Abstract
In vivo, neutralizing antibodies are critical for viral clearance. A high-throughput serum neutralization (HTSN) assay was developed to antigenically categorize Swine influenza virus (SIV) isolates. Uncategorized viruses were tested using a panel of antisera representing the H3N2 SIV subtypes and the results expressed as a serum neutralization ratio. Antisera were generated against contemporary isolates representing circulating H3N2 SIV subtypes (clusters I, III, IV). Reference viruses and the corresponding antisera were evaluated using traditional hemagglutination inhibition (HI) and the HTSN assays and good correlation (r = 0.84) was observed between the 2 tests. Categorical clustering of 40 recent (2008-2009) SIV isolates was assessed using the HTSN assay. The H3N2 SIV isolates with amino acid similarity >97% to the commonly used H3N2 cluster IV reference strain A/Swine/Ontario/33853/2005 (ON05) showed strong reactivity with cluster IV antisera. Isolates with <97% amino acid similarity to ON05 sporadically or completely failed to react with any antiserum. A cluster of 3 isolates with weak reaction with cluster III antiserum may be a potential emerging cluster of H3N2 with moderate genetic similarity to cluster II H3N2 (93% similarity). Potential uses of the HTSN assay include identification of broadly cross-reactive or antigenically distinct SIV isolates for use in vaccine virus selection or as part of surveillance efforts monitoring antigenic drift.
Introduction
Over the last decade, Swine influenza virus (SIV) has demonstrated marked genetic diversity. 23 From its first recognition in 1918, classical H1N1 remained the dominant subtype of influenza until 1998 when a triple reassortant H3N2 SIV, a combination of human, avian, and swine lineage influenza, rapidly spread throughout the United States. 15,25 Further reassortment between H3N2 and classical H1N1 SIV has led to H1N1 and H1N2 reassortants. 2,24 In addition, H3N1 and H2N3 reassortants have been isolated from swine, although these have not become widely distributed. 12,13 Genetic and antigenic analysis of H3N2 SIV led to further categorization of viruses into 4 clusters based on their unique human-like hemagglutinin (HA) gene lineages. While clusters I, II, and III were common in swine herds in the late 1990s through early 2000s, cluster IV, typified by A/Swine/Ontario/33853/2005, is now the dominant cluster. 16,18,25
The increasing genetic diversity of SIV makes serologic evaluation of host immune response challenging. 10,11 The hemagglutination inhibition (HI) assay has long been used to quantify SIV antibodies. However, there are problems with the HI assay, which include differing abilities of viruses to agglutinate erythrocytes leading to loss of assay sensitivity, presence of HA inhibitors in some samples, and the subjective nature of determining hemagglutination. 7,19 Despite these drawbacks, the HI assay remains the gold-standard for SIV antibody detection. Commercial enzyme-linked immunosorbent assays (ELISA) are also available to detect H1 and H3 SIV antibodies. Results from comparisons of commercial ELISA assays to HI have found correlations ranging from poor to good in terms of sensitivity and specificity. 1,20,26 A limitation for commercial ELISA assays is the lack of antigen diversity in the test. Serum neutralization (SN) is another assay used to measure anti-influenza antibodies. The SN assay measures the ability of nonneutralized virus to induce cytopathic effects in cell cultures. Consequently, the SN assay specifically measures antibodies capable of preventing viral infection. However, the SN assay is not widely used owing to its labor-intensive nature and technical difficulty. A modification of the SN assay to increase its throughput, objectivity, and ease of use has been reported. 14 The objective of the current study was to evaluate a high-throughput serum neutralization (HTSN) assay utilizing subtype-specific antisera to antigenically categorize SIV isolates. This approach is complementary to conventional genetic analysis and provides additional anti-genic information necessary for the characterization of new SIV isolates.
H3N2 Swine influenza virus reference isolates comparison to common cluster type strains.
