Abstract
A large, firm, multi-cystic mammary gland mass grew slowly over 4 y in a 12-y-old, female Finn–Shetland cross sheep. A diagnosis of epithelial malignancy was suspected following fine-needle aspiration cytology at 30 mo after initial observation. The sheep was euthanized when the flock was downsized 18 mo later. A field postmortem examination revealed a large mammary mass, but an absence of metastases to internal organs. Imprint cytology of the mammary tissue supported a benign proliferative process. Histologically, mammary tissue was obliterated by cystic, tubular, and papillary adenomatous arrangements of mammary epithelium, with an anaplastic component, consistent with mammary carcinoma arising in an adenoma. IHC showed strong nuclear positivity to the antibody against progesterone receptor and minimal positivity to the antibody against estrogen receptor alpha expression. Intrinsic subtyping for basal or luminal epithelial origin was attempted through adaptation of companion animal IHC classification panels; high- and low-molecular-weight cytokeratins (CK5, CK8, CK18) failed to stain, but p63 expression for basal epithelium was positive.
Keywords
Limited numbers of species-specific neoplastic conditions are reported in sheep, including lymphoma, nasal adenocarcinoma, intestinal adenocarcinoma, and pulmonary carcinoma.5,14,21,25 A review of 673 tumors from sheep in South Africa failed to document mammary neoplasia. 3 Descriptions of mammary gland neoplasia in sheep include a fibroadenoma,9,15 an adenoma, 1 a hemangiopapilloma of the gland cistern, 28 and a low-grade carcinoma and adenocarcinoma.19,28 All 5 reported ovine cases had benign to low-grade progression. Neoplasia in goats has been reviewed, and 7 of 102 tumors arose in the udder 17 ; all 7 tumors were mammary adenocarcinomas, and 2 of 2 animals examined had metastatic disease, suggesting aggressive behavior in goats. 17
A ewe developed a slow-growing, expansile mass in the left mammary gland. The ewe was one of a small farm flock whose fleeces were harvested yearly for wool. This ewe had not been bred or had any offspring for the past 5 y. The ewe was deemed healthy. The flock was maintained on ~1 ha (2 acres) of pasture year-round, with a diet supplemented by corn and hay during the colder months. Water and mineral supplement (Sheep mineral with ZinPro; Southern States) were available free choice. The mass distorted the entire gland and produced an undulating skin surface given its multi-lobular nature (Fig. 1).
