Abstract
The quantification of circulating plasma immunoglobulins represents a valuable diagnostic tool in human and veterinary immunology, although its application is very limited in reptile medicine to date. The objectives of our study were the development and standardization of a competitive enzyme-linked immunosorbent assay (cELISA) for the measurement of total plasma immunoglobulins (Igs; both IgM and IgY) in loggerhead sea turtles (LST; Caretta caretta; n = 254) and green turtles (GT; Chelonia mydas; n = 111), the establishment of reference intervals for Ig for both species, and the examination of associations between Ig and total protein (TP), condition index, and water temperature. The cELISA for Ig was successfully developed and optimized. Reference intervals for Ig were 0.38–0.94 g/dL in LST (median: 0.59 g/dL; range: 0.16–2.15 g/dL) and 0.40–0.85 g/dL in GT (median: 0.58 g/dL; range: 0.18–1.80 g/dL). In LST, there were positive linear relationships of Ig with TP, and TP with Ig and condition index, and a negative relationship of Ig with condition index. The positive linear relationships of Ig with TP, and TP with Ig were also identified in GT. These positive associations of Ig and TP were expected, as Ig represents fractions of TP, and TP reportedly increases with straight carapace length and weight. The negative association of Ig with condition index may indicate potential biological variations. The cELISA and reference intervals for total Ig of LST and GT presented herein have the potential to be useful as a diagnostic and research tool for sea turtle immunology.
Introduction
Sea turtle populations are in global decline from causes including subsistence hunting, bycatch and accidental entanglement in commercial fisheries, ingestion of foreign material, natural diseases, and loss of essential nesting habitat. 22 The International Union for the Conservation of Nature Marine Turtle Red List has assessed the following 6 of the 7 species of sea turtles as either: 1) critically endangered [hawksbill (Eretmochelys imbricata) and Kemp’s ridley (Lepidochelys kempii)]; 2) endangered [loggerhead sea turtle (Caretta caretta) and green turtle (Chelonia mydas)]; or 3) vulnerable [olive ridley (Lepidochelys olivacea) and leatherback (Dermochelys coriacea)] (http://iucn-mtsg.org/about/structure-role/red-list/). Data is deficient for assessing the status of the flatback sea turtle (Natator depressus). Federal Recovery Plans, which are required for each species listed under the U.S. Endangered Species Act, call for the development of integrated health assessment programs for these animals (http://www.nmfs.noaa.gov/pr/recovery/).
Comprehensive health assessment studies in wild animal populations frequently include collection of blood, which allows for analysis for hematology, biochemistry, blood gases, and immunology. The assessment of immune function in veterinary medicine can be performed through protein electrophoresis, lymphocyte proliferation assays, immunoglobulin quantification, and other advanced immunodiagnostics. 11 Lymphocyte proliferation in sea turtles has been described, 23 but is time- and cost-intense and not practical for a diagnostic setting. Analysis and evaluation of plasma electrophoretogram (ELP) fractions is frequently used in reptile medicine and has been proposed as a means of assessing and monitoring immune function. 24 Reference intervals for total protein (TP) and ELP fractions for loggerhead sea turtles have been documented and thus represent 1 useful diagnostic for sea turtle immunology.8,16 Quantification of immunoglobulins (Igs) can be achieved by radioimmunoassay (RIA) or enzyme-linked immunosorbent assay (ELISA). Studies on the performance of RIA, ELP, and ELISA in reptiles have reported the ELISA to be comparable to RIA and ELP in the ability to measure Igs, with the added benefit of convenience and practicality in clinical settings.10,20 However, these assays are currently not available for sea turtles because specific reagents are required for the development of competitive ELISA (cELISA) systems. 6 Available information on sea turtle Igs and their detection is limited, to the authors’ knowledge, to 2 publications in the current literature.3,9 The development of monoclonal antibodies (mAbs) for green turtle immunoglobulins M and Y (IgM and IgY, respectively) that cross-react with other sea turtle species 9 was a major milestone for identification of Ig in sea turtles.
The major Igs in chelonians described to date include IgY and IgM, which are presumed to be similar to mammalian IgG and IgM, respectively. 1 Genes encoding IgA, IgD, and IgD2 have been recognized in the American alligator, saltwater crocodile, and leopard gecko.7,13 Although structural homologues of IgA, IgD, and IgD2 have not been identified in chelonians, the presence of functional homologues of these molecules cannot be ruled out. Still, total Ig in the plasma of chelonians are presumably mainly composed of IgY and IgM (hereafter, total Ig). Two types of IgY heavy chains in sea turtles, either with full-length or truncated heavy chain, respectively, have been described. 9 Both IgY and IgM antibodies carry light chains that are detected by mAb HL673. 19 The quantification of Ig can provide valuable information for diagnosis, monitoring, prognosis, and/or treatment in individual sea turtles, as well as in epidemiologic studies in sea turtle populations. However, the use of mAbs for quantification of total Ig in sea turtle species has not been published in any format, including ELISA, to date.
Extrinsic and intrinsic factors can have significant effects on various hematologic and biochemical analytes in marine turtles, as documented for water temperature, season, and life stage on peripheral protein concentrations. 16 Because Igs occur in various protein fractions, similar effects on Igs can be anticipated, but have not been investigated in various reptile species to date. Knowledge of these effects on various analytes is essential for their accurate interpretation of the health of the individual animal or the population.
