Abstract
While Bovine herpesvirus 1 (BoHV-1) has been known as a cause of bovine abortion for nearly 50 years, information is limited on the current prevalence of BoHV-1 abortion in the United Kingdom, or about the mode of virus dissemination to cause infection of the fetus. The present study aimed to investigate these issues by surveying the prevalence of BoHV-1 in abortion cases in the United Kingdom, and comparing diagnostic methods to determine which are most efficient in BoHV-1–induced abortion. Where BoHV-1 DNA was detected, viral load was compared in fetal tissues, using real-time polymerase chain reaction (PCR), supported by histopathology and immunohistochemistry (IHC) to investigate virus dissemination in bovine abortions. A total of 400 U.K. bovine abortion cases were studied; PCR detected BoHV-1 nucleic acids in 10 cases, suggestive histopathological lesions were observed in 8, and positive IHC staining was observed in 9. In routine diagnosis, BoHV-1 was identified in 2 of these cases, highlighting the utility of using molecular diagnostic tests such as real-time PCR to achieve high sensitivity in potentially autolyzed tissues. The study of different fetal samples showed the highest viral load in the liver, along with severe multifocal necrotic hepatitis, suggesting either a clear tropism of the virus for this organ or that it is the first location to be reached in the fetus. Presence of viral antigen in endothelial cells of the placenta, brain, or heart suggest a hematogenous spread of virus from placenta to the liver, through the umbilical vein, and then to the rest of the organs via fetal blood vessels.
Keywords
Introduction
Bovine herpesvirus 1 (BoHV-1; order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus) is most commonly known as the causative agent of infectious bovine rhinotracheitis (IBR), a disease of the upper respiratory tract in cattle. However, if initial exposure to the virus, reactivation of the virus from latency, or vaccination using live virus occurs during pregnancy, it can cause abortion.18,23,29 Bovine herpesvirus 1–associated abortion in natural conditions usually occurs in the third trimester of pregnancy, 31 although an incubation period of 15–64 days has been observed in experimental infections, regardless of the gestation stage.8,29 Fetal death occurs 24–48 hr later, but it can be up to 7 days until the fetus is expelled. 10 Virus isolation (VI), histopathology, fluorescent antibody tests, and polymerase chain reaction (PCR) have been employed; fetal and/or maternal serology may also be considered.5,16 The success and suitability of these tests can be limited by the available samples and compromised by autolysis. As secondary bacterial infection is common in BoHV-1 abortion, bacterial causes may be misdiagnosed as the primary etiology. This is reflected in several surveys suggesting a higher rate of bacterial abortion in the United States. 2 The use of real-time PCR, which does not rely on the infectivity of the virus, could increase the diagnostic rate for BoHV-1. Furthermore, the closed system is less subject to risks of cross-contamination than conventional PCR.13,21 Further information can also be extracted using this method such as estimated viral copy number using absolute quantification methods. 20
The pathology related to BoHV-1–induced abortion has been well described.8,16,24 As the placenta does not always develop lesions, an early pathological study hypothesized that placental changes were secondary to fetal infection, 16 with a slow cell-to-cell virus spread between cotyledons. 8 However, based on the extensive multifocal necrotic lesions that are consistently observed in the liver, 24 hematogenous spread via the umbilical vein appears the likely route, with virus spreading from the placenta to the fetus. 29 This is supported by an experimental challenge study published in 2007 where viral antigen was associated with extensive lesions in fetal liver and also with blood vessels in the fetal placenta, suggesting a hematogenous spread from the mother to the fetus. 26 However, the 2007 study was based on an intravenous challenge, so should be interpreted with caution. To date, there has been no molecular support for this theory in natural cases of BoHV-1 abortion.
The current study aimed to detect evidence of BoHV-1 involvement in naturally occurring bovine abortion, in a case series comprised of 400 diagnostic submissions from Scotland and northern England. Samples were tested using both conventional VI and a real-time PCR assay.32,33 In addition, the transplacental spread and distribution of the virus through the fetus was studied by immunohistochemical staining of viral antigen and quantification of the viral load by real-time PCR in those cases where BoHV-1 was identified.
Materials and methods
Samples
Archived samples comprised of fetal tissues from 400 bovine abortions, received from veterinary laboratories across Scotland and northern England between 2007 and 2009, were tested. This was a retrospective study, analyzing cases that had been subjected to routine diagnostic testing, with the results of other tests and a diagnosis of the cause of abortion readily available (Table 1); however, the samples were tested blindly. The requested samples from these abortions were fresh and formalin-fixed fetal liver, lung, heart, brain, fetal fluid, and placenta. However, not all tissues were available from each case because the studied abortions occurred under natural conditions, where predation or degree of autolysis resulted in the failure to submit all tissues. Samples were taken using precautions to avoid PCR contamination and submitted promptly following fetal necropsy. On arrival, a sample from all submitted fresh tissues for each individual case were then pooled for real-time PCR testing as follows: 5-mm cubes of each tissue plus 250 µl of fetal fluid (if available) were pooled and homogenized in virus transport medium (1× 199 maintenance medium, a 0.5% bovine serum albumin, 2.95 g/l tryptose phosphate broth, 0.24 g/l sodium bicarbonate, 0.02 M N-2-hydroxyethylpiperazone-N-2-ethanesulfonic acid [HEPES], 0.01% yeast extract, 0.003 M sodium hydroxide, and distilled water with 2% fetal bovine serum, 1 mM glutamine, 100 U/ml penicillin–streptomycin, 2.5 µg/ml amphotericin, and 100 U/ml polymixin B sulfate added prior to use) b using automated homogenization. c Individual tissues and homogenate pools were stored at −80°C prior to testing. If BoHV-1 DNA was detected in the pooled samples, individual tissues from the positive case were then investigated by several diagnostic tests: VI, real-time PCR, histopathology, and immunohistochemistry (IHC).
