Abstract
To eliminate cytotoxic effects of colostrum on cells, a modified virus neutralization test (VNT) for the detection of
The level of antibodies in colostrum can be measured directly or measured in calf serum following intake of colostrum. Specific antibodies can be detected by enzyme-linked immunosorbent assay (ELISA) or through use of a virus neutralization test (VNT). Total bovine class-specific immunoglobulins or the quantities of antigen-specific immunoglobulin can be determined using ELISA techniques. However, the biologically relevant activity of virus-neutralizing antibodies can be determined only by application of a VNT. The primary difficulty of measuring specific neutralization antibodies by VNT is the cytotoxic effects of colostrum on cells, stemming from contamination of the colostrum with bacteria3,4,14,15,18 and the composition and the density of fresh colostrum. Colostrum is also 1 of the earliest potential sources of exposure for dairy calves to infectious agents,3,5,10,15 and it has been suggested that the presence of bacteria in the calf ’s small intestine at the time of colostrum consumption can indirectly harm calf health by interfering with the systemic absorption of immunoglobulin molecules, contributing to the failure of passive transfer of maternal antibodies.8,13 One such infectious agent is
Blood and colostrum samples were collected from 56 heifers and cows after calving from 6 independent dairy farms where vaccination against BVDV was not conducted. Samples were stored at 4°C and transported to the Croatian Veterinary Institute (Zagreb, Croatia), where the blood samples were centrifuged at 200 ×
Prior to carrying out the testing experiment, Madin–Darby bovine kidney (MDBK) tissue culture cells and a fetal bovine serum (FBS) supplement were tested for the presence of BVDV RNAs using reverse transcription polymerase chain reaction (RT-PCR) as described previously. 16 The FBS supplement was also tested for BVDV-specific antibodies by ELISA. Both MDBK cells and the serum supplement tested negative for BVDV, and bovine serum tested negative for BVDV-specific antibodies. The mVNT was performed using 96-well plates. a A suspension of 100 µl of MDBK cells (300,000/ml) was placed in each well, and the plate was incubated at 37°C with 5% CO2 for 3 hr. The MDBK cells were grown in Dulbecco modified Eagle medium (DMEM) b supplemented with 10% FBS. c In a second 96-well plate, DMEM was used in 2-fold serial dilutions of either fresh colostrum or serum in 100 µl of total volume. The dilution series ranged from 1:2 to 1:4,096 and were prepared in quadruplicate. Fifty microliters containing 102 TCID50/50 µl of reference strain NADL d were added to each well. A reference strain for BVDV genotype 1 was used, as it is the only genotype found to date in Croatia. 2 The serial dilutions containing virus were placed in a CO2 incubator at 37°C for 1 hr. After incubation, the DMEM was removed from the first plate containing MDBK cells, and the serial dilutions of serum or colostrum in virus suspensions from the second plate were transferred into the wells of the first plate. The plates were incubated at 37°C for 1 hr, followed by removal of the liquid suspension, and the wells were washed 5 times with DMEM. After washing, 200 µl of medium plus 10% FBS with antibiotics e was added to each well, and the plates were incubated at 37°C for 4 days.
After 4 days, the medium was removed, and the wells were rinsed once with 250 µl of phosphate buffered saline (PBS) f and once with 250 µl of 80% acetone. g Cells in each well were fixed with 250 µl of 80% acetone g for 20 min. After the cells were dried, 50 µl of BVDV-specific monoclonal antibodies h diluted in distilled water (1 mg/ml) were added per well and incubated for 30 min. After incubation, the wells were washed 3 times with 250 µl of PBS, f and 50 µl of antimouse immunoglobulin antibodies conjugated with fluorescein isothiocyanate i were then added to each well and the samples incubated for 30 min. After incubation, wells were washed 3 times with PBS and examined by fluorescent microscopy. Wells with any cells displaying green fluorescence were considered to be negative for neutralizing antibodies, whereas wells without green-fluorescing cells were considered to be positive for neutralizing antibodies.
To validate the mVNT procedure, all sera samples were also tested in both the mVNT and the World Organization for Animal Health (OIE)–recommended VNT (standard VNT). 19 Positive serum, negative serum, virus control, control cells, and the back titration of the NADL test virus were included in each run. The acceptable range of virus was 50–400 TCID50 per 50 µl. 19 The test was considered valid only if all control sera and the back titration of virus gave results within the acceptable ranges and if cytotoxic effects were not observed in wells containing control cells.