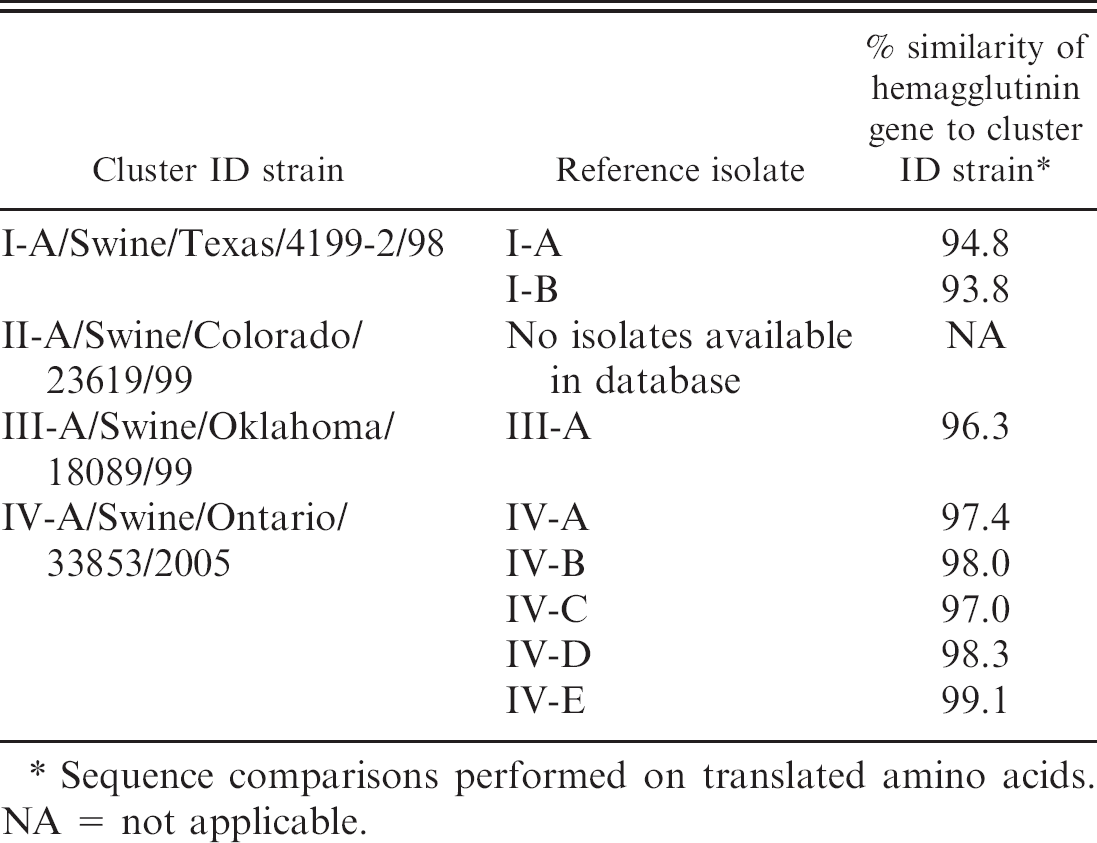
Sequence comparisons performed on translated amino acids. NA = not applicable.
Materials and methods
Viruses
Viruses representing H3N2 SIV circulating in North American swineherds were used in this study. All viruses were isolated from clinical samples submitted to Newport Laboratories (Worthington, MN) for isolation and genetic analysis in 2008 and 2009. Samples originated from North Carolina (n = 13), Minnesota (n = 10), Canada (Manitoba, n = 6), Indiana (n = 5), Iowa (n = 3), Illinois (n = 1), Missouri (n = 1), and Oklahoma (n = 1). The reference viruses were selected based on cluster analysis. Standard reference SIV sequences were used to identify the 4 known clusters of H3N2: Cluster I-A/Swine/Texas/4199-2/98, Cluster II-A/Swine/Colorado/23619/99, Cluster III-A/Swine/Oklahoma/18089/99, and Cluster IV-A/Swine/Ontario/33853/2005. Representative isolates were selected from each cluster and were chosen based on intracluster and intercluster HA sequence variability and according to prevalence in swine. Five viral isolates were selected from cluster IV (labeled as IV-A, IV-B, IV-C, IV-D, and IV-E) as this is the largest and most prominent H3N2 subtype circulating in swine. Two isolates represented cluster I (I-A and I-B) and 1 isolate for cluster III (III-A). Cluster II was not included in the current study as no representatives were identified in the authors' isolate collection. Table 1 shows the cluster reference strains and the percentage similarity of the chosen viral isolates.
