Abstract
We characterized bovine viral diarrhea virus (BVDV)-related abortions in cattle and identified the species and subgenotypes in the state of Santa Catarina, southern Brazil. Our RT-PCR assay was positive for BVDV in 5 fetuses from different farms; however, 3 of the 5 fetuses were also PCR-positive for Neospora caninum. In the 5 BVDV-positive fetuses, gross lesions included fetal mummification (1), hepatomegaly (1), subcutaneous edema (1), and perirenal edema (1). Predominant histologic lesions included epicarditis and mild-to-moderate lymphoplasmacytic myocarditis (5), mild multifocal lymphoplasmacytic interlobular pneumonia (4), nephrosis associated with moderate multifocal interstitial nephritis (1), moderate multifocal lymphoplasmacytic necrotic hepatitis (1), and mild multifocal lymphoplasmacytic meningitis (1). The amplification products from the Pestivirus 5′UTR region of 4 of the 5 fetuses had 96.3–100% similarity between fetal strains and reference strains. The samples were distributed into 2 branches of the phylogenetic tree; strains UDESC:01, UDESC:02, and UDESC:05 clustered in the BVDV-1e branch, uncommon in the Americas, and strain UDESC:04 clustered in the BVDV-2b branch. The three 1e strains had 96.9–97.4% similarity.
Bovine viral diarrhea virus (BVDV; Flaviviridae, Pestivirus) has a worldwide distribution and wide genetic and antigenic variability, and is classified into 2 distinct species, BVDV-1 (Pestivirus bovis) and BVDV-2 (Pestivirus tauri) and a separate species that infects cattle identified as HoBi-like pestivirus (Pestivirus brazilense).19,22
BVDV can cause bovine abortions at any stage of pregnancy. 7 Diagnosis can be difficult as a result of tissue autolysis, viral RNA integrity, 14 and the nonspecificity of histologic lesions, characterized mainly by nonsuppurative inflammation in various organs. 17 Comprehensive reports on bovine abortions involving BVDV in Brazil are scarce.6,13 There is no description of abortions caused by BVDV in cattle in Santa Catarina, even though there is evidence of virus circulation following the first detection in the 1990s. 10 We characterized the anatomopathologic aspects of BVDV detected in 5 aborted bovine fetuses in the state of Santa Catarina, and determined the involved subgenotypes.
Cases 1–5 originated from different farms in Santa Catarina state, southern Brazil, in 2020 and 2021. Four fetuses were from dairy herds: 2 of which were Holstein (cases 2, 4), 1 Jersey (case 3), and 1 crossbred (case 1) herd. One fetus was from a mixed-breed beef herd (case 5). Four farms purchased replacement pregnant heifers from other farms. All farms used inactivated vaccines containing BVDV-1a and BVDV-2a strains during the breeding season.
Postmortem examinations were performed in the Laboratório de Patologia Animal (Centro de Ciências Agroveterinárias, Universidade do Estado de Santa Catarina [UDESC], Lages, Santa Catarina, Brazil). The crown-rump lengths of the fetuses were measured. Three fetuses (cases 1, 2, 5) were aborted during mid-gestation; one fetus (1) was aborted in early gestation and one (4) in late gestation. Two fetuses had gross findings. Fetus 3 was mummified (Fig. 1A). Fetus 4 had hepatomegaly as a result of chronic passive congestion (Fig. 1B) and systemic edema. The placenta was available for examination in 2 cases, with a wrinkled and dry appearance in the mummified fetus (3) and no gross findings in the other (1).