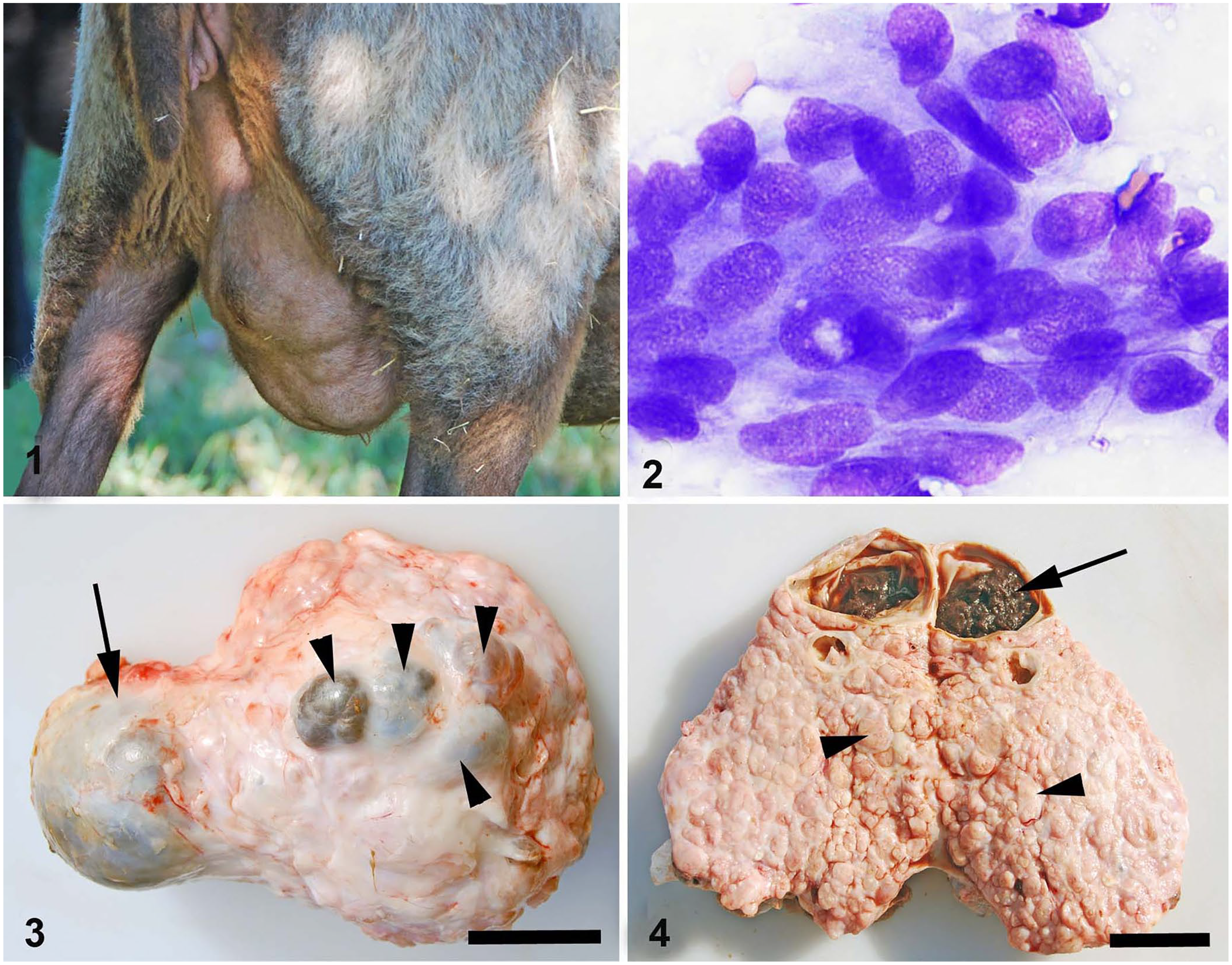
Mammary neoplasia in a ewe.
Thirty months following initial detection of the mass during annual shearing, the mass was aspirated. Air-dried smears were stained with a modified Wright–Giemsa stain. Cytologic findings included multiple disarrayed clumps of atypical polygonal-to-cuboidal epithelial cells with indistinct cytoplasmic borders, oval slightly eccentric nuclei with 1.5× anisokaryosis, a variable 1:1 nuclear-cytoplasmic ratio, course granular chromatin, occasional single and multiple nucleoli, and basophilic cytoplasm with occasional eosinophilic cytoplasmic inclusions as a result of cellular invagination (Fig. 2). The background contained erythrocytes, leukocytes, platelets, and pale basophilic fluid. The cytologic diagnosis was malignant epithelial neoplasia likely arising from the mammary gland epithelium, with the qualifier that cytologic criteria of malignancy in mammary neoplasms do not correlate strongly with biologic behavior. The recommendation was that histology be pursued to further characterize the mass and confirm the diagnosis.
Eighteen months following the initial cytologic examination, the animal was euthanized by captive bolt (Type KS; Karl Schermer) and exsanguinated from the carotid artery. A field postmortem examination was performed. The carcass was in excellent body condition, with excellent body fat stores and hydration status; there was slight roughening of the right femoral head, interpreted as osteoarthritis. The left mammary gland measured 27 × 14.5 × 10 cm and weighed 1,346 g without skin; the right gland weighed 784 g. The affected gland was enlarged, firm, multinodular, and could not be milked. Two large fluctuant cysts were present, as well as 4 small coalescing fluid-filled nodules of 2 × 2 × 0.8 cm to 10 × 10 × 3 cm that compressed tissue across the midline to the less affected gland (Fig. 3). Upon sectioning, the mass was nodular and tan, and the nodules and cysts were separated by firm white fibrous tissue septa (Fig. 4). The largest dorsal cyst contained ~50 mL of brown fluid admixed with brown clotted material (Fig. 4). Supramammary lymph nodes were grossly normal, but the internal iliac lymph nodes contained multiple light-tan foci throughout the cortex. A 0.3 × 0.3 cm pale-red unencapsulated nodule was identified in the medulla of the left adrenal gland.