Virus isolation was performed in swine testicle (ST) cells grown in Dulbeccos Minimum Essential Medium (DMEM) containing 5% fetal bovine serum at 37°C with 5% CO2. For virus propagation, DMEM without fetal bovine serum was used. Viral titers were determined by quantitative reverse transcription polymerase chain reaction (qRT-PCR) and hemagglutination using 0.5% turkey red blood cells in phosphate buffered saline (PBS). 3,17
Swine influenza virus reference isolates were grown in ST cells to a high titer (1280–2560 HA units) and inactivated using 5 mM binary ethyleneimine for 24 hr at 37°C. Vaccines were formulated using killed virus and Trigen, a a proprietary adjuvant.
Generation of antisera
Three-week-old pigs were obtained from a commercial high-health herd, and their SIV-negative status was confirmed individually by a commercial ELISA b and by HI and SN assays using the 8 reference viruses from the present study (Table 1). 3 Groups of 3 pigs were used for antisera generation. Sera were pooled prior to testing. Pigs were vaccinated on days 0, 7, 14, and 21 with 2 ml of vaccine delivered intramuscularly. Serum was collected on days 0 and 35. Because sera collected from vaccinated pigs had very high HI titers (homologous HI from 1,280 to >20,480), it was diluted from 4- to 16-fold in blood serum from cesarean-derived, colostrum-deprived (CDCD) pigs to achieve homologous HI titers of 1,280. The diluted reference antisera stocks were aliquoted and frozen at −80°C. In addition, negative control antiserum was generated by injecting pigs with ST cell debris and adjuvant. Antiserum generated from the ST cell debris was not diluted.
Serologic assays
Cross-reactivity was determined by HI and HTSN assays. All sera were heat inactivated at 56°C for 30 min prior to use. The HI assay was performed using standard assay procedures as previously described. 3 In brief, sera were first treated with receptor-destroying enzyme c for 24 hr at 37°C. Treated sera were next hemadsorbed with a 20% solution of washed turkey erythrocytes in PBS for 30 min at room temperature. Serial 2-fold dilutions of sera were used in the HI assay (1:10–1:20,480). Hemagglutination inhibition was assessed using 0.5% washed turkey erythrocytes with viral titers of 4–8 HA units/well. Hemagglutination inhibition titer was determined as the reciprocal of the highest dilution that showed complete inhibition of hemagglutination. Hemagglutination inhibition assays were run in duplicate, and results were reported as arithmetic mean titers.
The HTSN assay was developed by modification of a high-throughput Alamar blue assay. 14 In brief, reference antisera were diluted 1:800 in DMEM, and 100 μl was dispensed into 1 column of a 96-well microtiter plate. Ten diluted antiserum samples were run on a single plate (columns 1–10). Columns 11 and 12 contained CDCD pig sera diluted 1:800 in DMEM. A 1:800 dilution of sera was experimentally determined to yield strong homologous and poor heterologous reactions using the reference viruses and antisera in the present study (Table 2). Viruses were grown on ST cells as described above and harvested by a single freeze-thaw cycle. Viruses were diluted in DMEM to 100 median tissue culture infectious dose (TCID50)/100 μl as determined by titration on a monolayer of ST cells. Viral titers were further verified by real-time RT-PCR (threshold cycle [Ct] = 27–30). 17 One hundred microliters of diluted virus was added to wells 1–11 in duplicate rows. The DMEM was added to column 12 to serve as a no virus control. The virus and antiserum mixture was incubated at 37°C for 1 hr, and then 100 μl was transferred to a 1-day-old monolayer (100% confluent) of ST cells. Plates were incubated for 4 days at 37°C with 5% CO2. Cell culture fluids were discarded, and 100 μl of 10% Alamar blue d in PBS was added to all wells of the plates. Plates were incubated for 3 hr at 37°C with 5% CO2, and then fluorescence was measured on a fluorescent plate reader with excitation/emission filters of 544/590nm. e Mean fluorescence for the duplicate wells was determined and used to calculate the SN ratio. 9
Average serum neutralization (SN) ratios of homologous, intracluster, and intercluster reactions following serial serum dilution of 8 reference viruses and corresponding antisera.