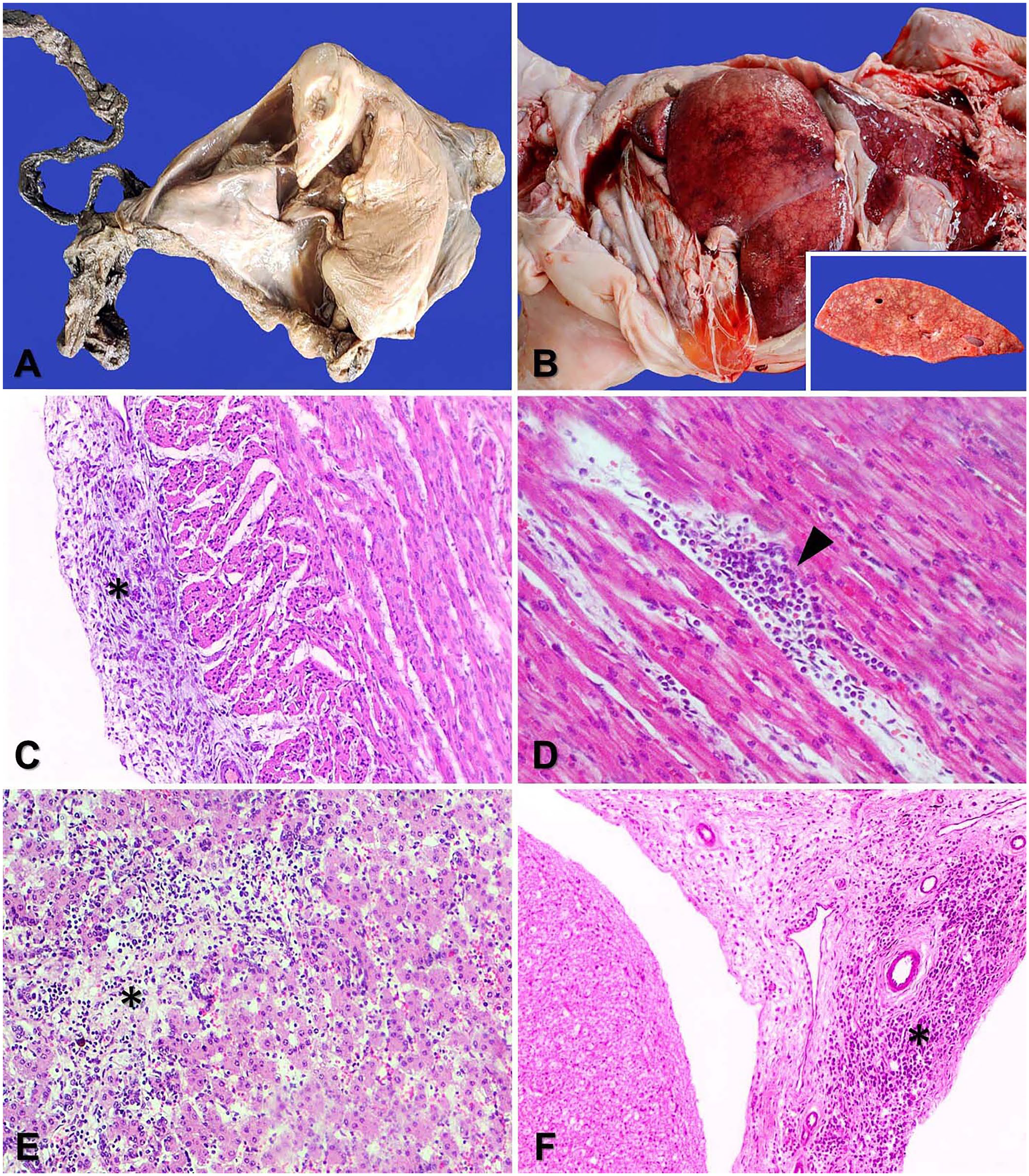
Gross and histologic lesions in bovine fetuses with BVDV infection in the state of Santa Catarina, Southern Brazil.