Two-fold dilutions of the samples were represented as a log (log2) scale. The titer was expressed as 50% endpoint of antibodies (D50), which was calculated for each sample by the Spearman–Kärber formula.9,12 Titers greater than 0.15 were considered positive. Correlations between the standard VNT and mVNT antibody titers in sera samples and antibody titers obtained in sera and colostrum samples by mVNT were determined by correlation analysis using commercial software. j
Forty sera samples out of a total of 56 tested were identified as positive for BVDV-specific antibodies by both the mVNT and standard VNT. Fourteen sera samples were determined to be negative by both tests. Two sera samples testing positive by mVNT exhibited cytotoxic effects on cells when tested by the standard VNT. Sensitivity and specificity of the mVNT compared to standard VNT were 100%. The results of the validation of the mVNT are shown in Figure 1. Antibody titers in sera samples (measured as D50) determined by the standard VNT showed a good correlation with titers provided by the mVNT (
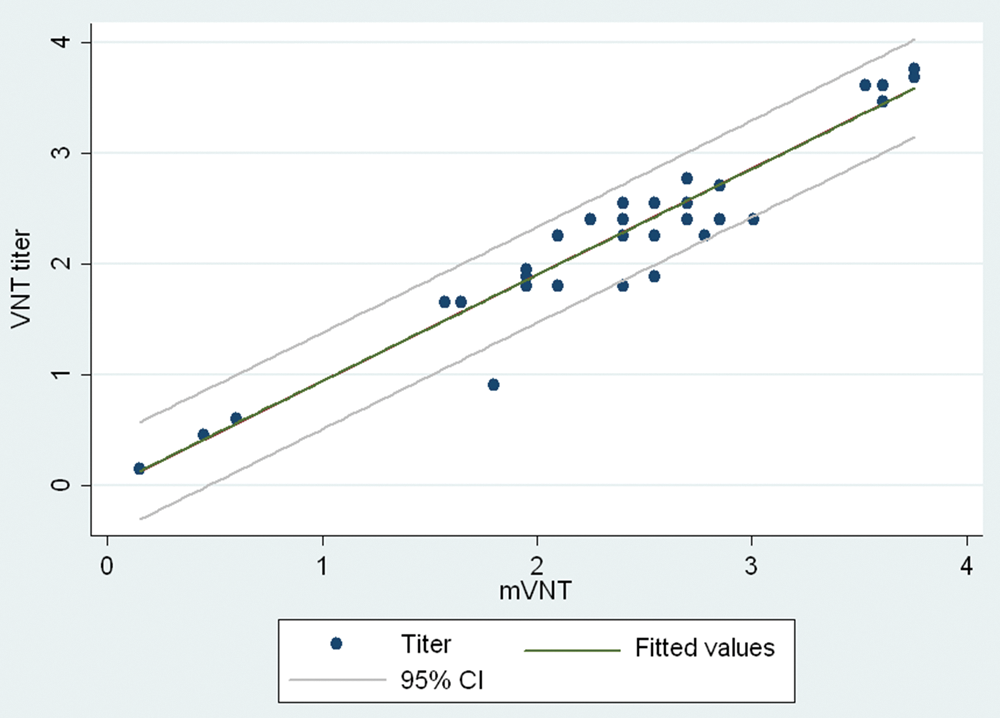
Correlation between titers in sera samples (log2D50) measured by the standard virus neutralization test (VNT) and the modified (m)VNT. CI = confidence interval.
