Abstract
The current report describes a complex canine mammary adenoma with a rare histological feature characterized by sebaceous differentiation of tumor cells. A 13-year-old, mixed-breed, intact female dog had mammary tumors on the right mammary chain. Histologically, one of the masses was composed of bilayered ductal structures with luminal epithelial cells together with basaloid or myoepithelial cell components. Within the tumor, there were a number of lobules and nests of large foamy cells associated with basaloid reserve–like cells similar to sebaceous gland. Squamous metaplasia was also seen within the tumor. Immunohistochemical staining indicated that the tumor cells with sebaceous differentiation were positive for cytokeratin (CK)14 and that the associated basaloid reserve–like cells were positive for p63. In contrast, other luminal epithelial tumor cells were positive for CK18 and CK19, but not for CK14 and p63. The myoepithelial cells were positive for α-smooth muscle actin and p63. The expression of p63 in both sebaceous basaloid reserve–like cells and myoepithelial cells, and their structural continuity within the tumor tissue, suggested a common origin of these 2 components.
The mammary gland is a modified apocrine sweat gland, 1 and its embryonic ectodermal anlagen is common with the cutaneous adnexa. 14 It is known that squamous metaplasia may occur in canine benign mammary tumors. 10 However, to the authors’ knowledge, sebaceous differentiation in mammary adenomas has not been documented in the veterinary literature although a few cases have been reported in human beings. 2,9 A rare case of canine mammary adenoma with sebaceous differentiation and squamous metaplasia is presented herein.
Cytokeratins (CKs) are classified according to their molecular weight and isoelectric point, and are numbered from 1 to 20. 4 Examination of CK isoforms in cutaneous tumors may be useful for identification of tumor origin and differentiation of the neoplastic cells, since expression of CK isoforms differs among the epithelial cells of various cutaneous adnexa. 17 The expression of a number of cell differentiation markers, such as CKs, p63, and α-smooth muscle actin (α-SMA), has been described in canine mammary tissues. Cytokeratins 18 and 19 are considered to be luminal epithelial cell markers, whereas CK14 and p63 are expressed in basaloid or myoepithelial cells. 6,8,15 Alpha-SMA is exclusively expressed in myoepithelial cells. 5 Therefore, the immunohistological characteristics of the tumor cells in the current case were analyzed to determine the origin of the sebaceous gland–like structure.
A 13-year-old, mixed-breed, intact female dog had 3 mammary tumors on the right side. The masses were found 1 year before surgery by the dog’s owner, and had gradually grown larger from first discovery. The masses were located in the right second, third, and fourth mammary gland, measuring 1.0 cm × 1.0 cm × 0.5 cm, 2.0 cm × 2.0 cm × 1.0 cm, and 1.0 cm × 1.0 cm × 1.0 cm, respectively. The regional lymph nodes showed no enlargement. Mastectomy was performed to remove the masses, together with the local lymph nodes. The tumor masses and lymph nodes were then submitted for histopathological examination. In cut sections, all masses were white to light brown with partial hemorrhage and were well demarcated.
Excised tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin and periodic acid–Schiff (PAS). Immunohistochemical staining was performed by the immunoenzyme polymer method using the primary antibodies shown in Table 1. Peroxidase-conjugated anti-mouse immunoglobulin (Ig)G g was used as a secondary antibody. After immunoreaction, the sections were colorized with diaminobenzidine (DAB) and counterstained with Mayer hematoxylin. Double-labeled immunohistochemistry was also performed using anti-CK18 and anti-CK14 antibodies. In this assay, CK18 and CK14 were visualized using DAB and a commercial substrate kit for peroxidase. h
Primary antibodies and immunostaining protocol in the current study.