Samples of brain, spinal cord, heart, lung, liver, kidney, thymus, spleen, urinary bladder, rumen, reticulum, omasum, abomasum, intestines, bone marrow, skeletal muscle, and diaphragm were collected from each fetus, fixed in 10% neutral-buffered formalin, processed routinely, and sections stained with H&E. The major histologic lesion of each fetus was mild-to-moderate multifocal lymphoplasmacytic epicarditis (Fig. 1C) and myocarditis (Fig. 1D). Four fetuses (cases 1, 2, 4, 5) had mild multifocal lymphoplasmacytic skeletal myositis and mild multifocal interlobular pneumonia. Fetus 4 also had moderate, multifocal, subpleural hemorrhage and pulmonary thrombosis.
Placental lesions were evident in 2 cases and were characterized by moderate diffuse lymphoplasmacytic placentitis (case 1) and lymphoplasmacytic placentitis with moderate diffuse infiltrates of macrophages and neutrophils (case 3). Fetus 4 had lesions in kidney, liver, and spinal cord, namely mild multifocal tubular necrosis and interstitial lymphoplasmacytic nephritis, mild multifocal hepatocellular necrosis with lymphoplasmacytic infiltrates and hepatocellular degeneration (Fig. 1E), and mild multifocal lymphoplasmacytic meningitis (Fig. 1F).
Spleen and thymus samples were tested with a reverse-transcription PCR (RT-PCR) BVDV assay using primer pairs 324F and 326R (5′-ATGCCCTTAGTAGGACTAGCA-3′; 5′-TCAACTCCATGTGCCATGTAC-3′), which amplified a 288-bp product from the 5′UTR region of the Pestivirus genome. 20 Samples from all 5 bovine fetuses were positive.
For BVDV gene sequencing and phylogenetic analysis, amplification products were purified and then Sanger sequenced. BioEdit v.7.2 (https://thalljiscience.github.io/) was used to assemble the 288-bp partial sequences of the obtained 5′UTR, which were later analyzed using BLAST. Pestivirus strain sequences deposited in GenBank, and strains obtained from the 5 fetuses, were aligned for phylogenetic analysis using CLUSTAL W. The phylogenetic tree was constructed with MEGA 11 (https://www.megasoftware.net/) using the maximum-likelihood method, with bootstrap analysis of 500 repetitions, and the Kimura 2-parameter model.
It was not possible to sequence the amplification product obtained from fetus 3 because of the poor quality of the amplified product in the RT-PCR assay. Gene sequencing followed by phylogenetic analysis showed that the strains detected in 4 fetuses clustered into 2 distinct clades, corresponding to BVDV-1 and BVDV-2 (Fig. 2). Most strains (3 of 4; UDESC:01, -02, -05) clustered in the BVDV-1 branch, together with subgenotype 1e reference strains. Only one strain (UDESC:04) clustered in the BVDV-2b branch.
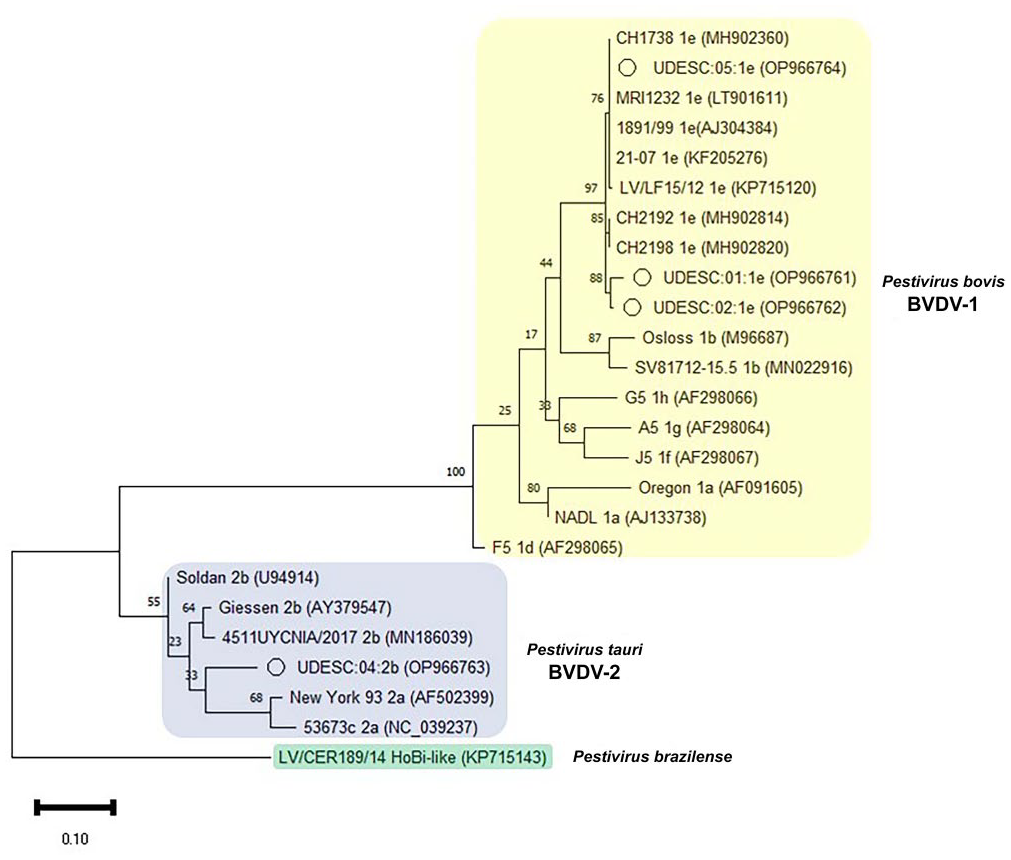
