Abstract
Tuberculosis pathology was studied on 19 African buffalo (
The current study provides a systematic and detailed description of the macroscopic and histopathologic lesions caused by natural
The buffalo used in the present study were obtained from a test-and-cull operation, aimed at reducing the prevalence of bTB in the species. The program started in 1999 in HiP and includes annual buffalo capture for standard bovine comparative intradermal test, using both avian and bovine Dutch tuberculin, a followed by culling of bTB-positive buffalo. 12 All positive reactors in 1 test-and-cull operation (24 buffalo) were killed by a gunshot to the brain and transported to the HiP abattoir. At slaughter inspection, 19 out of 24 animals showed visible gross lesions suggestive of tuberculosis, and these buffalo were selected for further pathology studies.
The age category, subadult or adult (Table 1), was determined based on tooth eruption patterns. The lungs were sliced at 2-cm intervals, and each slice was inspected and palpated. The following LNs were sliced (approximately 2-mm thick slices), and each slice was visually inspected: head-associated LN (paired mandibular, parotid, and medial retropharyngeal), thoracic LN (mediastinal and bronchial), abdominal LN (mesenteric, hepatic, omasal, and abomasal), and paired peripheral superficial LN (superficial cervical, axillary, and popliteal). Tissues with lesions were fixed in formalin for histopathology.
Nineteen African buffalo (
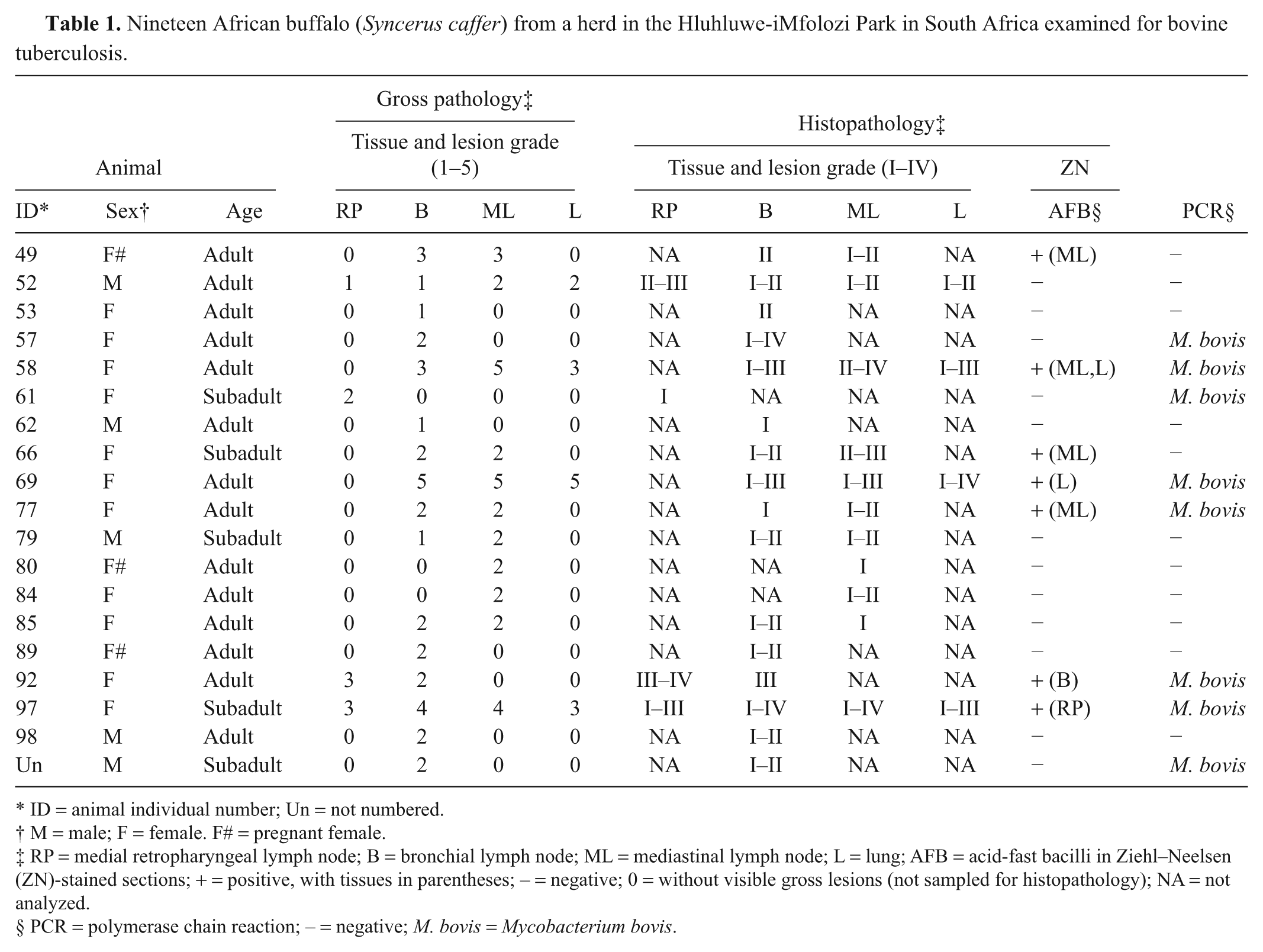
ID = animal individual number; Un = not numbered.
M = male; F = female. F# = pregnant female.
RP = medial retropharyngeal lymph node; B = bronchial lymph node; ML = mediastinal lymph node; L = lung; AFB = acid-fast bacilli in Ziehl–Neelsen (ZN)-stained sections; + = positive, with tissues in parentheses; – = negative; 0 = without visible gross lesions (not sampled for histopathology); NA = not analyzed.
PCR = polymerase chain reaction; – = negative;
Any focus, foci, or nodules, either as a single lesion, or multifocal or confluent, that were yellow–white, circumscribed, and solid, or necrotic, with or without apparent caseation and mineralization, were considered and graded as macroscopic tuberculous lesions. The lung and LN lesions were categorized in grades 0–5 according to the size and number of their