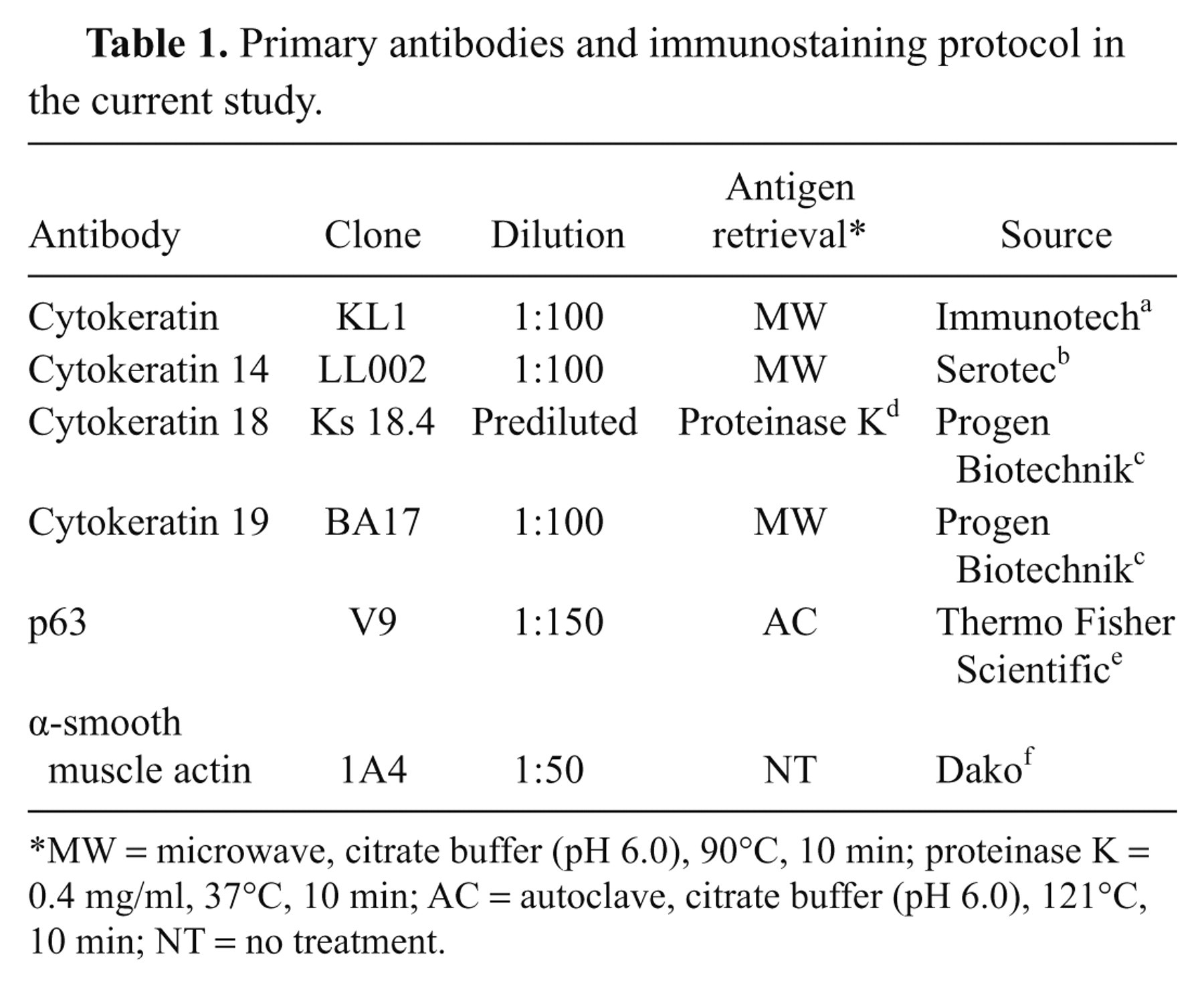
MW = microwave, citrate buffer (pH 6.0), 90°C, 10 min; proteinase K = 0.4 mg/ml, 37°C, 10 min; AC = autoclave, citrate buffer (pH 6.0), 121°C, 10 min; NT = no treatment.
