Abstract
Haemophilus parasuis is an economically important swine pathogen with 15 recognized serovars. An enzyme-linked immunosorbent assay (ELISA) was developed that detects serum antibodies to the oligopeptide permease A (OppA) polypeptide membrane protein present in the reference strains for 13 of the H. parasuis serovars. Using the OppA-ELISA, H. parasuis serologic profiles were assessed on 2 swine farms, with seroconversion defined as an OppA-ELISA sample-to-positive (S/P) ratio ≥0.5. Ten gilts from each farm were vaccinated for H. parasuis using either a live avirulent culture vaccine (farm 1) or an inactivated autogenous vaccine (farm 2). Seroconversion occurred in 100% of farm 1 gilts and 90% of farm 2 gilts, with a mean S/P ratio (MSPR) of 3.36 and 1.43, respectively. The OppA-ELISA MSPRs were determined for 2 piglets, 1 male and 1 female, randomly selected from 10 first-parity (P1), 10 second-parity (P2), and 10 third-parity (P3) litters farrowed by respective vaccinated gilts on each farm. On both farms, postfarrowing MSPRs and rate of seropositivity were highest in P1 versus P2 and P3 dams. Parity 1 piglets had higher MSPRs and rates of seropositivity versus later parities, with the difference being significant (P < 0.05) on farm 2. Polymerase chain reaction analysis of nasal swabs indicated that 100% of farm 1 piglets and 47–84%, depending on parity, of farm 2 piglets were H. parasuis–colonized at weaning. The results indicated that H. parasuis vaccination of gilts will not maintain serologic responses in the OppA-ELISA over their reproductive lifetimes, and that maternally derived antibodies do not prevent H. parasuis colonization of piglets.
Haemophilus parasuis, an opportunistic commensal pathogen of swine that can cause Glässer disease and other acute and chronic syndromes, is commonly found worldwide in pigs of all ages. 6 The economic impact of H. parasuis, including high treatment costs and productivity losses, and its potential for sporadic but high-mortality outbreaks are well known.6,10 The importance of H. parasuis in swine production underscores the need for accurate diagnosis of suspected cases and determination of herd immune status so that effective treatment and prevention strategies can be implemented and monitored.
Various laboratory methods are used to obtain a specific H. parasuis diagnosis. Such methods include bacterial culture, immunohistochemical staining, or polymerase chain reaction (PCR) detection from clinical samples, and serotyping using hemagglutination assays (Oliveira S: 2007, Diagnostic notes: Haemophilus parasuis diagnostics. J Swine Health Prod 15:99–103).6,8 However, H. parasuis diagnosis can be challenging for several reasons. Diagnosis of clinical disease can be complicated by coinfections with other pathogens. There are 15 recognized H. parasuis serovars, some of them nonvirulent. 6 Available serovar-specific assays do not identify all serovars circulating in the swine population, including nontypable isolates (Oliveira S: 2007). 4 Because H. parasuis is fastidious, culturing can be difficult as well as labor intensive. Given the limitations of other methods, PCR is the most widely used diagnostic test. However, because H. parasuis is often isolated from the respiratory tract of healthy pigs,11,12,14 positive PCR results from nasal swabs should be considered in conjunction with gross lesions, histology, and epidemiologic patterns that characterize clinical disease.
Protection against H. parasuis systemic infection has been associated with the presence of circulating antibodies. 13 Passive protection provided to litters via colostrum is a principal defense mechanism for protection of piglets against clinical H. parasuis disease.1,7 Prior to 2011, there were a limited number of serovar-specific tests available to monitor H. parasuis serologic status, none of them able to detect all H. parasuis serovars. An enzyme-linked immunosorbent assay (ELISA) using a species-specific oligonucleotide permease A (OppA) polypeptide was developed in 2011 (Macedo N, Oliveira S, van Esch E, Rush P: 2011, Development of a novel universal and species-specific Haemophilus parasuis ELISA test. In: Proceedings of the American Association of Swine Veterinarians, pp. 71–74. Phoenix, AZ, March 5–8) and has been commercially introduced. a The OppA polypeptide was present as a membrane protein in 13 reference H. parasuis serovars tested b (Macedo N, Oliveira S: 2010, Identification of a novel immunogenic and species-specific protein in Haemophilus parasuis. In: Proceedings of the 21st International Pig Veterinary Society Congress, p. 825, Vancouver, Canada, July 18–21). The OppA-ELISA appears to be a universal assay, and researchers have suggested that it can be used to track systemic exposure response regardless of the H. parasuis isolate or its serovar (Macedo N, et al.: 2011). The H. parasuis OppA-ELISA has not been validated in peer reviewed literature to date.