Phylogenetic tree based on the 5′UTR region of the Pestivirus genome. Sequences obtained from aborted bovine fetuses and reference Brazilian and international strains referring to BVDV-1, BVDV-2, and Hobi-like pestivirus, analyzed by the neighbor-joining method and Kimura 2-parameter model with 500 bootstraps. The strains of our study are highlighted with the (o) symbol. The GenBank accession for each strain is in parentheses.
Brain samples were also tested for Neospora caninum with a PCR assay using Np21 and Np6 primers, 5 and for Toxoplasma gondii using SAG2.F4 and SAG2.R4 primers. 21 Brain samples from fetuses 1–3 were positive for N. caninum on PCR; samples from fetuses 4 and 5 were negative for N. caninum on PCR. All fetal brain samples were negative for T. gondii. Kidney and liver fragments underwent PCR assay at Vertà Laboratory (Brazil; methodology not informed) for Leptospira spp. detection, with negative results. Liver, lung, and abomasal fluid samples underwent aerobic culture with absence of bacterial growth.
Establishing the causes of abortion is complex. 15 We used a RT-PCR assay to detect BVDV, which is the detection technique of choice in fetal tissues.6,16 BVDV can be difficult to detect in this type of sample because of tissue collection and storage quality as well as the level of tissue autolysis and consequent viral RNA degradation.7,15 We found no previous descriptions of abortions in cattle attributed to BVDV in the state of Santa Catarina in a PubMed search; there are scarce reports of BVDV abortion in Brazil6,13 as well as in the United States and Argentina.1,17
Most fetuses were in the second trimester of gestation, unlike in other reports in which the most common occurrence was in the third trimester, 17 which supports the belief that BVDV can cause fetal death at any stage of pregnancy. 7 Macroscopic changes commonly associated with BVDV include malformations, especially in cases of maternal infection at 80–150 d of pregnancy. 2 However, one of our macroscopic findings was fetal mummification, in the case of the fetus concomitantly infected with BVDV and Neospora caninum. The involvement of BVDV in fetal mummification has been verified in cases of spontaneous mummification in dairy cattle. 6 N. caninum has also been associated with naturally occurring fetal mummification in cattle. 5
Another relevant macroscopic finding was hepatic congestion with a lobular pattern, and edema in the subcutis and other organs. These changes may have resulted from the myocardial and epicardial lesions found in one of the fetuses and suggests congestive heart failure. 16 Myocarditis and epicarditis were the main changes found in the fetuses, as previously observed in 6 bovine fetuses aborted in late gestation, associated with viral antigen immunostaining 3 and also in fetuses aborted by N. caninum in any gestational period. 