Results of the different diagnostic tests for Bovine herpesvirus 1.*
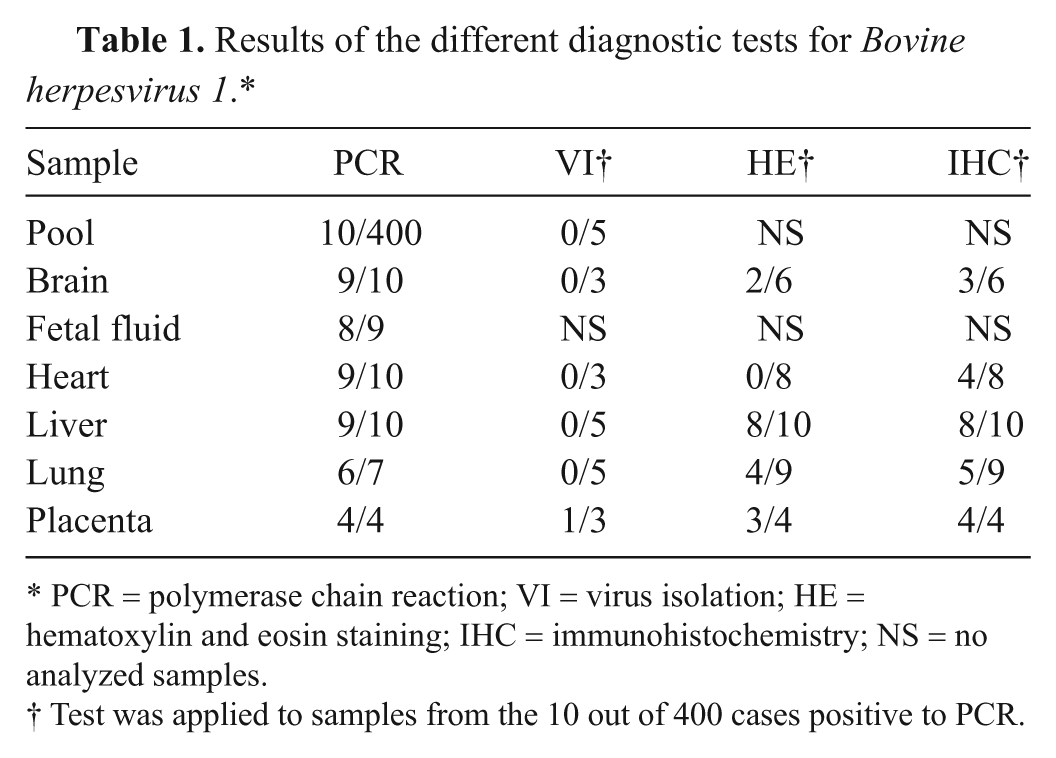
PCR = polymerase chain reaction; VI = virus isolation; HE = hematoxylin and eosin staining; IHC = immunohistochemistry; NS = no analyzed samples.
Test was applied to samples from the 10 out of 400 cases positive to PCR.
Virus isolation
For VI, individual tissues from cases in which BoHV-1 DNA was detected by real-time PCR were homogenized as above, filtered through 0.45-nm filters d and added to bovine turbinate (BT) cell monolayers growing in 12-ml TC-tubes, e following standard methods, 25 in Iscove modified Dulbecco medium a supplemented with 10% fetal bovine serum and 8 mM glutamine. Briefly, cells were washed twice in Hanks salt solution, and 0.2 ml of filtered inoculum was added. An uninfected control tube contained Hanks solution only. Virus inocula were adsorbed onto cell cultures by incubation at 37°C for 1 hr, prior to the addition of 199 maintenance me-dium a containing supplements as for virus transport medium above. Cultures were checked daily for any evidence of cytopathic effect.
Real-time polymerase chain reaction
DNA was extracted from unfiltered homogenized samples using an automated system f using the animal blood and cells protocol, according to the manufacturer’s instructions. An international ring trial–validated real-time PCR assay targeting a 96-bp fragment of the BoHV-1 glycoprotein B (gB) gene was used to detect BoHV-1 DNA.32,33 The assay was optimized for use with tissue-derived DNA samples. Reactions were carried out using a commercial master mix, g gB primers (500 nM) and probe (200 nM; gB-F: TGTGG ACCTAAACCTCACGGT; gB-R: GTAGTCGAGCA GACCCGTGTC; gB-probe: FAM-ACGACCGCGAGTTCT TGCCGC-TAMRA); β-actin primers (250 nM); and probe (250 nM) in a final volume of 25 μl (Actin_F: CACCTTC CAGCAGATGTGGA; Actin_R: CTAGAAGCATTTGCG GTGGAC; Actin_Probe: VIC-AGCAAGCAGGAGTACG-MGB). All reactions were run in triplicate on 96-well plates, h and each plate included a BoHV-1–positive control (IBR6660 reference strain), DNA extraction negative control, and a no-template control reaction. The reactions were run on an automated system h under the following conditions: 50°C for 2 min uracil-DNA glycosylase (UDG) incubation, 95°C for 2 min, followed by 40 cycles of 95°C for 15 sec and 60°C for 45 sec. All stages of template extraction, master mix preparation, template addition, and real-time PCR were carried out in separate laboratories using dedicated equipment to prevent contamination.