The OppA protein is a transmembrane protein and a component of the adenosine-5′-triphosphate (ATP)-binding cassette (ABC) transporter system. 3 Whole genome analysis of H. parasuis has made possible the identification of a large number of transporter-associated genes, suggesting a remarkable ability for environmental adaptation. 15 Three novel immunogenic ABC-type transporter proteins, including OppA, were identified in H. parasuis based on genomic analysis. 3 The immunological relevance of OppA has been examined in whole cell protein preparations from H. parasuis isolates recovered from convalescent pigs that experienced Glässer disease and probed with convalescent sera using Western blots (Macedo N, et al.: 2011, Development of a novel universal and species-specific Haemophilus parasuis ELISA test). Results from that study indicated that an immunodominant protein, later identified by sequencing as OppA, was present in all 10 H. parasuis isolates recovered from the outbreak and also in 13 H. parasuis reference strains. The specificity of OppA was tested by Western blot analysis using whole cell proteins from 10 bacterial species commonly found in swine tissue; the OppA protein was absent in all the bacteria tested including Actinobacillus spp. (A. porcinus, A. indolicus, A. minor, A. pleuropneumoniae, and A. suis), Streptococcus suis, Bordetella bronchiseptica, Pasteurella multocida, Escherichia coli, and Salmonella enterica subsp. enterica serovar Choleraesuis (Macedo N, Oliveira S: 2010, Identification of a novel immunogenic and species-specific protein in Haemophilus parasuis). b Because of its highly immunogenic and species-specific characteristics, the H. parasuis OppA was cloned into E. coli, expressed, purified, and used as the coating antigen for the development of the novel ELISA used in the present study (Macedo N, et al.: 2011).
The immunogenic and protective capacity of OppA was evaluated in pigs vaccinated with a highly purified H. parasuis OppA subunit vaccine and subsequently challenged with H. parasuis. Serum immunoglobulin G (IgG) responses were detected in immunized pigs after the first and second vaccine doses, reaching greater responses after challenge, while no specific IgG responses were found in control pigs. However, the subunit OppA vaccine was not protective against challenge with a virulent strain (Macedo N, Oliveira S, Torremorell M, Rovira A: 2012, Evaluation of the immunogenic and protective capacity of a transmembrane protein of Haemophilus parasuis. In: Proceedings of the 22nd International Pig Veterinary Society Congress, p. 142, Jeju Island, Korea, June 10–13).
Using the H. parasuis OppA-ELISA, the current study sought to determine the postvaccination serologic response in vaccinated gilts and the passive transfer of H. parasuis maternal antibodies to their litters. The effect of sow parity on passive antibody transfer was determined for first- (P1), second- (P2), and third-parity (P3) litters. The PCR assay was used to determine H. parasuis colonization in piglets and the relationship of colonization to serologic status.
Haemophilus parasuis serologic profiles were assessed on 2 swine farms. Farm 1 had a history of confirmed Glässer disease in nursery pigs, which is the population typically most often affected in disease outbreaks.9,12 Farm 2 had no clinical history of H. parasuis. The study design, as implemented on each farm, is shown in Figure 1.