5 Cardiac lesions were also reported singly or jointly in 53% of BVDV-positive aborted fetuses analyzed in a retrospective study in Argentina. 17 Although congestive heart failure is infrequent in animal fetuses, signs of abnormal cardiac function, such as chronic passive congestion and generalized edema, have been reported elsewhere in fetuses aborted as a result of BVDV infection. 17
Although cardiac and skeletal myositis are commonly seen in fetal N. caninum infections, the brain is the organ that is affected most consistently.5,8 In a study of bovine abortions caused by N. caninum in Santa Catarina state, brain lesions were found in 71.4% of the fetuses analyzed. 5 However, in our study, we did not find typical lesions of necrotizing encephalitis in fetuses with either positive or negative PCR assays for N. caninum. Coinfections by BVDV and other agents potentially causing reproductive failures are reported, favored by the immunosuppression caused by BVDV, which can promote the reactivation of latent infections, 2 including by N. caninum. 8 Therefore, the joint action of both agents in the development of the lesions observed in our cases cannot be ruled out.
Histologic lesions in the lungs, meninges, kidneys, and liver are frequently observed in BVDV persistently infected (PI) animals and aborted fetuses infected with BVDV, and are likely related to viral replication.11,17 Virus-associated placentitis has been reported in bovine BVDV abortions, and appears to be related to viral tropism and replication in placental tissue. 11 The multisystemic distribution of BVDV has been demonstrated by immunohistochemistry in placenta, liver, heart, CNS, and in epithelial cells in the lungs and kidneys. 3 The severity of the lesions in fetus 4 might be related to infection by a more pathogenic strain of BVDV, given that some BVDV-2 strains can have different degrees of pathogenicity. 9
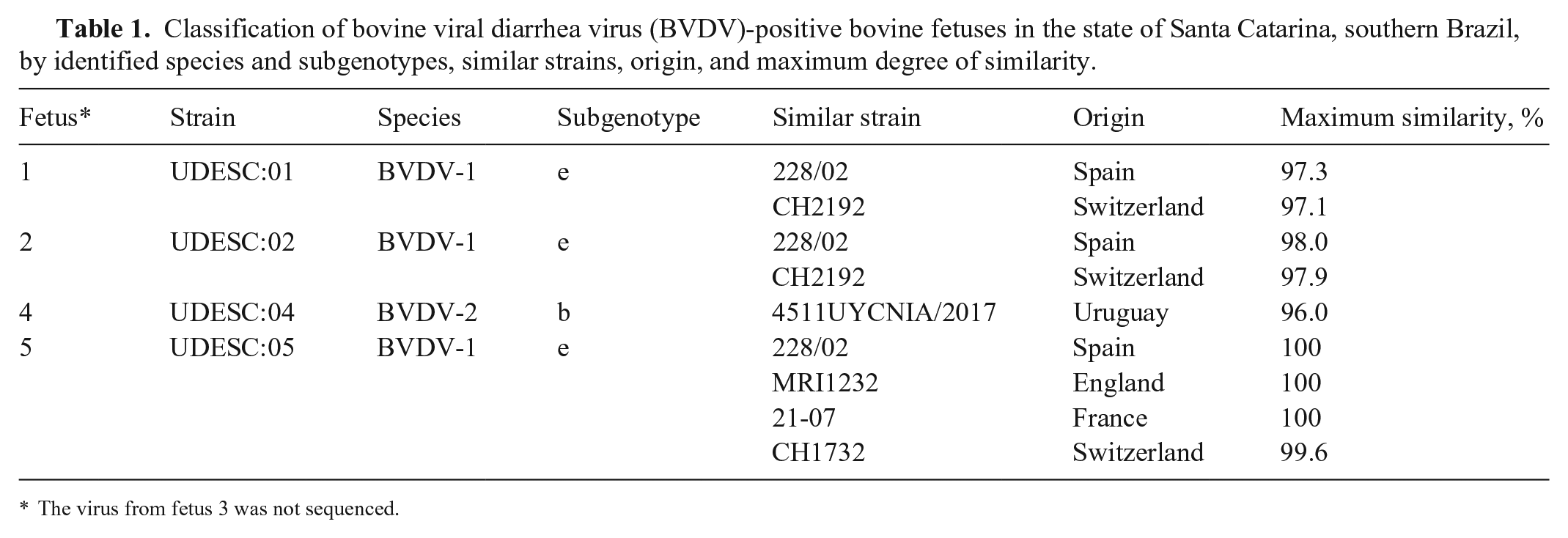
Subgenotype 1e, which we found in 3 of 4 aborted fetuses, has a restricted worldwide distribution, with a high concentration of reports in European countries; 1e has been detected only in Europe and South America to date. 22 In Brazil, this subgenotype is sporadic, and all reports of BVDV-1e have occurred only in Santa Catarina state, with 2 strains identified in 2012 and 2018. 10 It is unclear why Santa Catarina state is one of the few places besides Europe in which subgenotype 1e is present. All of our BVDV-1e strains had high similarity with European strains, especially from Switzerland (97.1–99.6%; Table 1). Surveys in Brazil showed a strong correlation between BVDV-1e and Simmental cattle, 18 a common breed in Santa Catarina. The raising of pure European breeds, such as Simmental, is facilitated in southern Brazil by favorable climatic conditions, which have allowed the successful importation of cattle from the Northern Hemisphere, and which may have enabled the localized introduction of this subgenotype into Brazil. 10