The main objective of our study was the development and standardization of a cELISA for the measurement of total plasma Ig in loggerhead sea (LST) and green (GT) turtles and the establishment of reference intervals for total Ig for both species. A secondary objective was the examination of associations between Ig and TP, condition index, and water temperature, as well as between TP and Ig, condition index, and water temperature.
Materials and methods
Sea turtles, condition index, and total plasma protein
In this study, plasma samples of 273 LST and 119 GT were selected from a repository of plasma samples, maintained in an ultra-low temperature freezer at −70°C, that were obtained from sea turtles entering the intake canal of the Port St. Lucie Power Plant, St. Lucie County, Florida between July 2004 and May 2006, as previously documented.10,16 Briefly, 20–23-gauge needles were used to collect up to 6 mL of blood from the dorsal cervical sinus into lithium heparin anticoagulant tubes at an appropriate volume. Data recorded for each turtle included the collection date, body weight (WT; kg), minimum straight carapace length (SCLmin; cm; notch-to-notch length measured from the mid-point of the nuchal scute to the notch where the 2 most posterior marginal scutes meet), and overall health status. All turtles in our study were considered clinically healthy by standard field physical examination. A condition index was calculated using the following formula: WT/SCL 3 × 104. 4 Blood was collected from each turtle, and TP (g/dL) was measured as previously described using the biuret method. 16 Daily water temperature was recorded for the power plant intake canal.
Antibody selection and purification
A mouse mAb specific for the light chain of the desert tortoise Ig, HL673, was produced from a high-density hybridoma supernatant and, subsequently, 25 mg were purified using a protein G sepharose column. a Cross-reactivity of HL673 with LST and GT plasma was assessed by Western blot performed according to an established protocol.9,19
Approximately 10 mg of purified HL673 was coupled to a protein G column b in accordance with manufacturer recommendations in order to affinity purify LST Ig from LST plasma. A 2-mL volume of LST plasma was then applied to the column. Following washing and elution, ~2 mg of LST Ig was obtained from the column eluate as previously described. 15 The purification of the LST Ig was evaluated using sodium dodecyl sulfate–polyacrylamide gel electrophoresis and was considered sufficiently pure (~90%) to be used in this study. A 4-mg aliquot of purified mAb HL673 was labeled with biotin to be used in the cELISA as previously described. 15
Assay development and optimization
Optimal competition reaction conditions for the cELISA were established for the LST Ig and biotinylated HL673 using an indirect ELISA, according to previously published methodologies.9,17 Several permutations of concentration ranges of purified LST Ig (0.1, 0.3, 0.5, and 1.0 μg/mL) and biotinylated HL673 (6.25–0.0031 μg/mL, serial 2-fold dilutions) were used, with optimal competition reaction conditions defined as the mAb concentration at 70% of maximum binding.6,20 The optimum purified LST Ig coating concentration of 0.5 μg/mL and optimum biotinylated HL673 concentration of 0.78 µg/mL were determined at those concentrations of antigen-antibody competing mixture.
Using previously reported methodology 9 and the optimal concentrations of purified LST Ig and competing biotinylated HL673 as established above, a cELISA was used to establish the standard curve (Fig. 1). Serial 2-fold dilutions of purified LST Ig (12.5–0.006 µg/mL) were allowed to compete in duplicate reactions with 0.78 µg/mL biotinylated HL673. The optical density at 405 nm (OD405) was measured at 1 hr, and values were plotted against the known Ig concentrations to establish the standard curve equation using commercial software. c This standard curve was included on each 96-well plate during Ig sample quantification as positive standard controls, as well as 1% bovine serum albumin as a negative control.