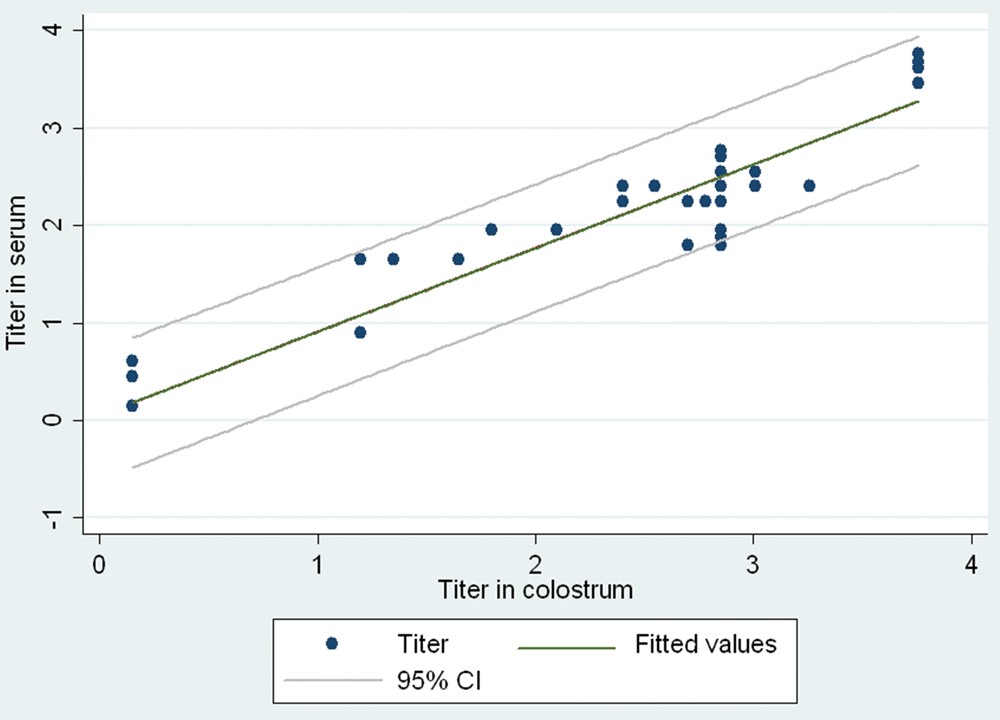
Correlation between the modified virus neutralization test titers in sera and colostrum samples (log2D50). CI = confidence interval.
The primary difficulty of using VNT for specific antibody detection in colostrum is the cytotoxic effects of the sample on the indicator cells. In the current study, an mVNT was developed to eliminate these cytotoxic effects. First, to prevent detachment of indicator cells from the surface of the wells, a higher density of MDBK cells than is employed in the standard VNT was used in the mVNT, and the cells were incubated for 3 hr. To eliminate nonspecific reactions caused by complement, the colostrum was heat-inactivated. Previous studies have found that inactivation does not affect the levels of antibodies. 6 To reduce the negative effects of colostrum on cells, the colostrum–virus mixture was removed after 1 hr of incubation, and cells were washed 5 times with medium to minimize the contact between any toxic substances present in the colostrum and the cells. To determine if the incubation and washing procedures affected the ability of the test virus to attach to the indicator cells, sera samples were tested in parallel with the standard VNT and mVNT. Such additional tests also allowed an evaluation of the mVNT itself compared to the standard VNT. When compared, the correlation between both tests for sera samples was 98%. In 2 sera samples, cytotoxic effect was observed in the standard VNT but not by mVNT, suggesting that the modifications to the standard procedures eliminated the cytotoxicity. In 7 colostrum samples in which specific antibodies were detected, the antibody titers was equal to or lower than in sera samples taken from the same cow (Table 1). Furthermore, antibodies were not detected in 4 colostrum samples from cows with detected specific antibodies in sera samples (Table 1). This suggests there may be some threshold level of serum antibodies that is required before immunoglobulins are successfully transferred into colostrum. However, further studies are required to establish that possibility, especially because large volumes of milk created by high milk production cows affect the dilution effects as well.
Titers of

In conclusion, a modification of the standard VNT for detection of BVDV-specific antibodies in colostrum was developed, and the results using this modified test exhibited a good correlation with the standard OIE-recommended VNT. The modified method will provide simple and accurate detection of specific antibodies to a variety of pathogens in fresh colostrum. When applied to colostrum and replacement colostrum products, the mVNT can improve protection against infectious diseases, such that replacement colostrum products with high antibody titers can be easily selected.
Footnotes
a.
96-well plates for cell culture, Nunc A/S, Roskilde, Denmark.
b.
Dulbecco’s modified Eagle’s medium, Sigma-Aldrich, St. Louis, MO.
c.
Fetal bovine serum, PAA Laboratories GmbH, Pashing, Austria.
d.
NADL, Friedrich Loeffler Institute, Insel Riems, Germany.
e.
Zellshield, Minerva Biolabs GmbH, Berlin, Germany.
f.
Phosphate buffered saline (pH 7.4), Sigma-Aldrich, St. Louis, MO.
g.
Acetone, Kemika d.d., Zagreb, Croatia.
h.
BVDV-specific mouse monoclonal antibodies, Veterinary Laboratory Agency, Weybridge, UK.
i.
Fluorescein isothiocyanate conjugate antimouse antibodies, Sigma-Aldrich, St. Louis, MO.
j.
STATA 10, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grant no. 048-0481186-1183 from the Ministry of Science, Education and Sports, Republic of Croatia.