Classification of bovine viral diarrhea virus (BVDV)-positive bovine fetuses in the state of Santa Catarina, southern Brazil, by identified species and subgenotypes, similar strains, origin, and maximum degree of similarity.
The virus from fetus 3 was not sequenced.
Strains UDESC:01:1e (fetus 1), UDESC:02:1e (fetus 2), and UDESC:05:1e Brazil (fetus 5) were 96.9–97.4% similar in BLAST analysis. Fetuses 1 and 2 were from the same region of the state. The geographic proximity between dairy farms may have facilitated the trade of animals and transmission of the virus among properties, mainly through the acquisition of pregnant replacement heifers. 14 Strain UDESC:05:1e was detected in a region distant from the other BVDV-1e strains in our study. The high similarity between strains from different regions shows the circulation of this subgenotype among different herds in the state.
Subgenotype 2b has been detected frequently in Brazil compared to other subgenotypes, especially in the southern region.20,22 The UDESC:04:2b strain (fetus 4) was 96% similar to the 4511UYCNIA/2017 (MN186039.1) strain originally reported in Uruguay. The occurrence of BVDV-2b in fetus 4 is likely related to the proximity to the neighboring state of Rio Grande do Sul where the detection of this subgenotype is frequent. 20
Dairy herds predominated in our report, likely because of the greater propensity to high seroprevalence, reproductive disorders, and the emergence of PI animals, as well as the risk of virus introduction by frequent external visitors, such as deadstock collectors and muck spreaders. 4 Herd replacement through the acquisition of females potentially pregnant with PI progeny represented a risk of introducing BVDV to naïve herds. 14
Only inactivated vaccines were used in the herds in our report, and these vaccines tend to induce low-to-medium immunity against various viral strains. Subtypes 1a and 2a included in these vaccines did not prevent infection of the aborted fetuses by subtypes 1e and 2b. This inability to promote cross-immunity between virus variants is related to genetic and antigenic differences, allowing the occurrence of abortions and persistent infections by BVDV subtypes that differ from those used in vaccines. 12
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Fundação de Amparo à Pesquisa e Inovação do Estado de Santa Catarina, Brazil (FAPESC, 05/2018 - Nº: 2019TR702) and by the (PROAP/CAPES, Finance code 001), Brazil.