gross lesions. Grade 1 was used for a single minimal lesion, up to 1 mm; grade 2 for 2–4 lesions of 2–5 mm; grade 3 for 5–8 lesions, up to 10 mm, or many small lesions affecting approximately 50% of a LN; grade 4 for confluent and extensive lesions in most slices but with some normal looking tissue left; and grade 5 for abundant lesions, with none or nearly no apparently healthy tissue left. The lungs and LNs that did not show visible lesions were classified as grade 0 and were not sampled for further histopathology (Table 1). Histological sections were stained with hematoxylin and eosin, with Ziehl–Neelsen stain to identify acid-fast bacilli (AFB) and with Masson trichrome to visualize fibrous tissue.
The tuberculous granulomas in the LNs and lungs were microscopically classified into 4 categories of development according to criteria previously described for cattle. 22 The lesions were evaluated for their size, presence of AFB, type and extension of necrosis and mineralization, abundance and distribution of inflammatory cell components, and degree and architecture of fibrosis. Briefly, the granulomas were classified into 4 categories, namely: stage I (initial or early lesions); stage II (solid granulomas); stage III (minimal necrosis); or stage IV (necrosis and mineralization).
DNA was extracted from formalin-fixed, paraffin-embedded tissue sections as previously described.
11
Real-time polymerase chain reaction (PCR) was performed by using a commercial system.
b
The real-time PCR mixtures for amplification of DNA consisted of 50-μl total volume containing: 25 μl of 2× power SYBR Green,
c
5 μl of template, and 10 pmol of each oligonucleotide primer (myc1 and myc3)
d
in a final 50-μl reaction volume. These primers simultaneously amplify the
Results showed all animals to be in good body condition. Postmortem lesions consistent with tuberculosis were found in the LNs of the head, thoracic cavity, and/or lungs of the 19 animals (Table 1). Lesions suggestive of tuberculosis were not detected in other organs or LNs. The spectrum (macroscopic grades 1–5) of lesions in the LNs ranged from small (1 mm in diameter) white foci, to single or multiple encapsulated granulomas of different sizes, with central caseation and mineralization, or to diffuse, nonencapsulated lesions effacing and replacing normal lymphoid tissue.