Results were analyzed using the supplied system software. h A baseline and threshold level was set according to manufacturer’s guidelines. Samples were considered positive when BoHV-1 DNA was detected in at least two-thirds of replicates with a threshold cycle (Ct) <35.
DNA was also extracted, as described above, from individual tissues of sample pools in which BoHV-1 DNA was detected. Extracted DNA was quantified and diluted so that all reactions contained 10–20 ng of DNA to normalize the amount of starting material between the different tissue types. Real-time PCR reactions were performed for BoHV-1 and β-actin, as described above, in 2 separate reactions. All samples were run in triplicate for both gB and β-actin with a DNA extraction negative control, no-template control, and BT cell DNA as a positive control for β-actin. Efficiency of the 2 primer/probe sets were evaluated and found to be comparable (gB = 104%; β-actin = 100%), allowing β-actin to act as a suitable reference for relative quantification analysis using the ΔΔCt method.12,28 This method uses the amount of β-actin (a normal cellular gene, present in the same amount in each tissue tested) signal detected as a basis for comparison of the amount of BoHV-1 gene present. These normalized values can then be compared to each other for different tissues, allowing assessment of the tissues that contain the highest virus load. Brain showed a consistent low virus load and was used as a calibrator sample (i.e., was assigned a value of 1) to estimate relative viral load as a proportionate increase in BoHV-1 gB copy number compared to the copy number detected in the brain. Data were statistically analyzed using a General Linear Model with post hoc Tukey test using the Minitab i statistical package, with P < 0.05 being considered statistically significant.
Histopathology and immunohistochemistry
Tissue samples from all cases where BoHV-1 DNA was detected by real-time PCR were analyzed using histopathological methods. Samples were fixed in 10% formalin for at least 24 hr and then embedded in paraffin wax blocks. Five-micron sections were cut using a microtome, placed on slides, and stained with hematoxylin and eosin (HE) by standard methods. Where the brain was available, 10 different sections (from internal capsule to medulla oblongata, including cerebellum) were studied. From the other organs, only 1 section was analyzed. Sections from the same paraffin blocks as above were cut and labeled using a commercial kit j with a commercial monoclonal mouse antibody targeting glycoprotein C of BoHV-1 k as previously described. 26
Results
Real-time polymerase chain reaction
BoHV-1 DNA was detected by real-time PCR in 10 (2.5%) of the 400 tissue pools from abortion cases (Tables 1, 2). The original diagnostic procedures had identified just 2 of these cases as BoHV-1 related, 4 were considered likely to have a bacterial cause, 1 was due to a noninfectious cause, and no diagnosis was reached for the remaining 3 cases (Table 3). In one case, clinical signs of IBR had been observed in the dam 1 month before the sample was submitted. Another case had received an IBR vaccination.
Results for each test for the Bovine herpesvirus 1 polymerase chain reaction–positive abortion cases (identified as A–J).*
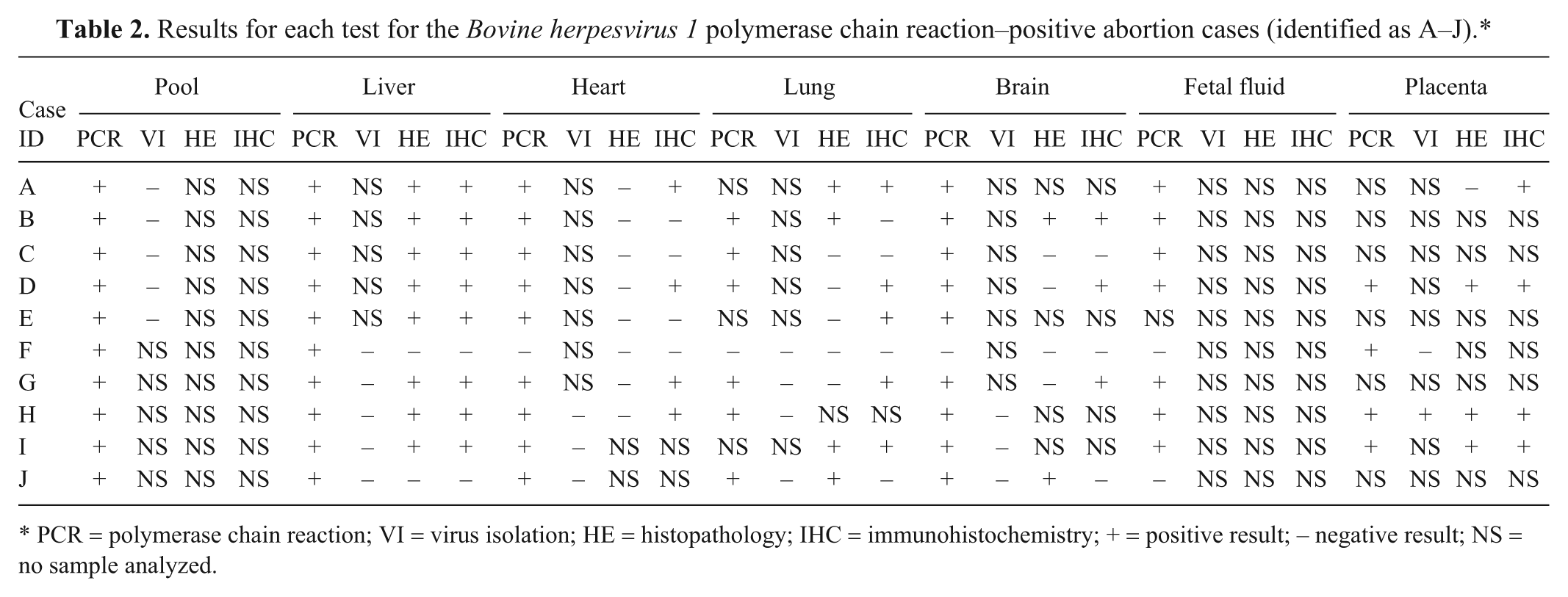
PCR = polymerase chain reaction; VI = virus isolation; HE = histopathology; IHC = immunohistochemistry; + = positive result; − negative result; NS = no sample analyzed.