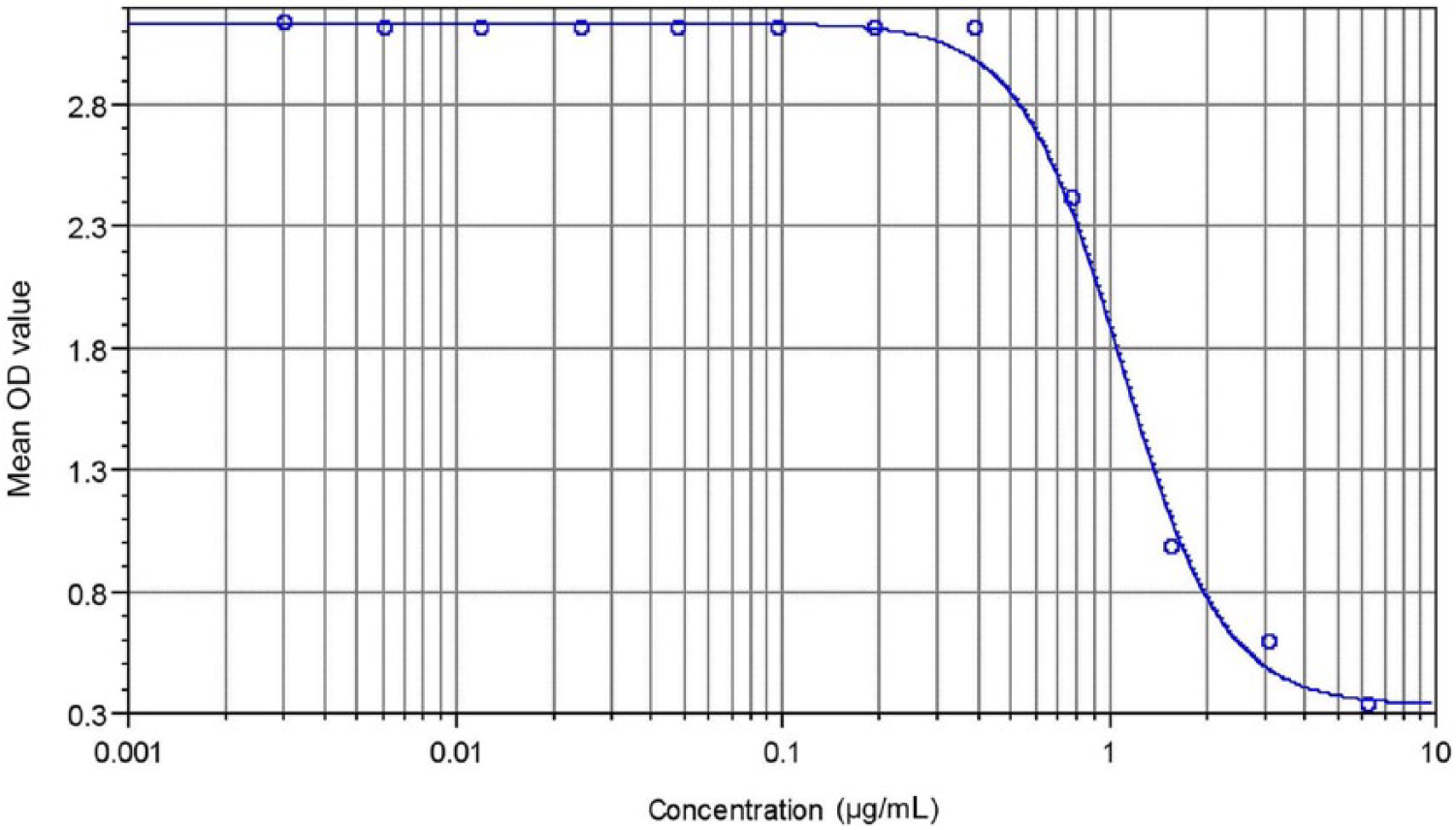
Standard curve for quantification of sea turtle total immunoglobulin in µg/mL (R2 = 0.933).
Optimal competing dilutions of the sea turtle plasma samples were determined using serial dilutions of a low TP sample of 1.6 g/dL and a high TP sample of 6.3 g/dL. An optimal plasma dilution range of 1:4,000–1:16,000 was selected based on the location of corresponding OD405 readings on the linear aspect of the slope of the standard curve. 6 Total Ig of all unknown sea turtle samples run in duplicate at these dilutions was determined against the standard curve control and reported in g/dL.
Three randomly selected plasma samples were run in triplicate within 1 plate and in triplicate within 4 consecutive plates to determine inter- and intraplate variability. The inter- and intraplate assay variability (%) was calculated as the mean of the variability (%) of each of the 3 samples between and within plates, respectively. The mean variability for each sample was calculated as the mean % deviation of each replicate from the average of each sample.
Data analysis
The null hypotheses that Ig concentrations (g/dL) were not different in LST or GT with TP (low, medium, high), condition index (low, medium, high), and water temperature (low, medium, high) were tested by using the nonparametric Kruskal–Wallis test. The 3 categories (low, medium, high) for each independent variable were based on their 33rd percentile distribution.
Similarly, the null hypotheses that TP concentrations (g/dL) were not different between LST and GT with Ig (low, medium, high), condition index (low, medium, high), and water temperature (low, medium, high) were tested by using the nonparametric Kruskal–Wallis test. Pairwise comparisons were conducted by using the Scheffé method.
Finally, because TP, condition index, and water temperature were associated with Ig, these 3 factors were examined using logistic regression analysis to address possible confounding effects that these factors may have had on Ig. The continuous (response) variable for Ig was dichotomized as low (0.16–0.59 g/dL) or high (0.60–2.15 g/dL), based on the median distribution. Similarly, the variables for Ig, condition index, and water temperature were examined using logistic regression analysis to address possible confounding effects that these factors may have had on TP. Logistic regression analysis (instead of linear regression) was used because the outcome variables for Ig and TP did not meet assumptions of normality (including when data was ranked or log 10, natural log, or squared root transformations were performed). In all analyses, values of p < 0.05 were considered significant.
Results
Sea turtles, plasma samples, SCLmin, weight, and condition index
Of the 273 LST and 119 GST plasma samples selected for testing, 254 LST (242 immature and 12 mature) and 111 GT (110 immature and 1 mature) had measurable Ig. The total Ig of 19 LST and 8 GT samples fell outside the measurable range of the Ig cELISA, and therefore these samples were excluded. Hemolysis, other discoloration (i.e., lipemia, biliverdinemia), or visible contamination with lymph during blood collection were absent by visual examination in any of the plasma samples.
The weight and SCLmin of each individual were used in the calculations of condition index. For this group, body size (SCLmin) of LST ranged from 50.3 to 96.8 cm, with a median of 65.4 cm. Body weight of LST ranged from 17.8 to 101.9 kg, with a median of 40.2 kg. Condition index of LST ranged from 0.97 to 1.96, with a median of 1.43.