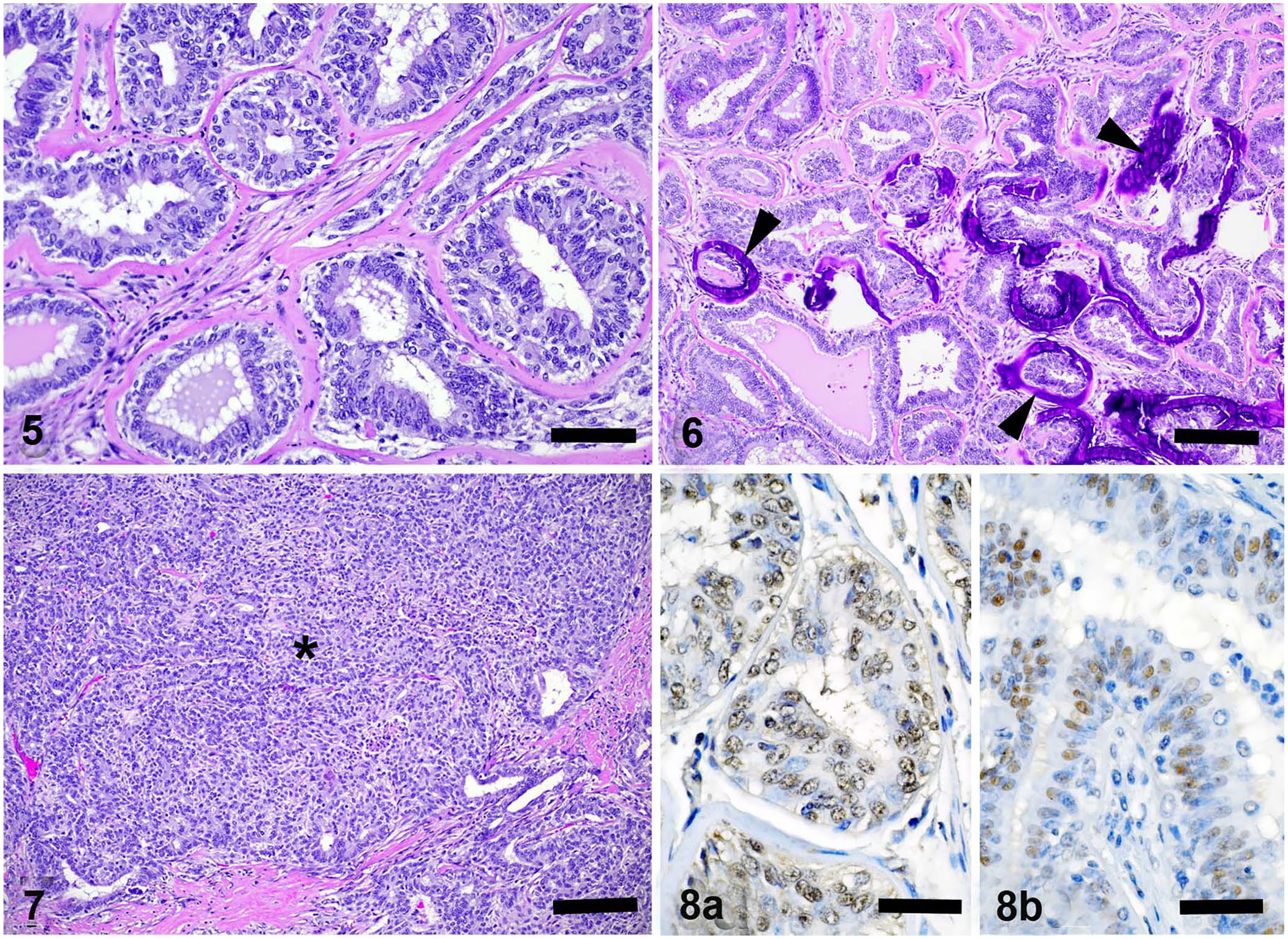
Samples of liver, lung, heart, spleen, kidney, lymph nodes, large intestine, small intestine, adrenal, uterus, ovaries, mammary mass, and urinary bladder were collected, fixed for 24 h in 10% neutral-buffered formalin, trimmed, processed routinely, and slides stained with hematoxylin and eosin. On histologic examination, the largest cystic structure was lined by pseudostratified epithelium, and contained groups of red blood cells enmeshed in fibrin, serum, and acicular clefts. Surrounding mammary gland lobules were encapsulated, focally compressed by the cyst, and varied between multiple nodules and multiple cysts obliterating normal tissue architecture through expansion (Fig. 5). Expansile lobules comprising 95% of the encapsulated mass contained benign proliferative epithelium arranged in tubular and papillary patterns, separated by abundant fibrous connective tissue septa, and were interpreted as adenoma (Fig. 5). Luminal sizes varied as did the presence of luminal fluid. Typically, the lining epithelium was one-to-several layers, vacuolated, with basal vesicular nuclei, occasional nucleoli, and apical blebbing. Additionally, the lobules were characterized by scattered apoptotic cells, variable central necrosis and hemorrhage, scattered foci of squamous differentiation with mineralization, hyalinization and less commonly mineralization of basement membranes, and interstitial inflammation composed of lymphocytes and plasma cells (Fig. 6). The mitotic count in the areas of malignant transformation, representing <5% of lobules, was 1 mitosis per 10 high-power fields (2.37 mm2). In rare lobules, there was a solid arrangement of neoplastic cells, with mild peripheral invasion with or without desmoplasia, consistent with a histologic diagnosis of early carcinoma arising in an adenoma (Fig. 7).
Left mammary mass in a ewe.
Other histologic lesions included an unencapsulated left adrenal medullary nodule composed of packets of epithelial cells, rimmed by fine fibrovascular stroma, confirmed as a medullary (neuroendocrine) tumor. Additional findings included multiple ovarian follicles, small ventral cyst-like structures, and 3 raised nodules within the left ovary consistent with ovarian follicles and corpora lutea, and lymphoid hyperplasia within the cortex of the internal iliac lymph nodes. Multiple firm tan-to-brown nodules throughout all lung lobes, raised tan nodules throughout all liver lobes, and yellow gritty nodules within and adjacent to the mesenteric lymph nodes were all consistent with eosinophilic granulomas, some of which contained larval nematodes.
