Abstract
Measuring trace mineral concentrations can be an important component of assessing the health of free-ranging deer. Trace mineral concentrations in liver most accurately reflect the trace mineral status of an individual, but, in live animals, whole blood or serum are the most commonly used sample types. Trace minerals measured in serum, such as copper, zinc, and iron, do not always accurately correlate to liver concentrations, and supplementary samples for evaluating the trace mineral status in live deer would be useful. We evaluated the utility of body and tail hair for measuring selenium, copper, zinc, iron, and manganese in free-ranging mule deer (
Introduction
Evaluation of trace mineral concentrations can be an important component of assessing individual or population health in free-ranging wildlife. Deficiencies have been detected in association with disease syndromes, unthriftiness, and poor reproductive performance in deer, elk, and moose populations.10,12,13,15,16,18,24–26 In California, selenium and copper deficiencies have been detected in deer7,10,25 and elk,15,18 respectively. Trace mineral values in liver are considered to most accurately reflect the nutritional status of the animal, but, in live animals, concentrations are most commonly measured in whole blood or serum. For some trace elements such as copper, the correlation between serum and liver concentrations is only moderate, 33 making accurate estimations in live animals difficult. Similarly, manganese levels are difficult to measure in live animals, as serum manganese is a poor indicator of the trace mineral status. 32
Hair has been evaluated as an indicator of trace mineral status in livestock, 4 and the reliability of hair as a measure of mineral levels varies with animal species, geographic location, hair color, age, sex, and element. 4 Furthermore, laboratory methods to eliminate external contaminants from biologically incorporated elements vary and can result in altered mineral concentrations. 4 To our knowledge, the utility of hair to measure trace mineral concentrations has not been assessed in free-ranging deer. If trace mineral concentrations in hair correlates with the concentration in other sample types, it can potentially be used as a complementary sample in live deer. Hair trace mineral concentrations were previously studied in Alaskan moose 14 and in small samples of Idaho and Washington mule deer,8,9 but the relationship between the concentrations in hair and in other sample types was not evaluated.
The goal of this study was to determine the utility and value of various biological sampling variables for measuring trace mineral concentrations in deer in California. We compared tail and body hair concentrations of selenium, copper, zinc, iron, and manganese with the concentrations in whole blood (selenium), serum (copper, iron, and zinc) and liver (selenium, copper, zinc, iron, and manganese). The concentrations from this investigation are compared with levels reported in other deer populations in the United States.
Materials and methods
Samples were collected from a total of 109 live- and 54 postmortem-sampled deer from the Sierra Nevada Mountains, Sierra Nevada foothills, and areas of coastal and southern California (Fig. 1). Deer in the Sierra Nevada and Sierra Nevada foothills consist of migratory Rocky Mountain mule deer (
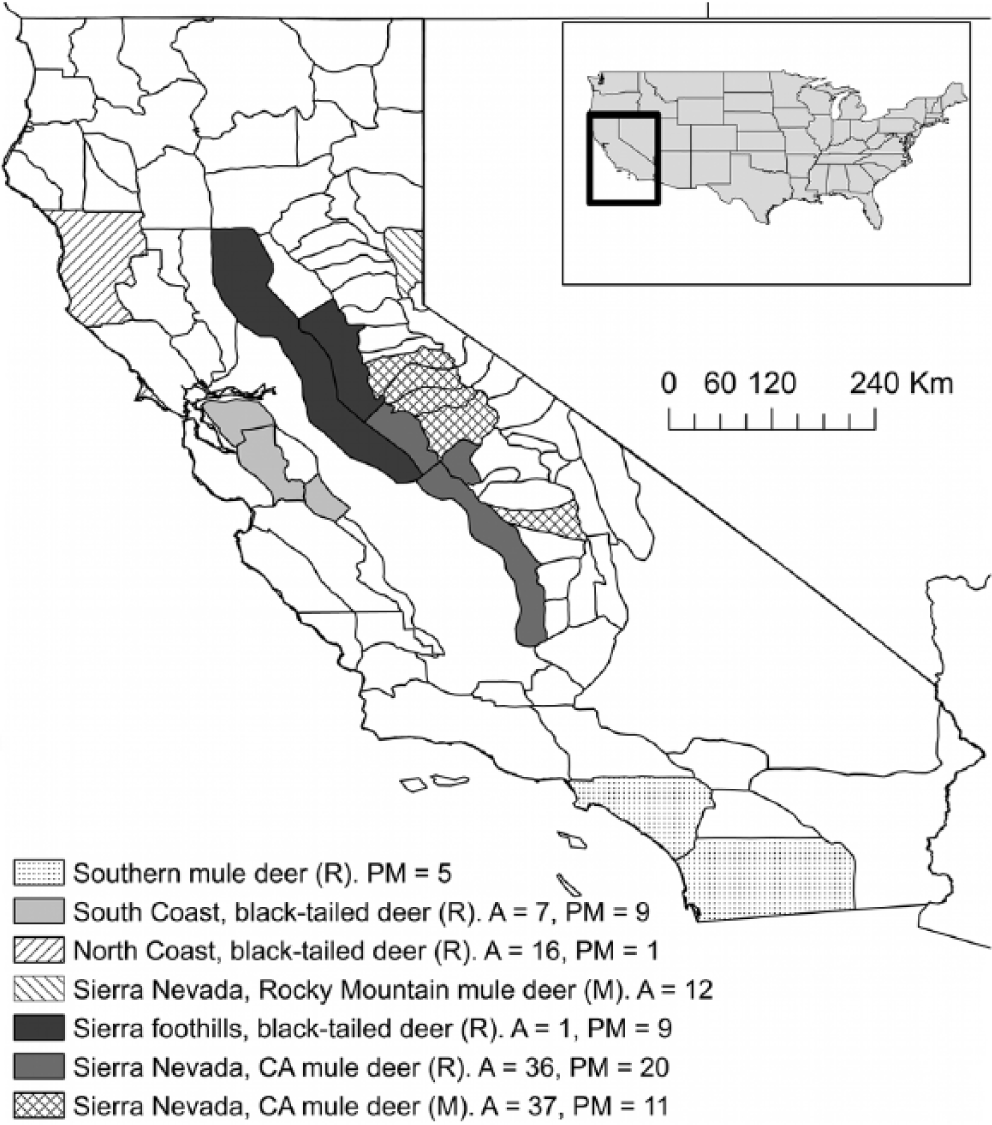
Location of live- and postmortem-sampled free-ranging mule deer (
Whole blood selenium concentrations and serum zinc, copper, and iron concentrations were measured by inductively coupled argon plasma–atomic emission spectrometry f (ICP-AES) as described previously.21,31 Accuracy of ICP results for these elements was measured by analyzing quality assurance sera obtained from the Veterinary Laboratory Association Quality Assurance Program. g Data was accepted if the analyzed quality assurance serum values were within 2 standard deviations of the reference values.