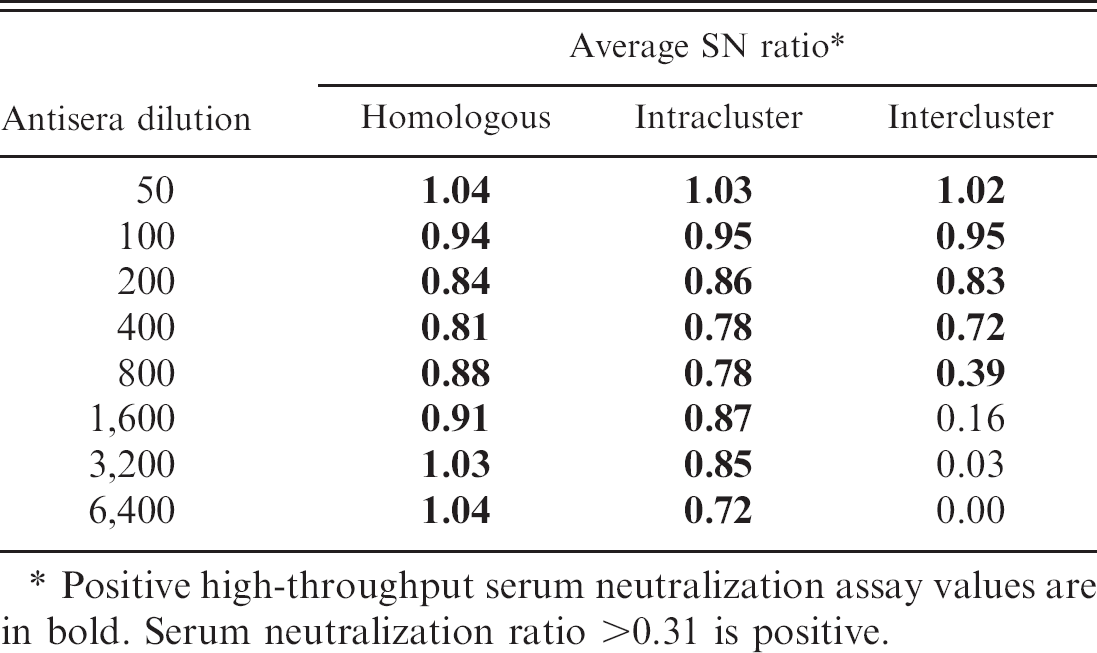
Positive high-throughput serum neutralization assay values are in bold. Serum neutralization ratio >0.31 is positive.

Serum neutralization ratios were calculated for each virus and antiserum combination run in duplicate and reported as a mean SN ratio. The HTSN assays were repeated on 2 separate days, and values were reported as means (Tables 3, 4).
Determination of the optimal antiserum dilution for HTSN assay
A 2-fold dilution series (1:50–1:6,400) of each reference antiserum generated against reference H3N2 subgroup isolates was evaluated against each reference virus to determine the optimal dilution of antiserum for the HTSN assay. Average homologous, intracluster, and intercluster SN ratios were determined for each antiserum dilution using data from all 8 reference viruses (Table 2).
Genetic analysis
Six hundred base pairs of the HA gene (base pairs 199–798) were determined by DNA sequencing. DNA sequences were translated into amino acids and aligned by pairwise cluster analysis using commercial software. f The following SIV subtype reference strains were included in the analysis: H3N2 cluster I, A/Swine/Texas/4199-2/98; H3N2 cluster II, A/Swine/Colorado/23619/99; H3N2 cluster III, A/Swine/Oklahoma/18089/99; and H3N2 cluster IV, A/Swine/Ontario/33853/2005.
Results
Determination of the optimal antiserum dilution for HTSN assay
At antiserum dilutions of 1:50–1:400 SN ratios were similar for homologous, intracluster, and inter-cluster reactions (Table 2). At antiserum dilutions of 1:1,600–1:6,400, homologous and intracluster HTSN ratios were positive but heterologous ratios were negative. Antiserum dilution of 1:800 gave high homologous and intracluster SN ratios (0.88 and 0.78, respectively) but gave a weak positive average intercluster HTSN ratio (0.39). Based on these data, an antiserum dilution of 1:800 was chosen for the HTSN assay, as this value consistently gave positive homologous and intracluster SN ratios and sporadic positive intercluster values.