Summary of previous testing and diagnosis for 10 Bovine herpesvirus 1 (BoHV-1)-positive cases by BoHV-1 real-time polymerase chain reaction.*
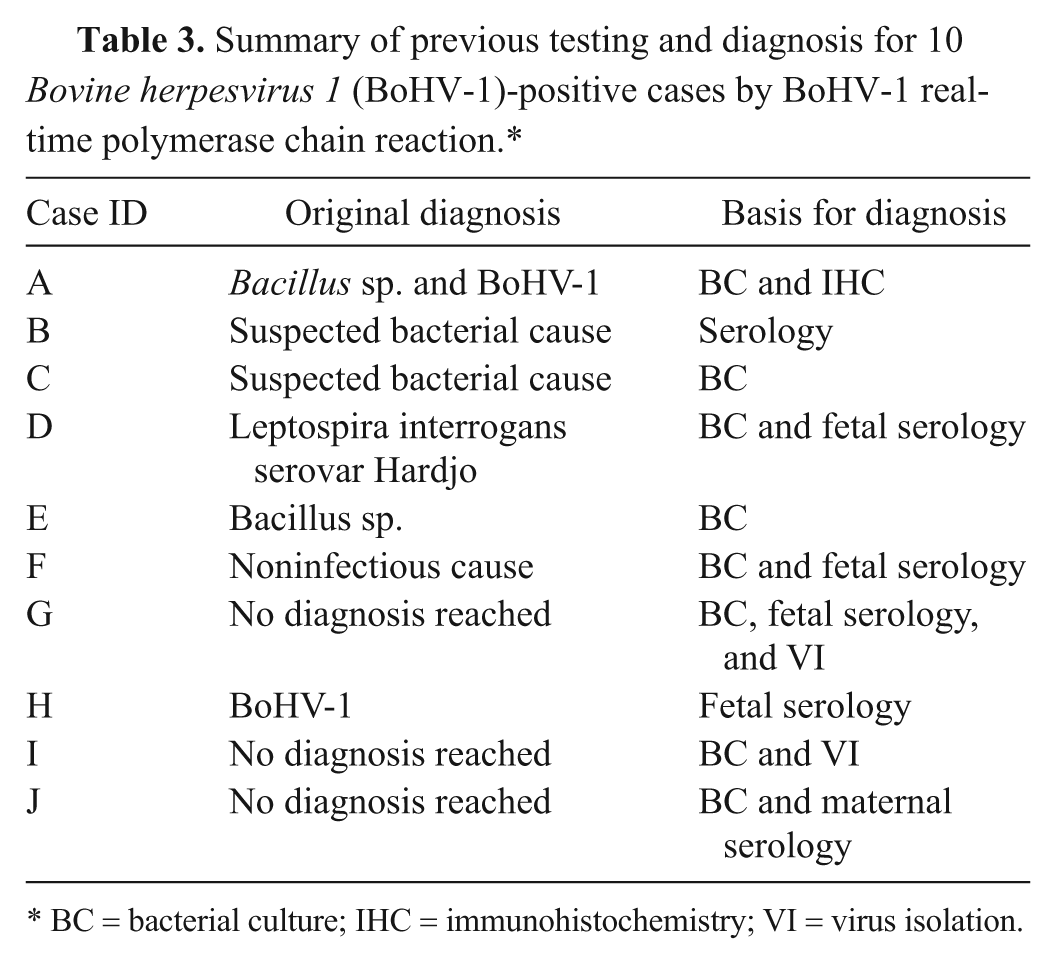
BC = bacterial culture; IHC = immunohistochemistry; VI = virus isolation.
Real-time PCR detected BoHV-1 DNA in all of the individual tissues in 9 out of 10 cases; in the other case (animal F) BoHV-1 DNA was detected only in the placenta. The relative quantification of the viral load between the individual tissues showed consistent differences in each of the cases with an average of 86,000-fold higher viral load in the liver than in the brain (Fig. 1), with a significantly higher viral load in the liver relative to all the other tissues (P < 0.01). No other tissues showed significantly different viral loads but there is some indication that the placental samples contained a higher viral load than the heart. The outliers showing high viral loads in the fetal fluid and heart were from the same animal.
