Abstract
The objective of the current study was to determine whether detection of Canine distemper virus (CDV)-specific serum antibodies correlates with resistance to challenge with virulent virus. Virus neutralization (VN) assay results were compared with resistance to viral challenge in 2 unvaccinated Beagle puppies, 9 unvaccinated Beagle dogs (4.4–7.2 years of age), and 9 vaccinated Beagle dogs (3.7–4.7 years of age). Eight of 9 (89%) unvaccinated adult dogs exhibited clinical signs after virus challenge, and 1 (13%) dog died. As compared to adult dogs, the 2 unvaccinated puppies developed more severe clinical signs and either died or were euthanized after challenge. In contrast, no clinical signs were detected after challenge of the 9 adult vaccinated dogs with post-vaccination intervals of up to 4.4 years. In vaccinated dogs, the positive and negative predictive values of VN assay results for resistance to challenge were 100% and 0%, respectively. Results indicate that dogs vaccinated with modified live CDV can be protected from challenge for ≤4.4 years postvaccination and that detection of virus-specific antibodies is predictive of whether dogs are resistant to challenge with virulent virus. Results also indicate that CDV infection in unvaccinated dogs results in age-dependent morbidity and mortality. Knowledge of age-dependent morbidity and mortality, duration of vaccine-induced immunity, and the positive and negative predictive values of detection of virus-specific serum antibodies are useful in development of rational booster vaccination intervals for the prevention of CDV-mediated disease in adult dogs.
Introduction
Canine distemper virus (CDV; order Mononegavirales, family Paramyxoviridae, subfamily Paramyxovirinae, genus Morbillivirus) is a highly contagious, vaccine-preventable organism that infects dogs.2,3 Canine vaccination protocols have been under review since the 1990s because of an increased awareness of the risks associated with revaccination and acceptance that duration of immunity (DOI) provided by vaccines against many pathogens is likely >1 year.4,5,11,12,15,20,23 DOI in dogs following administration of a number of killed Rabies virus vaccines has been demonstrated by challenge and the presence of virus-specific antibody has been shown to be at least 3 years.13,14 Many researchers believe the DOI induced by other viral vaccines is similar or even >3 years.1,9,17,18,21,25 In 2003, the American Animal Hospital Association’s Canine Vaccine Task Force (AAHA-CVTF) recommended that puppies ≥6 weeks old be vaccinated against CDV every 3–4 weeks until they are 12–14 weeks of age, be administered a booster vaccination 1 year later, and, after the 1-year booster, dogs at minimal risk of infection be administered additional boosters no more than every 3 years. 19 In 2011, the AAHA-CVTF released revised guidelines that reaffirm these recommendations and conclude that no negative medical ramifications have resulted from implementation of these guidelines. 26
Modified live CDV vaccines are thought to elicit both humoral and cell-mediated immune responses. Humoral immune responses can readily be measured by detecting virus-specific antibodies. In contrast, quantifying cell-mediated immune responses is difficult, thus cell-mediated immune responses are not routinely measured. CDV antibodies derived from colostrum have been shown to be sufficient for protection from challenge. 7 However, it is not currently known to what extent humoral or cell-mediated immunity is responsible for the protection elicited by vaccination against CDV. Nevertheless, if the CDV-specific antibody titer can be shown to be correlated with protection from challenge, then serologic screening of individual vaccinated dogs can be used as an alternative to booster vaccination at arbitrary intervals. 24 In a previous study, CDV serum antibody titers could be detected in 95% of dogs 48 months after the last vaccination. 16
At the time of the experiments described herein, the approved U.S. Department of Agriculture (USDA) CDV challenge model was to inoculate the virus intrathecally. We considered this model to be too invasive and argue that it does not mimic natural infection. Thus, the first objective of our study was to determine whether intranasal inoculation of CDV could be used in vaccine challenge studies. The other objectives of the study were to use the new challenge model to determine whether dogs with a vaccine interval of >3 years are resistant to CDV challenge and whether CDV serologic titers could be used to predict resistance or susceptibility to disease.
Materials and methods
Research dogs
A total of 9 purpose-bred, unvaccinated beagles (4–7 years of age) and 9 vaccinated beagles (3–4 years of age) were housed separately in a closed facility until used in the experiments described herein. For the final CDV challenge study, an additional 2 purpose-bred, unvaccinated beagles (11–12 weeks of age) were purchased. The vaccinated adult dogs had been administered a modified live multivalent (canine distemper, adenovirus type 2, parainfluenza virus, and parvovirus) vaccine a subcutaneously with an average interval as the last vaccination was, on average, 4.2 years (range: 3.3–4.4 years) prior to challenge in our study. All of the experiments were approved by the Institutional Animal Care and Use Committee at the research facility.