Hemagglutination inhibition (HI) antibody titers and high-throughput serum neutralization (HTSN) ratio of H3N2 subtype reference isolates.*
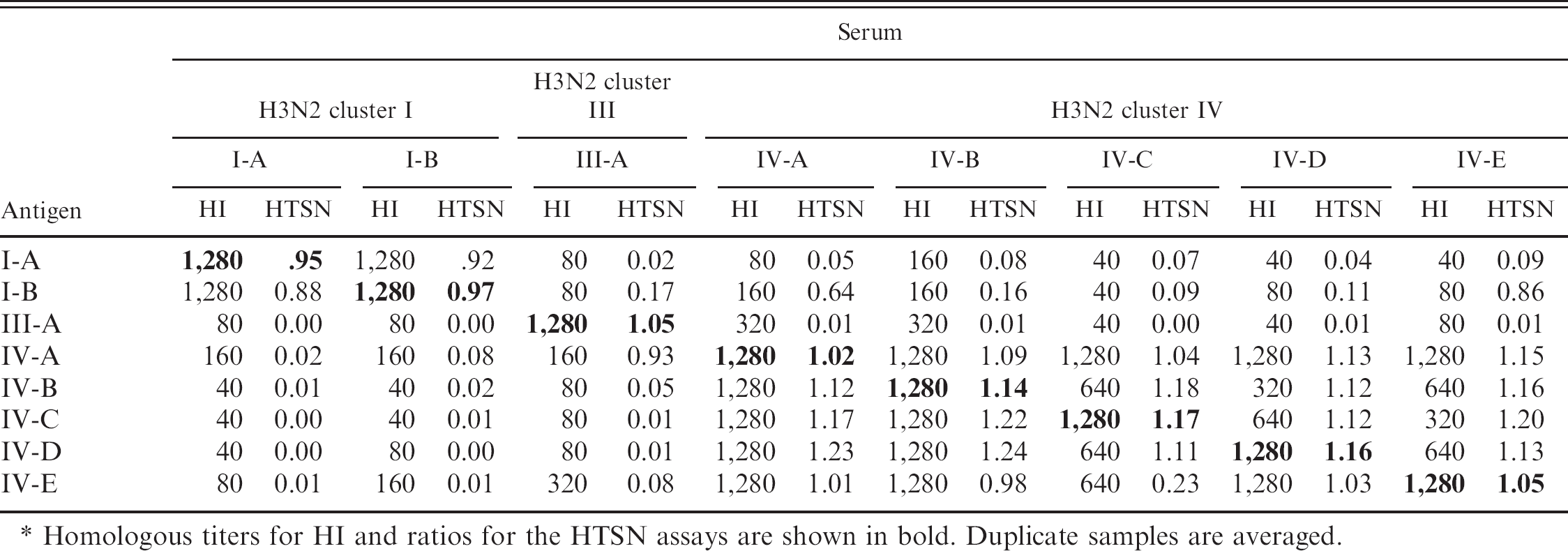
Homologous titers for HI and ratios for the HTSN assays are shown in bold. Duplicate samples are averaged.
High-throughput serum neutralization assay ratios of 40 contemporary H3N2 isolates.
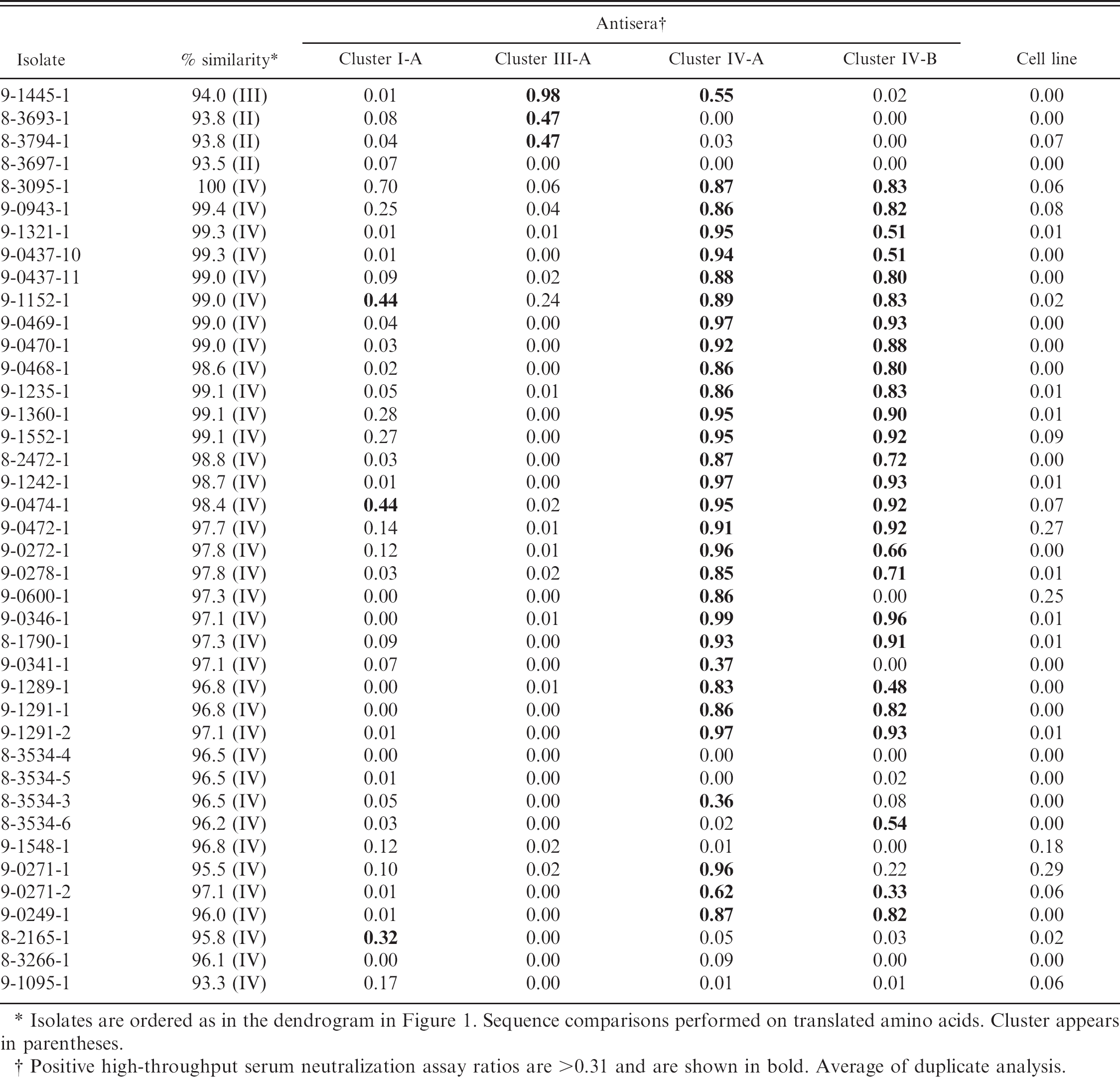
Isolates are ordered as in the dendrogram in Figure 1. Sequence comparisons performed on translated amino acids. Cluster appears in parentheses.
Positive high-throughput serum neutralization assay ratios are >0.31 and are shown in bold. Average of duplicate analysis.
Comparison of HI and HTSN assays using reference viruses and antisera
The H3N2 SIV reference isolates and their corresponding antisera were compared by HI and HTSN assays (Table 3). Intracluster cross-reactivity was strong for all clusters, with HI titers of 320–1280 and HTSN ratios greater than 0.87 for all comparisons except virus IV-E with antiserum IV-C. Inter-cluster cross-reactivity was moderate in the HI assay, with titers ranging from 40 to 320. Conversely, intercluster HTSN ratios were generally low (<0.20), with 1-way cross-reactivity seen between virus I-B and cluster IV-A E antisera and virus IV-A with III-A antiserum. The Pearson correlation coefficient between the HI and HTSN assays was determined using commercial software g to be r = 0.84 with P < 0.0001. Repeatability was calculated based on HTSN assay results for 2 independent runs. Assay repeatability was 86.3%. The HI assay repeatability of 91.1% was calculated from results of 2 independent runs.
HTSN assay analysis of contemporary H3N2 swine influenza virus isolates
Forty H3N2 SIV isolates (Fig. 1) were evaluated in the HTSN assay using antiserum generated against the ST cell line to establish a positive SN ratio threshold (Table 4). Analysis of the data by nonparametric prediction interval with a 97.5% confidence limit established a positive SN ratio threshold of >0.31.