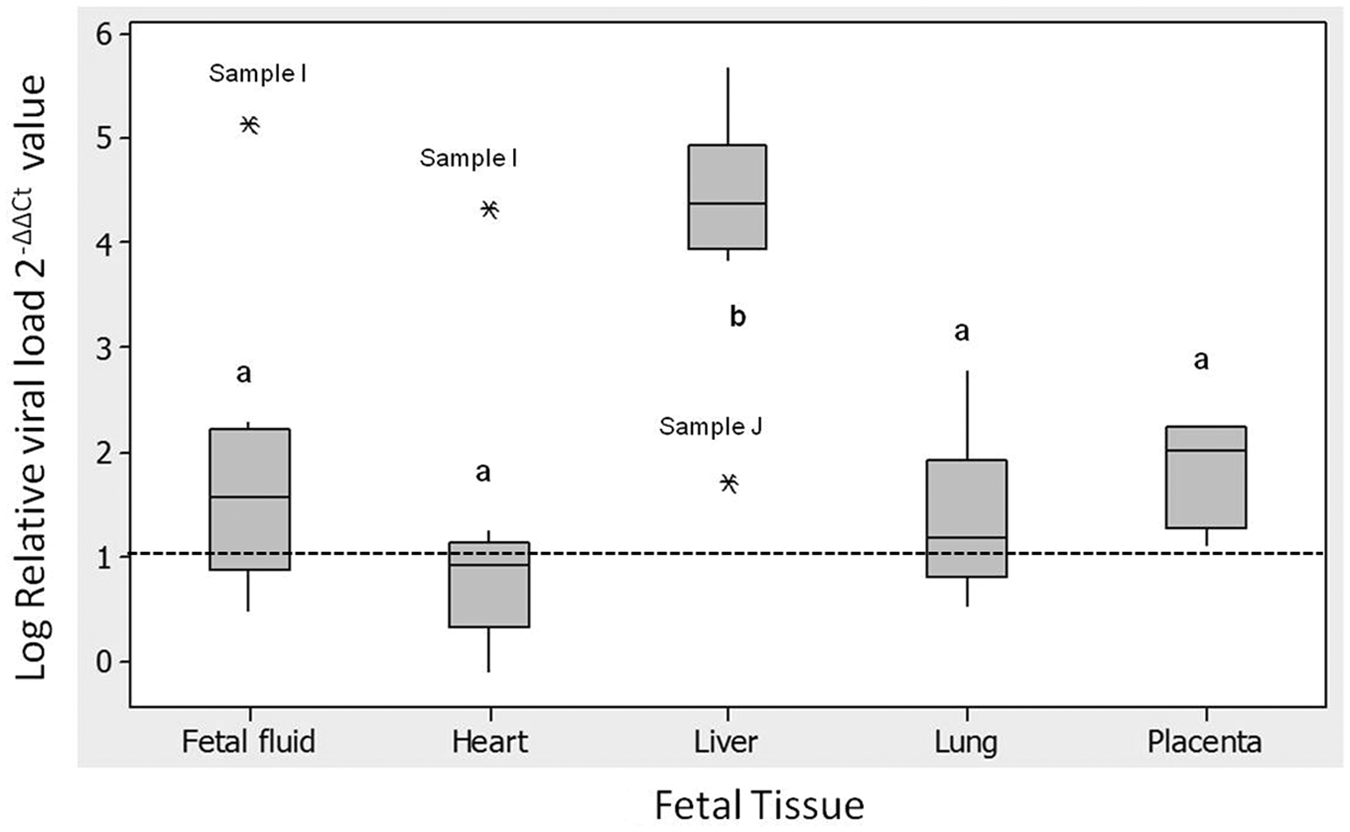
Box plot showing Bovine herpesvirus 1 (BoHV-1) viral load found in fetal tissues relative to viral load in fetal brain calculated by the ΔΔCt method from real-time polymerase chain reaction targeting BoHV-1 glycoprotein B (gB). 2-ΔΔCt represents the fold difference of BoHV-1 gB copy number between each tissue compared to copy number detected in the fetal brain, from the same case (fetal fluid, n = 8; heart, n = 9; liver, n = 9; lung, n = 7; placenta, n = 3). Asterisk (*) represents outlier samples; lowercase letters indicate statistical test grouping; dotted line represents gB BoHV-1 DNA in the calibrator sample, the brain.
Virus isolation and histopathology
Virus isolation was attempted on all cases in which BoHV-1 DNA had been detected. Cytopathic effect typical of BoHV-1 was observed only in 1 sample, the placenta of case H (Table 2).
Records from the original submission forms showed no gross lesions in any of the cases, and only advanced autolysis was reported in some cases. Most of the cases (6/10) examined by histopathology showed moderate to advanced autolysis, which impaired interpretation, especially of the placenta.