The SCLmin of the GT ranged from 25.4 to 86.9 cm, with a median of 39.8 cm. Body weight of green turtles ranged from 1.9 to 89.2 kg with a median of 7.7 kg. Condition index of GT turtles ranged from 0.97 to 1.66, with a median of 1.26. Water temperature ranged from 16.7°C to 29.6°C. The morphometric parameters SCLmin, weight, and condition index are also summarized by month in Supplementary Tables S1 and S2 (available at http://vdi.sagepub.com/content/by/supplemental-data), which demonstrate no obvious variations for these parameters during the study period.
Reagent validation of the cELISA and inter- and intraplate variability
Cross-reactivity of HL673 with LST and GT plasma was confirmed by Western blot as demonstrated in Figure 2. The Ig detection range of this assay, as calculated by the standard equation of the standard curve, was 0.15–2.15 g/dL. Of the 273 LST and 119 GST samples selected for testing, 19 LST and 8 GT total Ig were outside the measurable range of this cELISA. Seven of the LST and 2 GT had total Ig above the measurable range of the standard curve, and 12 LST and 6 GT had total Ig below detection level. The calculated inter- and intraplate variability of the cELISA were 8% and 5%, respectively.
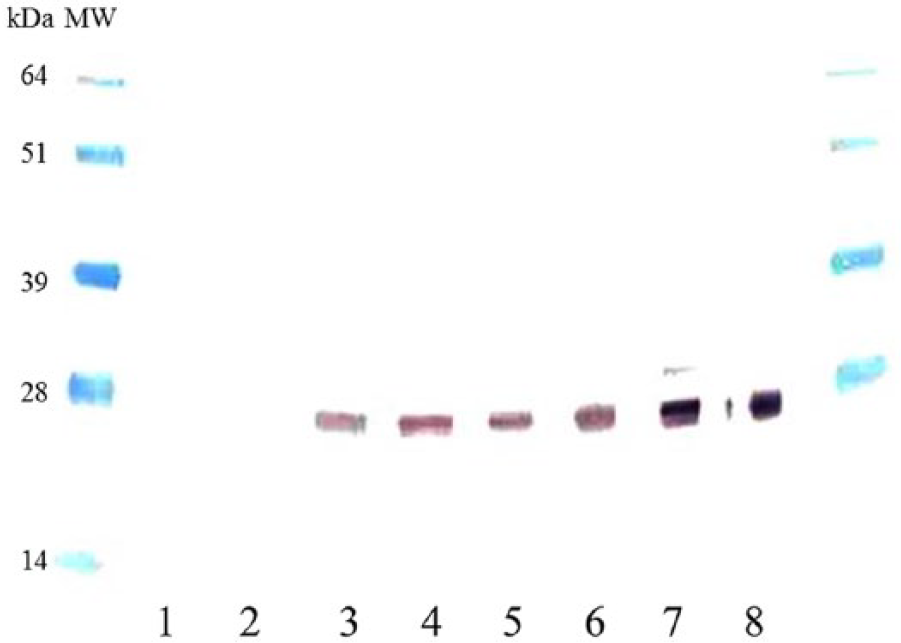
Western blot of 2 individual samples each of desert tortoise (DT), green turtle (GT), and loggerhead sea turtle (LST) plasma samples detected with monoclonal antibody HL673 as the primary antibody. Plasma samples were used at a 1:50 dilution, HL 673 was used at a concentration of ~5.0 μg/mL. Lanes 1 and 2: negative control with DT (phosphate buffered solution and goat anti-mouse immunoglobulin (Ig)G; lanes 3 and 4: LST with HL673 (1st antibody) and goat anti-mouse IgG conjugate; lanes 5 and 6: GT with HL673 (1st antibody) and goat anti-mouse IgG conjugate; lanes 7 and 8: DT with HL673 (1st antibody) and goat anti-mouse IgG conjugate. The goat anti-mouse IgG conjugate was used at a dilution of 1:4,000. The detected band is located at ~23 kDa.
Total plasma protein and immunoglobulin
The Ig and TP data was not normally distributed. Total plasma protein ranged from 1.8 to 5.2 g/dL, with a median of 3.2 g/dL for LST, and from 1.7 to 5.9 g/dL, with a median of 3.5 g/dL for GT. Reference ranges for total Ig were 0.38–0.94 g/dL in LST (median: 0.59 g/dL; range: 0.16–2.15 g/dL) and 0.40–0.85 g/dL in GT (median: 0.58 g/dL; range: 0.18–1.80 g/dL). Total Ig (g/dL), TP (g/dL), straight carapace length (cm), weight (kg), and condition index (median, first and third quartiles) per month for LST and GT are presented in Supplementary Tables S1 and S2, respectively.
Associations between Ig and TP, condition index, and water temperature in LST
In the univariable analysis, we observed a positive linear relationship between Ig and TP and water temperature, and a negative linear relationship between Ig and condition index in LST (p < 0.05; Table 1). Using logistic regression, the relationship between Ig with TP and condition index remained significant. The odds (likelihood) of LST testing with high total Ig (0.79–2.15 g/dL) were 3.9 higher in LST with high TP (3.5–5.2 d/dL), after controlling for condition index (p < 0.05). In addition, the odds of LST testing with low total Ig (0.16–0.44 g/dL) were 3.3 lower (odds ratio = 0.3) in LST with a high condition index (1.48–1.96), after controlling for TP (p < 0.05).