Genetic analysis of HA sequences categorized 35 of the 40 viruses as cluster IV H3N2. These viruses had amino acid sequences >95% similar to the reference A/Swine/Ontario/2005. One cluster III isolate, 9-1445-1, was identified. In addition, 4 isolates (8-3693-1, 8-3794-1, 8-3697-1, 9-1095-1) failed to show significant homology (<94% homology) with reference isolates.
Thirty of the 35 cluster IV isolates (86%) were neutralized by at least 1 cluster IV antiserum used in the HTSN assay. Cluster IV isolates not neutralized by cluster IV antisera were less than 97% similar to the reference A/Swine/Ontario/2005. Two of the cluster IV isolates not neutralized by cluster IV antisera, 8-3534-4 and 8-3534-5, were 99.7–100% similar to 2 isolates (8-3534-3 and 8-3534-6) that were neutralized by cluster IV antisera. These 4 isolates were further analyzed by HI and had highest titers with cluster IV antisera (HI titers 80–160, Table 5).
Cluster I antiserum neutralized only 3 cluster IV isolates (8.6%). Cluster III antiserum was unable to neutralize any cluster IV isolates. The sole cluster III isolate, 9-1445-1, was neutralized by cluster III antisera and, to a lesser extent, by one of the cluster IV sera. Also interesting was a group of isolates (8-3693-1, 8-3794-1, and 8-3697-1) with only 93% similarity to previously sequenced SIV. The reference virus with the highest similarity to this group belonged to cluster II H3N2. Isolates 8-3693-1, 8-3794-1, and 8-3697-1 showed greater than 99% amino acid similarity to each other. Isolates 8-3794-1 and 8-3693-1 were weakly neutralized by cluster III sera. Isolate 8-3697-1 was not neutralized by any antisera used in the HTSN assay. The 3 isolates originated from Minnesota, Iowa, and North Carolina, respectively, suggesting a broad distribution.
One additional isolate, 9-1095-1, failed to fall within established clusters by sequence analysis and was not neutralized by any reference antisera used in the HTSN assay. This isolate had only 92% amino acid similarity and was most homologous to previously sequenced cluster IV H3N2 SIV.
Discussion
The remarkable degree of constantly changing genetic diversity makes control of SIV infection challenging. 8 The gene encoding the HA protein is typically partially sequenced, and particular isolates are chosen for vaccine production such that the genetic diversity present at a site is represented by the isolates used in the custom vaccine. This approach assumes that genetically similar isolates are antigenically similar.

Amino acid dendrogram of the 40 contemporary H3N2 Swine influenza virus isolates used in the present study. Commonly used reference strains A/Swine/Texas/98 (cluster I), A/Swine/Oklahoma/99 (cluster III), A/Swine/Colorado/99 (cluster II), and A/Swine/Ontario/2005 (cluster IV) are included.
Hemagglutination inhibition (HI) antibody titers and high-throughput serum neutralization (HTSN) assay ratio of isolates 8-3534-3, 8-3534-4, 8-3534-5, and 8-3534-6.

The SN assay measures the ability of antibodies in a serum sample to neutralize a particular virus. The assay described herein has been modified to categorize SIV isolates using reference SIV antisera generated using viruses representing the various subtypes of SIV. While similar in principle, the HI assay differs significantly from SN. The HI assay measures the ability of antibodies to prevent virus-mediated red blood cell agglutination, which is mediated by the HA protein. The SN assay measures virus neutralization and consequently can detect viral-antibody interactions, which can involve a number of proteins and can be thought of as providing a more global antigenic perspective. 6,21 This is especially important given the recent finding of Eurasian-lineage SIV matrix and neuraminidase genes in the human pandemic H1N1 virus isolated from both humans and pigs. 4 In principle, the HTSN assay can measure the contribution of neutralizing antibodies against the gene variants that may be missed by the HI assay.