Histopathological lesions typical of BoHV-1 were observed in the liver and lung, and IHC demonstrated localization of viral antigens both within lesions and at sites where no histopathological lesions were evident, such as brain or placenta (Table 2, Fig. 2). The liver showed extensive multifocal necrotic hepatitis (Fig. 2A) in 8 out of the 10 samples studied. In this organ, necrotic foci showed an apparent random distribution within the parenchyma. Inflammatory infiltration in these lesions was scarce and, when present, it was formed by lymphocytes and plasma cells located at the periphery of the necrotic focus. Those liver samples without lesions were from cases F and J, where case F liver was also negative by PCR and case J demonstrated a comparatively low viral load by PCR. All placental samples showed an advanced degree of autolysis, which hindered the interpretation whether desquamation of the epithelial cells in the surface of the microvilli was due to the autolysis or to viral infection. There were also some vessels in which the lumen was filled by desquamated endothelial cells. No evidence of necrosis or inflammation could be discerned. Lung samples were available for 9 animals, 4 of these showed a small number of scattered foci of necrosis of the alveolar wall with no inflammatory infiltration. Brain was available for only 6 animals; 2 showed mild focal nonpurulent encephalitis characterized by scarce glial foci in the white matter. No lesions were visible in any of the heart samples. In some cases, the advanced degree of autolysis compromised the interpretation of histological findings.
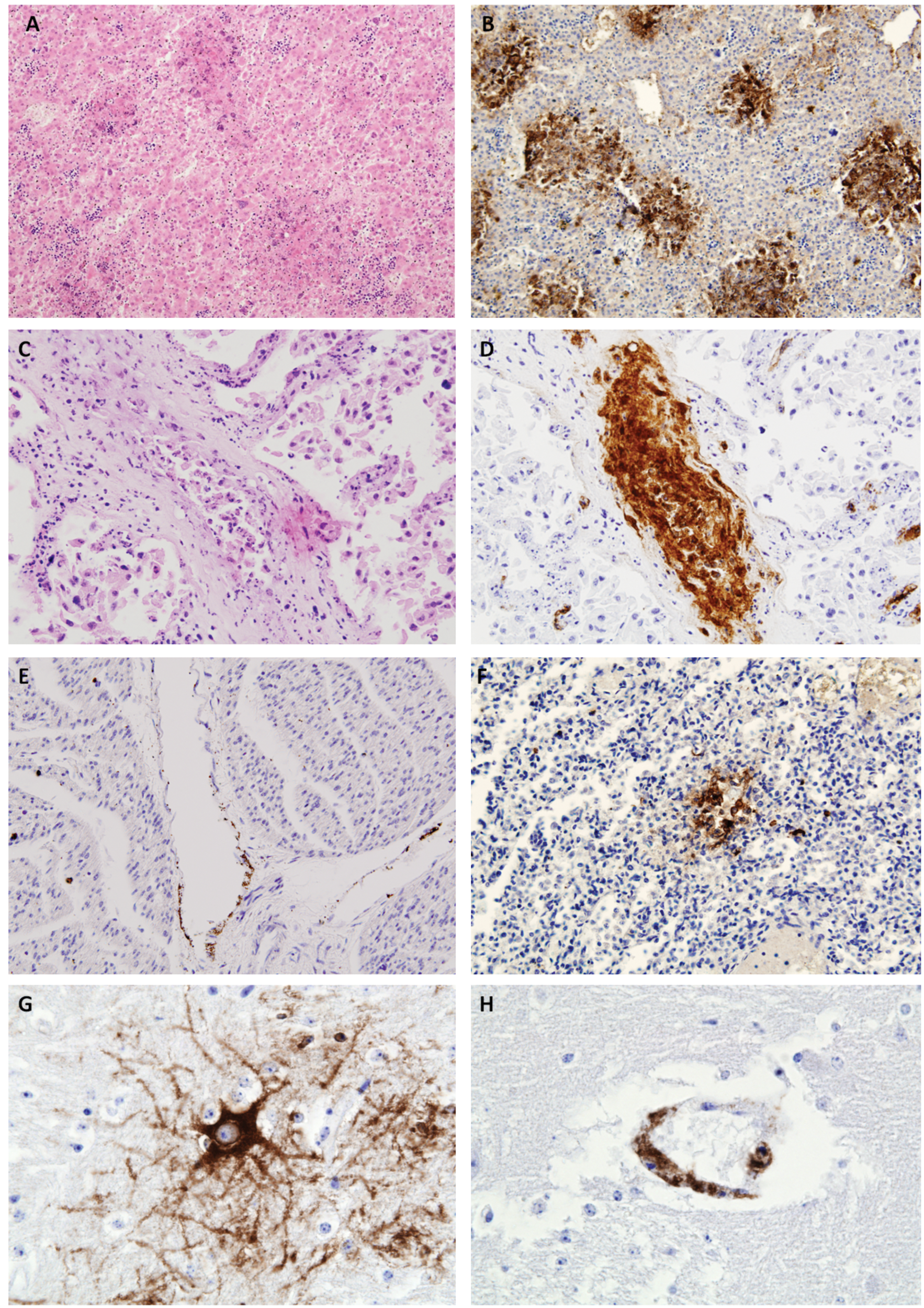
Hematoxylin and eosin (HE)-stained fetal tissue showing lesions typical of a Bovine herpesvirus 1 (BoHV-1) infection.