The analysis of hair samples was a slight modification of a previously published methodology. 5 Hair was initially separated from any associated skin and debris. The hair sample was then rinsed with type 1 reagent–grade water (18.3 Mohm-cm water) and placed into a 50-mL polypropylene centrifuge tube. After the addition of 10 mL of acetone, the tube was shaken vigorously for 30 sec. The contents of the tube were poured into a Büchner funnel to separate acetone from hair. Hair was then washed twice with type 1 reagent–grade water before being dried in a drying oven at 95°C for 24 hr. One half to 1 g of dried hair was placed into a 10-mL digestion tube, h and the exact weight was recorded. After the addition of 3 mL of trace metal–grade nitric acid, i the sample was digested at 190°C for 90 min using block digesters. j After digestion, 2 mL of trace metal–grade hydrochloric acid k were added, and the digestate was diluted with type 1 reagent–grade water to a final volume of 10 mL. The solution was thoroughly vortexed for 10 sec. A subsample of this solution was subsequently analyzed for copper, zinc, iron, and manganese by ICP-AES. f
For selenium analysis, a 4-mL subsample of the digestate was transferred to a new 10-mL digestion tube. After addition of 1 mL of trace metal–grade perchloric acid k and 1 mL of ultrapure sulfuric acid, l the sample was digested at 350°C for 180 min. After completion of digestion and cooling of the sample to room temperature, 7 mL of 5 M ultrapure hydrochloric acid k were added slowly before further digestion at 95°C for 15 min. The digestate was diluted with type 1 reagent–grade water to a final volume of 10 mL and analyzed for selenium by ICP-AES. f
For each batch of samples, method blanks were included in the analyses. Method blank samples were prepared by adding boiling chips to an acid-cleaned 10-mL digestion tube. h For metal analysis other than selenium, 3 mL of trace metal–grade nitric acid were added followed by digestion at 190°C for 90 min. After digestion, 2 mL of trace metal–grade hydrochloric acid were added, and the digestate was diluted with type 1 reagent–grade water to a final volume of 10 mL. Methods blanks for selenium analysis were prepared by adding boiling chips to an acid-cleaned 10-mL digestion tube. After addition of 3 mL of trace metal–grade nitric acid, 1 mL of trace metal–grade perchloric acid, and 1 mL of ultrapure sulfuric acid, the sample was digested at 350°C for 180 min. After completion of digestion and cooling of the sample to room temperature, 7 mL of 5 M ultrapure hydrochloric acid were added slowly before further digestion at 95°C for 15 min. The digestate was diluted with type 1 reagent–grade water to a final volume of 10 mL.
Liver samples were prepared for selenium analysis by digestion of the samples in an oxidizing mixture of nitric, sulfuric, and perchloric acids at 350°C, followed by reduction with 5 M hydrochloric acid at 95ºC. Selenium concentrations were determined by hydride vapor generation inductively coupled plasma spectrometer.f,31
Accuracy of ICP results was measured by analyzing certified standard reference materials (SRMs), as well as liver and hair fortified with all measured metals. Control hair (human) was prepared in duplicate and fortified before acid was added. The SRMs included dogfish liver (National Research Council of Canada, DOLT-4) and lobster hepatopancreas (National Research Council of Canada, TORT-2). The method reporting limits were determined by a series of dilutions of reference materials. The lowest reporting limit was determined as the lowest level at which the relative standard deviation of 3 replicates of a diluted reference material was <10%. The limit of quantitation for selenium in liver and hair was 0.02 mg/kg (wet weight). For copper, manganese, zinc, and iron analysis, liver samples were digested with nitric acid at 180°C and subsequently analyzed by ICP-AES.f,20 Accuracy of ICP results for these elements was measured by analyzing SRMs and fortified liver tissue. The SRMs used were DOLT-4 and TORT-2. The limits of quantitation were 0.01 mg/kg for copper, 0.04 mg/kg for manganese, 0.1 mg/kg for zinc, and 0.2 mg/kg for iron (all on wet weight basis). Data for these analyses was accepted if SRM results were within 2 standard deviations of the certified reference values and spike recoveries were within an 80–120% recovery range.