Associations between immunoglobulin and total protein, condition index, and water temperature in loggerhead sea turtles (Caretta caretta).
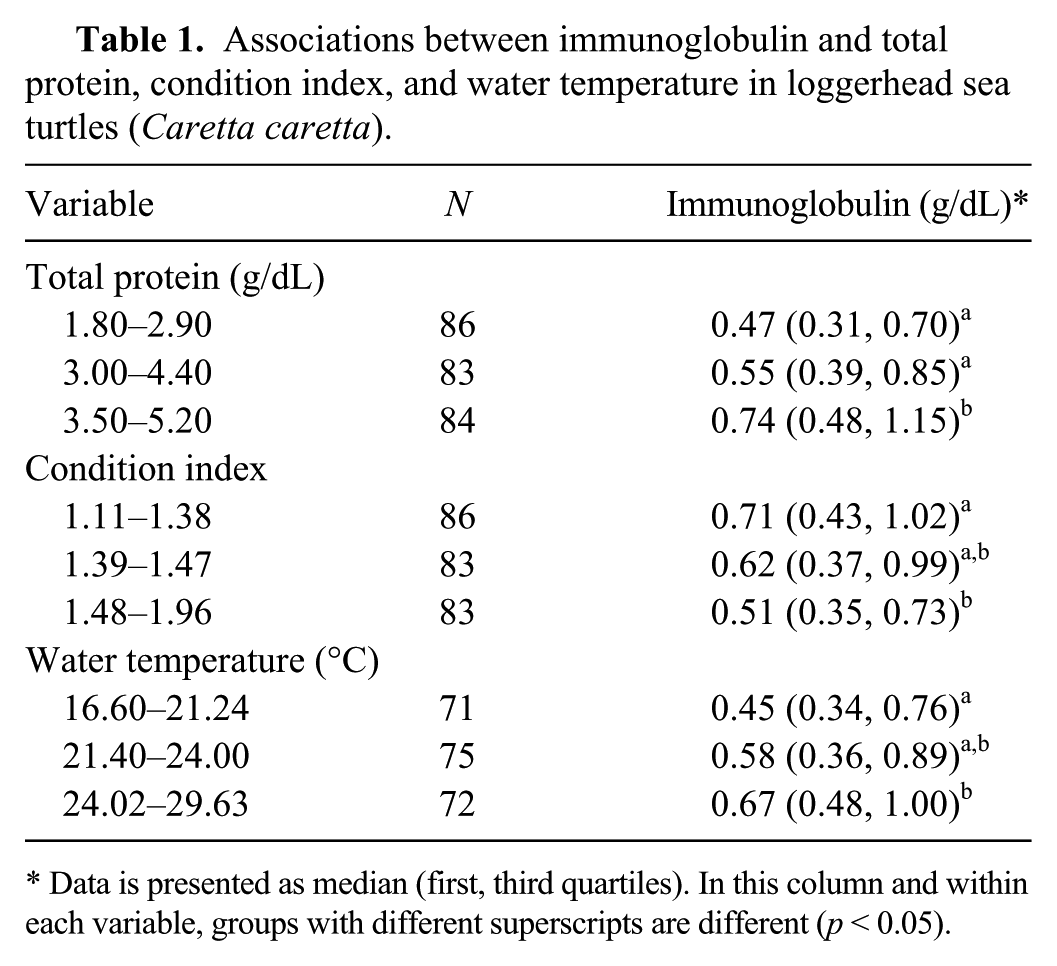
Data is presented as median (first, third quartiles). In this column and within each variable, groups with different superscripts are different (p < 0.05).
Associations between TP and Ig, condition index, and water temperature in LST
In the univariable analysis, we observed a positive linear relationship between TP and Ig, condition index, and water temperature in LST (p < 0.05; Table 2). Using logistic regression, the relationship between TP with Ig and condition index remained significant. The odds of LST testing with high total Ig (0.79–2.15 g/dL) were 3.9 higher in sea turtles with high TP (3.5–5.2 g/dL), after controlling for condition index (p < 0.05). In addition, the odds of LST testing with low total Ig (0.16–0.44 g/dL) were 3.3 lower (odds ratio = 0.3) in LST with a high condition index (1.48–1.96), after controlling for TP (p < 0.05).
Associations between total protein and immunoglobulin, condition index, and water temperature in loggerhead sea turtles (Caretta caretta).