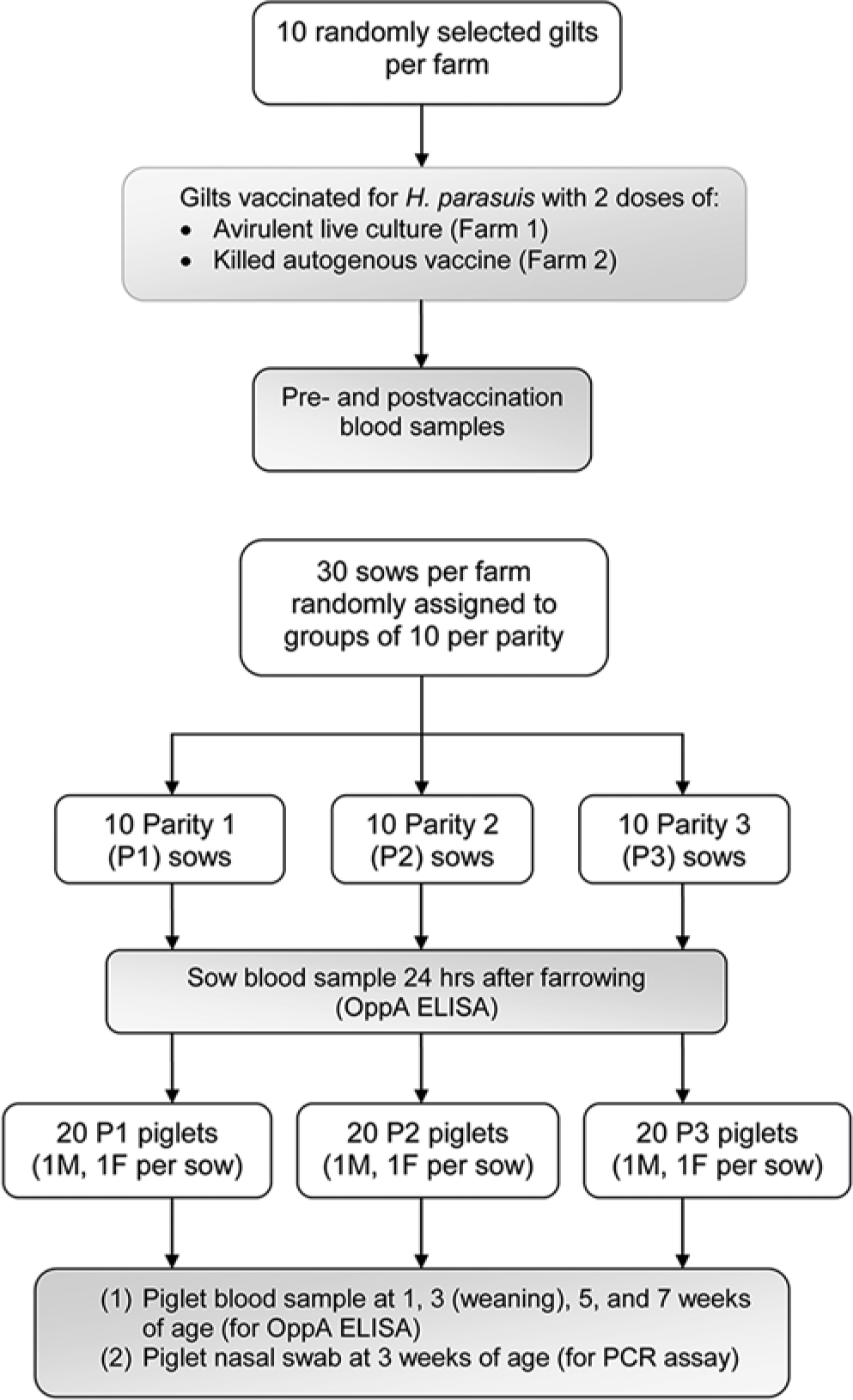
Study design for determining the Haemophilus parasuis serologic profile of vaccinated sows and their litters on each of 2 endemic farms. To evaluate serologic response to vaccination, blood samples were obtained from 10 randomly selected gilts from each farm before and after vaccination. Serologic status was determined for 10 randomly selected, vaccinated sows from each farm and for piglets from parity 1, parity 2, and parity 3 litters from respective sows. Haemophilus parasuis colonization was determined for piglets at week 3.
Ten clinically normal gilts on each farm were randomly selected to measure serologic status and postvaccination serologic response. Gilts on farm 1 were vaccinated with 2 doses of an H. parasuis avirulent live culture vaccine, given, respectively, at 13 and 17 weeks of age. c Gilts on farm 2 were vaccinated with an autogenous, noncommercial killed vaccine, with the first dose given at 18–23 weeks of age and the second dose at 28 weeks of age. To determine seroconversion rate and duration, blood samples were obtained before vaccination and at 2 weeks after the second vaccine dose.
A further 30 sows on each farm were randomly assigned to P1, P2, and P3 groups of 10 sows each. Farm 1 sows farrowed their first litter 21–24 weeks after receiving the second vaccine dose. Farm 2 sows farrowed their first litter 29 weeks after the second vaccine dose. No further vaccinations were given to sows in subsequent parities. Cross-fostering among litters was discouraged for the first 24 hr after farrowing in all cases. All litters were weaned at 3 weeks of age. Blood samples for OppA-ELISA testing were obtained from respective sows 24 hr after the sows farrowed their P1, P2, or P3 litters.