Sections of the mammary gland mass were submitted for IHC for estrogen receptor–alpha (ERα) and progesterone receptor (PR) to the University of Tennessee Histology Laboratory (Knoxville, TN). Primary antisera were rabbit polyclonal antibodies against ERα (ER-α clone H-184; Santa Cruz Biotechnology) and PR (PR clone C-19; Santa Cruz Biotechnology), incubated for 30 min at 1:50 dilutions, using canine and ovine positive and negative controls. PR expression was strong in the ovarian tissue of the dog (system control) and less intense in the sheep ovary (species control), and showed minimal but diffuse nuclear immunoreactivity to the antibody against PR throughout the well-differentiated tubular regions of the mass (Fig. 8a). In the more solid region of the lobule (Fig. 7), nuclear immunoreactivity against PR antibody was reduced. ERα expression was strong in the uterine tissue of the dog (system control) and intense in the ovarian tissue of the sheep (species control), and showed regional, moderate-intensity nuclear immunoreactivity against ERα antibody in ducts and tubular regions within the mammary mass (Fig. 8b). Scattered smooth muscle nuclei were also positive.
Additionally, sections of well-differentiated and more anaplastic regions of the mammary gland mass (without normal mammary tissue) were submitted for IHC for CK5 and CK8/18 (Vector), calponin (Dako), and p63 (Santa Cruz Biotechnology) to the Indiana Animal Disease Diagnostic Laboratory (Purdue University, West Lafayette, IN). Ovine positive controls included thymic Hassall corpuscles, small intestinal epithelium, vascular smooth muscle, and thymic lymphocytes, respectively. Antibody expression was absent for all of the cytokeratins tested. Of the basal cell markers, p63 showed strong nuclear positivity in basal epithelium of ductular, tubular, and more solid areas of the mass. Calponin showed strong positivity in the cytoplasm of the smooth muscle of the vascular walls, and in spindle-shaped cells within the stroma between mammary epithelial lobules.