Monitoring
Serum was collected for CDV virus neutralization (VN) assay on days 0, 7, 14, and 21. Temperature, body weight, and clinical signs (e.g., anorexia, coughing, dehydration, depression, diarrhea, lethargy, ocular discharge, and/or nasal discharge) were recorded daily for 14 days after challenge. The daily clinical monitoring was performed by individuals blinded to the dog’s vaccination status using the USDA CDV challenge protocol. b Dogs developing clinical signs of CDV infection were to be administered supportive care as indicated, with euthanasia performed for dogs exhibiting severe, irreversible clinical signs.
Canine distemper virus antibody assay
Sera were tested for CDV antibodies by means of a VN assay performed at a commercial laboratory. c Results for the CDV VN assay were considered by the laboratory to be positive if titers were >16. The CDV strain used in the VN assay was not reported by the laboratory at the time of testing.
Development of an intranasal CDV challenge model
Three initial experiments were performed in adult unvaccinated dogs to determine an effective dose of virulent CDV for intranasal challenge. The CDV samples used for challenge were obtained from the Center for Veterinary Biologics Laboratory d (CVB-L) and were maintained frozen at −70°C until used. Prior to challenge, the VN titers were ≤4 for each of the unvaccinated dogs.
In experiment 1, the Snyder Hill CDV strain (CVB-L lot 90-18) was thawed, diluted 1:20 in serum-free Dulbecco modified Eagle medium and administered by atomizer into each nostril (1.0 mL) of 2 unvaccinated adult dogs (4.4 and 5.9 years of age, respectively) that were lightly anaesthetized with a short-acting barbiturate. In experiment 2, two unvaccinated adult dogs (6.7 and 7.0 years of age, respectively) were challenged using the same design as experiment 1 except a 1.0 mL of a 1:3.3 dilution of CDV (CVB-L lot 90-18) was administered into each nostril (2.0 mL total). In experiment 3, three unvaccinated adult dogs (4.8, 6.5, and 7.2 years of age, respectively) were challenged using the same design as experiment 1 except 1.0 mL of a 1:5 dilution of a different lot of the Snyder Hill CDV strain (CVB-L lot 93-03) was administered into each nostril (2.0 mL total). The primary clinical and laboratory findings from these 3 experiments (Table 1) were used to determine the CDV challenge dose in vaccinated dogs.