All results for blood selenium concentrations were reported in mg/kg (ppm). Serum zinc, copper, and iron concentrations were reported in mg/L (ppm). Liver mineral concentrations were reported as mg/kg wet weight, illustrating that liver mineral concentrations were not corrected for % moisture. Hair mineral concentrations were reported as mg/kg dry weight because all hair samples were completely dried prior to analysis.
Statistical analysis
The median and ranges of trace element concentrations in blood and serum, body hair, tail hair, and liver were tabulated by age, sex, and migratory behavior and geographical area. Because trace mineral concentrations in hair do not change after death, the trace mineral results from both live- and postmortem-sampled deer were pooled for the descriptive statistics. Mineral concentrations below the detectable limit were included in the calculations by assigning values of half the detection limit.
34
Correlations between trace mineral concentrations in hair, blood and serum, and liver were assessed using Spearman rank correlations for nonnormally distributed data. Because the correlations were expected to be stronger around time of hair growth, samples from live deer were pooled into the seasonal groups December, February–March, April–June, and July–August, and samples from postmortem-sampled deer into the groups August–November and April–June for the correlation analysis. For live-sampled deer, data was further categorized into resident and migratory deer, as correlations were assumed to be stronger in resident deer that have a more consistent diet. Only a small number of migratory deer sampled postmortem, and therefore, were not divided into a migratory and resident category. For correlation significant at
Results
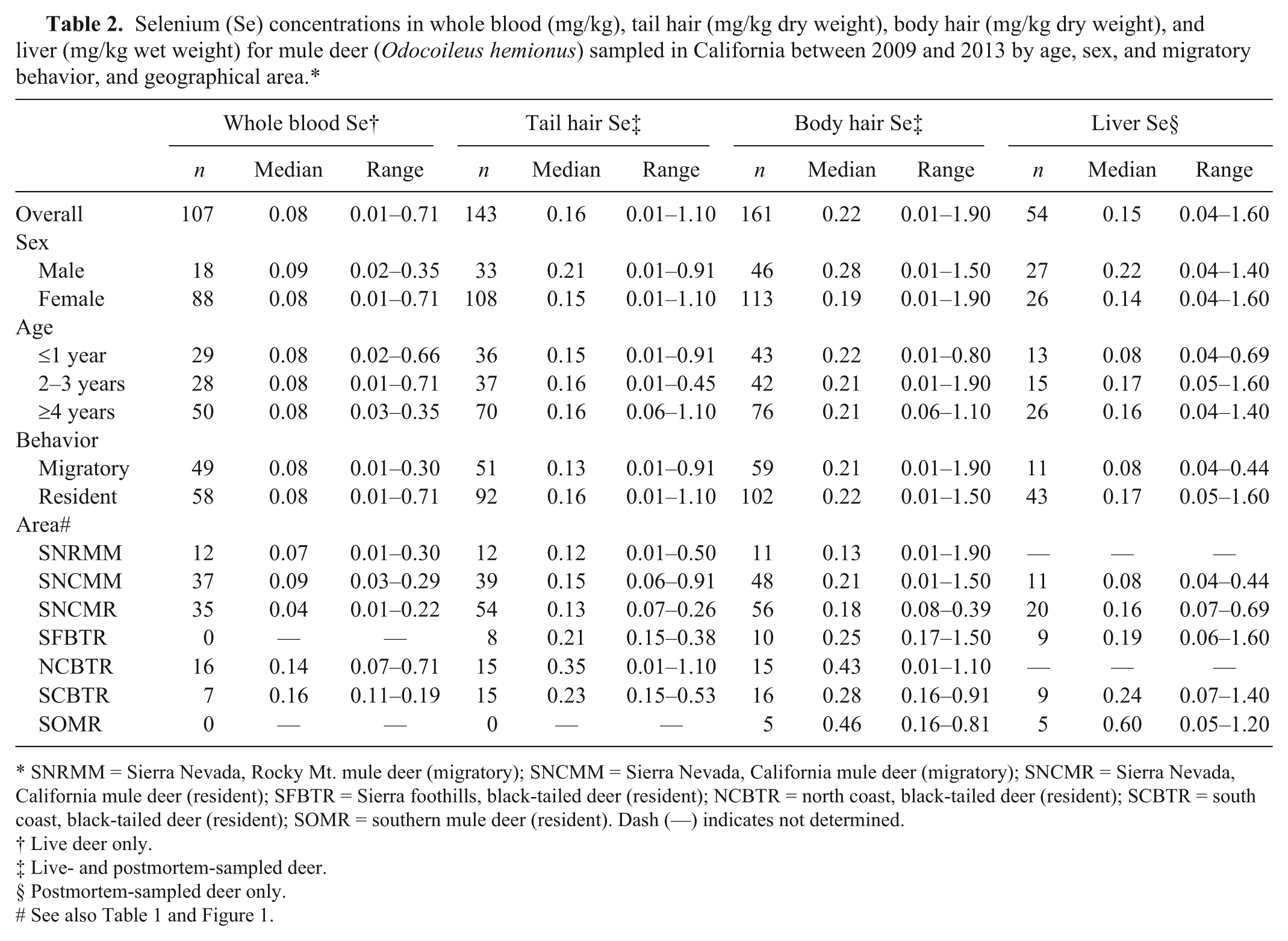
In both the coastal and Sierra Nevada and Sierra foothills areas, the majority of live-sampled deer were female, whereas for postmortem-sampled deer, the majority were male in the coastal and southern California areas and female in the Sierra Nevada and foothills areas (Table 1). In the Sierra Nevada and foothills areas, live- and postmortem-sampled deer were sampled across all age classes. In the coastal areas, the majority of live-sampled deer were ≥4 years old, whereas most postmortem-sampled deer from the coastal and southern California areas were 2–3 or ≥4 years old (Table 1). The distribution of selenium, copper, zinc, iron, and manganese in blood, tail hair, body hair, and liver are shown in Tables 2–6. Trace mineral concentrations were difficult to compare between areas because samples were collected at different times of year from several herds and age classes, and the live and postmortem samples did not originate from the same animals. In general, the median whole blood, body hair, tail hair, and liver selenium concentrations, as well as median liver concentrations, were lowest in the Sierra Nevada and foothills areas compared to the coastal area and southern California (Tables 2, 3). Liver zinc and iron were highest in the resident California mule deer and black-tailed deer of the Sierra Nevada and foothills areas (Tables 4, 5), but no similar pattern could be detected for serum or hair samples for these trace minerals. Median liver and body hair manganese was lowest in southern California and in the resident California mule deer of the Sierra Nevada (Table 6).
Number of live- and postmortem-sampled free-ranging mule deer (
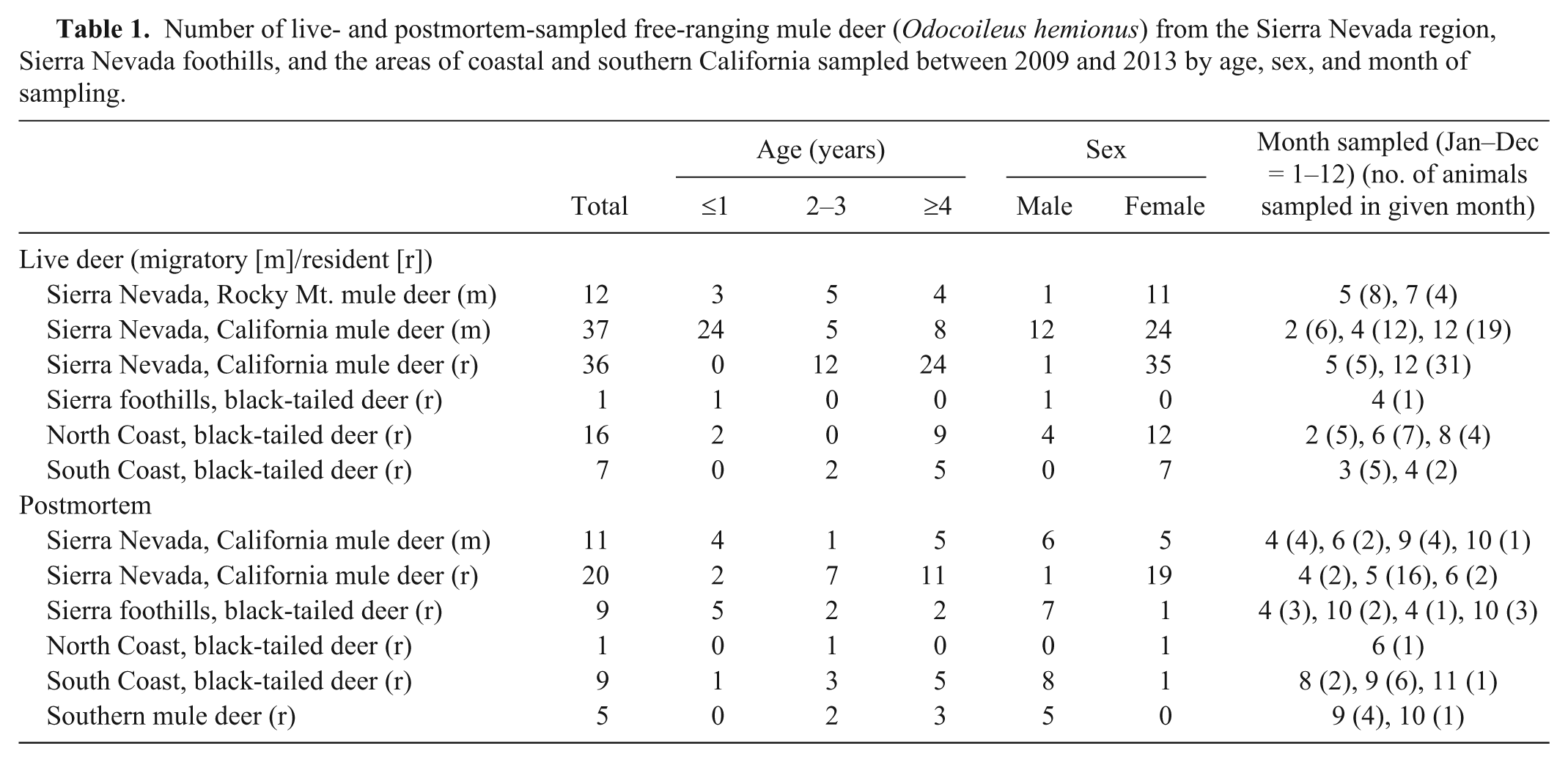
Selenium (Se) concentrations in whole blood (mg/kg), tail hair (mg/kg dry weight), body hair (mg/kg dry weight), and liver (mg/kg wet weight) for mule deer (
SNRMM = Sierra Nevada, Rocky Mt. mule deer (migratory); SNCMM = Sierra Nevada, California mule deer (migratory); SNCMR = Sierra Nevada, California mule deer (resident); SFBTR = Sierra foothills, black-tailed deer (resident); NCBTR = north coast, black-tailed deer (resident); SCBTR = south coast, black-tailed deer (resident); SOMR = southern mule deer (resident). Dash (—) indicates not determined.
Live deer only.
Live- and postmortem-sampled deer.
Postmortem-sampled deer only.