Two piglets, 1 male and 1 female, were randomly selected from each of the 10 P1, 10 P2, and 10 P3 litters farrowed by respective, vaccinated dams on each farm (vaccinated as gilts). Blood samples were obtained from the piglets at 1, 3, 5, and 7 weeks of age for OppA-ELISA testing. Nasal swabs were collected from the piglets at 3 weeks of age for PCR analysis to determine local H. parasuis colonization.
Blood samples were tested for H. parasuis OppA-ELISA antibodies, and nasal swabs were tested with a H. parasuis PCR 8 by a veterinary diagnostic laboratory. d Serologic response was expressed as a sample-to-positive (S/P) ratio of ELISA optical density values, with a S/P value ≥0.5 being defined as H. parasuis seropositivity. The PCR results were expressed as positive or negative.
To determine seroconversion after vaccination, mean S/P ratios (MSPRs) and the percentage of positive samples were compared in gilts pre- and postvaccination and in P1 sows. To determine a parity effect on serological responses, sow and litter serological results were analyzed with commercial software, e where log-transformed antibodies were analyzed using a repeated measures analysis with a model that considered parity, week, and the interaction of parity-by-week as fixed effects while sow and the residual error were considered random effects. Week was the repeated factor. The covariance structure in the repeated measures analysis was investigated using compound symmetry. Sow parity, week, and the interaction of parity-by-week were all tested at the 5% level of significance (P < 0.05). The PCR-positive tests were expressed as a percentage of the tested population.
The MSPR results indicated that on farm 1 all gilts were negative prevaccination and that on farm 2, 1 sample was positive (10%) and the rest of the samples were negative (90%). Postvaccination OppA-ELISA S/P ratios indicated that both vaccines induced H. parasuis seroconversion in a high percentage of vaccinated gilts: 100% of farm 1 gilts and 90% of farm 2 gilts. In both farms, 50% P1 sows had detectable H. parasuis systemic antibodies at the time their first litter was delivered.
On both farms, the immediate post-farrowing MSPR was highest in P1 sow compared to P2 and P3 sows, with the rate of seropositivity generally declining at successive parities (Table 1). Farm 1 sows maintained a MSPR above the seropositive threshold for the P1 and P2 parities, while farm 2 sows were seropositive only for P1. The MSPRs and rates of seropositivity generally declined in litters from successive parities (Table 1). The difference in MSPR between parities was not significant on farm 1. On farm 2, P1 litters had a significantly higher MSPR versus the MSPR for P3 litters at each postfarrowing time point and for P2 litters at 1 and 5 weeks postfarrowing only. Results from the present study indicate that the OppA-ELISA detected systemic IgG antibodies following H. parasuis vaccination and antibodies acquired via colostrum intake.