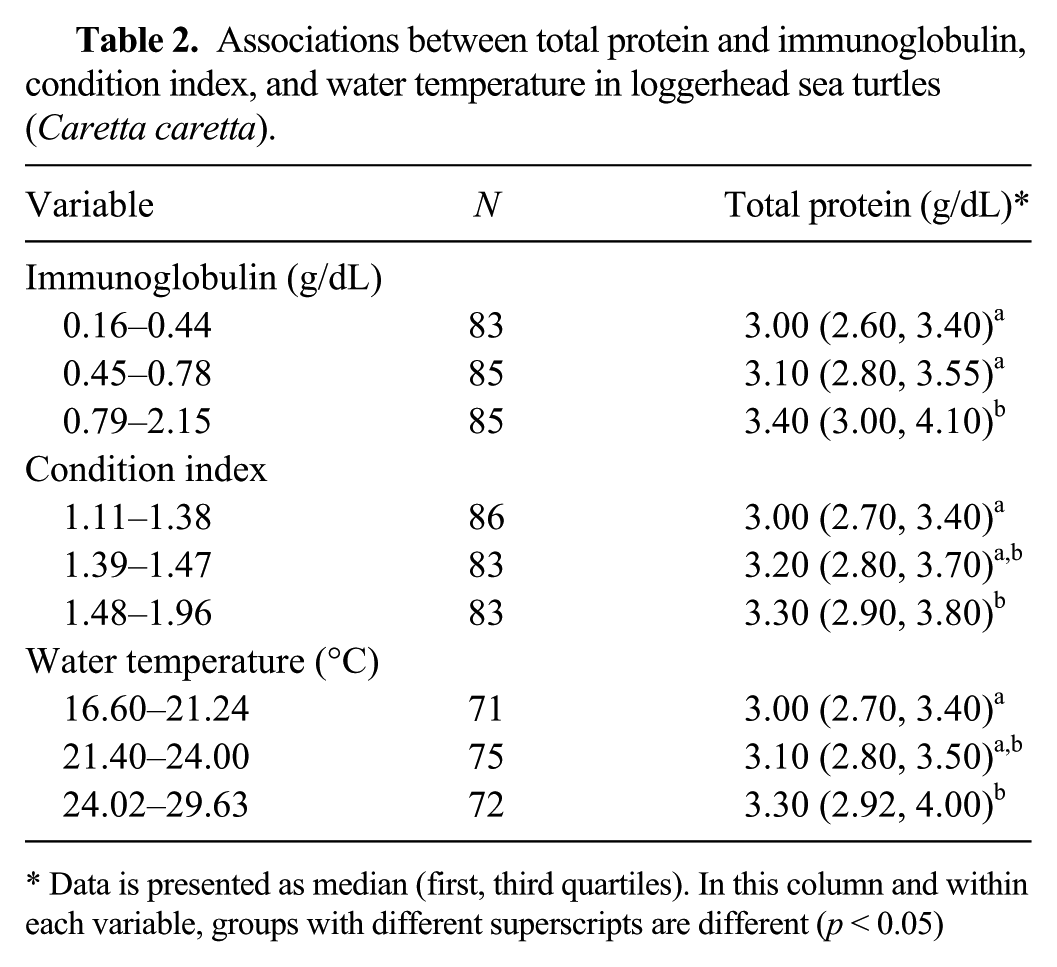
Data is presented as median (first, third quartiles). In this column and within each variable, groups with different superscripts are different (p < 0.05)
Associations between Ig and TP, condition index, and water temperature in GT
In the univariable analysis, we observed a linear relationship between Ig and TP in GT (p < 0.05; Table 3). We also observed a linear relationship between TP and Ig in GT (p < 0.05; Table 4).
Associations between immunoglobulin and total protein, condition index, and water temperature in green turtles (Chelonia mydas).
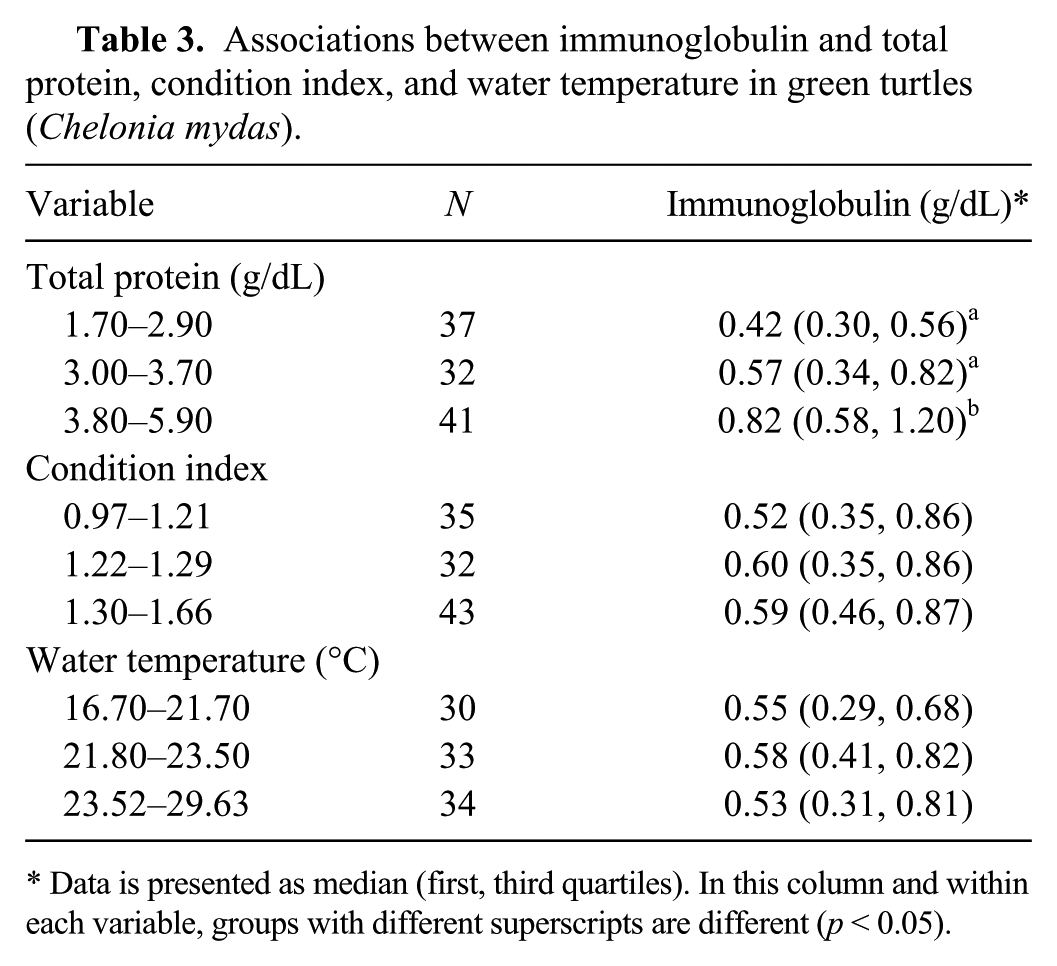
Data is presented as median (first, third quartiles). In this column and within each variable, groups with different superscripts are different (p < 0.05).