Intranasal Canine distemper virus challenge model experiments.*
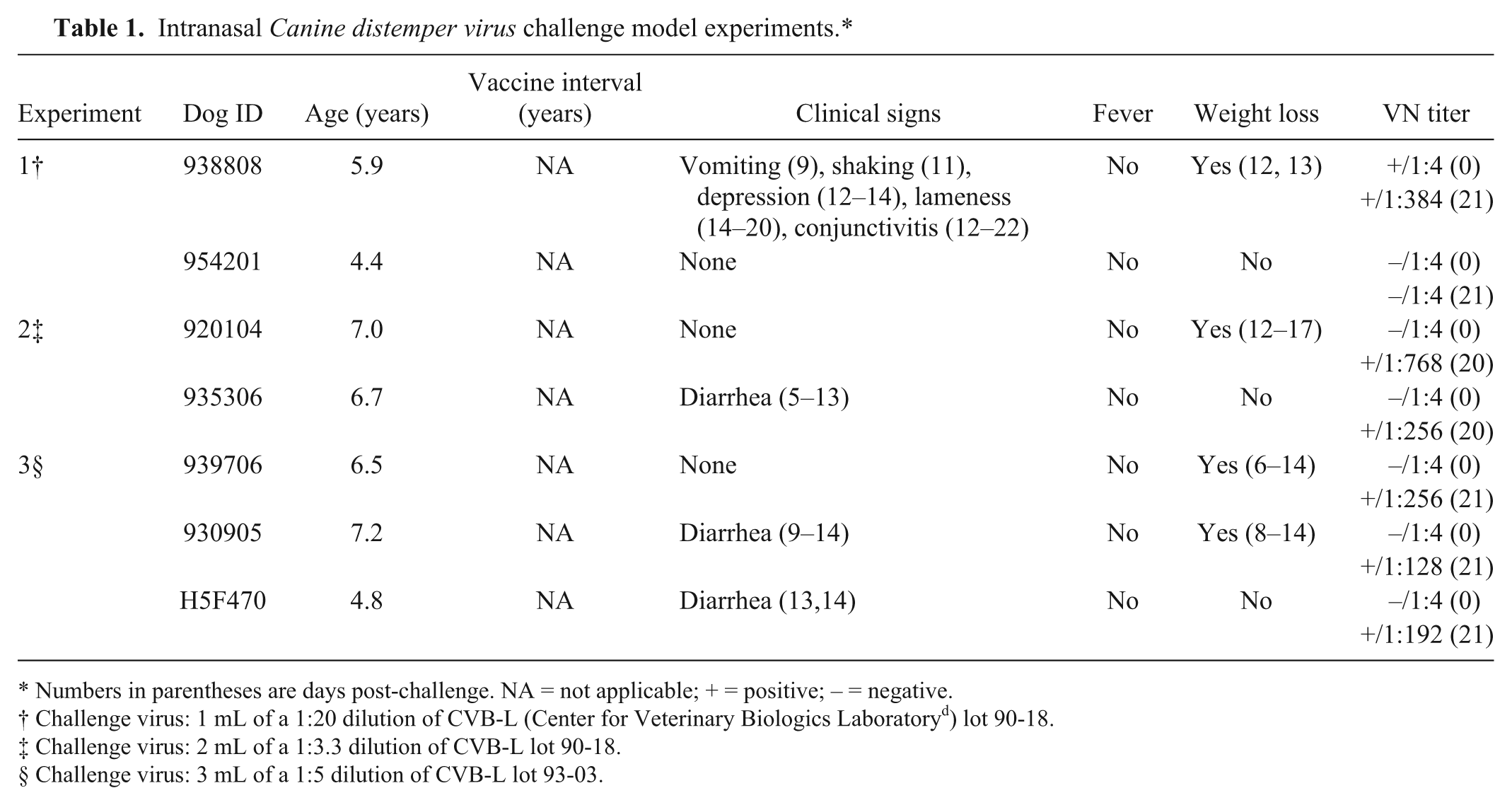
Numbers in parentheses are days post-challenge. NA = not applicable; + = positive; – = negative.
Challenge virus: 1 mL of a 1:20 dilution of CVB-L (Center for Veterinary Biologics Laboratory d ) lot 90-18.
Challenge virus: 2 mL of a 1:3.3 dilution of CVB-L lot 90-18.
Challenge virus: 3 mL of a 1:5 dilution of CVB-L lot 93-03.
Intranasal CDV challenge of vaccinated dogs
A final experiment was performed to determine the susceptibility of adult vaccinated dogs to challenge with virulent CDV. Two unvaccinated puppies (both 0.25 years of age) and 2 adults (5.2 and 6.7 years of age, respectively) were used in the final experiment as controls to document viability of the CDV challenge viruses. The unvaccinated dogs and 9 vaccinated dogs (3.7–4.7 years of age) were challenged with CDV as described in the challenge model optimization experiments except that 1 mL of a 1:5 dilution of a pool of equal volumes of CVB-L lot 90-18 and 93-03 was administered into each nostril (2.0 mL total). Prior to challenge, the unvaccinated dogs had VN titers ranging from 6 to 16 and so were considered susceptible (VN titer ≤16) based on the testing laboratory definition (Table 2). The 9 vaccinated dogs had prechallenge VN titers ranging from 8 to 96, with 5 of the 9 dogs having titers of ≤16 (Table 2).
Susceptibility of vaccinated dogs to Canine distemper virus challenge.*
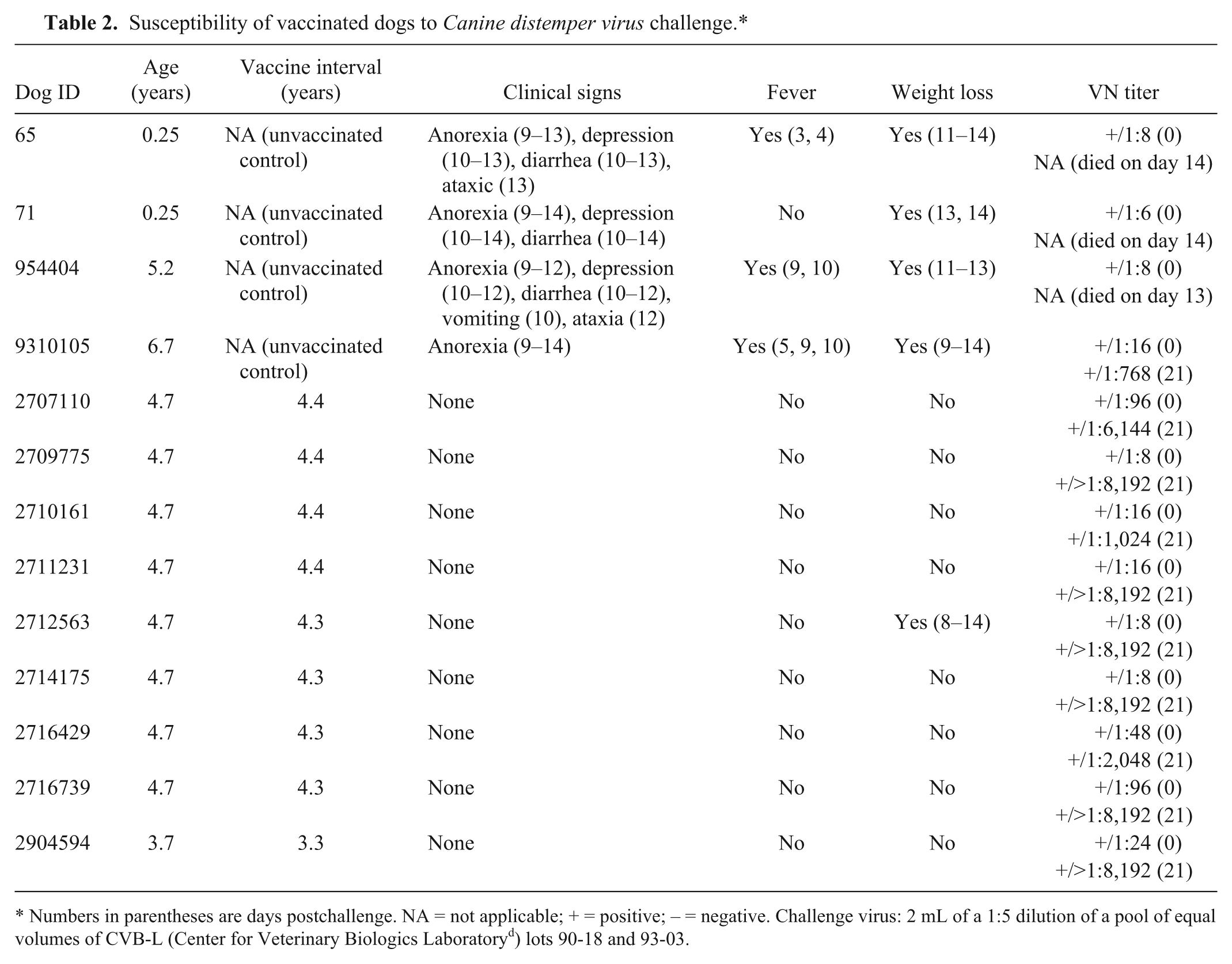
Numbers in parentheses are days postchallenge. NA = not applicable; + = positive; – = negative. Challenge virus: 2 mL of a 1:5 dilution of a pool of equal volumes of CVB-L (Center for Veterinary Biologics Laboratory d ) lots 90-18 and 93-03.
Data analyses