Haemophilus parasuis serological profiles in sows from parities 1, 2, and 3 and their litters, and the percentage of colonization in piglets at 3 weeks of age.*
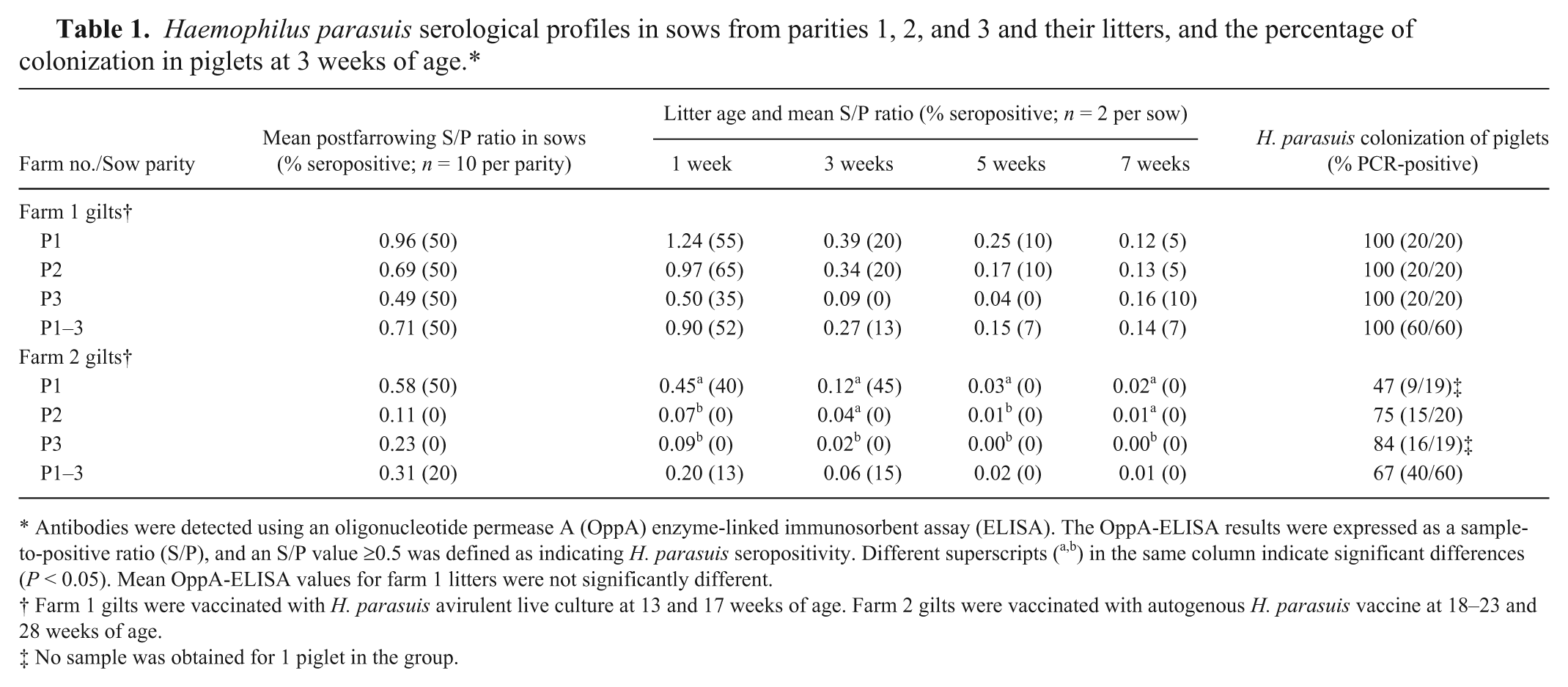
Antibodies were detected using an oligonucleotide permease A (OppA) enzyme-linked immunosorbent assay (ELISA). The OppA-ELISA results were expressed as a sample-to-positive ratio (S/P), and an S/P value ≥0.5 was defined as indicating H. parasuis seropositivity. Different superscripts (a,b) in the same column indicate significant differences (P < 0.05). Mean OppA-ELISA values for farm 1 litters were not significantly different.
Farm 1 gilts were vaccinated with H. parasuis avirulent live culture at 13 and 17 weeks of age. Farm 2 gilts were vaccinated with autogenous H. parasuis vaccine at 18–23 and 28 weeks of age.
No sample was obtained for 1 piglet in the group.
The PCR analysis of nasal swabs indicated that all piglets on farm 1 were colonized with H. parasuis at the time of weaning at 3 weeks of age, regardless of parity (Table 1). On farm 2, the rate of colonization at weaning ranged from 47% to 84% depending on parity (Table 1). The serologic and colonization data provided several insights on the dynamics of the H. parasuis immune response following vaccination or natural exposure, some of which were unexpected. The PCR results confirmed that both farms were endemic for H. parasuis, with the organism able to colonize a high percentage of weaned piglets that showed no evidence of clinical disease. The usual source of H. parasuis infection is from sow to litter during the suckling period. 6 Colonization of litters from all parities in the current study indicates that the dams were infected with H. parasuis and were the primary source of infection. Haemophilus parasuis colonization occurred in a high percentage of piglets 3 weeks after farrowing even in cases where they were seropositive. This indicates that colostrum-derived serum antibodies as detected in the OppA-ELISA from vaccinated sows do not prevent nasal colonization. These colonized pigs have the potential to expose other pigs or for recrudescence into clinical disease under conditions of stress or coinfection. Results from the current study differed from a prior study that reported low levels of colonization in litters of unvaccinated young sows at 14 and 28 days of age compared to multiparous sows. 5 In another study, piglets from multiparous sows vaccinated in gestation had higher antibody levels at early time points and subsequently were colonized later and to a lower degree than piglets from nonvaccinated sows, suggesting that sow vaccination modulates colonization of the upper respiratory tracts in piglets. 2 In contrast, in the current study, nasal colonization occurred in the presence of systemic antibodies (IgG) detected by the OppA-ELISA.