Copper (Cu) concentrations in serum (mg/L), tail hair (mg/kg dry weight), body hair (mg/kg dry weight), and liver (mg/kg wet weight) for mule deer (
SNRMM = Sierra Nevada, Rocky Mt. mule deer (migratory); SNCMM = Sierra Nevada, California mule deer (migratory); SNCMR = Sierra Nevada, California mule deer (resident); SFBTR = Sierra foothills, black-tailed deer (resident); NCBTR = north coast, black-tailed deer (resident); SCBTR = south coast, black-tailed deer (resident); SOMR = southern mule deer (resident). Dash (—) indicates not determined.
Live deer only.
Live- and postmortem-sampled deer.
Postmortem-sampled deer only.
Zinc (Zn) concentrations in serum(mg/L), tail hair (mg/kg dry weight), body hair (mg/kg dry weight), and liver (mg/kg wet weight) for mule deer (
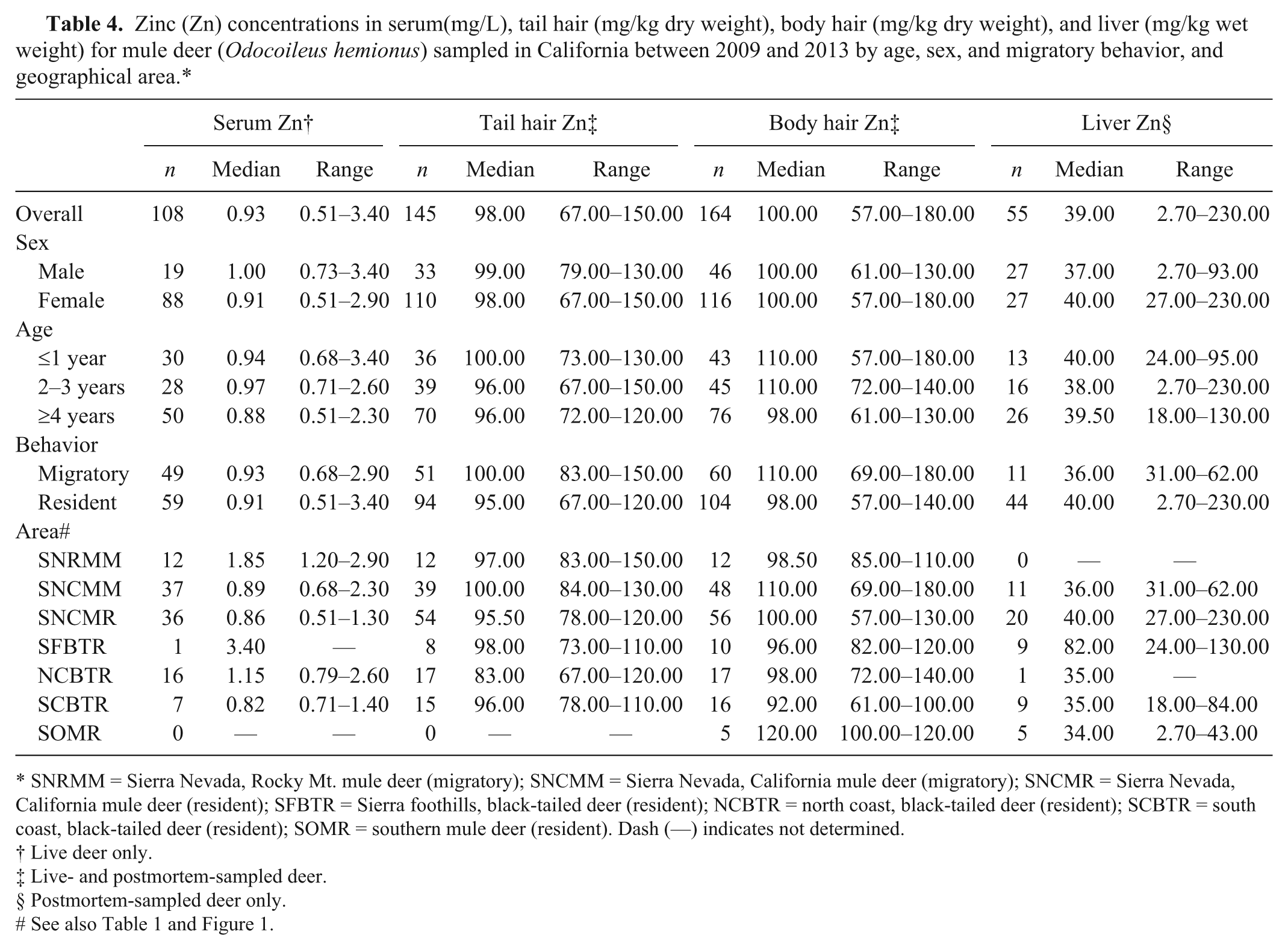
SNRMM = Sierra Nevada, Rocky Mt. mule deer (migratory); SNCMM = Sierra Nevada, California mule deer (migratory); SNCMR = Sierra Nevada, California mule deer (resident); SFBTR = Sierra foothills, black-tailed deer (resident); NCBTR = north coast, black-tailed deer (resident); SCBTR = south coast, black-tailed deer (resident); SOMR = southern mule deer (resident). Dash (—) indicates not determined.
Live deer only.
Live- and postmortem-sampled deer.
Postmortem-sampled deer only.