Immunohistochemistry on the fetal liver showed consistent positive detection of BoHV-1 antigen in 8 of the 10 studied cases, in accord with the histological lesions. In all cases, the positive staining was localized to the sites of the necrotic lesions (Fig. 2B), where the brown signal covered the entire focus. In the placenta (Fig. 2C), positive staining for BoHV-1 antigen by IHC was mainly observed in the microvilli, where signal was localized in the endothelium of the vessels in 3 out of the 4 samples studied. Some of the positive vessels showed extensive desquamation of the endothelial cells into the lumen (Fig. 2D). Despite lesions not being detected in the heart by HE, specific staining for BoHV-1 antigen was localized in 4 out of the 8 cases analyzed by IHC. As in the placenta, the positive signal appeared in the endothelial cells of isolated vessels (Fig. 2E). In the lung, the 4 cases that showed histological lesions were also positive by IHC. In these cases, the necrotic foci showed a strong positive signal, similar to that found in the hepatic lesions, but IHC staining was also found in the alveolar lining with no associated histological lesions (Fig. 2F). In the brain, 3 positive cases were detected, both cases in which lesions had been detected and 1 further case with no histological lesions. The distribution of BoHV-1 antigen varied between these cases, staining individual neurons in 2 cases (Fig. 2G) and the endothelium in the third (Fig. 2H).
Discussion
The prevalence of BoHV-1 in the current study was found to be 2.5%. Bovine herpesvirus 1 is generally not considered an important bovine abortifacient agent in the United Kingdom, although it has been regularly reported since it was first described in the 1960s. 23 Between 1984 and 1986, BoHV-1 was recorded (using fetal pathology and/or maternal serology) in 19 out of 149 (13%) abortion cases in the northwest of England 17 and a more recent Scottish survey (2004–2008) recorded less than 2% caused by BoHV-1. 27 In the United States, a meta-analysis study of 7 surveys conducted between 1950 and 2003 covering 3 geographical regions demonstrated varying levels of BoHV-1 diagnosis between 0% and 45%, depending on the time period and region. 2 A 10-year study in the United States that sampled more than 8,000 cases found a BoHV-1 prevalence of 5.41%. 10
When investigating natural abortion cases, use of the most sensitive and reliable methods is important for rapid and accurate diagnosis. Comparison of the results obtained by real-time PCR with previous diagnosis (Table 3), histopathology, IHC, and VI show the advantage of using rapid and highly sensitive molecular diagnostic techniques to support a diagnosis of BoHV-1, particularly in highly autolyzed tissue. Virus isolation was successful in only 1 of the 10 cases in which real-time PCR demonstrated the presence of BoHV-1 DNA, similar to other studies (Caldow GL, Buxton D, Spence JA, et al.: 1996, Diagnoses of bovine abortion in Scotland. In: Proceedings of British Cattle Veterinary Association, pp. 191–194. Edinburgh, UK); it has been estimated that BoHV-1 is detected by VI in only 33–50% cases where the diagnosis is confirmed by histology and fluorescent antibody test. 10 While detection of viral DNA in tissues from aborted fetuses does not necessarily mean that the virus is the causative agent, the ability to quantify the viral load can give an indication of the relevance of the finding.
Under the conditions of the current study, only PCR-positive cases were further evaluated by other techniques, and histopathology was also found to be reliable, although less sensitive than real-time PCR, for the diagnosis of BoHV-1 abortion cases. Liver lesions were evident in 8 out of 10 cases where BoHV-1 DNA was detected in fetal tissue pools, and while autolysis of some tissues compromised their histological examination, liver usually provided sufficient information for diagnosis without specific labeling by IHC. Immunohistochemistry added to the findings of HE staining, providing specific labeling for BoHV-1, and demonstrated good correlation with real-time PCR results. The limits of sensitivity for IHC are demonstrated in case J, where PCR demonstrated only a low viral load, presumably below the detection threshold of IHC. Histological examination and IHC labeling together showed a similar sensitivity as PCR in the 10 cases; only in animal F did both histological techniques fail to detect BoHV-1, although it should be noted that the presence of the virus DNA by PCR in this case was identified only in the placenta (which was not examined histopathologically), suggesting either acute abortion due to placentitis, limited spread of the virus within the fetus, or the co-involvement of causes other than BoHV-1 infection.
Mechanisms involved in systemic spread from the upper respiratory tract to the uterus and/or other organs in the case of systemic disease in calves 6 are also only poorly understood. 18 While a cell-associated viremia has been clearly demonstrated in closely related herpesviruses such as Equid herpesvirus 1 1 (EHV-1) and Suid herpesvirus 1 (Pseudorabies virus), 19 attempts to demonstrate the same process in BoHV-1 infection have been inconclusive. A single study has demonstrated a cell-free viremia 4–9 days post infection with 2 highly virulent strains. 7 Some evidence of cell association with lymphocytes and monocytes in vitro has been reported, 22 but the detection of BoHV-1 in blood buffy coats has predominantly been reported in studies using an intravenous challenge.14,15 A single study has demonstrated BoHV-1 in buffy coat following an intranasal 4 route of infection, though immunosuppression due to Bovine viral diarrhea virus (BVDV) may have influenced the BoHV-1 infection as BVDV had been inoculated intravenously prior to the BoHV-1 challenge. 4
The samples collected in the current survey also provided an opportunity to investigate the spread of the virus in natural cases of bovine abortion. Quantitative real-time PCR and histological techniques were successfully combined to investigate the distribution of the virus in various fetal tissues and allow the consideration of potential mechanisms of viral spread within the fetus. The highest viral load was detected in the fetal liver, consistent with the severity of the multi-focal necrotic lesions found by histopathology and by IHC. This finding is in accordance with several published descriptions of the fetal lesions associated with BoHV-1 abortion,2,9,10,16,26 and provides support for BoHV-1 as the causative agent of abortion in the cases identified herein. While one description of BoHV-1 abortion found more lesions in the lung and chorioallantoic membrane than in the liver, 17 care must be taken in interpreting these results, as they were based solely on histopathology without confirmatory etiological testing.