The results of the current study also indicate that H. parasuis upper respiratory tract colonization within the herd does not necessarily result in a serological response in the colonized pig or in contact pigs. To illustrate, farm 2 P2 and P3 sows and their litters were serologically negative in the OppA-ELISA at all time points despite H. parasuis colonization in ≥75% of the piglets that were tested by PCR assay. Stated another way, natural H. parasuis exposure in an endemic setting did not produce or maintain a serologic response detectable in the OppA-ELISA in clinically normal pigs.
Sows vaccinated as gilts had OppA-ELISA MSPRs for H. parasuis that declined over time, which corresponded to declining levels of maternally derived antibodies in litters from later parities. Thus, similar to previous reports, it is incorrect to assume that vaccination of gilts will provide protective levels of colostral antibodies throughout the reproductive life of the sow. 6 Periodic revaccination of female breeding stock, perhaps prior to farrowing across all parities on high-risk farms, is advisable to maintain preweaning protection in successive litters. Given the short duration of passive immunity in piglets that was demonstrated in the current study, vaccination of litters to confer active postweaning immunity may be equally relevant as sow vaccination for protecting the herd against a clinical outbreak. The presence of maternal antibodies has been found not to interfere with vaccine efficacy in piglets vaccinated prior to weaning. 13 However, care must be taken, as the vaccine efficacy in the presence of maternal antibodies may be influenced by vaccine type and vaccination protocol. 13
Several conclusions can be drawn from the current study. Colonization of piglets on both farms indicates that the premises were endemic for H. parasuis and that litters were exposed by clinically normal dams. The current findings indicated that H. parasuis respiratory colonization does not elicit a systemic antibody response detectable with the OppA-ELISA. The H. parasuis colonization rates in the present study were higher than previously reported, with as many as 100% of pigs becoming carriers by weaning age in one of the herds, even in the presence of maternally derived serum antibodies. Seroconversion patterns in gilts indicate that either live or inactivated H. parasuis vaccines will induce active immunity detectable with the OppA-ELISA (Fig. 2). The current findings dispel the assumption that clinically normal sows will maintain active immunity in an endemic setting where repeated exposure occurs. Previous studies have determined that H. parasuis maternally derived antibodies begin to decline in litters by the age of 5–8 weeks.6,9 Results from the current study indicate that colostral antibodies, as detected in the OppA-ELISA, may decline much more rapidly, with seropositivity disappearing in a high percentage of piglets by weaning at 3 weeks of age, even on farms where sows were vaccinated for H. parasuis and where endemic exposure occurs and in a farm with no cases of Glässer disease reported (farm 2).