Iron (Fe) concentrations in serum (mg/L), tail hair (mg/kg dry weight), body hair (mg/kg dry weight), and liver (mg/kg wet weight) for mule deer (
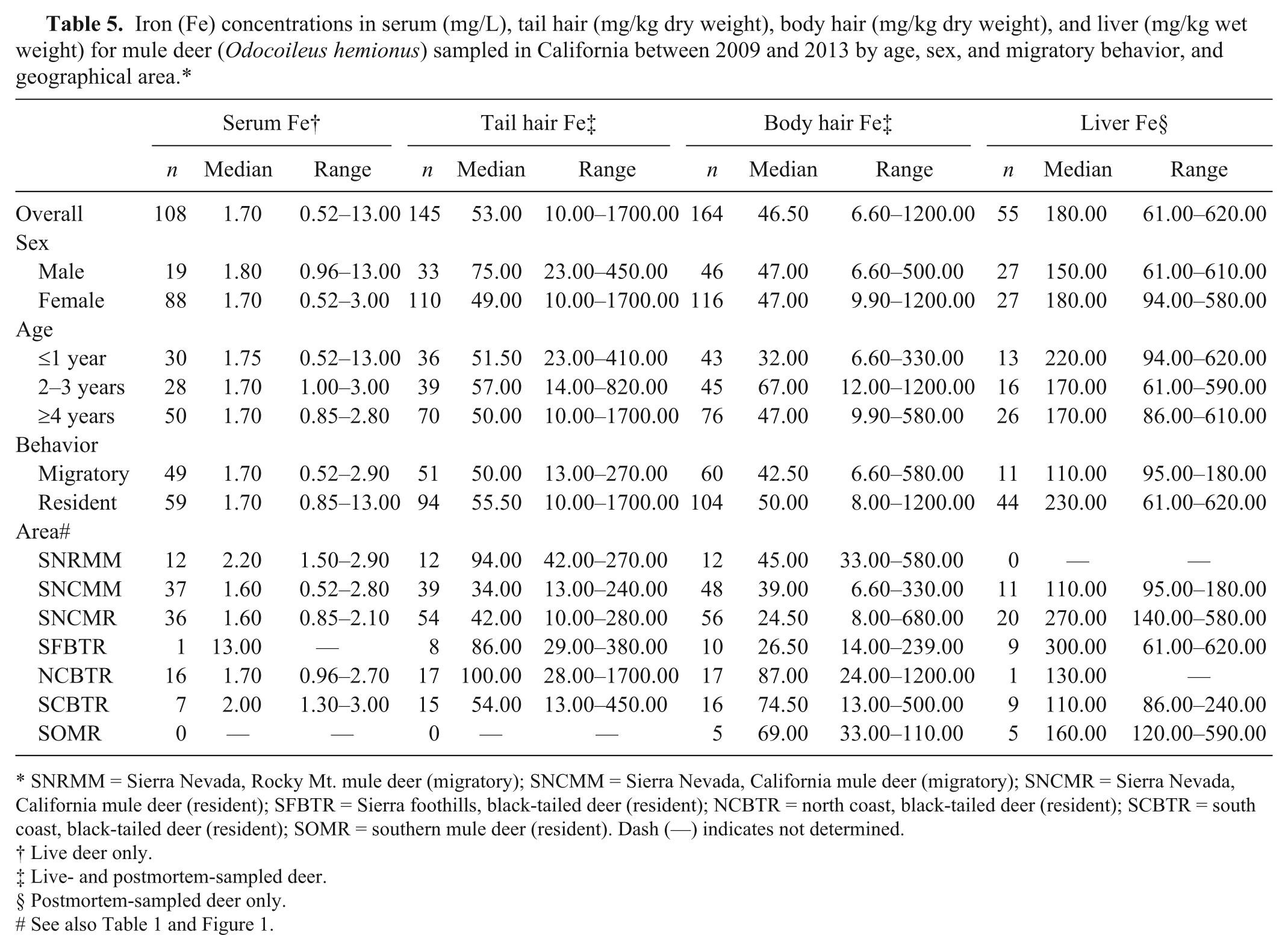
SNRMM = Sierra Nevada, Rocky Mt. mule deer (migratory); SNCMM = Sierra Nevada, California mule deer (migratory); SNCMR = Sierra Nevada, California mule deer (resident); SFBTR = Sierra foothills, black-tailed deer (resident); NCBTR = north coast, black-tailed deer (resident); SCBTR = south coast, black-tailed deer (resident); SOMR = southern mule deer (resident). Dash (—) indicates not determined.
Live deer only.
Live- and postmortem-sampled deer.
Postmortem-sampled deer only.
Manganese (Mn) concentrations in tail hair (mg/kg dry weight), body hair (mg/kg dry weight), and liver (mg/kg wet weight) for mule deer (
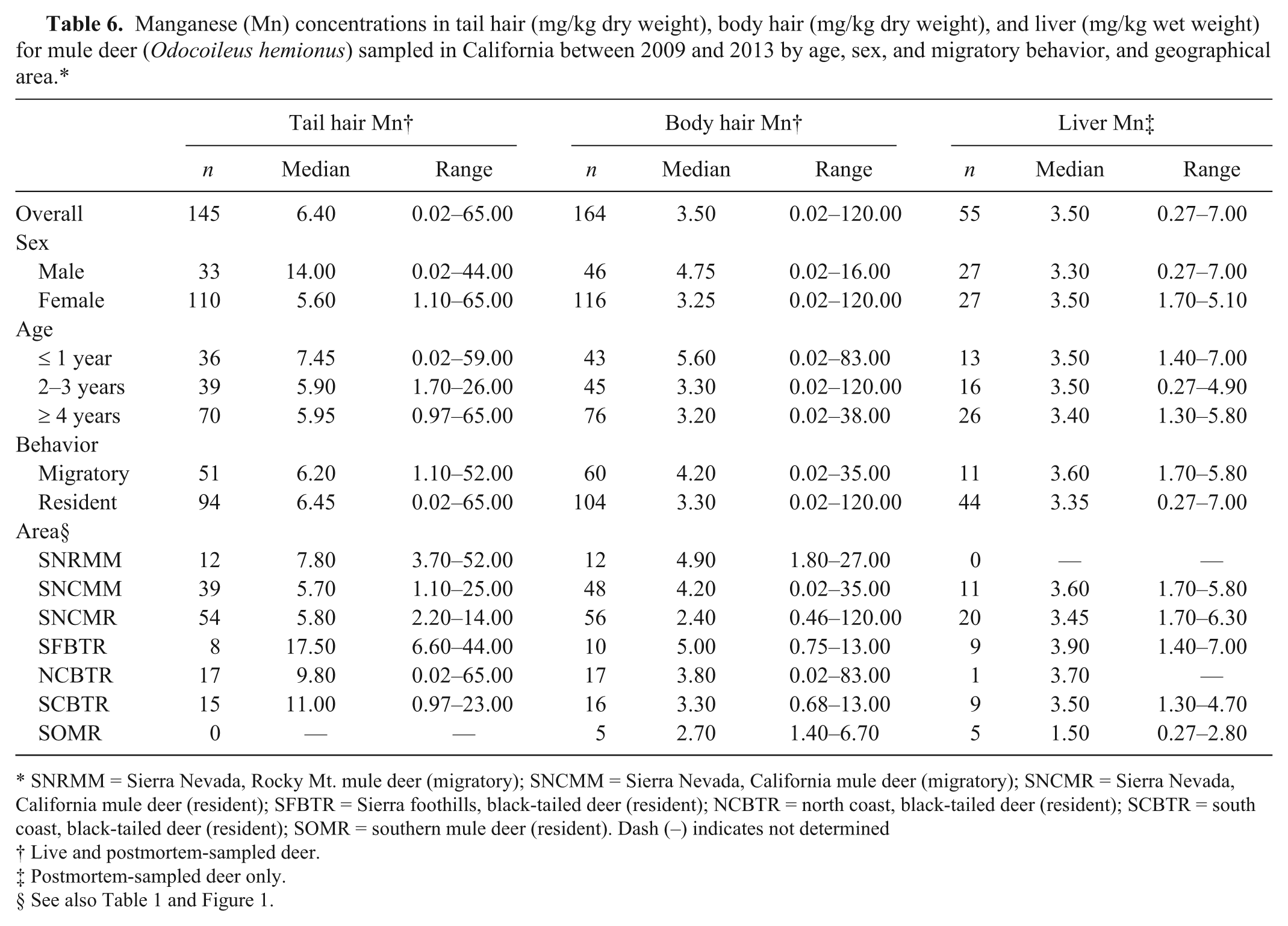
SNRMM = Sierra Nevada, Rocky Mt. mule deer (migratory); SNCMM = Sierra Nevada, California mule deer (migratory); SNCMR = Sierra Nevada, California mule deer (resident); SFBTR = Sierra foothills, black-tailed deer (resident); NCBTR = north coast, black-tailed deer (resident); SCBTR = south coast, black-tailed deer (resident); SOMR = southern mule deer (resident). Dash (–) indicates not determined
Live and postmortem-sampled deer.
Postmortem-sampled deer only.