Limited numbers of neoplasms have been reported in sheep in the veterinary literature.5,14,21 Reproductive tissue tumors are of low incidence; those reported include uterine leiomyosarcoma, 29 ovarian granulosa tumor, 12 and mammary gland tumors1,15,19; the mammary masses were reported to have low-grade progression. This is similar to our case, which was present for at least 4 y without noticeable ill effect. Cytologic aspirates mid-course suggested a possible malignant phenotype, but the ultimate biologic behavior was more compatible with low-grade clinical progression given that the ewe was alive without ill effect 18 mo later. The postmortem findings, lack of metastases, and generally well-differentiated mammary epithelium arranged in tubular and ductular patterns within lobules, all within an encapsulated mass in the left mammary gland, supported adenomatous and a variably cystic process, with lobules of more solid phenotype, which we diagnosed as carcinoma arising in an adenoma. At 4 y since inception, a few lobules within the mass had more solid epithelial cell arrangements, invasive growth with only slightly more obvious mitotic activity, and reduced PR expression, which likely represent a more malignant phenotype indicative of localized early malignant transformation. We felt that the diagnosis of carcinoma developing in an adenoma best reflected the morphology and phenotype of the neoplasm. 13 The small number of reported ovine mammary tumors and the absence of aggressive behaviors, even in those named as carcinomas or adenocarcinomas,8,19 may suggest that mammary masses typically have a slow clinical progression in sheep.
Causes of mammary gland neoplasia are varied and species dependent. In some species, the occurrence is so infrequent that risk factors and etiologic agents have not been determined. In general, ruminants have only sporadic mammary neoplasia, with a reported incidence in sheep of 7 in 3,693 (0.2%).13,28 In bitches, reaching the first heat prior to ovariohysterectomy increases the risk of development of mammary neoplasia. 27 Ovariohysterectomy before the first heat cycle is preventive; that protective response declines after 1 y of age. 27 This finding strongly supports a hormonal basis for mammary neoplasia in dogs and cats. 23 In ruminants, the role of hormones in stimulating proliferation and transformation is undetermined and speculative; ruminants typically have shorter lifespans and are production animals raised through multiple heat cycles, parturitions, and lactations, and with low mammary neoplasia outcomes. Diet is thought to play a role in some species, including humans; the high-fiber, low-fat diet eaten by sheep and other ruminants may diminish risk. Rarely, mammary tumors result from infection with a retrovirus, such as in mice infected with mouse mammary carcinoma virus. 10 Sheep are particularly prone to retrovirus-induced neoplastic conditions, albeit a link to mammary neoplasia has not been determined. 21
Hormone imbalances underly many proliferative mammary lesions, primarily from serum excesses, altered biosynthesis, altered metabolism, and/or changes in hormone receptor presence.20,22,23 Imbalances may result from endogenous or exogenous sources. The ewe of our case had ovarian nodules present at the time of postmortem, confirmed to be corpora lutea on histology. The contribution of corpora lutea to the development of this mammary mass, if any, is unknown. Variability in gene expression for hormones, such as estrogen (ESR1), has been associated with mammary tumor development in dogs 7 and cats. 11
Exogenous sources of hormone, linked to mammary tumors, include estrogen-based contraceptives in dogs 26 and megestrol acetate in captive exotic felids. 18 In livestock, as in people, the influence of exogenous sources of hormone, such as phytoestrogen-containing forages, have been postulated to have estrogenic effects and potentially contribute to mammary tumor development.6,24 In our case, clover was not a significant part of the forage on the farm.