Histologically, the right fourth mammary gland mass was well-circumscribed and was composed of bilayered ductal structures with an inner cuboidal to columnar luminal epithelial cell layer and outer myoepithelial cells or basaloid cells (Fig. 1). The nuclei of the luminal epithelial cells and outer cells appeared large with prominent round nucleoli but mitotic cells were extremely rare. Papillary proliferation of epithelial components within the glandular lumens was occasionally observed. Within the tumor, there were a number of lobules and nests of large foamy cells similar to the sebaceous gland (Figs. 1, 2). The nuclei of these cells were centrally located, and their cytoplasm was finely vacuolated and was negative for the PAS reaction. These sebaceous gland–like nests were surrounded by basaloid reserve–like cells with scant cytoplasm and high chromatic nuclei (Fig. 1 inset). The sebocyte-like neoplastic cells and basaloid reserve–like cells frequently showed continuity with the neoplastic ductal structures (Fig. 3). Several foci of squamous metaplasia were also seen within the tumor. Most squamous cells were intimately associated with sebocyte-like cells within the tumor (Fig. 2).
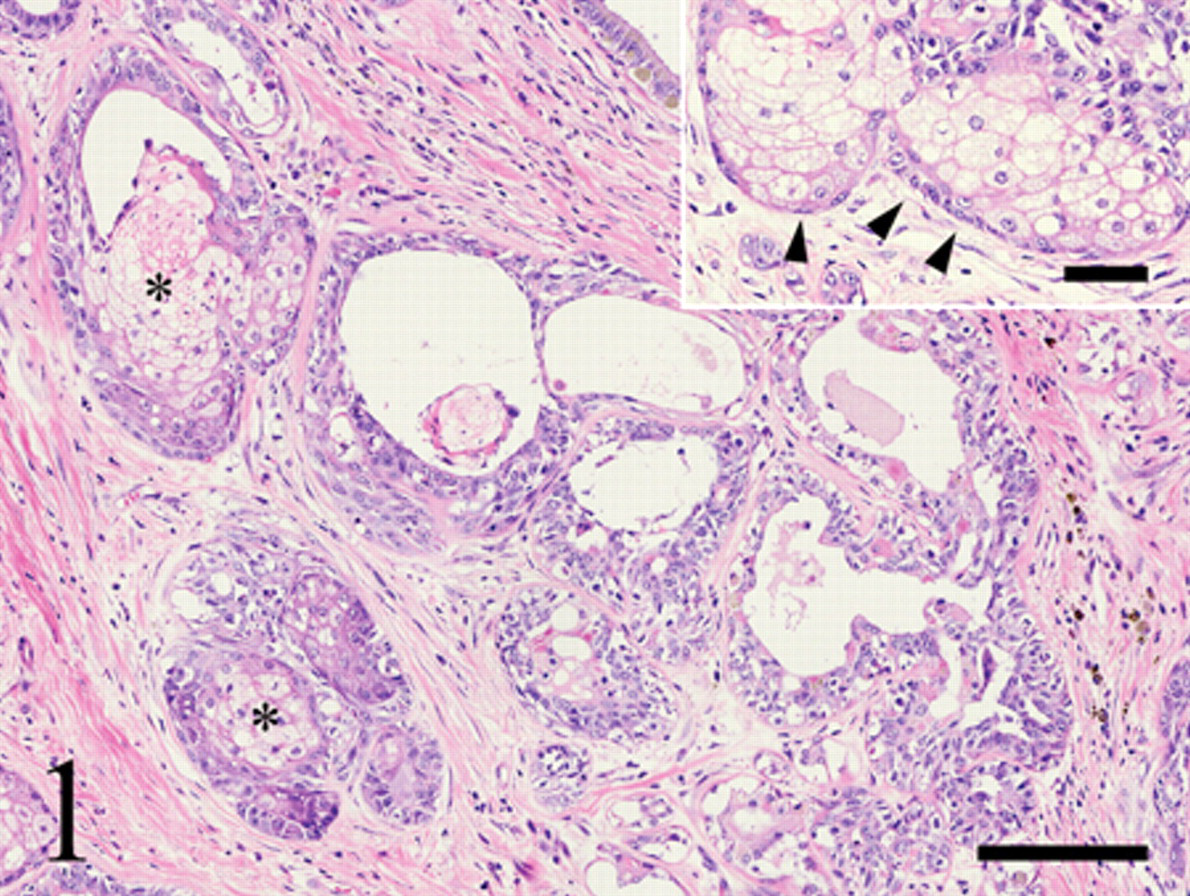
Histological appearance of the mass in the right fourth mammary gland from a 13-year-old, mixed-breed, intact female dog. Lobules of clear cells similar to sebaceous glandular epithelium (asterisks) are observed within the tumor. Hematoxylin and eosin stain. Bar = 150 µm. Inset: Sebaceous gland–like nests are surrounded by small cells resembling basaloid reserve cells (arrowheads). Bar = 50 µm.