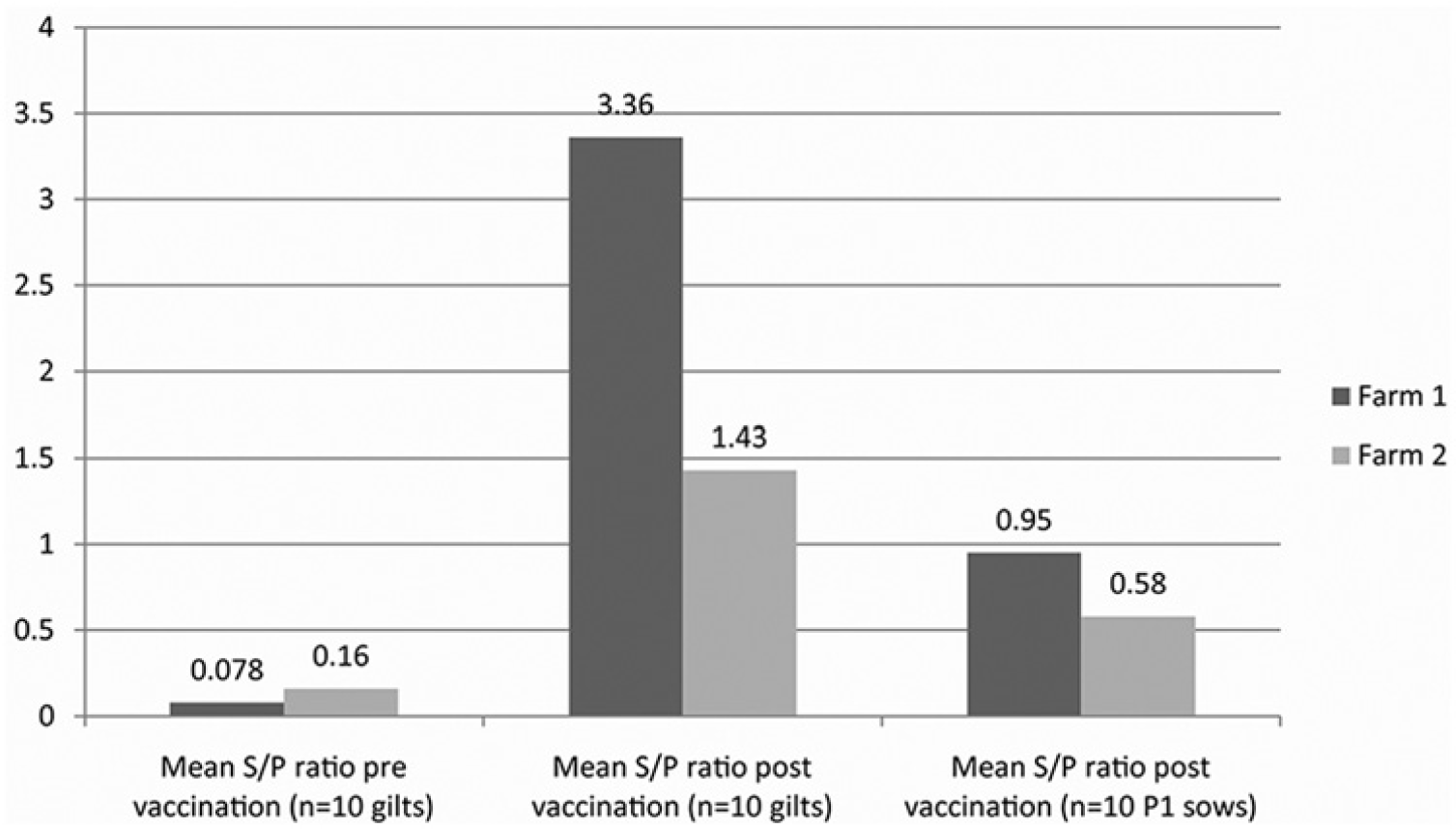
Haemophilus parasuis seroconversion in dams vaccinated as gilts using an avirulent vaccine (farm 1) or an autogenous vaccine (farm 2) and antibody decline in parity 1 (P1) sows, approximately 6 months postvaccination. The cutoff for seropositivity (sample-to-positive [S/P] ratio ≥0.5) was determined by enzyme-linked immunosorbent assay using a species-specific oligonucleotide permease A polypeptide.
The results of the present study have several practical implications. Gilts should be vaccinated and exhibit seroconversion before entering an H. parasuis–positive herd as reproductive stock. In high-risk herds, sow vaccination prior to each farrowing is more likely to protect preweaned litters via colostral antibodies (Oliveira S: 2007, Diagnostic notes: Haemophilus parasuis diagnostics). Because passively acquired antibodies acquired from colostrum may be short-lived, vaccination of litters at or near weaning is advisable to minimize clinical disease and mortality in the animals at greatest risk.
Availability of a universal H. parasuis serologic assay such as the OppA-ELISA gives veterinarians a means of assessing herd serological status, confirming a serological response following vaccination, timing the introduction of vaccinated gilts into H. parasuis–positive herds, monitoring passive transfer of antibodies to litters, and identifying the best time to implement anti-infective therapies.
Footnotes
Acknowledgements
The authors acknowledge the contributions of Mark Dana of Scientific Communications Services, Limited Liability Company in the preparation of the manuscript. The authors thank Drs. Albert Rovira, Simone Oliveira, and Nubia Macedo of the University of Minnesota, and Dr. Eric van Esch of BioChek USA for technical assistance in use of the OppA-ELISA.
a.
Haemophilus parasuis antibody test kit, BioChek USA, Scarborough, ME.
b.
Haemophilus parasuis polypeptides and methods of use, WIPO Patent Application WO/2011/056954.
c.
ParaSail, Newport Laboratories, Worthington, MN.
d.
University of Minnesota Veterinary Diagnostic Laboratory, College of Veterinary Medicine, St. Paul, MN.
e.
SAS 9.2 (Proc Mixed procedure), SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
Two authors (Galina and Amodie) work for Zoetis. Zoetis manufactures antimicrobials and vaccines for H. parasuis, none of which were evaluated in this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Zoetis.
