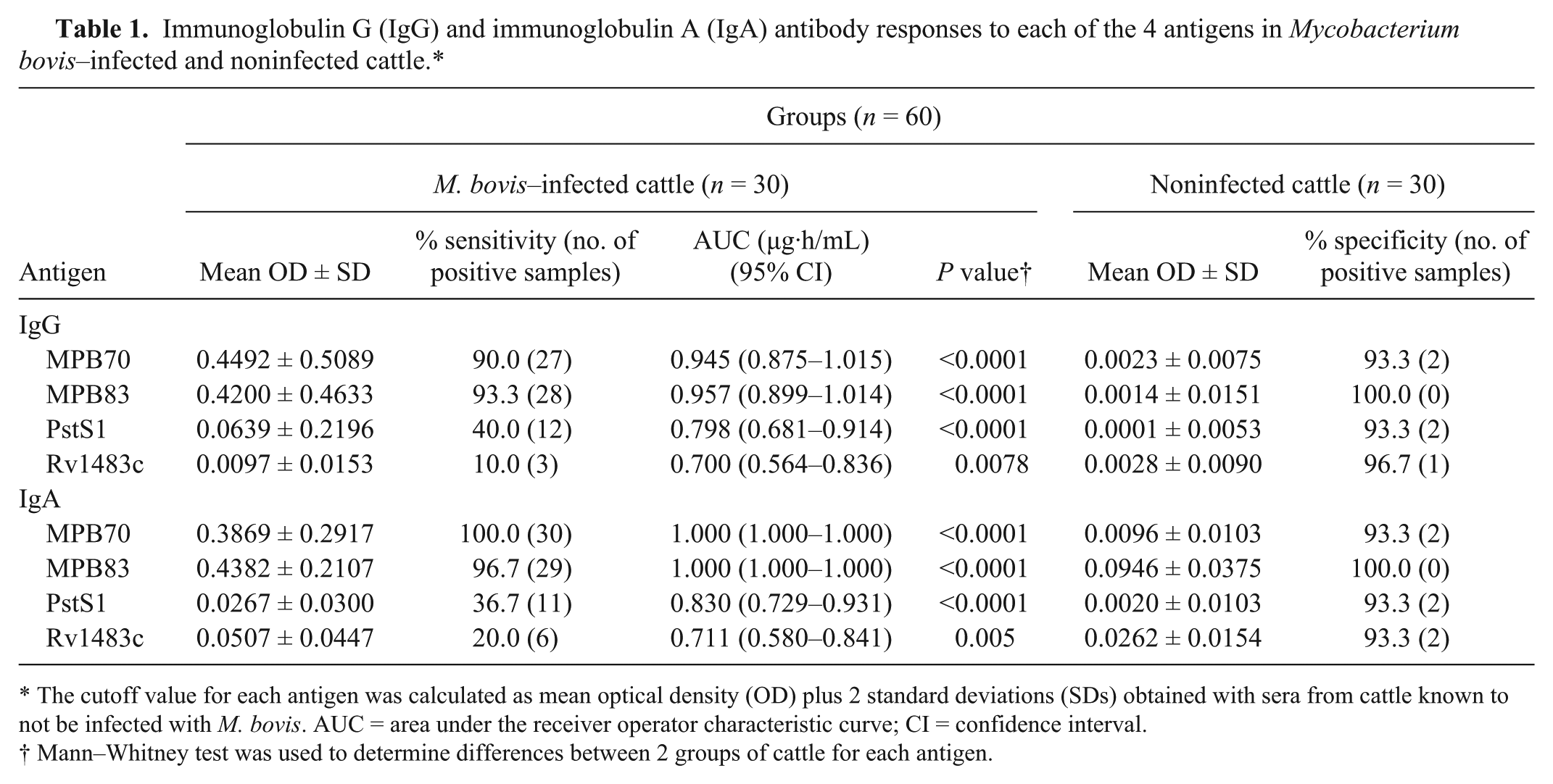
The cutoff value for each antigen was calculated as mean optical density (OD) plus 2 standard deviations (SDs) obtained with sera from cattle known to not be infected with M. bovis. AUC = area under the receiver operator characteristic curve; CI = confidence interval.
Mann–Whitney test was used to determine differences between 2 groups of cattle for each antigen.