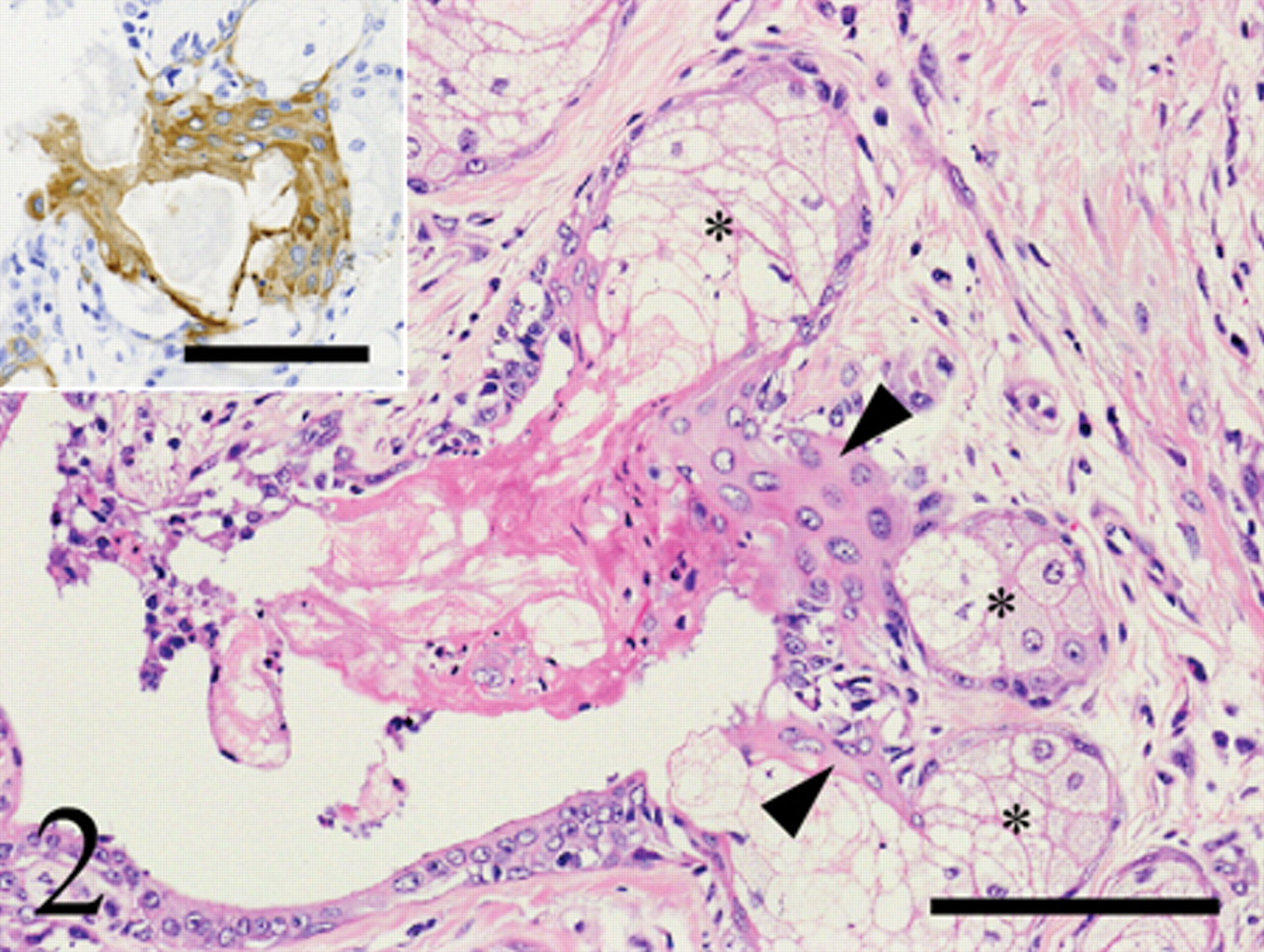
Squamous metaplasia and sebaceous differentiation observed within the canine mammary complex adenoma. Metaplastic squamous cells (arrowheads) were intimately associated with sebaceous gland–like structures (asterisks). The tumor section was stained with hematoxylin and eosin, and immunostained for cytokeratin (CK). Bar = 100 µm. Squamous cells are positive for CK clone KL1. Inset: Bar = 100 µm.
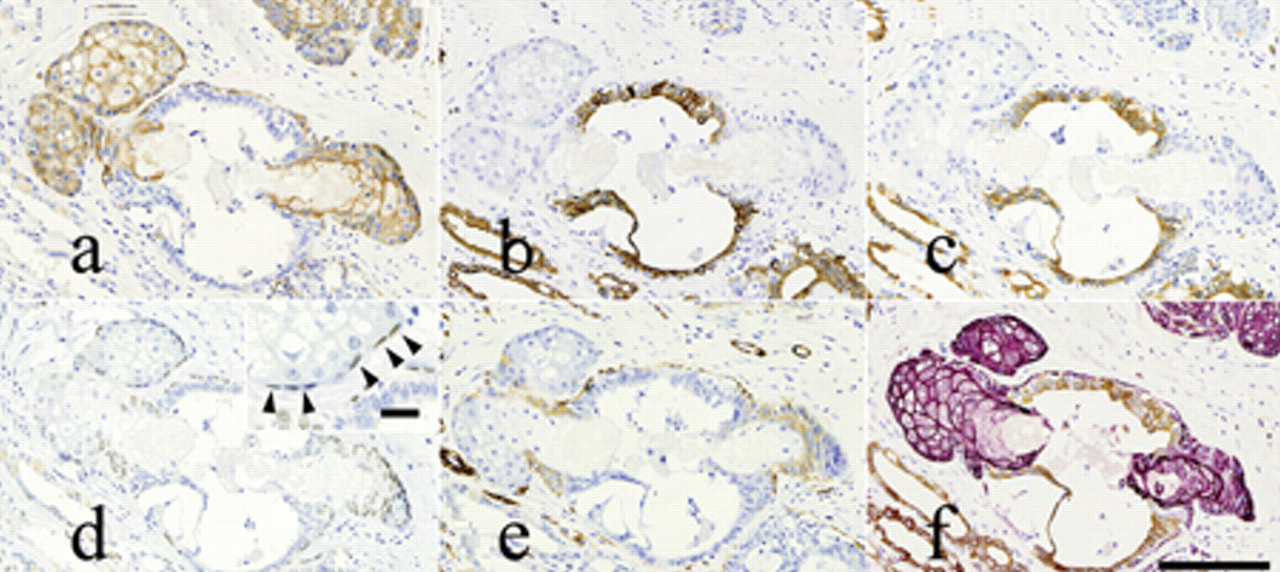
Immunostaining of serial sections of the canine mammary complex adenoma with sebaceous differentiation. Tumor tissues were immunostained for:
The results of the immunohistochemical staining of tumor cells are summarized in Table 2 and Figure 3. Sebocyte-like cells were positive for CK14 (Fig. 3a), while luminal epithelial cells without such differentiation were positive for the mammary epithelial cell makers CK18 and CK19 (Fig. 3b, 3c). Myoepithelial components were mostly positive for the mammary myoepithelial markers α-SMA and CK14 (Fig. 3a, 3e). Myoepithelial cells and basaloid reserve–like cells surrounding the sebaceous gland–like structures were positive for p63 (Fig. 3d). Based on double-labeling immunohistochemistry, tumor cells were clearly either CK14- or CK18-positive, and no double-positive cells were observed (Fig. 3f). The tumor was ultimately diagnosed as a complex adenoma with sebaceous differentiation and squamous metaplasia. The second and third mammary masses were diagnosed as a complex adenoma and a benign mixed tumor, respectively, and no sebaceous differentiation and/or squamous metaplasia was observed within these 2 masses. Metastasis was not observed in the local lymph nodes.