A majority of the animals showed tuberculous lesions in the bronchial (16/19) and mediastinal (11/19) LNs. Twenty-one percent (4/19) of the animals presented tuberculous lesions in the lungs and 21% (4/19) in the retropharyngeal LNs. Forty-seven percent (9/19) of the animals presented lesions in only one LN, 31.6% (6/19) in 2 LNs, 10.5% (2/19) in 2 LNs as well as in the lungs, and 10.5% (2/19) showed lesions in 3 different LNs and in the lung. Most buffalo (73.7%) presented macroscopic tuberculous lesions of early stages (grade 1 and/or 2) while 26.3% (5/19) presented moderate to high severity gross lesions (grades 3–5). Grade 5 lesions were observed in only 2 (10.5%) animals, no. 58 (in the mediastinal LN) and no. 69 (in the bronchial and mediastinal LNs and lung; Table 1). All 4 microscopic stages were found. Stage I lesions were characterized by irregular, unencapsulated clusters of predominantly epithelioid macrophages, lymphocytes, and few multinucleated giant cells, without necrosis (Fig. 1); stage II were partly or completely thinly encapsulated granulomas formed by predominantly epithelioid macrophages, Langhans giant cells, and lymphocytes surrounding minimal areas of necrosis; stage III were encapsulated granulomas with central necrosis, often caseous and mineralized, surrounded by epithelioid cells, Langhans giant cells, and lymphocytes at the periphery (Fig. 2); and stage IV were large, thickly encapsulated, multicentric granulomas with extensive caseous necrosis and often multiple areas of mineralization surrounded by epithelioid cells, giant cells and lymphocytes. Extensive lesions formed by multifocal to coalescing stage I granulomas were frequently observed. Some granulomatous lung lesions involved the wall of the bronchioles, occasionally extending into the lumen. Most animals presented microscopic TB lesions of stage II. There were often several lesions of different stage within a single LN. The most chronic or advanced microscopic stage (IV) was seen in 5 animals. One buffalo with severe macroscopic lesions presented microscopic granulomas of stage III. In LNs with macroscopic lesions of grades 4 and 5, it was common to find lesions of microscopic stage IV in that tissue. Neovascularization was a frequent finding in the peripheral connective tissue in stages III and IV granulomas. Acid-fast bacilli were observed in tissues from 7 animals (36.8%), most with macroscopic lesions of grades 3–4. The AFB were detected in mediastinal LNs (4/7), lung (2/7), bronchial LNs (1/7), and retropharyngeal LNs (1/7). When present, AFB were found in low numbers, mostly as a single bacillus in multinucleated giant cells (Table 1). Amplification of DNA by real-time PCR identified the genome of