Dogs in our study were considered susceptible to challenge with CDV if they developed weight loss of ≥10% of prechallenge body weight and clinical signs of CDV infection (e.g., anorexia, cough, depression, diarrhea, fever, lethargy, vomiting, nasal discharge, and/or ocular discharge). Results of the VN assay were compared with results of CDV challenge, and predictive values of positive and negative tests were then calculated using results for vaccinated dogs. 10
Results
Development of an intranasal CDV challenge model
Only 1 of the 2 adult unvaccinated dogs exhibited clinical signs of CDV infection in the first challenge experiment and so the inoculation dose was increased in the second challenge experiment (Table 1). As this challenge was also considered inadequate, a different lot of CDV was assessed in the third challenge experiment, which resulted in clinical signs of disease in all 3 adult beagles (Table 1). In an attempt to assure that clinical illness would develop, a combination of the 2 lots of CDV was chosen for the final challenge model.
Prediction of CDV resistance or susceptibility on challenge
In this experiment, all 4 unvaccinated dogs developed clinical signs consistent with CDV infection (Table 2). One of the adult unvaccinated dogs and both unvaccinated puppies challenged with the combination of 2 CVB-L challenge viruses died or were euthanized as a result of CDV infection. Only 1 of the 9 vaccinated dogs experienced weight loss, and none of the dogs developed clinical signs of CDV infection, so all were considered to be resistant to CDV on challenge. The surviving unvaccinated dog and all 9 of the vaccinated dogs seroconverted to CDV as determined by a minimum of a 4-fold increase in their CDV VN titers.
None of the 4 vaccinated dogs with positive VN assay results developed clinical signs of CDV infection following challenge, giving a positive predictive value of 100%. However, all 5 vaccinated dogs with CDV VN assay results suggesting susceptibility (≤16 titer) were also resistant to CDV infection and so the negative predictive value of the VN assay results was 0%.