In live-sampled deer, there was a significant correlation (
Spearman rank correlations between selenium (Se), copper (Cu), zinc (Zn), and iron (Fe) in whole blood (for Se), serum (for Cu, Zn, and Fe), and tail and body hair of free-ranging, live California mule deer (
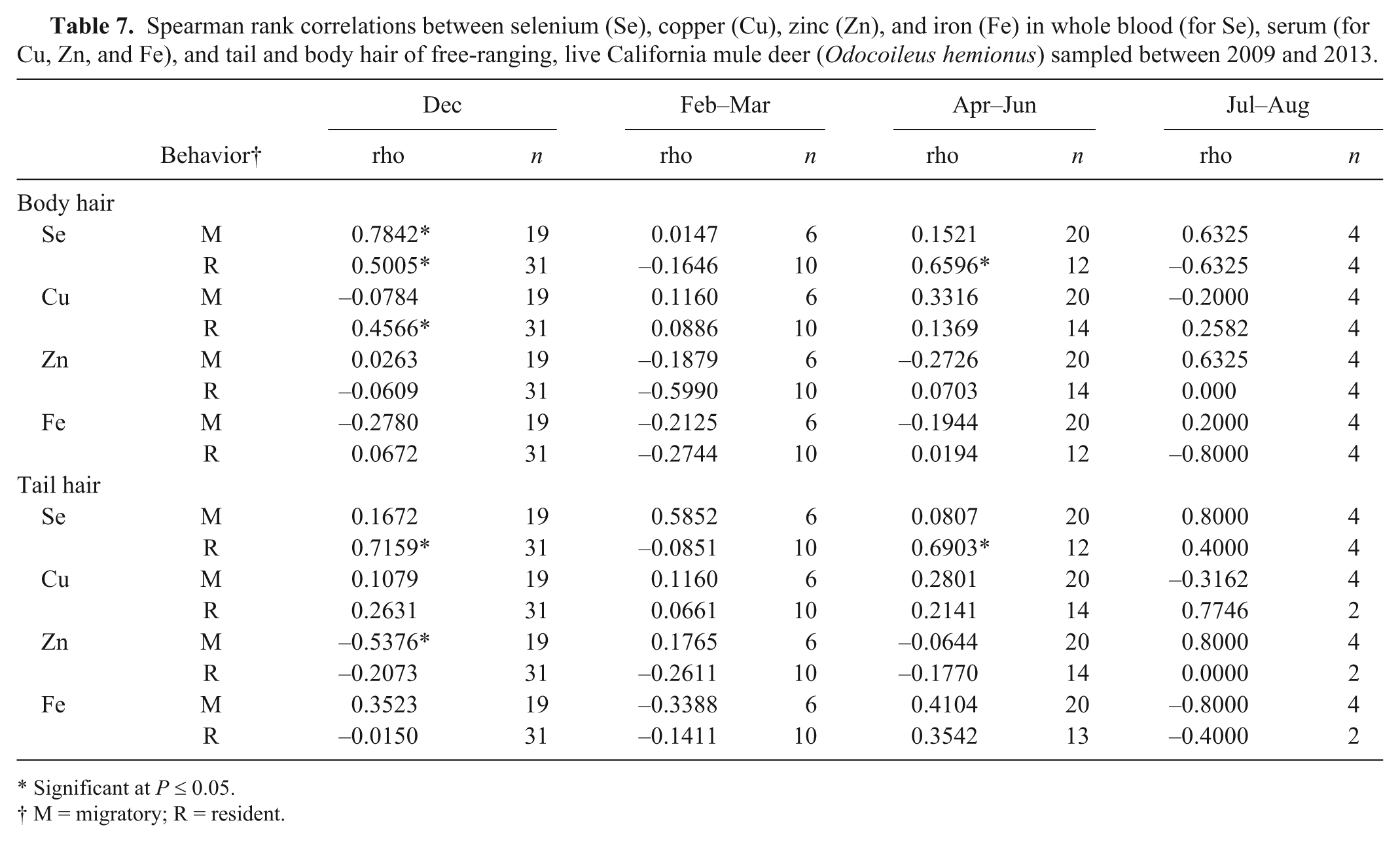
Significant at
M = migratory; R = resident.
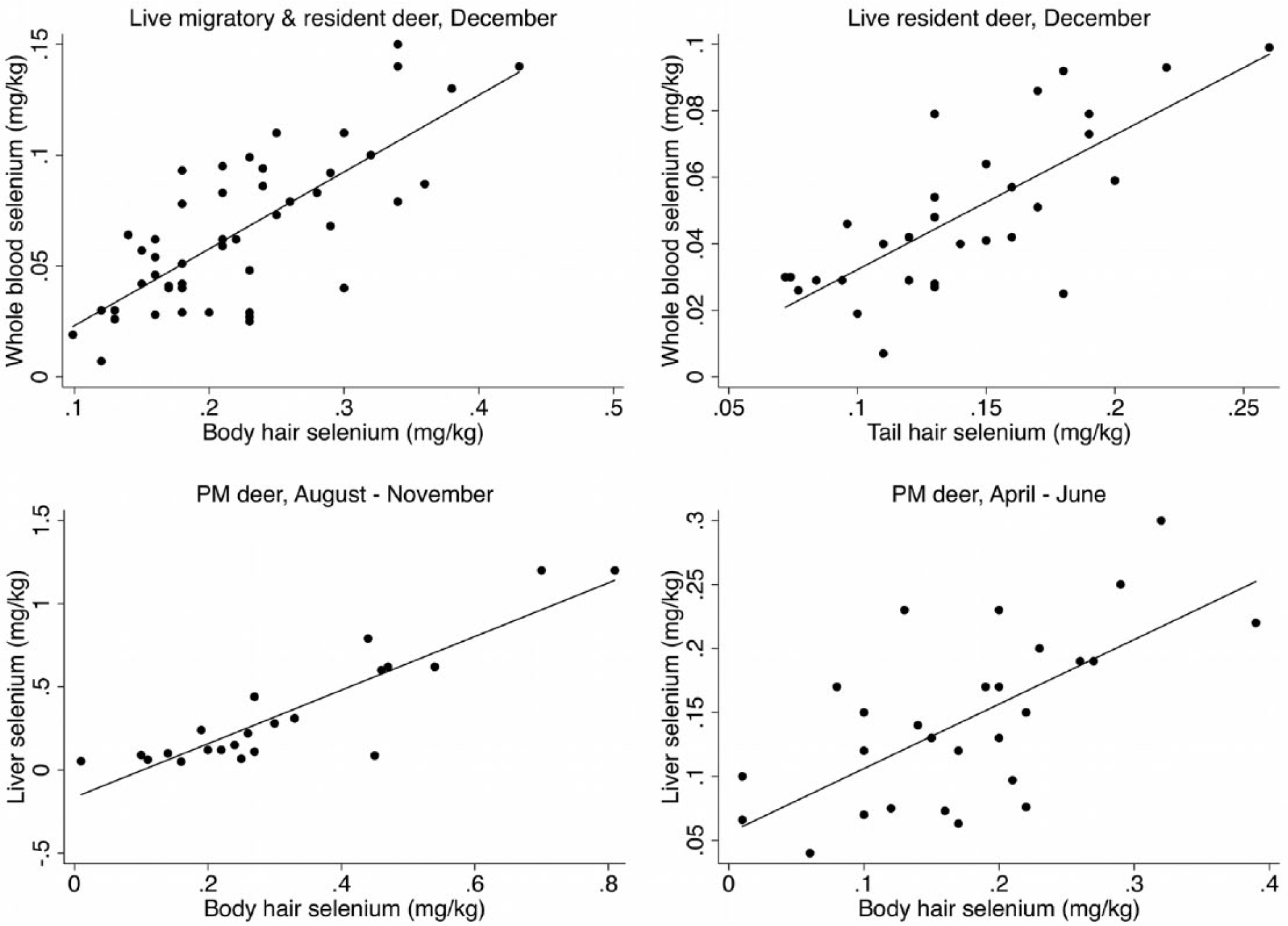
Linear relationship between whole blood selenium and body and tail hair selenium in live-sampled free-ranging mule deer (
In postmortem-sampled deer, there was a significant correlation and strong linear relationship between liver and body hair selenium concentrations in deer sampled in August–November (ρ = 0.8194,