Dissemination to the fetal lungs, heart, and brain was demonstrated in 9 out of 10 cases as has also been previously reported in both experimental 26 and natural case studies. 17 The single case where BoHV-1 DNA was only detected in the placenta supports the hypothesis that placental damage may precede spread to the fetus,24,26,29 and occasionally that viral insult is enough to cause abortion, in contrast to the hypothesis that placental lesions are a result of spread from fetal infection. 16 The presence of BoHV-1 within the endothelium of the blood vessels in the placenta has been previously observed17,26 and, in an experimental challenge, virus was detected by the fluorescent antibody test despite the absence of placental lesions. 8 Abortion occurring in absence of a fetal infection has also been noted in cases of EHV-1 equine abortion, suggested to be due to failure of the vascular supply caused by EHV-1 infection.11,30
The close relation between BoHV-1 and blood vessels is shown in the current study by the immunostaining of the virus in vessels of several organs. These findings, together with the high viral load in the liver, strongly support a hematogenous spread of the virus from the placenta to the fetus via the umbilical vein, as has been previously suggested. 29 In a previous challenge study, the highest number of lesions observed in the placenta were in the chorioallantoic membrane and the placentome 17 ; further quantitative real-time PCR and IHC studies could help to elucidate the placental tissues most important in BoHV-1 abortion.
Interestingly, BoHV-1 has been associated with fetal encephalitis. 3 In the current study, consistent, though relatively low, levels of viral DNA were found in the brain accompanied by mild lesions and positive IHC labeling of both vessels and neurons, demonstrating that virus not only reached the brain via a hematogenous route but also spread and replicate within neural tissue.
The original diagnoses (Table 3) included only one that was considered to be caused by BoHV-1 based on histopathology and IHC. Five were originally considered to be due to bacterial causes, presumably as bacterial contamination or secondary infection was detected, suggesting BoHV-1 is often overlooked as a possible cause of abortion. It is therefore important to consider testing for evidence of BoHV-1 even where bacterial pathogens have been cultured. These results also emphasize the importance of correlating all the information obtained in abortion cases, as histological examination often indicates a presumptive diagnosis, which could be then confirmed by etiologic diagnostic test such as PCR or IHC.
The present study also highlights the importance of considering the disease status of the farm. At least 1 of the cases came from a vaccinated herd; most vaccines prevent clinical signs after wild-type challenge but cannot prevent latent infection and subsequent reactivation. 18 Only 1 case was observed in relation to a recent outbreak of IBR. This suggests that either clinical signs of primary IBR are rarely noticed in the dam or that reactivation during pregnancy may be associated with minimal clinical signs but can still allow BoHV-1 to spread to the fetus. No serological testing of the dams for evidence of exposure to IBR was carried out; however, BoHV-1 seroprevalence has not been found to be predictive of abortion in herds. 5
In conclusion, BoHV-1 has an estimated prevalence of 2.5% in bovine abortions in the United Kingdom, but will be underdiagnosed unless specifically sought. Both real-time PCR analysis and IHC provide a reliable test for the presence of BoHV-1 in bovine abortion cases even in autolyzed tissue; molecular diagnostic techniques have the advantage of high sample throughput and can generate rapid results. Further, quantitative PCR studies demonstrate that the liver is a key organ for both the etiological diagnosis of abortion and for the study of pathogenesis of the disease, supporting previous hypotheses of transplacental spread and fetal dissemination of BoHV-1. However, the mechanisms involved in the spread of the virus from the respiratory tract, or the site of latency, of the infected dam to the developing fetus in the uterus remain unclear.
Footnotes
Acknowledgements
Thanks to Linda Marriot, Ellie Laming, and Dylan Turnbull for technical assistance in the Virus Surveillance Unit, and to Clare Underwood and Val Forbes in the Histopathology Unit of the Moredun Research Institute and BioSS for advice on statistical analysis. The authors are also grateful to many colleagues at Scottish Agricultural Colleges Disease Surveillance Centres for providing the samples and access to original diagnostic data.
a.
Sigma-Aldrich Company Ltd., Dorset, UK.
b.
E&O Laboratories Ltd., Bonnybridge, Scotland.
c.
gentleMACS M tubes on a Dispomix drive; Miltenyi Biotec Ltd., Bisley, Surrey, UK.
d.
Sartorius Ltd., Epsom, Surrey, UK.
e.
Greiner Bio-One GmbH, Kremsmunster, Austria.
f.
DNeasy kit running on the QIAcube, Qiagen Ltd., West Sussex, UK.
g.
Platinum qPCR supermix-UDG mixed with 0.1 μl ROX reference dye, Life Technologies Ltd., Paisley, UK.
h.
ABI Prism7500, Life Technologies Ltd., Paisley, UK.
i.
Minitab Inc., State College PA.
j.
Envision HRP system, Dako UK Ltd., Ely, Cambridgeshire, UK.
k.
VMRD Inc., Pullman, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Moredun Scientific Limited. The Virus Surveillance Unit and Pathology Surveillance Unit at the Moredun Research Institute are funded by the Scottish Government.