Discussion
Intranasal administration of CDV using the challenge protocol developed herein was able to induce clinical signs of CDV disease in all 4 unvaccinated dogs in the final experiment. Three of the 4 unvaccinated control dogs (1 adult and both puppies) died or were euthanized. The severity of illness was similar to the intrathecal CDV inoculation model used by the USDA at the time, where it was defined that 80% of control dogs must die for a CDV challenge experiment to be considered successful. Thus, we believe that the final challenge model used herein to determine whether the vaccinated beagles were resistant or susceptible to CDV-induced disease was valid.
In our study, adult vaccinated Beagles were resistant to virulent CDV on challenge even though the post-vaccination interval was >4 years in 8 of the 9 dogs. All 9 vaccinated dogs developed at least a 4-fold increase in CDV VN titers in the 3-week post-challenge serum samples relative to pre-challenge serum samples documenting adequate exposure to the CDV challenge virus. These findings support the current AAHA-CVTF recommendations. 26
The predictive value of positive antibody test results for vaccinated dogs was 100% for the CDV VN assay in that all dogs with positive results were resistant to challenge. Thus, although it has not been determined whether CDV-specific antibodies are responsible for protection from challenge, results of the study confirm that detection of CDV-specific antibodies is predictive of whether dogs are resistant to challenge with virulent CDV. Because a positive CDV VN assay result is predictive of protection from disease in vaccinated dogs, use of the assay described in the current study to detect virus-specific antibody in previously vaccinated dogs could be applied in the development of rational booster vaccination regimens for adult dogs.
For some unvaccinated dogs in our study, results of the CDV VN assay were positive at titers ≤16. These results were likely due to nonspecific binding of unknown substances in the serum to test reagents and do not necessarily indicate the presence of virus-specific antibody. Therefore, we suggest that the CDV VN assay not be used to predict resistance to CDV infection in dogs for which the vaccination history is unknown unless the VN titer is at least 2-fold above the assay cutoff (i.e., ≥64 for this VN assay).
The percentage of adult vaccinated dogs that lacked detectable CDV-specific antibodies (VN titers ≤ 16) was higher than previously reported. 25 However, in our study, the absence of detectable antibody in vaccinated dogs did not result in susceptibility to disease on CDV challenge. These results confirm that cell-mediated immunity and/or the ability to rapidly regenerate humoral immunity can maintain protection even when serum antibodies have declined to low or undetectable levels as previously described. 22 Thus, if serologic testing for CDV-specific antibodies is used to determine whether dogs require booster vaccination, some dogs will be vaccinated unnecessarily. However, the number would be less than the number of dogs that would be vaccinated unnecessarily if an arbitrary booster vaccination interval were used.
Of the 9 unvaccinated adult dogs used in the challenge experiments in our study, clinical signs of CDV occurred in 8 dogs. One dog in the first challenge experiment failed to develop clinical signs, most likely due to an inadequate challenge as evidenced by a lack of seroconversion. The clinical signs of CDV resolved in 7 of the other 8 (87%) adult unvaccinated dogs challenged with CDV. The observed mortality of 1 out of 8 (13%) is lower than what has been previously reported in adult unvaccinated dogs. 6 However, the studies cannot be directly compared as the previous study used the intrathecal CDV administration route. Although all but 1 of the adult unvaccinated dogs in our study developed self-limited clinical signs of CDV infection, both unvaccinated puppies developed severe clinical signs and either died or were euthanized. These finding could support the hypothesis that an age-associated decrease in the severity of disease caused by CDV infection in dogs exists. 8 However, this conclusion should be interpreted cautiously as different lots and doses of CDV were used over the course of the experiments described herein.
Footnotes
Authors’ contributions
WA Jensen, JS Totten, and MR Lappin contributed to conception and design of the study. RD Schultz contributed to the design of the study. WA Jensen and JS Totten contributed to acquisition, analysis, and interpretation of data. MR Lappin and RD Schultz contributed to analysis and interpretation of data. WA Jensen drafted the manuscript. JS Totten, MR Lappin, and RD Schultz critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Either Duramune Adult (Fort Dodge Animal Health, Fort Dodge, IA) or Progard-5 (Intervet Inc., Millsboro, DE).
b.
Code of Federal Regulations. Animals and Animal Products (revised January 1, 2003); Canine distemper vaccine (codified at 9 CFR 113.306).
c.
New York State Diagnostic Laboratory, Ithaca, NY.
d.
USDA, Animal and Plant Health Inspection Services, Veterinary Services, Center for Veterinary Biologics Laboratory, Ames, IA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Heska Corp.