Spearman rank correlations between selenium (Se), copper (Cu), zinc (Zn), iron (Fe), and manganese (Mn) in liver, and tail and body hair of free-ranging, postmortem-sampled California mule deer (
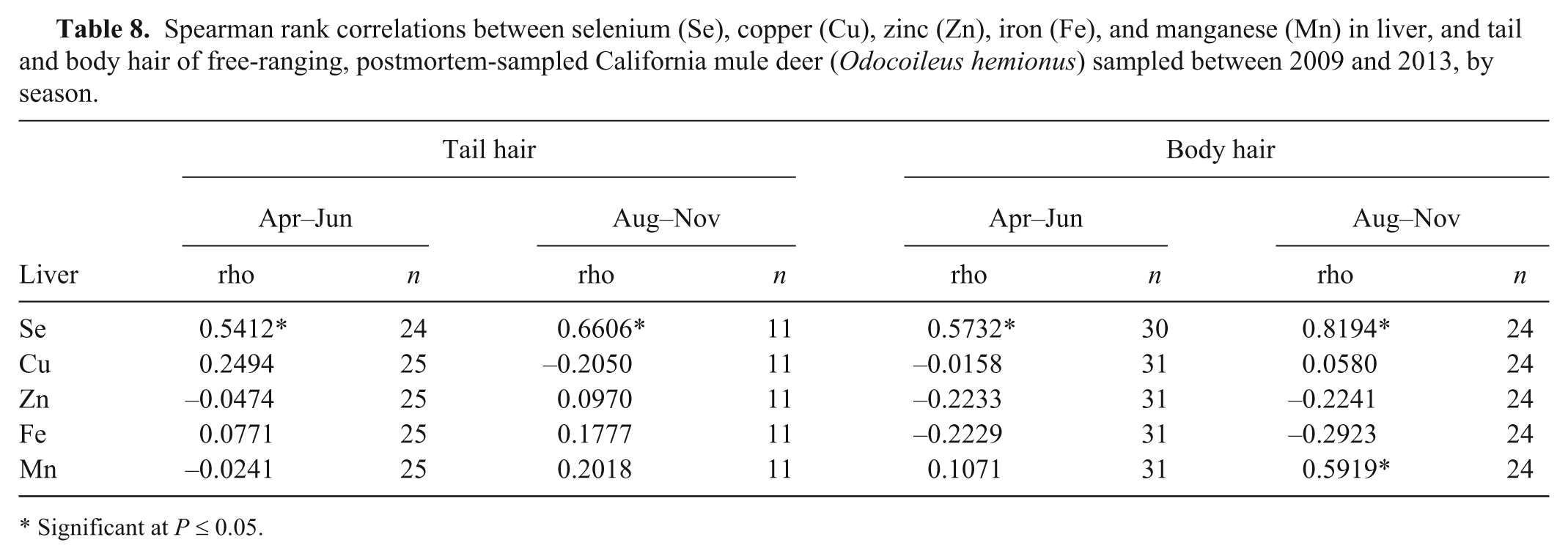
Significant at
Linear regression equations for significant correlations between trace element concentrations in live- and postmortem-sampled free-ranging California mule deer (
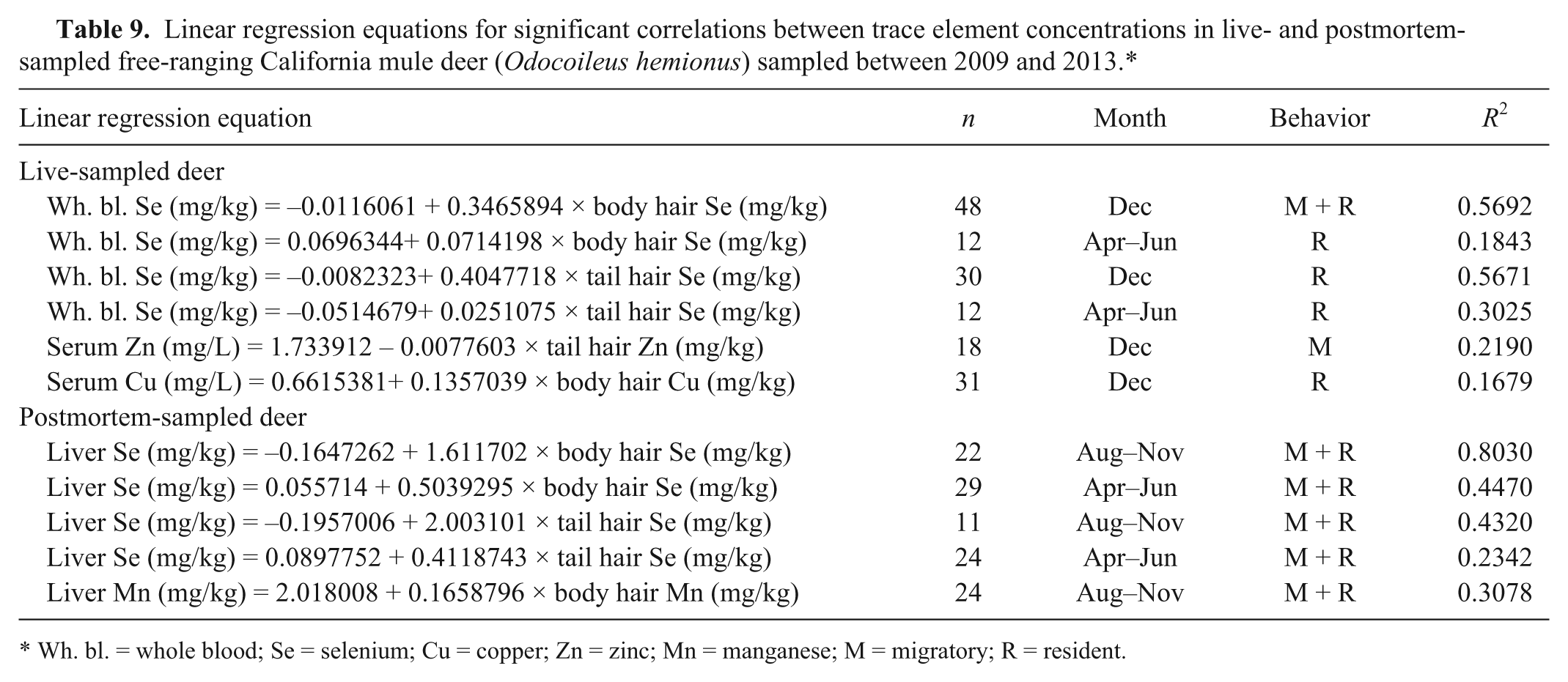
Wh. bl. = whole blood; Se = selenium; Cu = copper; Zn = zinc; Mn = manganese; M = migratory; R = resident.