Results of immunostaining of the tumor cells for cytokeratins (CKs), p63, and α-smooth muscle actin (α-SMA).*
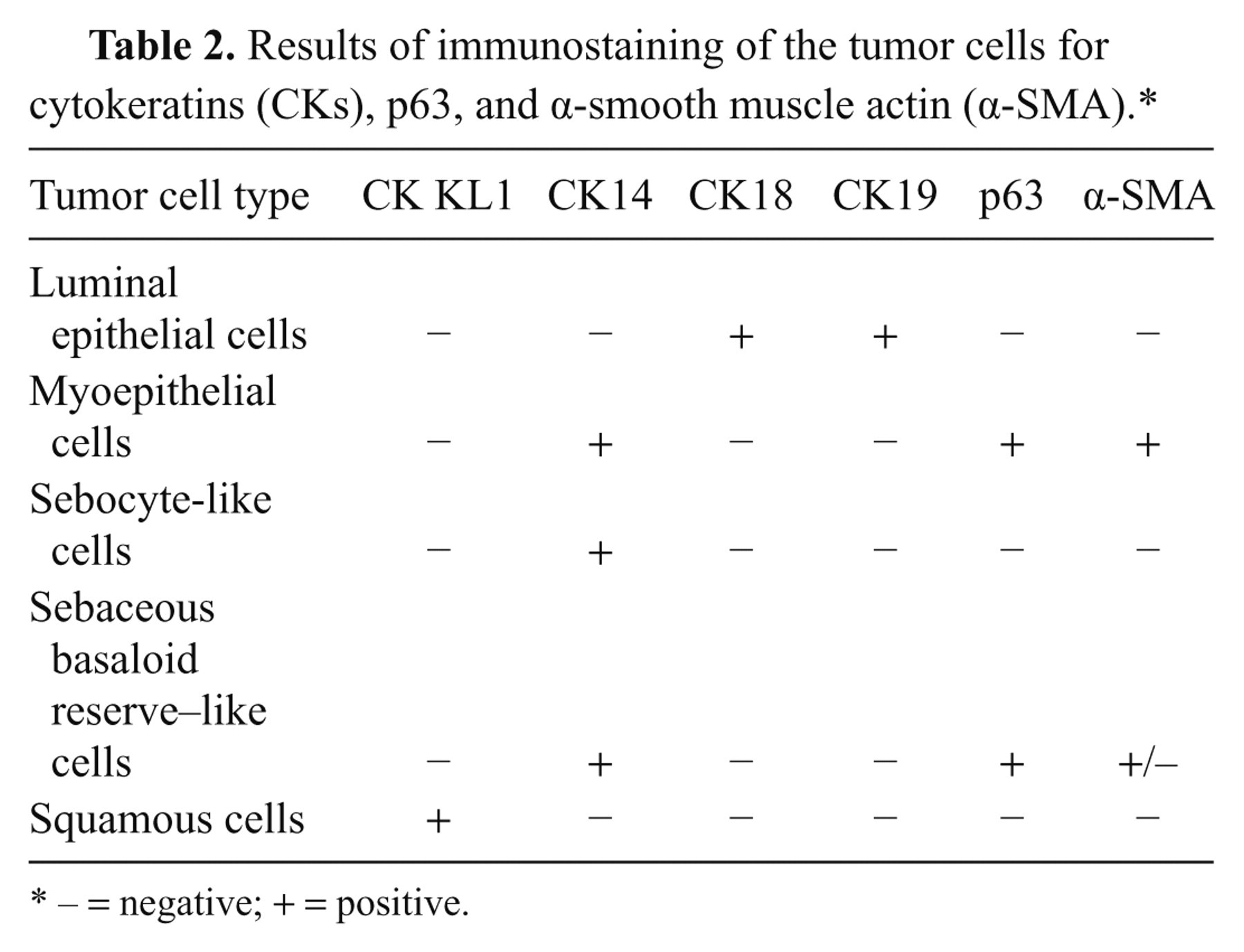
− = negative; + = positive.