We performed IHC for 2 hormone receptors (ERα and PR) on the basis of speculation that these hormones might play a role in proliferation of mammary epithelium in certain circumstances, such as retained corpora lutea and/or multiple reproductive cycles. The role of these receptors in the development of benign and malignant mammary neoplasia has been established in dogs and rabbits, in which receptors are often expressed in benign forms and lost as cells undergo malignant transformation.4,16,20,23 Much less is known about this relationship in sheep because the number of reported neoplasia cases is so few. In a 3-mo-old lamb with a mammary fibroadenoma, estrogen and progesterone receptors were negative. 15 This might be related to the young age and pre-pubertal state of this animal, the benign nature, and/or the mixed cellular proliferation in this type of mass. An additional report of a lamb with a mammary fibroadenoma showed >50% positivity by IHC for estrogen receptor but not progesterone receptor, and plasma estrogen was elevated. 9
Mammary gland would have been a more ideal IHC control tissue for our study, but the canine control used stained appropriately, and conclusions could still be drawn. In our case, there was diffuse, albeit mild, nuclear immunoreactivity against PR antibody with slight reduction in intensity in areas with more solid cellular arrangements. This finding of immunoreactivity against PR may be related to the age of the ewe and the presence of corpora lutea. Additionally, unlike the positivity for ER determined in lambs, our geriatric ewe had only multifocal regions of positive nuclei within pre-existent ducts and foci of proliferation within areas of the sampled mass (<5%).
The total number of ovine mammary neoplasia cases is small, and the total number of cases assessed by IHC even smaller, and hence the associations are only speculative. Our IHC results, in addition to those below, can be used to further subtype mammary tumors, based on criteria utilized in humans and introduced with less success in companion animals. 13 The molecular classification of canine and feline mammary carcinomas is possible given that the histologic and IHC technology exists for those species. Cytokeratins and myoepithelial cell markers can be used to determine if cells are of luminal or basal and/or myoepithelial cell origin. Luminal epithelial cells are distinguished by low-molecular-weight cytokeratins (CK7, CK8, CK18, CK19); basal cells express high-molecular-weight cytokeratins (CK5, CK6, CK14). Because basal and/or myoepithelial cells have other functions, they express other markers, which can include alpha–smooth muscle actin, calponin, vimentin, p63, and others. In humans, mammary neoplasms are classified into 4 clearly recognized intrinsic subtypes, which are based on prognostic outcomes: luminal A, luminal B, basal-like, and erb-B2 overexpressing. 13 Based on IHC, each of these subtypes has surrogate definitions based on expression of additional proliferation markers, hormone receptors, or tyrosine kinase receptors (Ki67, ER, PR, and/or HER 2). Applying IHC panels to canine mammary tumors has not always been successful given that the panels cannot always distinguish between the expression of the epithelial and myoepithelial components, which are a major portion of the neoplasms in dogs. 13 The results from the ovine-specific IHC available at both University laboratories that we used was limited, or produced negative results despite appropriate positive controls, and hence did not allow for complete characterization of this solitary neoplasm into this “human adapted to animal classification” scheme. 13
There are always issues when adapting IHC for an unusual species. The following reagents were used in our case; cytokeratins (CK8, CK18, CK5), p63, calponin, ERα, and PR. Erb-B2, HER2, and Ki67 were not utilized. Expression for markers of myoepithelium (p63) were positive, and that could be interpreted as representing basal or myoepithelial origin in our case. Calponin expression was also noted, but only in stroma and not in the epithelial cells of the neoplasm.
With regard to the surrogate definitions for the each of the subtypes, our additional IHC results give us some information, in that there was nuclear immunoreactivity to the antibody against PR and multifocally against the antibody to ER. Basal subtypes could express p63, as in our case, but these subtypes do not typically have hormone receptor positivity. These findings do not let us classify this mass easily into any of the mammary tumor subtypes of other species. Based on the repeatable bi-layered appearance of the cells, made clearer by p63 expression, the histologic pattern alone perhaps confirmed mammary ductular origin.
The adrenal mass was confirmed to be a neuroendocrine medullary tumor, possibly a pheochromocytoma, but its functionality was not confirmed. This too is a rare finding in ruminants but reported previously in sheep. 2
Footnotes
Acknowledgements
We thank the ViTALS laboratory at the Virginia Maryland College of Veterinary Medicine for sectioning and processing of the histology sections, the University of Tennessee histology laboratory for processing of the hormone receptor IHC slides, and the Indiana Animal Disease Diagnostic Laboratory for processing the mammary subtype IHC panel.
Authors’ Note
Shelley J. Newman is now affiliated with Newman Specialty VetPath, Hicksville, NY.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