Associations between total protein and immunoglobulin, condition index, and water temperature in green turtles (Chelonia mydas).
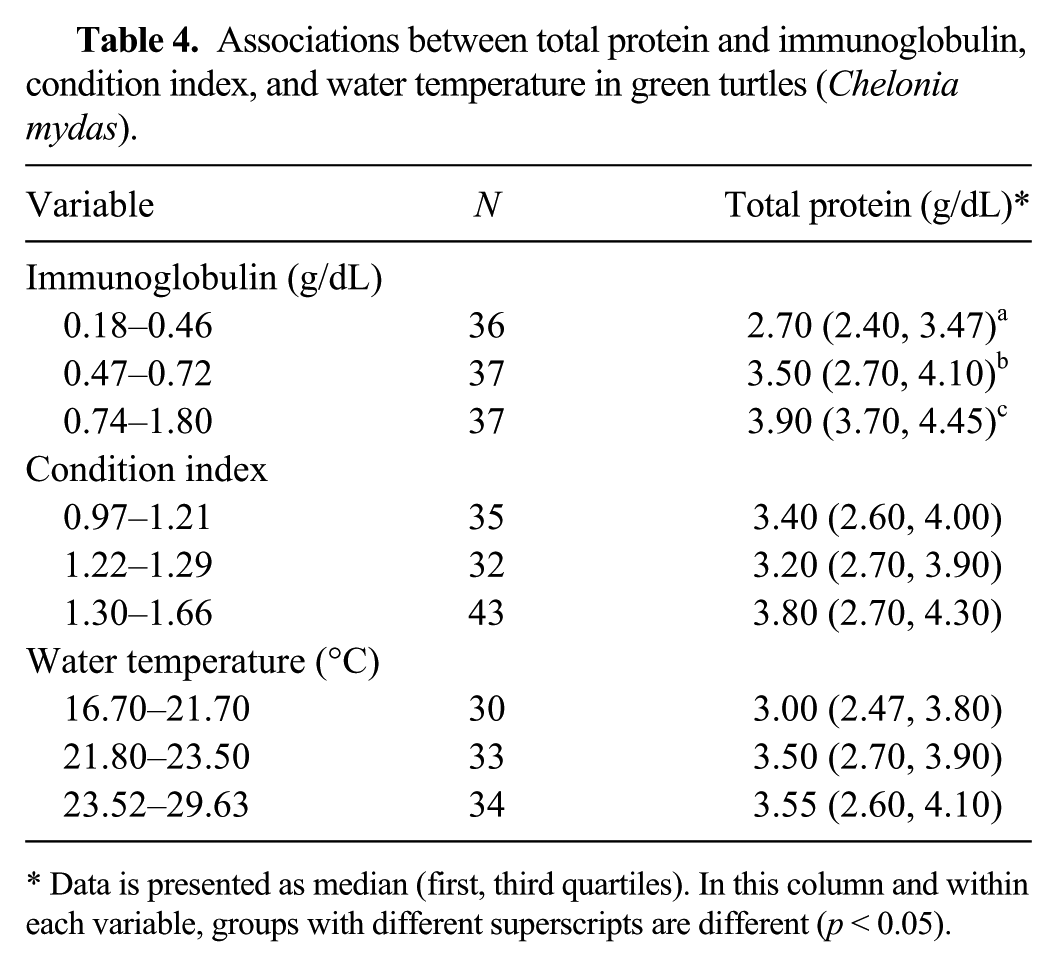
Data is presented as median (first, third quartiles). In this column and within each variable, groups with different superscripts are different (p < 0.05).
Discussion
The format of a cELISA was chosen for our study because of its practicality and the availability of a mAb that specifically recognizes the light chain of the desert tortoise (Gopherus agassizii) Igs with confirmed cross-reactivity with LST and GT Ig as confirmed by Western blot (Fig. 1). Competitive ELISA assays have successfully been utilized for the measurement of Igs in a number of mammal and reptile species for the assessment of immune function, diagnosis of disease, and monitoring of treatment.9,14,25
The cELISA is considered superior compared the direct ELISA, as the latter is titer based and converting a titer to actual concentrations of a protein may not be as accurate and sensitive as a cELISA.2,10 The presented cELISA was successful in the quantification of total Ig LST and GT plasma samples. Possible considerations for the samples that were excluded because their total Ig was either above or below the measurable range include presence of subclinical disease, individual variance in baseline circulating total Ig, undetectable Ig in some individuals, the possibility of high antibody concentrations causing a falsely decreased result (i.e., prozone effect), binding of Ig to sites other than receptors of interest (i.e., nonspecific antibody binding), or lymph contamination during blood sample collection as can occur infrequently in sea turtles. The latter can be observed when clear fluid enters the syringe during blood withdrawal. It is common practice to then discard the sample and collect a new one from another site. Some instances of lymph contamination may not be visibly evident and should be suspected if results are inconsistent.