To the authors’ knowledge, only a single case of primary mammary carcinoma 3 with sebaceous differentiation has been reported in a dog. The present study is the first report of a sebaceous gland differentiation that developed within a canine complex adenoma, which is a benign lesion of the mammary gland.
In the present case, cellular differentiation into sebocytes and squamous metaplasia was suggested not only by the morphological features but also by the immunohistochemical characteristics of the cells. Sebocyte-like cells and basaloid reserve–like cells expressed CK14 and p63, which are also expressed in normal mature sebaceous gland cells 17 and sebaceous basaloid reserve cells. 12 Cytokeratin (KL1) is expressed in differentiated keratinocytes, 16,17 and the squamous cells in the present case were also positive for CK (KL1). The findings strongly support the conclusion that the present case developed into mature sebocytes and squamous cells.
The expression of p63 both in basaloid reserve–like cells and in myoepithelial cells within the tumor tissue suggested a common origin for these 2 components. The p63 gene is expressed in basal cells of several epithelial organs, and it has been suggested that the p63 gene plays a major role in the preservation of the stem cell component of these organs, including the breast. 7 A previous study 6 indicated a putative role of p63-positive basal epithelium (myoepithelium) in mesenchymal metaplasia, since metaplastic cells within complex and mixed tumors included p63-positive cells. Therefore, the authors propose that the sebocyte-like tumor cells were probably derived from p63-positive basaloid cells with a pluripotency of differentiation. Importantly, there were no CK14 and CK18 double-positive cells within the present tumor, supporting the proposal that sebaceous differentiation was not derived from a glandular epithelial component, but from a basaloid or myoepithelial component. Squamous metaplasia is suggested to be associated with sebaceous differentiation in human mammary tumors. 2,9,11,13 Furthermore, canine squamous metaplasia has not been reported to be accompanied by sebaceous differentiation in a case of mammary carcinoma. 3 In the current case, the squamous epithelium was partially continuous with a sebaceous structure, indicating close association between these 2 components. However, some previous studies have also concluded that mammary stem cells convert into sebaceous cells or adenosquamous cells, 9,13 suggesting that these 2 cells were derived from common cells within the tumor.
Footnotes
a.
Immunotech SAS, Marseille, France.
b.
Serotec, Wiesbaden, Germany.
c.
Progen Biotechnik, Heidelberg, Germany.
d.
Wako Pure Chemical Industries Ltd., Osaka, Japan.
e.
Thermo Fisher Scientific Inc., Waltham, MA.
f.
Dako Denmark A/S, Glostrup, Denmark.
g.
Histofine Simple Stain MAX-PO(M), Nichirei Corp., Tokyo, Japan.
h.
Vector® VIP substrate kit, Vector Laboratories Inc., Burlingame, CA.
The authors declared that they had no conflicts of interests in their authorship and publication of this contribution.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by a project grant (Creative Research Project, 2009) awarded by Azabu University Research Services Division.