Antibody responses in field samples
The antibody responses in field samples, which were classified into 2 groups based on a skin test, were determined. As shown in Table 2, the mean IgG and IgA OD values against the MPB70 and MPB83 antigens were significantly higher in sera from PPD(+) cattle compared to those from PPD(−) cattle. The sensitivity of the anti-MPB70 IgA antibody was significantly higher than that of the anti-MPB70 IgG antibody (P = 0.016). However, no significant differences were observed between mean OD values of the IgG and IgA antibodies to MPB83 in PPD(+) samples. The mean OD values of the anti-PstS1 IgG antibody were significantly higher in sera of PPD(+) cattle compared to those in PPD(−) cattle, but not the anti-PstS1 IgA antibody. The antibody titers to PstS1 were significantly lower than those to MPB70 and MPB83 (P < 0.0001). No significant differences were observed between antibody responses to Rv1483c in both PPD(+) and PPD(−) cattle. Among the 89 PPD(+) subjects, 10 and 9 sera that were negative for IgG against MPB70 and MPB83, respectively, were positive for IgA. As well, 8 sera that were negative for IgA against MPB83 were positive for IgG. There were no sera IgA negative and IgG positive against MPB70. When compared to the sensitivity of IgG antibody class alone, combining the results for IgG and IgA against MPB70 significantly increased sensitivity to 93% (P = 0.016) in the PPD(+) field samples. An increase in sensitivity for the combined IgG and IgA results for MPB83 to 79.7% (P = 0.031) was also seen in these field samples. These results suggest that simultaneously determining both IgA and IgG may help increase sensitivity, such as when using multiple antigens.