The HI and HTSN assays were compared using isolates representing clusters I, III, and IV of H3N2 and were in good agreement (r = 0.84). Strong homologous and intracluster reactions were typically seen by both HI and HTSN assays (HI titers 320–1,280; HTSN ratios 0.88–1.24). Only 1 intracluster HTSN ratio was negative (virus IV-E with antiserum IV-C; Table 3). Not surprising, reference viruses IV-E and IV-C were the most divergent of the 5 cluster IV reference isolates used (97.1% similarity; Fig. 1). Despite the strong correlation between the HI and HTSN assays, a lack of HI titers did not always equate with a lack of neutralizing titers. This latter situation could be explained by the presence of neutralizing epitopes on proteins other than HA. 6 Investigation of cross-reaction afforded to pigs that were previously infected with H1N1 or H3N2 and then challenged by a European H1N2 isolate suggested that internal influenza proteins may play a significant role in protection. 21 In addition, presence of HI titers did not ensure positive HTSN ratios. The HI assay measures abilities of antibodies to bind viral HA, and results from the HTSN assay suggest that this is not necessarily indicative of virus neutralization. Previous studies have also noted inconsistent serologic cross-reactivity with modern H1 influenza. 6,22
Despite forming a relatively cohesive cluster with approximately 5% amino acid differences, antigenic differences clearly exist among cluster IV H3N2 isolates. Four isolates were not neutralized by either cluster IV antiserum. In addition, genetic analysis of these 4 isolates revealed 96.1–96.8% amino acid similarity to the cluster IV reference strain A/Swine/Ontario/33853/2005. Taken together, these results suggest that antigenic drift has occurred in that cluster.
Further evidence of antigenic differences within cluster IV is seen with isolates 8-3534-3, 8-3534-4, 8-3534-5, and 8-3545-6. These isolates all originated from the same farm and diagnostic submission and were 99.7–100% similar to each other and 96.2-96.5% similar to A/Swine/Ontario/33853/2005 at the amino acid level as determined by partial HA gene sequencing. Isolates 8-3534-4 and 8-3534-5 were not neutralized by any antisera in the HTSN assay, while 8-3534-3 and 8-3534-6 were neutralized by (albeit different) H3N2 cluster IV antiserum with relatively low SN ratios of 0.36 and 0.54. These 4 isolates were further characterized by HI, and all isolates had titers of 80–160 with cluster IV antisera. These HI titers are lower than observed for cluster IV reference isolates (HI 320–1280, mean intracluster HI titer 992; Table 3) and, in conjunction with the HTSN results, suggest antigenic drift has occurred within cluster IV. Sequencing of other viral genes is planned to investigate the source of antigenic variation seen in the HTSN assay with these isolates.
The cluster of isolates composed of 8-3693-1, 8-3794-1, and 8-3697-1 appears relatively antigenically distinct from clusters I, III, and IV. It would be interesting to test cluster II antiserum for its ability to neutralize these isolates. Given their genetic and antigenic uniqueness, these isolates could be considered a potential newly emerging cluster of H3N2. Additional isolates with high genetic similarity to this cluster have recently been isolated in the authors' laboratory from pigs displaying clinical signs of SIV.
Overall, genetic analysis appears to be a good predictor of antigenic type. Isolates with high genetic similarity tend to react similarly in the HTSN assay. In the present work, 31 of the 35 cluster IV isolates analyzed in the HTSN assay were neutralized by at least 1 cluster IV antiserum. Despite the large majority of cluster IV isolates neutralized by cluster IV antisera in the HTSN assay, 4 exceptions were observed. This finding suggests that genetic analysis alone is an imperfect predictor of antigenic sameness. Similarly, others have noted that large genetic differences do not always result in a lack of antigenic cross-reaction. 5 The HTSN assay developed as part of the current study is a fast, relatively simple method to qualitatively assess antigenic differences between isolates and offers an additional tool for SIV characterization. The HTSN assay could also be useful for identifying broadly cross-reactive SIV isolates for vaccine candidates. In addition, the HTSN assay could be used for surveillance purposes. An example of this would be to screen new viral isolates in an HTSN assay incorporating vaccine antiserum. Future work will seek to establish the relationship between the SN ratio and protection of swine from SIV.
Acknowledgements
The authors would like to thank Jen Iverson and Amy Shirbroun for technical assistance.
Footnotes
a.
Newport Laboratories, Worthington, MN.
b.
HerdChek SIV H1N1 and SIV H3N2, IDEXX Laboratories, Westbrook, ME.
c.
Accurate Chemical Corp., Westbury, NY.
d.
Life Technologies Corp., Carlsbad, CA.
e.
Fluoroskan Ascent®, Thermo Electron Corporation, Waltham, MA.
f.
Applied Maths, Austin, TX.
g.
GraphPad Prism®, GraphPad Software Inc., La Jolla, CA.