Discussion
Geographical variations were most consistent across sample types for selenium. Selenium was also the only trace mineral evaluated where moderate to strong correlations and linear relationships were detected between sample types in both live- and postmortem-sampled deer. The associations were stronger in the fall and early winter than in the spring and early summer. The winter coat grows during August–September,
17
and personal observations by one author (G. Gerstenberg) confirm that deer in the Sierra Nevada have fully grown winter coats by November–December. Although deer sampled in April–June are changing into the summer coat, it is more likely that a collected sample contains some of the long winter coat in addition to the summer coat, which may explain the weaker correlation during this sampling period. Tail hair selenium was only significantly correlated with whole blood selenium in resident deer, likely because trace minerals in tail hair is reflective of hair grown at different times of the year, and the diet of resident deer is more consistent than that of migratory deer. Selenium concentrations in whole blood have previously been shown to significantly correlate with body hair selenium in Holstein cattle (
The correlation between serum copper and body hair copper in December was moderate, but the linear relationships was poor (
Similarly, there appeared to be a correlation between serum zinc and body hair zinc in migratory deer, but the relationship was not linear (
Based on this analysis with limited sample size, the strong linear relationship between liver selenium and body hair selenium in December could indicate that body hair may serve as a supplementary sample for measuring the selenium status of a herd if samples are collected during or shortly after the growth of the winter coat. If collected later in the winter, hair could possibly serve as a retrospective indicator of selenium levels at the time of entry into the winter. None of the other significant correlations appeared to be sufficiently strong to conclude body hair should be used as reliable supplementary samples.
Selenium concentrations are of interest in California deer, as several studies have reported low concentrations of this trace mineral.7,10,25 The exact reference ranges for selenium in deer have not been established but based on a literature review, it was suggested to categorize blood selenium values <0.05 mg/L as extremely deficient and 0.08–0.50 mg/L as adequate, and liver selenium values <0.15 mg/kg (wet weight) as deficient, 0.15–0.22 as marginal, and >0.22 mg/kg (wet weight) as adequate. 11 Based on the suggested reference values for blood, 28 out of 84 (33.3%) of Sierra Nevada or Sierra foothills deer, but none of the coastal deer, could be categorized as “extremely deficient” for selenium, and based on the criteria for liver, 6 out of 14 (42.9%) of the coastal and southern California deer and 23 out of 40 (57.5%) of the Sierra Nevada or Sierra foothills deer could be considered selenium deficient. The numbers from live- and postmortem-sampled deer are not directly comparable because they do not originate from the same deer. The whole blood selenium concentrations detected in both coastal and Sierra Nevada and Sierra foothills deer were comparable to findings in the 1980–1988 study of 1,695 mule deer from 42 herds in California that reported mean herd selenium values of 0.060–0.178 mg/kg in coastal herds and 0.068–0.084 mg/kg in Sierra Nevada and Sierra foothills deer herds. 25 The clinical implications of possible selenium deficiency in free-ranging deer are difficult to determine. One study found that selenium supplementation increased pre-weaning fawn survival, 10 but no reports of clinical white muscle disease due to selenium deficiency have been published in California.
Serum copper, zinc, and most serum iron concentrations fell within published suggested adequate ranges of 0.60–1.30 mg/L for copper, 0.50–1.00 mg/L for zinc, and 1.52–2.77 mg/L for iron, 27 whereas some serum iron values exceeded this range. Although 3 out of 15 (20%) coastal and southern California deer and 12 out of 40 (30%) Sierra Nevada and Sierra foothills deer had liver copper values categorized as “deficient” when using the ranges suggested by studies on red deer (<4 mg/kg wet weight = deficient, 4–6 mg/kg = marginal, and >6 mg/kg = adequate), 35 the ranges observed were comparable to reports of mule deer in Colorado,30,36 Montana, 23 and South Dakota. 37 A total of 8 deer could be categorized as deficient for both selenium and copper based on liver concentrations; 6 from the Sierra Nevada, and 2 from southern California. Liver zinc, manganese, and most liver iron concentrations fell within published suggested reference ranges (30–110 mg/kg wet weight for zinc, 3.00–3.40 mg/kg for manganese, and 120–300 mg/kg for iron). 27 The concentrations were similar to the ranges detected in mule deer in South Dakota, Montana, or Colorado,23,36,37 although especially in the Sierra Nevada areas, some iron values exceeded the suggested reference range and the median values were higher than what was detected in other studies.28,29
No prior information is available on hair trace mineral concentrations in California deer, but the body hair selenium concentrations were comparable to a Washington study that measured body hair selenium in 10 mule deer (mean = 0.150 mg/kg, range = 0.01–0.314 mg/kg). 9 Body hair copper was comparable to concentrations detected in mule deer in Idaho, 8 and body hair copper, zinc, and manganese were similar to the mean body hair zinc and iron concentrations detected in Alaskan moose (range of means: 1.4–14.7 mg/kg for copper, 54.3–135.6 mg/kg for zinc, and 0.1–15.7 mg/kg for manganese). 14 Body hair iron was variable, and median body hair iron concentrations exceeded the range of means (24.6–66.8 mg/kg) observed in Alaskan moose. 14
This study reports trace mineral concentrations in tail and body hair of California deer, and assesses the utility of this sample type in measuring trace mineral levels in free-ranging deer. Furthermore, new data is provided on copper, zinc, and iron concentrations in serum and liver of free-ranging California deer. Body hair may serve as a supplementary sample to measure selenium if collected during or shortly after growth of the winter coat, and if collected later, it could be a rough measure of the trace element status at entry into the winter. For other trace minerals, the correlations between concentrations in hair and other sample types were not strong enough to conclude that hair can be used as a reliable supplementary sample to evaluate the true trace element status of free-ranging deer, but limited sample sizes may have influenced the results. Further information on how trace mineral concentrations in hair, liver, and blood relate in an individual animal may contribute to a better understanding of how hair can be utilized to assess the trace element status. In addition, regionally relevant trace mineral reference values in different sample types need to be established.
Footnotes
Acknowledgements
We are grateful to the many California Department of Fish and Wildlife biologists, scientific aids, analytical chemists, volunteers, and other field personnel for technical assistance and expertise needed for deer capture, sample collection, sample processing, and sample analysis. We also thank Ian Holser, Larry Melton, and Sabine Hargrave from the California Animal Health and Food Safety Laboratory for their technical assistance.
a.
Coda Netgun, Coda Enterprises, Mesa, AZ.
b.
Pneu-dart, Williamsport, PA.
c.
Telazol, Fort Dodge Animal Health, Fort Dodge, IA.
d.
AnaSed, Lloyd Inc., Shenandoah, IA.
e.
Whirl Paks, Nasco, Fort Atkinson, WI.
f.
FISONS, Accuris Model, Thermo Optek Corp., Franklin, MA.
g.
Genzyme Diagnostics, Blaine, MD.
h.
Kimax graduated tube, Kimble Chase, Vineland, NJ.
i.
JT Baker Instra-Analyzed, Avantor Performance Materials Inc., Central Valley, PA.
j.
AIM 500 and AIM 600, Seal BD-50; SEAL Analytical Inc., Mequon, WI.
k.
TraceMetal grade, Thermo Fisher Scientific Inc., Waltham, MA.
l.
Optima Grade, Thermo Fisher Scientific Inc., Waltham, MA.
m.
Version IC 12.1, Stata Corp., College Station, TX.
n.
Version 10.1, ESRI Corp., Redlands, CA.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the California Department of Fish and Wildlife.