Immunoglobulin G (IgG) and immunoglobulin A (IgA) antibody responses to each of the 4 antigens in field samples.*
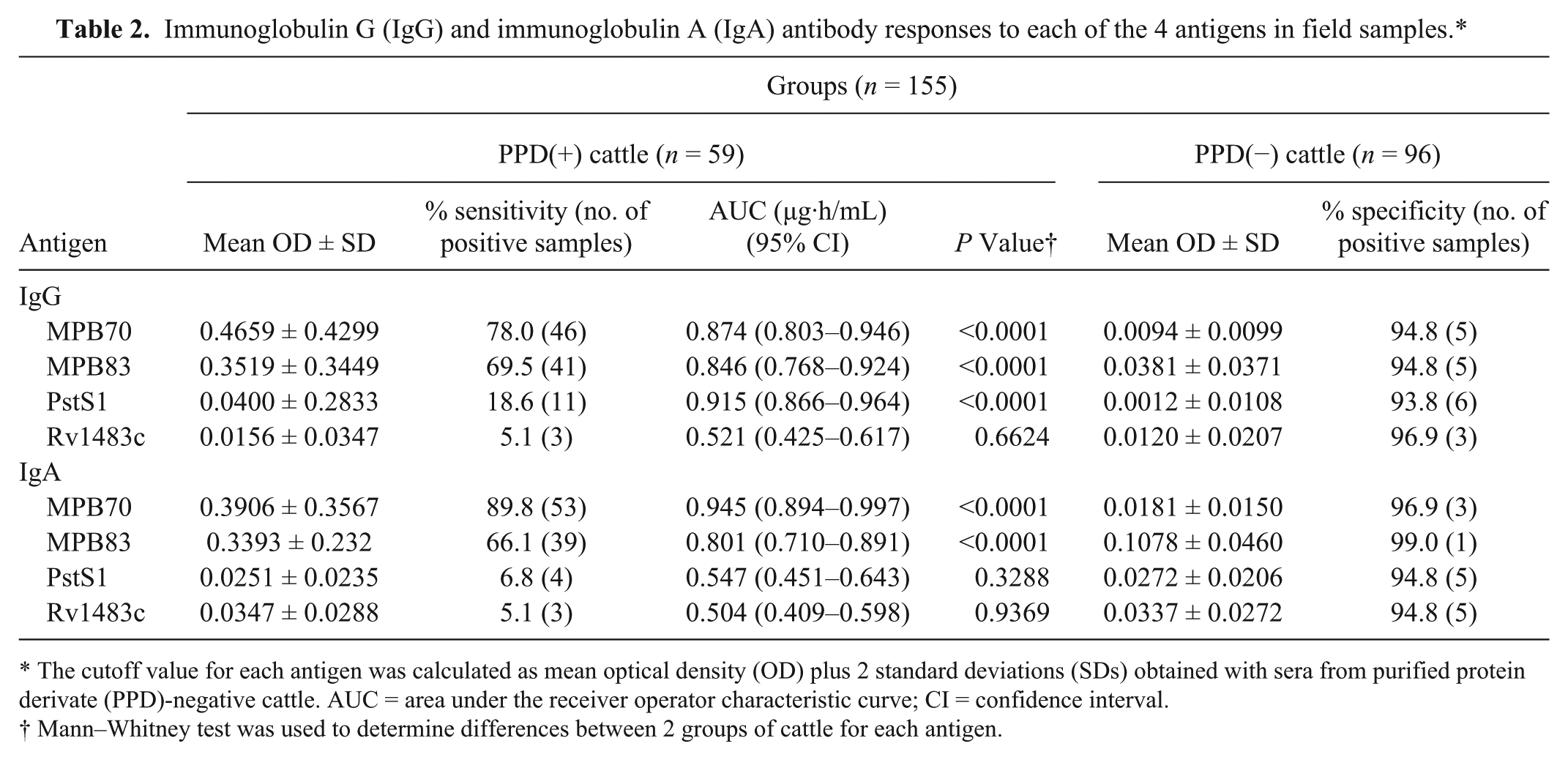
The cutoff value for each antigen was calculated as mean optical density (OD) plus 2 standard deviations (SDs) obtained with sera from purified protein derivate (PPD)-negative cattle. AUC = area under the receiver operator characteristic curve; CI = confidence interval.
Mann–Whitney test was used to determine differences between 2 groups of cattle for each antigen.
Multiantigen print immunoassay
As OD titers against the PstS1 and Rv1483c antigens in the ELISA were relatively low, only the MPB70 and MPB83 antigens were printed onto nitrocellulose membranes. The IgA and IgG antibody reactivities were then measured in cattle sera. Some typical reactions are shown in Figure 3. Both IgA and IgG reacted strongly with MPB70 and MPB83 in sera from culture-confirmed bovine TB (Fig. 3A) and PPD(+) field samples (Fig. 3B), but very weakly or not at all in noninfected cattle and PPD(−) field samples, with the exception of 1 serum sample (Fig. 3B, lane d). The PPD(+) field samples that were negative in the ELISA showed no prominent bands (Fig. 3B, lanes marked with b). Some sera that were positive in the ELISA, but with low IgG and IgA antibody titers, showed only a weak reactivity in the multi-antigen print immunoassay (MAPIA; Fig. 3, lanes a and c).