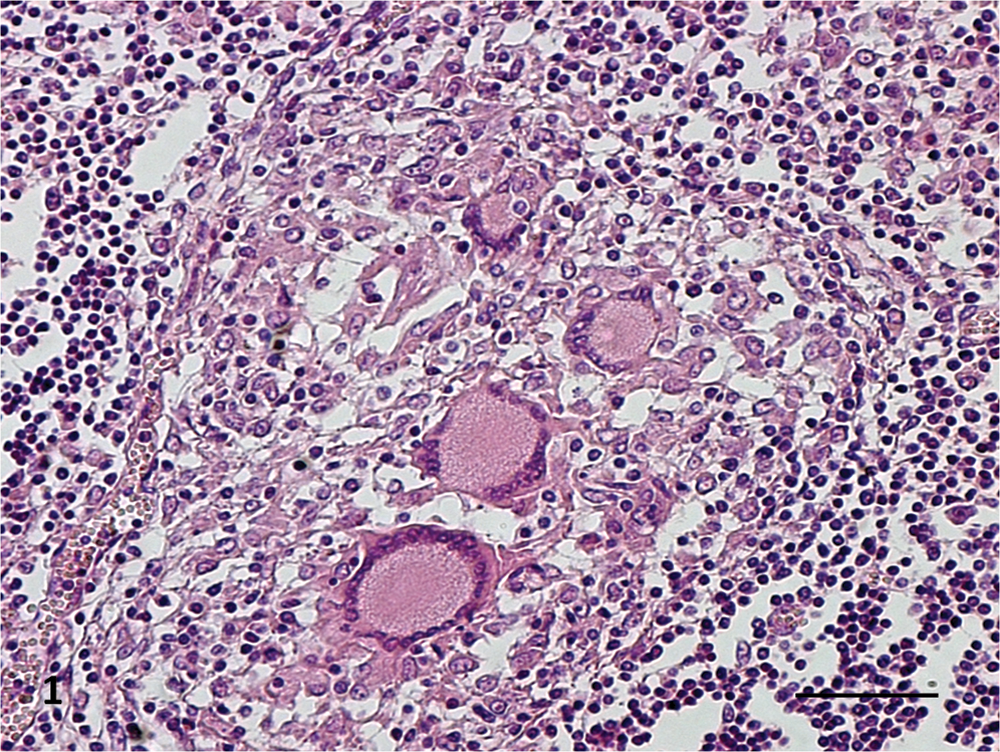
Histological section of a mediastinal lymph node of an African buffalo (
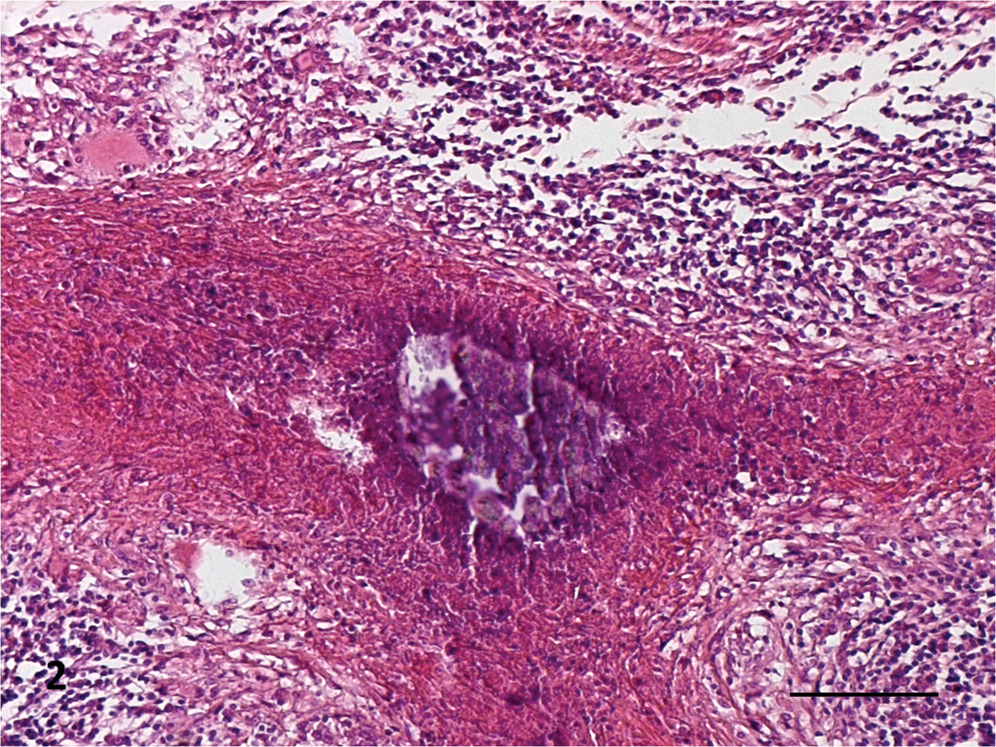
Histological section of a bronchial lymph node of an African buffalo (
Of the 24 animals that tested positive with the tuberculin skin test and were slaughtered, 19 (79.2%) presented macroscopic and microscopic tuberculous lesions. The animals were in good body condition, and the lesions affected only a few LNs, indicating an early or limited disease stage. Only 3 animals (nos. 58, 97, and 69) presented more severe lesions in 2 or 3 LNs, as well as in the lung. Advanced cases are less likely to be found in the herds subjected to test-and-cull program, since many of the positive animals that may have had advanced disease have probably been removed since the program in HiP began in 1999. A study in HiP demonstrated that approximately 70% of bTB cases in buffalo were mild, 12 and it was considered unlikely that they had shown clinical signs.2,6 This supports the assertion that bTB in buffalo is a disease of limited and slow development in the majority of the cases. 12
Studies have shown that gross examination at slaughter fails to detect some of the
The buffalo presented lesions most frequently in the LNs associated with the respiratory tract (mediastinal and bronchial LNs) and in the lungs, suggesting that the most likely route of infection was inhalation. The findings are consistent with previous descriptions in African buffalo,7,13 water buffalo, 9 and are also similar to bTB in cattle. 15 Similar to other studies in cattle 16 and buffalo, 8 the tuberculous lesions in the lungs were common in the caudal lobes. Most of the buffalo (80%) did not show bTB lesions in the lungs, but had gross lesions in bronchial and mediastinal LNs. This agrees with descriptions of early stages of bTB in cattle, and is most likely due to the difficulties of detecting very small lesions in the lungs at inspection, due to their large volume. 15 In the present study, pulmonary lesions were detected in 21% of the buffalo, and they were of the higher grades. The absence of lesions in the gastrointestinal tract suggests that the oral route is not the main route of infection in buffalo. 13