The use of this mAb in a cELISA provides measurement of total peripheral Igs and thus quantification of humoral immunity in chelonians from multiple families. In contrast to the cELISA, interpretation of plasma protein electrophoresis requires consideration of methodology, presence of hemolysis, fibrinogen concentration, and variability of complement and acute phase proteins in certain disorders. 11 However, cELISA and protein electrophoresis combined complement each other in the diagnostic information they provide, and together could be useful in the clinical setting.
The positive linear relationships between Ig and TP in both LST and GT were anticipated. Total Ig is composed of major fractions of the TP concentration, which presents the sum of all circulating proteins. 11 TP concentrations were similar to previous reports in LST and GT.8,16 The biuret method, as used in our study, is the only currently acceptable method for TP measurement in reptiles, as the bromocresol green method results in falsely increased results, especially in diseased animals. 12
The condition index for GT from our study was similar to values previously reported for this life stage from Caribbean GT. 4 The positive association of TP with condition index in LST is consistent with previous reports in which TP was correlated with SCL and weight. 16 Although TP was positively associated with condition index, a negative association of Ig with condition index was observed in LST. Given that the group of LST in our study included a proportion of larger animals compared to GT, this finding leads to the conclusion that TP in larger LST may contain less circulating Ig than in smaller LST. Another possible influencing factor may be sex; the group of LST may have included females close to sexual maturity. Although the reason for lower total Ig in larger LST cannot definitely be explained, this variation should be considered when interpreting results.
Statistical associations of total Ig with sex and life stage were not investigated in this study. Most of the study animals were too young to be sexed by visual evaluation, and additional methods of sexing were not applied. Various studies in other species observed seasonality and total Ig in association with the animal’s breeding cycle.5,18,21 Given the importance of seasonal and temperature effects on the physiology in reptiles, total Ig by month was presented in our study and did not reveal any obvious trends or variations. Relative age has been documented to share a relationship with immune function in chelonians and other species. In cetaceans, such as the killer whale, the relationship is a progressive increase in serum Ig with increasing age. 21 However, this relationship appears to be inversed in red-eared slider turtles and also in dolphins older than 1 year.17,25
The cELISA assay presented herein augments the diagnostic ability for the study of the humoral immune system and its disorders in sea turtles, and complements other currently available tests of immune function, such as the readily available protein electrophoresis. In the clinical setting, uses of the ELISA could include detection of subclinical or early onset of disease, immunodeficiency, evaluation of inflammatory responses, and monitoring of Ig trends of the sea turtle patient during treatment and rehabilitation. As a research tool, this assay would allow for the evaluation of humoral immunity on the level of sea turtle populations with the potential for extrapolation to aquatic ecosystem health. Additional studies investigating intrinsic and extrinsic factors on the immune system in sea turtles, and Ig quantification in diseased animals, are warranted.
Footnotes
Acknowledgements
We thank Mike Bresette, Inwater Research Group, for logistic support, and Diane Duke, Interdisciplinary Center for Biotechnology Research, for technical assistance. All sample collection protocols were approved by the University of Florida Institutional Animal Care and Use Committee (protocol D-370). Field sampling was conducted under Florida Fish and Wildlife Conservation Commission Marine Turtle Permit #086 issued to E Jacobson.
Authors’ contributions
NI Stacy, E Jacobson, and LG Green contributed to conception and design of the study. AJ Kaplan contributed to conception of the study. CR Le-Bert, HH Nollens, S Bootorabi, and A Bolten contributed to design of the study. NI Stacy contributed to acquisition, analysis, and interpretation of data. E Jacobson and CR Le-Bert contributed to acquisition and analysis of data. AJ Kaplan, HH Nollens, LG Green, and S Bootorabi contributed to analysis of data. JA Hernandez contributed to analysis and interpretation of data. FC Origgi and A Bolten contributed to interpretation of data. AJ Kaplan, NI Stacy, and E Jacobson drafted the manuscript. NI Stacy, E Jacobson, CR Le-Bert, HH Nollens, FC Origgi, LG Green, A Bolten, and JA Hernandez critically revised manuscript. AJ Kaplan, NI Stacy, E Jacobson, CR Le-Bert, HH Nollens, FC Origgi, LG Green, S Bootorabi, and JA Hernandez gave final approval. NI Stacy, E Jacobson, CR Le-Bert, HH Nollens, LG Green, and JA Hernandez agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
GE Healthcare Technologies, Piscataway, NJ.
b.
IgG Plus orientation kit, Pierce Biotechnology Inc., Rockford, IL.
c.
SoftMax Pro Software, Molecular Devices Corp., Sunnyvale, CA.
Declaration of conflicting interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research of this article: The study was funded by the Florida Sea Turtle Grants Program (administered by Caribbean Conservations Corp.) and by a Merck Merial Veterinary Scholars Research Grant to A Kaplan.