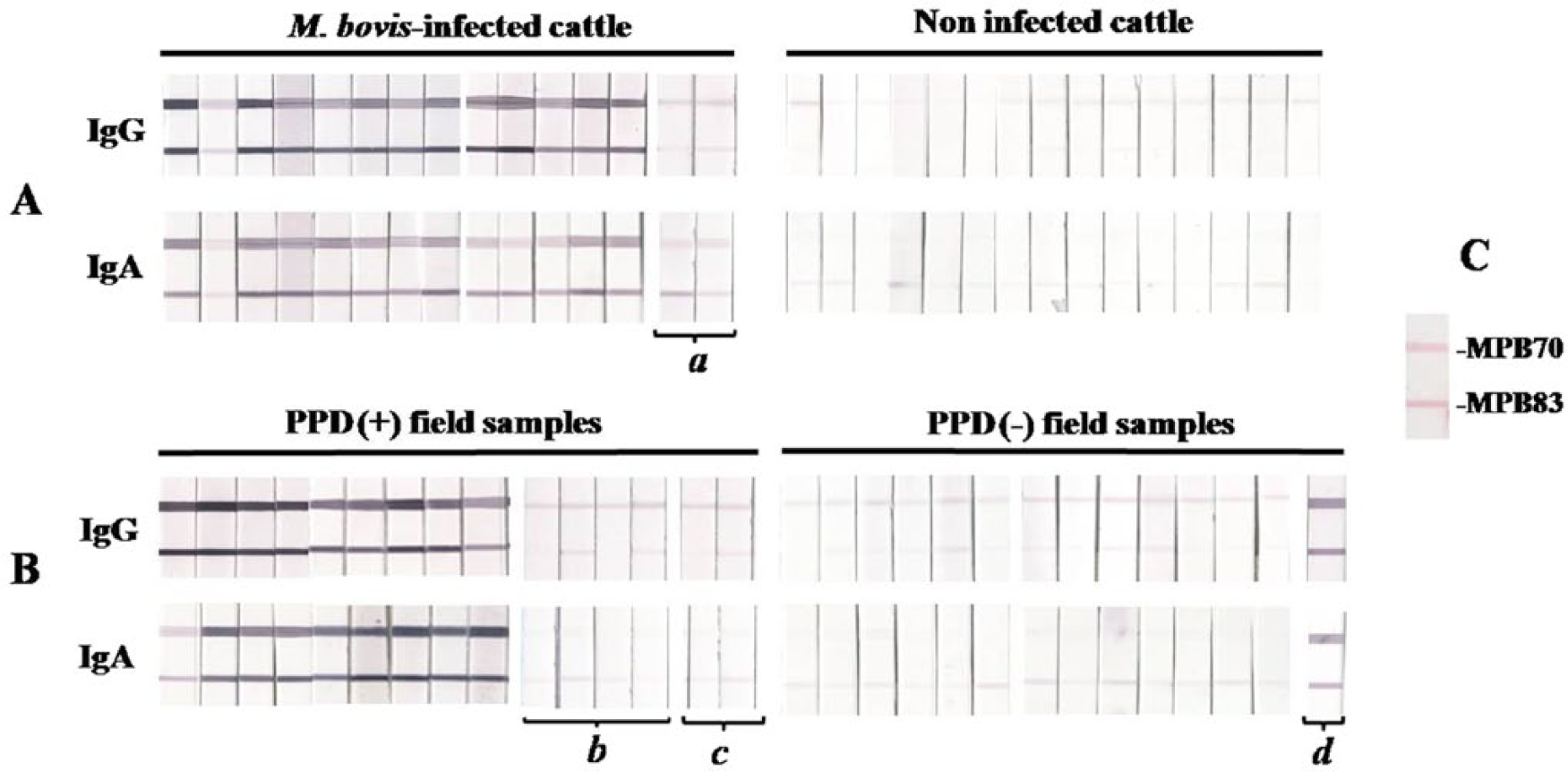
Representative immunoglobulin G (IgG) and immunoglobulin A (IgA) antibody reactivity results to MPB70 and MPB83 by multiantigen print immunoassay (MAPIA). Both antigens were printed onto nitrocellulose membranes. The strips reacted with sera from Mycobacterium bovis–infected or noninfected cattle (
Discussion
Animal reservoirs of M. bovis infection as a source for TB in cattle are a significant problem, and the availability of a rapid serodiagnostic test that can be used in a range of animals is of great importance. 29 However, serological tests have not demonstrated a sufficiently high performance,19,26 which has led to efforts to identify novel antigens and add them to multiantigen approaches. The M. bovis MPB70 and MPB83 proteins appear to be major targets for detecting bovine TB. Some antigens, including ESAT6, have been used as an additional protein in diagnostic kits 27 or as fusion proteins. 23 In addition, antibody classes (IgG, IgM, and IgA) that bind to a specific antigen have been analyzed for diagnosing human TB.3,11,14,20 However, to our knowledge, no reports have assessed the IgA antibody response to antigens for serodiagnosing bovine TB. In this study, both IgG and IgA antibody responses against MPB70, MPB83, PstS1, and the Rv1483c proteins were compared in M. bovis–infected and noninfected cattle, as well as in field samples. The results of the current study reveal that IgA had superior or comparable diagnostic utility to that of IgG depending on the antigens tested in the ELISA.