As in other studies, most of the animals (73.7%) included in the current study were adults.12,19 It has been reported that young buffalo have a lower risk of being positive for bTB than older buffalo, but that the risk for both age groups increases with increasing prevalence of the disease within the herd.
19
Microscopically, in 2 animals with severe lung lesions, the granulomas had eroded the wall of bronchioles, indicating that these animals could excrete
Animals that presented higher macroscopic lesions, grades 3–5, were the most likely to present up to 4 different microscopic grades in the same section. This result agrees with a previous report in naturally TB-infected cattle,
15
and it likely represents repeated seeding of the LNs by
Polymerase chain reaction identified 3 animals in which acid-fast bacteria were not detected, but conversely, acid-fast staining identified organisms in 2 animals that were negative by real-time PCR. Overall, the 2 methods combined identified DNA and/or acid-fast organisms in 10 out of 19 animals that had gross postmortem and histological lesions compatible with
In conclusion, the gross and microscopic morphology of TB in the African buffalo closely resembles TB in cattle. The current study presents a systematic evaluation and semiquantification of the severity and stages of development of tuberculous lesions in naturally infected buffalo. The results may contribute to 1) the understanding of the pathogenesis of the disease, 2) the evaluation of experimental models of
Footnotes
Acknowledgements
The authors thank Dr Dave Cooper, from HiP, for providing the excellent facilities and appropriate environment for the sampling. The staff of the division of Pathology, Veterinary Faculty, Eduardo Mondlane University is acknowledged for the work with cutting and staining of histological sections.
a.
PPD, Institute for Animal Science and Health, Lelystad, The Netherlands.
b.
7300, Applied Biosystems, Foster City, CA.
c.
Applied Biosystems, Warrington, United Kingdom.
d.
TIB Molbiol, Berlin, Germany.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was funded by SIDA-SAREC, with contribution of the European Union Seventh Framework Programme grant agreement no. 222633 (WildTech).