The sensitivity of IgM is too low in human patients with TB for diagnostic application.11,20 Therefore, only IgA and IgG antibody responses to MPB70 and MPB83 were measured in the current study. The sensitivity of the IgA antibody to MPB70 was 100% in culture-confirmed bovine TB. The sensitivity of the IgA antibody to MPB70 in PPD(+) field samples was also higher than that of IgG. However, mean IgA OD values against all tested antigens except Rv1483c were lower than those of IgG to these antigens in cattle with TB. This finding was reported in a study that evaluated IgA responses to the ESAT-6 and Rv2031c antigens in human TB. 12 The overall sensitivities of IgG and IgA in culture-confirmed bovine TB and PPD(+) field samples were 81.5% and 93.8%, respectively, for MPB70, and 74.1% and 75.3%, respectively, for MPB83. Comparable results to the current study have been reported in an evaluation of 2 commercial kits for detecting the IgG antibody to MPB70 and MPB83 where a sensitivity of 70–85% was found. 1 There is a promising role for IgA in the diagnosis of human TB as studies suggest that the serum IgA response to the 16-kDa antigen discriminates between patients with TB and controls,4,12 and that the IgA antibody to the P-90 antigen distinguishes individuals recently infected with M. tuberculosis. 3 The results of the current study suggest that the IgA antibody against MPB70 and MPB83 could be valuable for diagnosing bovine TB. As expected, the combination of the IgG and IgA results gave the higher diagnostic sensitivity for field samples than the use of IgG antibody class alone.
The M. tuberculosis PstS1 glycoprotein is the most frequently studied serological antigen in human TB. 10 In the current study, the native protein reacted with bovine TB sera, but the antibody titers were significantly lower than those to MPB70 and MPB83. This may have been due to the lower expression of the PstS1 antigen, although M. bovis has a gene encoding the protein. 7 In contrast to the MPB70 and MPB83 proteins, the sensitivity of the IgA antibody to the PstS1 antigen was significantly lower than that of IgG. Furthermore, no significant difference was observed between mean IgA OD values in either PPD(+) or PPD(−) field samples. These results suggest that the IgA antibody to the PstS1 antigen lacks sensitivity for detecting bovine TB in the field.
Identifying serologically active antigens and evaluating their usefulness to distinguish bovine TB from healthy non-infected cattle needs to be investigated. Extensive proteomic analyses of M. bovis CFPs to identify seroreactive proteins in cattle have been undertaken.2,5,6 Because M. tuberculosis and M. bovis have different antigenic profiles, the current study analyzed M. tuberculosis CFPs seeking a novel antigen with strong seroreactivity in cattle. In fact, the Rv1483c protein reacted strongly with M. bovis–infected cattle sera, but existed as a very faint band in total CFPs. This protein band was enriched by fractionation of total CFPs using 2-step chromatography. Because the Rv1483c protein gene sequence is identical in M. tuberculosis and M. bovis, a recombinant Rv1483c protein was produced, and the antibody responses of this protein were tested in cattle. The IgA antibody titers to Rv1483c were significantly higher than those of IgG in M. bovis–infected cattle. However, the sensitivities of the IgA and IgG antibodies to Rv1483c were very low. Furthermore, there was no diagnostic utility for Rv1483c in field samples.
The MAPIA approach, a novel method for a cocktail-based serological diagnosis, was originally developed to examine the diversity of heterogeneous antibody responses and to enhance the sensitivity for diagnosing infectious diseases.8,16 The application of MAPIA to diagnose human and bovine TB has been used because it is relatively easy to incorporate multiple antigens as compared to ELISA applications. In the current study, IgA antibodies also reacted strongly with antigens tested in the MAPIA, suggesting that they could be used to discriminate TB-infected cattle from healthy ones.
It has already been noted that an effective serological test can detect infected animals missed by the routine tuberculin skin test. 28 The results of the current study support the feasibility of using assays based on IgA antibodies against MPB70 and MPB83 to detect bovine TB.
Footnotes
a.
DEAE-sepharose CL-6B for ion-exchange chromatography and phenyl sepharose for HIC; GE Healthcare Biosciences, Piscataway, NJ.
b.
Pierce Biotechnology Inc., Rockford, IL.
c.
Novagen, Madison, WI.
d.
Qiagen Inc., Valencia, CA.
e.
KPL Inc., Gaithersburg, MD.
f.
Nalge Nunc International Corp., Rochester, NY.
g.
Catalog no. A7554, Sigma-Aldrich, St. Louis, MO.
h.
Catalog no. A10-121AP, Bethyl Laboratories Inc., Montgomery, TX.
i.
Molecular Devices Corp., Sunnyvale, CA.
j.
Matrix 1600, Kinematic Automation, Sonora, CA.
k.
Prism version 5, GraphPad Software Inc., San Diego, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Animal and Plant Quarantine Agency (Z-1541778-2012-13-01), Ministry of Agriculture, Food and Rural Affairs, Republic of Korea.
